Abstract
Imaging followed by endoscopic ultrasound (EUS)-guided therapy has become the preferred modality for treating pancreatic pseudocysts over surgical or radiological interventions. However, there continues to be a lack of consensus regarding the utility of endoscopic retrograde cholangiopancreatography (ERCP) before and after cyst drainage. We describe 4 cases of large pancreatic pseudocyst causing extrinsic biliary obstruction treated successfully with endoscopic cystogastrostomy decompression using a lumen-apposing self-expandable metal stent (LAMS) without ERCP. Endoscopic cystogastrostomy with LAMS was successful in all cases; none required ERCP. As endoscopic therapeutic systems continue to evolve, ERCP is becoming less essential to improve outcomes in patients with extrinsic biliary obstruction by pseudocysts and walled-off necrosis.
Introduction
Pancreatic pseudocysts are localized, enzyme-rich fluid collections which can form as sequelae from ductal disruption or necrosis in the setting of pancreatitis. 1 Although they are considered a common complication of chronic pancreatitis, the incidence is only 1.6 to 4.5 per 100 000 adults per year. 2 Pseudocysts are found via imaging, and patients with large pseudocysts may present with jaundice or elevated bilirubin due to extrinsic biliary obstruction. 3 Management was historically primarily surgical but has shifted to minimally invasive approaches (percutaneous and/or endoscopic drainage) in past years due to evidence supporting improved patient outcomes. 4
Case Series
Case 1
A 38-year-old woman with a prior history of necrotizing pancreatitis was admitted for a 1-day history of acute epigastric abdominal pain radiating to the right side and back, not associated with meals, diarrhea, or other bowel habit changes. The patient’s prior episode of necrotizing pancreatitis was only managed medically and not treated with any invasive interventions. Initial laboratory studies indicated pancreatic involvement (Table 1). Computed tomographic (CT) imaging revealed a large walled-off, heterogeneous collection in the pancreas resulting in extrahepatic biliary obstruction (Figure 1). Endoscopic retrograde cholangiopancreatography (ERCP) was attempted; however, the major papilla was not found due to mass effect against the first and second portion of the duodenum and the procedure was aborted. Endosonographic findings included an anechoic lesion in the pancreatic body, suggestive of a pseudocyst, measuring 45 mm × 26 mm with multiple compartments and a thick outer wall. There was no associated mass, but internal debris was noted within the fluid-filled cavity. It was decided to perform a cystogastrostomy using the AXIOS stent system. Once an optimal location was identified in the stomach, color Doppler imaging was used to check for any vessels between the stomach and the cyst. The AXIOS stent and electrocautery device were then introduced through the working channel, creating an ostomy with the cautery tip. The AXIOS device was advanced into the cyst, and a 15 mm × 10 mm AXIOS stent was deployed successfully, with the flanges closely approximating the walls of the cyst and stomach to form a cystogastrostomy. The patient’s symptoms resolved after 6 interventions (Figure 2). The patient eventually underwent a successful laparoscopic cholecystectomy.
Clinical and Endoscopic Data.


Large pancreatic pseudocyst measuring in the pancreatic neck, body, and tail (yellow arrow) abutting the posterior wall of the gastric body.

Interval cystogastrostomy with subsequent reduction in size of pancreatic pseudocyst (blue arrow).
Case 2
A 48-year-old man with a recent episode of necrotizing pancreatitis complicated by pseudocyst formation with extrahepatic biliary obstruction was admitted for evaluation of epigastric pain, decreased appetite, and jaundice. During admission, ERCP was attempted but unsuccessful due to a severely edematous duodenum. Initial laboratory studies indicated acute pancreatitis and jaundice (Table 1). The CT imaging revealed a large pseudocyst with multiple loculations in the pancreatic body and tail, the largest of which was extrinsically obstructing the duodenum and common bile duct (Figure 3). Endosonographic findings identified an anechoic lesion suggestive of a pseudocyst in the pancreatic body, measuring 70 mm × 60 mm in maximal cross-sectional diameter, which was too large to measure accurately. The lesion had a single compartment without septae, a thick outer wall, no associated mass, and internal debris within the fluid-filled cavity. It was decided to perform a cystogastrostomy using the AXIOS stent system. Once an optimal location was identified in the stomach, color Doppler imaging was used to check for any vessels between the stomach and the cyst. The AXIOS stent and electrocautery device were then introduced through the working channel, creating an ostomy with the cautery tip. The AXIOS device was advanced into the cyst, and a 15 mm × 10 mm AXIOS stent was deployed successfully, with the flanges closely approximating the walls of the cyst and stomach to form a cystogastrostomy. Afterward, 3 repeat endoscopic interventions/necrosectomy led to complete resolution. The patient underwent a successful robotic cholecystectomy (Figure 4).

