Abstract
Individuals with COVID-19 are prone to a variety of infections due to immune dysregulation. The present report presents a case of actinomycotic infection in the maxillary bone and sinus region in a patient with a history of COVID-19. This case report highlights the importance of considering bacterial infections including actinomycosis when encountering destructive lesions resembling more prevalent fungal infections due to different therapeutic medication protocols. In addition, a literature review of the existing reports of similar post-COVID-19 actinomycotic infection is presented.
Introduction
Severe acute respiratory syndrome coronavirus 2 (SARS-COV-2) causes the coronavirus disease 2019 (COVID-19) which led to a pandemic of more than 650 million cases and 6 million death as of January 2023 in the course of nearly 3 years. The most common signs of COVID-19 are respiratory distress, gastrointestinal (GI) and hepatic diseases, and neurological complications. 1 This infection could develop in fatal outcomes, especially in patients with underlying comorbidities, such as diabetes mellitus, hypertension, obesity, old age, trauma, organ transplant, hematopoietic malignancies, or cardiac, respiratory, or renal disorder. 2 3
The physiopathology suggests that in infected COVID-19 patients, the absolute number of T lymphocytes, and CD4+ and CD8+ T cells are lower than healthy individuals while various inflammatory markers, including interleukin (IL)-2, IL-6, IL-10, tumor necrosis factor-alpha (TNF-α) are at higher levels. 4
This immune dysregulation is exacerbated in patients with metabolic disorders, including hypertension and diabetes and other chronic medical conditions, such as respiratory system disease and cardiovascular disease as well as patients taking drugs, such as steroids and Tociluzimab during COVID treatment. 5 6
Following COVID-19 infection, individuals are prone to other infections, especially those infections for which cell-mediated immunity constitutes an important host defense. Recent studies suggest that some people may develop sinus infections after COVID. 7 The weakened inherent immunity predisposes the patient to fungal and bacterial infections. Fungal infections were observed in SARS patients in earlier studies and were considered the leading cause of death in 25% to 73.7% of patients.8-10
Other studies have found a higher percentage of secondary pulmonary infections (8%-15%) in COVID-19 patients, but it is not clear whether it is bacterial or fungal infection. 11 12 A rare kind of bacterial infection is caused by Actinomyces israelii that could be associated with COVID infection.
The aim of the present study is to present a rare case of actinomycosis and perform a review of literature on post-COVID-19 occurrence of actinomycotic infection.
Case Presentation
A 62-year-old Iranian female attended the office of an oral and maxillofacial surgeon with a chief complaint of pain in the maxillary and zygomatic region. The patient had a history of controlled hypertension and cardiac problems and was taking the following medications: aspirin 80 mg, clopidogrel 75 mg, bisoprolol 2.5 mg, indapamide 1.5 mg, and losartan 12.5 mg. She tested positive for COVID-19 in August 2021 using real-time polymerase chain reaction (RT-PCR) technique. After 10 days, she felt an intense, dull, pulsating pain in the maxillary and zygomatic region. She claimed that the pain was alleviated only with diclofenac. After 1 month, the patient was referred to an otolaryngologist who performed endoscopic sinus surgery. However, the pain did not resolve and was exacerbated. The patient was then referred to a dentist after 20 days who extracted the right maxillary second premolar. However, the pain was not alleviated and the extracted socket was not healed. Finally, 2 months after the diagnosis of COVID-19, the patient was referred to the oral and maxillofacial surgeon.
Extraoral clinical examination was normal with a normal range of mouth opening and no extraoral swelling. In the intraoral examination, necrotic bone was observed in the posterior right maxillary alveolar bone. The roots of the right maxillary first molar were exposed and inflammation was noted in the surrounding gingiva (Figure 1). The right maxillary first molar had severe mobility.
