Abstract
The Delta variant of COVID-19 has been associated with severe disease causing a surge in the second half of 2021. Atypical pathogens can be present in those in particular with severe ARDS and can contribute to excess morbidity and mortality. We must maintain a high level of suspicion for these pathogens as this can present an opportunity to dramatically improve the prognosis of a patient with COVID-19 ARDS. However, lend caution to Mycoplasma IgM serology as this can be a false-positive. If suspicion remains high for Mycoplasma pneumoniae infection, sputum polymerase chain reaction (PCR) for M pneumoniae is the gold standard for diagnosis. We present the case of a 42-year-old female with COVID-19 Delta variant presumed ARDS who had co-infection with M pneumoniae confirmed by endotracheal sputum aspirate PCR with rapidly improving oxygenation and extubation within 4 days of effective antibiotic therapy.
Keywords
Introduction
The Delta variant of COVID-19 became known to the world in the second half 2021 causing a surge in severe cases with many patients progressing to acute respiratory distress syndrome (ARDS) and mechanical ventilation requiring 100% oxygen and high positive end–expiratory pressure (PEEP). Almost 2 years into the pandemic, we had new tools to fight severe disease, such as tocilizumab making its way into Infectious Diseases Society of America (IDSA) guidelines. 1 Although there were reductions in mortality in COVID-19 ARDS with the use of tocilizumab and dexamethasone, Delta variant was associated with worse outcomes than its predecessor, Beta variant. 2 In an analysis at INOVA healthcare of recorded outcomes of mechanically ventilated patients with COVID-19, the mean duration of mechanical ventilation was 14.6 days. 3 This is consistent with the well-recognized fear of intubating these patients, as many do not come off of the ventilator for a long time if at all. In our community, we had many suspected COVID-19/Mycoplasma pneumoniae co-infections based on M pneumoniae IgM positivity, but all but 1 were false-positives that did not improve despite the addition of azithromycin. We report a case of co-infection of M pneumoniae and COVID-19 mimicking COVID-19 ARDS with rapid oxygenation improvement with effective antibiotic therapy with azithromycin for M pneumoniae once it was diagnosed.
Case
A 42-year-old female with a past medical history of morbid obesity, asthma on as-needed albuterol rescue inhaler, hypertension, and bipolar disorder presented to the emergency department with severe shortness of breath during the Delta variant surge. She was found to be severely hypoxic with oxygen saturation of 47% on arrival. She was placed on bi-level positive airway pressure ventilation with poor response; she ultimately required intubation with mechanical ventilation when transferred to the intensive care unit (ICU). Computed tomography angiography (CTA) chest was negative for pulmonary embolism but showed severe COVID-19 with ARDS as shown in Figure 1. The patient required PEEP of 10 cm H2O with PaO2/FiO2 ratio of 79 on the Horowitz Lung Function scale which is associated with 45% mortality. She was sedated with midazolam and propofol, with fentanyl for analgesia. She was started on dexamethasone 10 mg IV BID and remdesivir. She also had a C-reactive protein (CRP) of 17.4, and was a candidate for tocilizumab, which was given in addition to standard treatment. Subsequent testing revealed a positive IgM antibody to M pneumoniae, so she was started on azithromycin by the infectious disease consultant. Given the low sensitivity of testing for IgM M pneumoniae antibodies, sputum M pneumoniae polymerase chain reaction (PCR) was performed from an endotracheal aspirate, which was also positive. After starting azithromycin, she rapidly began oxygenating better as evidenced by decrease in oxygen requirements from 85% fraction of inspired oxygen (FiO2) on day 1 to 50% by day 3. By day 4, she was on 35% FiO2 with PEEP of 5. She was given lung protective ventilation by 4 mL/kg Tidal Volume (TV) by ideal body weight. Nevertheless, she did develop a pneumomediastinum. By day 4, she was on minimal ventilation settings and was successfully extubated. She did, however, continue to struggle with agitation and confusion likely due to sedation causing ICU delirium. She was subsequently moved to the medical/surgical floor on room air. She continued to be altered but biochemical workup was negative as well as magnetic resonance imaging (MRI) of the brain. She recovered to baseline nearly 2 weeks after her admission and was subsequently discharged.