Large pancreatic pseudocyst (blue arrow) compressing bile duct and leading to biliary dilation (yellow arrow).
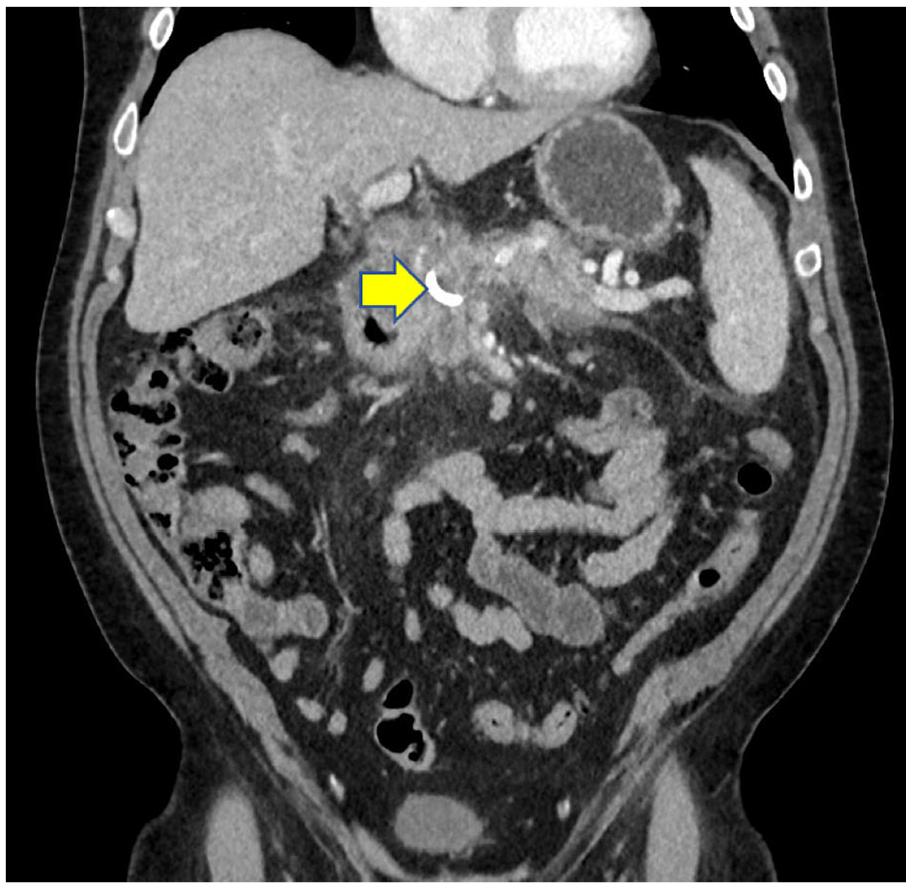
Post-cystogastrostomy (yellow arrow) decompression of pseudocyst with resulting relief of biliary obstruction.