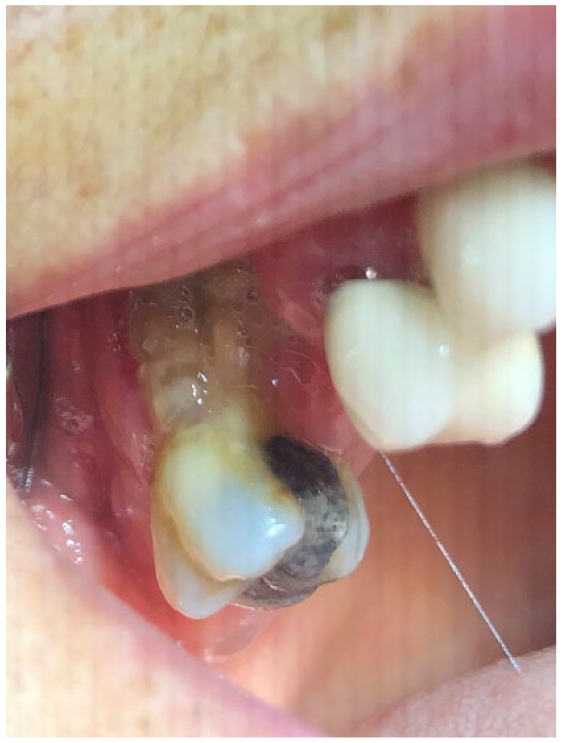
Intraoral photograph depicting exposed roots of the right maxillary first molar with surrounding inflammation.
Cone-beam computed tomography (CBCT) was obtained from the maxillary region and paranasal sinuses. In the CBCT views, a lytic and destructive lesion was observed on the right side of the maxilla. Destruction of the posterior alveolar process, medial wall, anterior wall, lateral wall, and floor of the right maxillary sinus was seen. The right maxillary sinus was completely opacified with soft tissue density and fragments of bone were also seen in the sinus. Evidence of endoscopic sinus surgery was observed in the nasal and paranasal regions. Loss of bone support of the involved teeth was detected without root resorption (Figure 2).

Cone-beam computed tomographic images in (A) coronal and (B) axial sections depicting the lytic and destructive lesion on the right side of the maxilla.
Under general anesthesia, the right maxillary first molar was extracted and a biopsy was obtained from the necrotic bone and granulation tissue in the maxillary sinus and sinus mucosa. The samples were stored in 2 different containers: one with normal saline for culturing and another one with formalin for histopathological examination. Bacterial smear and culture revealed a tangled mass of elongated, rod-shaped cells that branched into filaments about 1 to 2 µm in diameter. Gram staining revealed gram-positive rods and thioglycolate broth growth showed non-acid fast bacteria indicating Actinomyces. Histopathological evaluation using hematoxylin and eosin staining also demonstrated areas of bone necrosis along with inflammatory infiltration and bacterial rods (Figure 3).

Histopathological hematoxylin and eosin staining of the sample indicating focus of actinomycosis with surrounding inflammatory response.
The patient was prepared for surgery by a team containing an oral and maxillofacial surgeon, an oral and maxillofacial radiologist, and an otolaryngologist. Half an hour before the surgery, 1 g of cefazolin was administered via intravenous injection. First, endoscopic sinus surgery was performed to clear the nasal cavity and sinus ostium. Afterward, curettage and removal of necrotic bone was performed using intraoral open sinus surgery with buccal advancement flap and buccal fat grafting to close the oroantral connection. After the operation, cephalexin 500 was administered every 8 hours for 1 week. Routine non-steroidal anti-inflammatory drugs (NSAIDs) were administered in case of pain. Chlorhexidine 0.2% mouthwash was also administered 3 times a day for 1 week. The patient then received high-dosage penicillin G for a period of 6 months administered by the infectious diseases specialist.
The patient was followed 1 week, 1 month, 3 months, 6 months, and 8 months after the surgery. The clinical signs and symptoms started to improve after the surgery. The surgical site and the extraction socket have been completely healed. No evidence of pus discharge and secretions and oroantral fistula was observed in clinical examination (Figure 4).