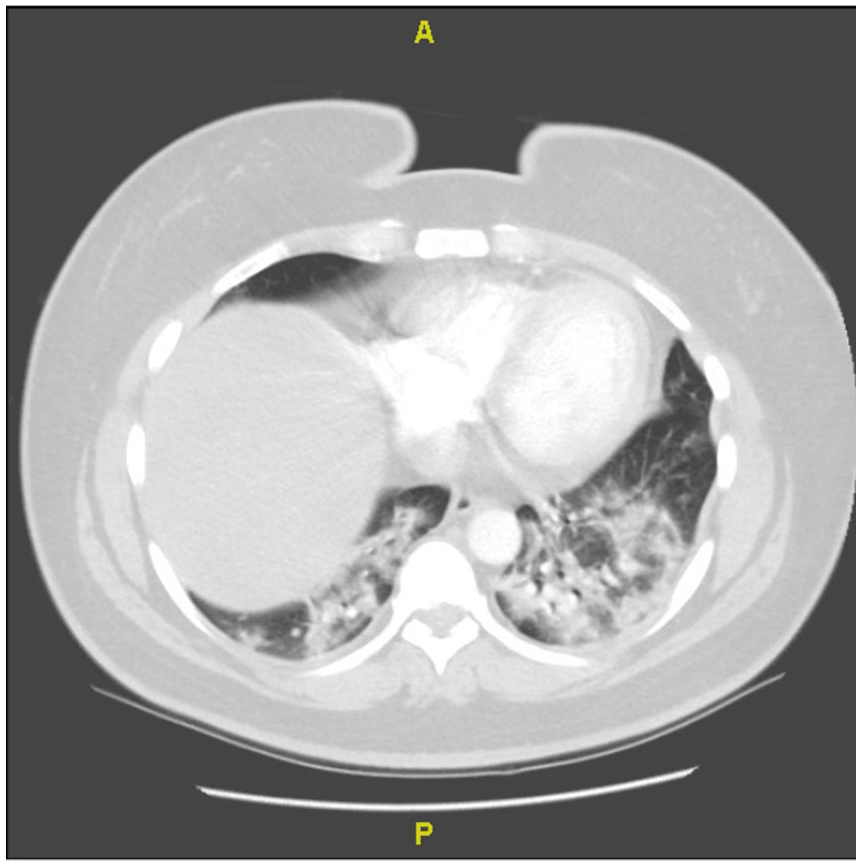
Computed tomography (CT) of the chest with contrast demonstrating diffuse ground glass opacities with dense consolidation from the bases to the apex on admission.
Discussion
Co-infection with M pneumoniae and COVID-19 has been described in the literature.4-7 There are no current guidelines on when to test for concomitant Mycoplasma infection, however, or when to use procalcitonin as a guide in COVID-19 infection. The described studies make a case for testing for other treatable infections; in those studies, serologic markers were used for diagnosis. There are, however, many false-positives with testing for the IgM M pneumoniae antibody. 8
There are concerns when using single serologic tests for diagnosis of M pneumoniae infection. Some individuals may not mount an IgM response and some will suffer from cross reactivity because a substantial amount of healthy blood donors have measurable IgG, IgA, and IgM antibodies. 9 Acute convalescent sera have been used in the past with IgM measured in the acute setting followed by IgG after the illness resolves, but there are limitations with this method as well. 9 The current gold standard for diagnosis of M pneumonia infection is real-time PCR.9,10 None of the above studies incorporated PCR as we did with this patient, demonstrating that this infection was a true infection. Diagnosis of M pneumoniae infection is not routinely used for patients but can be useful in severe pneumonia when the patient is not responding to standard antibiotics. 10 Current guidelines from American Thoracic Society(ATS)/IDSA recommend macrolides as part of the current regimen for routine community acquired pneumonia, thus negating the utility of testing in this particular group. 11 With COVID-19 introduction and the potential for severe pneumonia with ARDS and high associated mortality, we would like to make an argument in favor of checking for certain pathogens such as M pneumoniae with severe COVID-19 ARDS. This case illustrates the importance of effective antibiotic therapy for specific pathogens because co-infection may be present and may be a significant contribution to the morbidity of the patient. This case illustrates a patient with a modifiable factor which can be treated, unlike typical COVID-19 ARDS which has high mortality. There are, however, issues with this strategy, as a negative IgM for M pneumoniae does not necessarily rule out infection, nor does treating with azithromycin in COVID-19 ARDS have any mortality benefit.9,12 However, further clinical trials will need to be done as those were mainly retrospective studies showing no benefit. 12 The main purpose of this case report is to illustrate the high index of suspicion that we need to have as clinicians so we can treat our patients appropriately and improve outcomes, especially in a pandemic, when outcomes for COVID-19 ARDS have been so poor.
Conclusion
Maintain a high level of suspicion for other etiologies of pneumonia particularly in patients with severe COVID-19 ARDS as this can be the difference between life and death in these patients. Further trials will be needed to evaluate azithromycin in this specific subpopulation of severe COVID-19 ARDS.
Footnotes
Acknowledgements
We thank the patient for allowing us to share this with the medical literature.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Written informed consent was obtained from the patient for their anonymized information to be published in the article.
Patient Consent
Patient consent was obtained for this case report.
HCA Healthcare Disclaimer
This research was supported (in whole or part) by HCA Healthcare and/or an HCA Healthcare affiliated entity. The views expressed in this presentation represent those of the author and do not necessarily represent the official views of HCA Healthcare or any of its affiliated entities.