Case 3
A 30-year-old man with a history of heavy alcohol use presented to the emergency department with sharp epigastric pain, nausea, and vomiting. Initial laboratory studies suggested acute pancreatitis with evidence of cirrhosis (Table 1). The CT imaging revealed a nodular liver consistent with cirrhosis and a large pseudocyst with loculations in the pancreas, the largest of which was along the posterior wall of the stomach and causing extrahepatic biliary obstruction (Figure 5). Endosonographic examination revealed an anechoic lesion suggestive of a pseudocyst in the pancreatic body, measuring at least 40 mm × 39 mm in maximal cross-sectional diameter. The lesion had a single compartment without septae, a thick outer wall, no associated mass, and internal debris within the fluid-filled cavity. It was decided to perform a cystogastrostomy using the AXIOS stent system. Once an optimal location was identified in the stomach, color Doppler imaging was used to check for any vessels between the stomach and the cyst. The AXIOS stent and electrocautery device were then introduced through the working channel, creating an ostomy with the cautery tip. The AXIOS device was advanced into the cyst, and a 15 mm × 10 mm AXIOS stent was deployed successfully, with the flanges closely approximating the walls of the cyst and stomach to form a cystogastrostomy. The patient achieved complete resolution of symptoms within 4 days (Figure 6). The patient underwent 4 necrosectomies before removal of lumen-apposing self-expandable metal stent (LAMS).

Large pseudocyst along the posterior wall of the stomach (yellow arrow) with multiple pseudocysts and acute pancreatitis (blue arrow) causing extrahepatic biliary obstruction.

Post-cystogastrostomy changes with interval decrease in pancreatic pseudocyst size (yellow arrow).
Case 4
A 63-year-old man with a prior history of pancreatitis presented to the emergency department due to progressive icterus and jaundice. Initial laboratory studies revealed a cholestatic pattern of liver injury (Table 1). Magnetic resonance imaging identified a large walled-off necrotic collection replacing the pancreatic parenchyma with diffuse dilation of the intrahepatic biliary tree from extrahepatic biliary obstruction (Figure 7). Endosonographic examination revealed an anechoic lesion suggestive of a pseudocyst in the pancreatic body, measuring 42 mm × 38 mm in maximal cross-sectional diameter, without obvious communication with the pancreatic duct. The lesion had a single compartment without septae, a thick outer wall, no associated mass, and internal debris within the fluid-filled cavity. It was decided to perform a cystogastrostomy using the AXIOS stent system. Once an optimal location was identified in the stomach, color Doppler imaging was used to check for any vessels between the stomach and the cyst. The AXIOS stent and electrocautery device were then introduced through the working channel, creating an ostomy with the cautery tip. The AXIOS device was advanced into the cyst, and a 15 mm × 10 mm AXIOS stent was deployed successfully, with the flanges closely approximating the walls of the cyst and stomach to form a cystogastrostomy. After a total of 2 procedures, there was a resolution of symptoms (Figure 8) and near normalization of lab abnormalities and LAMS was removed.

Walled-off necrosis (yellow arrow) replacing pancreatic parenchyma with mass effect on the common bile duct resulting in intra/extrahepatic biliary dilation.
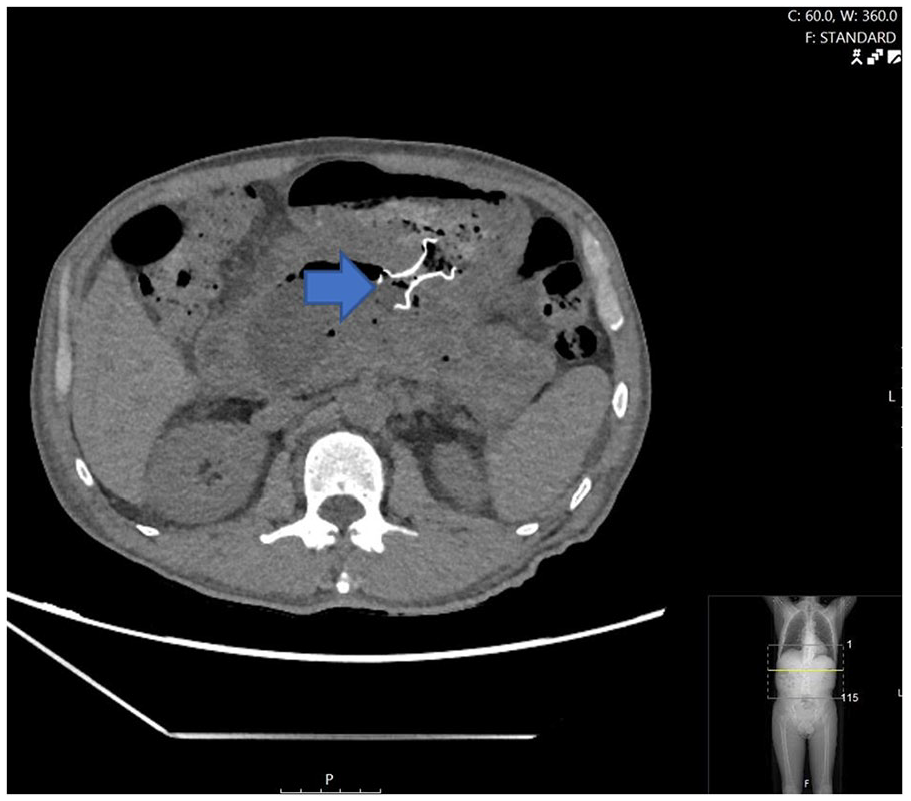
Post-cystogastrostomy with LAMS seated within the large walled-off necrosis (blue arrow).