Intraoral photograph depicting resolution of inflammation and favorable healing of the infection.
Discussion
Actinomycosis is a rare anaerobic bacterial infection caused by gram-positive, non-motile, non-acid fast filamentous bacterial rods. Actinomyces are part of the normal flora commonly seen in the human oral cavity, female urogenital tract, and GI tract. The organism has low virulence and only invades the body to cause deep-seated infections when there is tissue injury or following a break in the normal mucosal barrier. Clinically, actinomycotic infection has 3 subtypes; cervicofacial, thoracic, and abdominal. Cervicofacial type is the most common type of actinomycotic infection. Infection is mostly polymicrobial, established with the help of a companion bacteria by inhibiting host defense, reducing oxygen tension, or by toxin production that facilitates the inoculation of actinomycoses. 13
The infection is characterized by contagious spread, suppurative and granulomatous inflammation leading to multiple abscess formation, and sinus tracts that may discharge yellow-colored sulfur granules. In the jaw bones, it can result in osteomyelitis if untreated. Imaging findings are nonspecific and are non-contributory in diagnosing fungal or bacterial osteomyelitis, but will help in assessing the degree of soft tissue and bone involvement. Only the biopsy specimen of the involved tissue could confirm the diagnosis in which the filamentous shape of the microbes that resemble fungal hyphae could be observed.
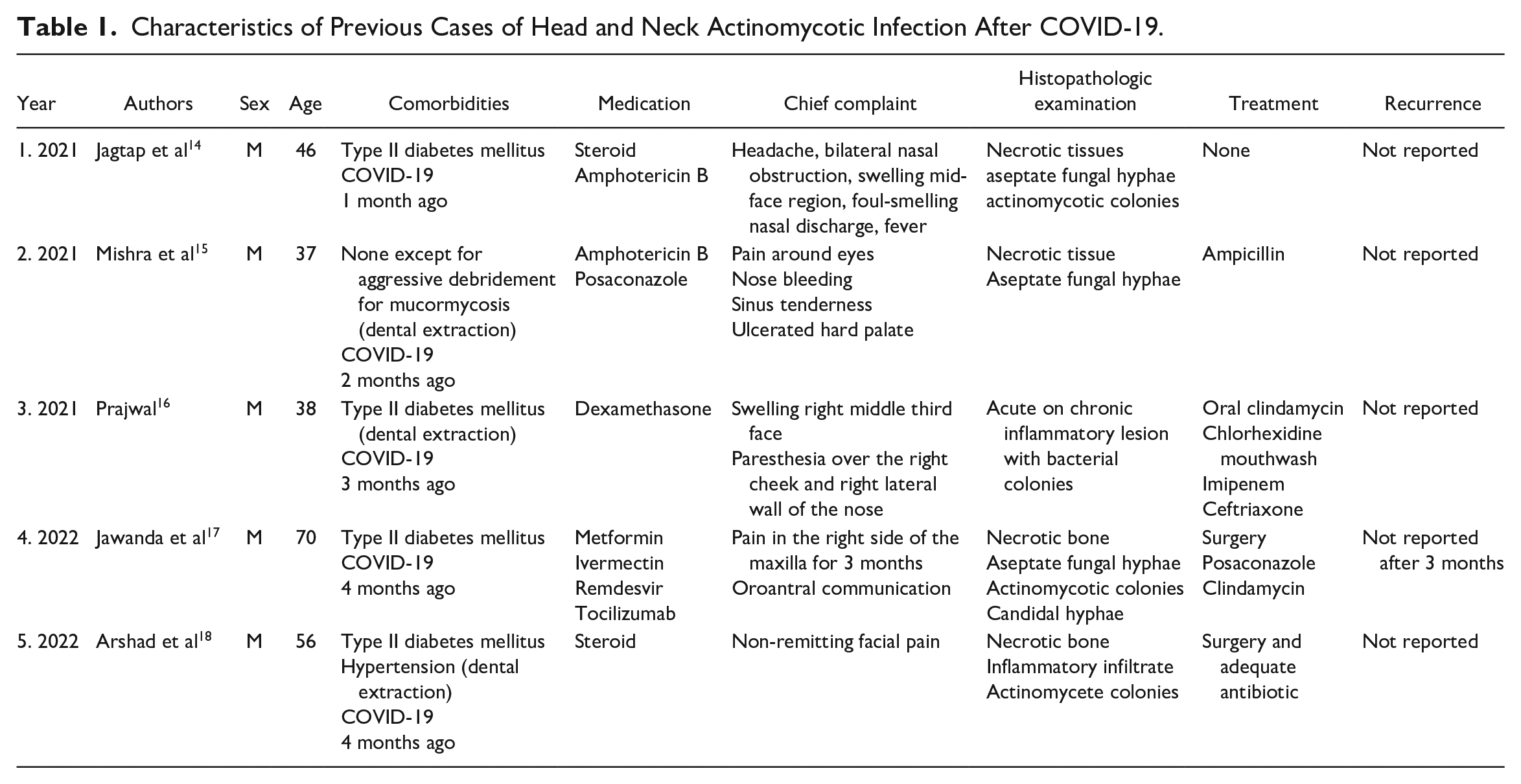
There are only a few reported cases of actinomycosis secondary to COVID-19 infection in the literature (Table 1). All of them report male patients, with a mean age of 49-year-old, with a history of COVID-19 for 1 to 4 months before the start of the bacterial infection and with other comorbidities, such as type II diabetes and other predisposing conditions, including dental extractions. Hyperglycemia in diabetic patients is associated with impairment of immune response and interference with the wound healing process. Therefore, resistance to bacterial infection is lower in diabetic individuals compared with those without diabetes.19,20 Similarly, administration of corticosteroids can lead to immune suppression, in turn increasing the susceptibility of the individuals to infective diseases. 