Discussion
The formation of pancreatic pseudocysts and walled-off necrosis is a well-known complication of pancreatitis. The overall incidence is low though (1.6%-4.5% per 100 000 adults per year). The prevalence of pseudocysts/walled-off necrosis in patients with chronic pancreatitis is 20% to 40% and acute pancreatitis is 10% to 26%. The development of endoscopic and laparoscopic techniques in management of these has helped decrease the mortality considerably. 5 Patients with chronic pancreatitis who develop these complications are represented in Figure 9. 6 As seen in the cases presented above, these patients may present with large, multiloculated lesions which can have a variety of presentations ranging from asymptomatic to abdominal pain, nausea, vomiting, early satiety, weight loss (in cases of gastric outlet obstruction), and jaundice (in case of biliary obstruction).7-13 These symptoms are largely attributed to mass effect on the stomach, duodenum, or biliary tree. These are unlikely to recover spontaneously. Common indications for endoscopic intervention include increasing size of the lesion (>6 cm), persistence of the lesion for more than 6 weeks with symptoms, and/or presentation of aforementioned symptoms at any time.

Rate of complications after chronic pancreatitis.
Currently, there are 3 major approaches for the management of pancreatic pseudocysts/walled-off necrosis: percutaneous techniques which require the creation of an external pancreatic fistula, abdominal surgery which consists of resection, external drainage, and/or formation of a Roux-en-Y gastric bypass, and endoscopic cystogastrostomy creation which requires cyst access and subsequent stent placement for drainage followed by endoscopic necrosectomy. Traditionally, management of pseudocysts had been surgical as past studies showed comparable rates of hospital stay and successful drainage rates with surgical intervention. 8 However, recent data trends all seem to support the use of minimally invasive approaches, such as endoscopic cystogastrostomy and necrosectomy, due to reports of lower mortality rates and hospitalization times. Repeat endoscopic interventions can also be done if required. 9 Percutaneous drainage is reserved as an alternative for patients who exhibit symptoms, have easy percutaneous access windows, and are poor endoscopic and surgical candidates. Lately, trends suggest surgery should be considered only for patients with complex anatomical involvement, where there is a higher risk of hemorrhage or complications with other treatment methods, failed endoscopic or radiologic management, suspected neoplasm, or venous occlusive disease. 10
Endoscopic cystogastrostomy is performed under endoscopic ultrasound (EUS) guidance due to better safety and access. Endoscopists may use various techniques, such as EUS-guided fine-needle aspiration, to access the pseudocyst or walled-off necrosis through the gastric or duodenal wall and into the fluid collection. A long, typically hydrophilic 0.035-inch guidewire is inserted through the needle and coiled within the fluid collection, with positioning confirmed by fluoroscopy. The needle is then withdrawn, leaving the wire in place. A fistula is subsequently created by gradually dilating the gastric mucosa and cyst wall using an endoscopic cannula, catheter dilators, and cautery devices. For pseudocysts, plastic or metal stents can be placed within the collection. 10 In the last couple of years, lumen-apposing metal stents (LAMS) have been preferred over plastic or fully covered biliary stents for management of these lesions and have shown several advantages over them. The bilateral double-walled anchoring flanges hold the gastric or duodenal wall in direct apposition to the wall of the pseudocyst, thus preventing leakage and migration. This can be deployed in simpler steps using integrated cautery. The larger luminal diameter (10-20 mm) allows subsequent direct endoscopic debridement in case of necrosis, often required for effective control of infections. 11