21 The reported cases mostly revealed co-infection of actinomycosis and mucormycosis in the individuals. A systematic review in 2021 revealed 101 cases of post-COVID-19 mucormycosis, of which 82 were from India. Diabetes, extensive use of corticosteroids, and a background of COVID-19 appears to increase incidence of mucormycosis. 19 Therefore, mucormycosis is one of the most important differential diagnoses to consider in such cases. Another bacterial infection that needs to be considered is nocardiosis. Nocardia are gram-positive aerobic filamentous bacteria with similar cytomorphologic features with actinomycete. However, they differ most prominently in their ability or lack thereof to retain acid-fast staining. 22 Infections with the two microorganisms also may show similar clinical manifestations. In addition, nocardiosis has been reported in COVID-19 patients. 23 However, since the bacterial tests in the present case revealed non-acid-fast features, a diagnosis of actinomycosis was made.
Characteristics of Previous Cases of Head and Neck Actinomycotic Infection After COVID-19.
The current report presented a case of actinomycosis in a 62-year-old female patient with a history of controlled hypertension and cardiac problems and showing clinical symptoms 10 days after testing positive for COVID-19. The radiographic appearance of bone destruction and necrosis in the sinonasal region of COVID-19 patients rises the more common possibility of fungal infections including mucormycosis. Improper diagnostic tests and culture can lead to misdiagnosis and thus hinder adequate and timely treatment with antibiotic therapy. Therefore, it is important to consider the possibility of imitating bacterial infections, such as actinomycosis in these cases.
Typical management of cervicofacial actinomycosis consists of conservative debridement and intravenous antibiotic therapy. 24 The same regimen used in our case report allowed for the resolution of infection.
Conclusion
This case reports highlights the importance of histopathological diagnosis in routine clinical practice. The prevalence of actinomycosis in COVID-19 patients is considerably less than mucormycosis. Definitive treatment cannot be provided in cases of misdiagnosis, which can lead to substantial morbidity and mortality. Histopathological examination of these lesions is crucial for a definitive diagnosis and prognosis of the lesion.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