Even with surgical management, reoperation is not uncommon, especially in patients with chronic pancreatitis or persistent alcohol abuse. In these situations, minimally invasive approaches, such as endoscopic cystogastrostomy, are remarkably effective as they allow for precise and effective drainage of collections while also permitting repeated minimally invasive necrosectomy procedures, often through placed covered metal stents such as LAMS. A recent randomized controlled trial showed that EUS-guided endoscopic cystogastrostomy achieved a comparable success rate (95% vs 100%) to surgical cystogastrostomy, while offering advantages such as a shorter hospital stay, lower costs, and improved quality of life scores.12,13
The ERCP, while no longer necessary in diagnosing pseudocysts and chronic pancreatitis, has become a primarily therapeutic tool to relieve biliary and/or pancreatic duct obstruction from choledocholithiasis, pancreatic duct stones, or pancreatic or biliary strictures (intrinsic and extrinsic). 14 Previously, therapy of pseudocysts involved ERCP-assisted transpapillary drainage and pancreatic duct stent placement; however, recent studies suggest the benefit of transpapillary drainage is equivocal in patients undergoing EUS-guided drainage, while the technical success rate of transmural drainage with cystogastrostomy (96.8%) was significantly higher than that of transpapillary drainage with ERCP (44%). 15 As endoscopic technology and techniques continue to evolve, the utility of ERCP in the management of these lesions will likely continue to decline and ERCP will only be reserved for cases in which biliary obstruction is present from ongoing choledocholithiasis or intrinsic biliary strictures.
Farr et al 16 described a case series of 5 children who had a pancreatic pseudocyst developed after trauma. They found that endoscopic cystogastrostomy was a safe and effective method for treatment of these lesions. Will et al 17 conducted a study comparing percutaneous drainage of pseudocysts with EUS-guided treatment and found that interventional endoscopic management of these lesions is a reasonable treatment option with low invasiveness compared with surgery and an acceptable outcome regarding the complication rate (11.1%) and mortality (3.7%). The GEPARD (German Pharmacoepidemiological Research Database) study also found that clinical success of direct endoscopic necrosectomy via cystogastrostomy ranges from 75% to 90% and success is often associated with the size of the walled-off necrosis. 18
In summary, pancreatic pseudocysts and walled-off necrosis are commonly known complications of pancreatitis whose management has evolved over the years as gastroenterologists become more experienced with minimally invasive approaches such as endoscopic cystogastrostomy creation and necrosectomy, if necessary. Here, we illustrate 4 cases with very large symptomatic pseudocysts/walled-off necrosis causing extrinsic biliary obstruction; these cases required only a few interventions over a relatively short interval for full resolution of symptoms without the assistance of biliary or pancreatic duct ERCP. All these interventions were completed entirely endoscopically, illustrating the effectiveness and utility of EUS-guided procedures, namely endoscopic cystogastrostomy, in the management of these lesions. We believe that these endoscopic techniques are the new gold standard for the management of symptomatic pancreatic pseudocysts/walled-off necrosis.
Footnotes
Authors’ Note
This abstract was presented as an oral presentation at the Digestive Diseases Week 2024 in Washington DC. The abstract is currently published in Gastrointestinal Endoscopy, 99(6), AB102. DOI:10.1016/j.gie.2024.04.844.
Author Contributions
LN, SD, and BT wrote the manuscript and conducted the literature review. RB critically reviewed and approved of the article and provided the radiology images. All authors reviewed and approved of the manuscript. RB is the article guarantor.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Verbal informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
