Abstract
We present the case of a 23-year-old man with a previous deceased-donor renal transplant maintained on tacrolimus and prednisone who developed culture-negative endocarditis (CNE) of the mitral and aortic valves. He was suspected of being co-infected with Bartonella henselae and Coxiella burnetii, confirmed with serology testing. He was successfully managed with appropriate antibiotics and dual valve replacement.
Keywords
Introduction
Infective endocarditis (IE) refers to infection of the endocardial surface of the heart, including one or more heart valves or infection of an intracardiac device. Typically, the in-hospital mortality rate is approximately 18% to 23%, and the 6-month mortality rate is slightly higher at 22% to 27%. Early appropriate antibiotic stewardship can reduce adverse sequelae from IE.1 -5
Culture-negative endocarditis is a huge diagnostic dilemma for the clinical team, as the ambiguity of not knowing the causative organism may result in untimely delays in instituting appropriate antimicrobial therapy. Bartonella henselae is an exceedingly rare cause of CNE and Coxiella burnetii, but to the authors’ knowledge, co-infection of both these pathogens has not been previously described.
Thus, we present a novel, suspected case of a deceased-donor renal transplant recipient with CNE who had positive serology analysis for both Bartonella henselae and Coxiella burnetii. Clinicians should be aware of the possible co-infection of rare, atypical organisms in an immunocompromised patient with CNE.
Case Report
A 23-year-old male medical student with a history of a previous deceased-donor renal transplant 5 years ago for end-stage renal disease (ESRD) due to membranoproliferative glomerulonephritis (MPGN) initially presented with a 2-week duration of constitutional symptoms of intermittent fever and fatigue. His medical regimen included tacrolimus and prednisone as prescribed by his consultant transplant nephrologist. He did not report any recent antecedent viral infection, pet, or travel history; however, he attended clinical rotations in both inpatient and outpatient settings. Prior to the onset of symptoms, he was physically active and regularly participated in noncontact, recreational sports. He never contracted coronavirus 2019 (COVID-19) infection.
His vital signs on presentation revealed a blood pressure of 112/67 mmHg, heart rate of 103 beats per minute and regular, with pulse oximetry of 98% on ambient air, temperature of 37.1°C, and body mass index of 22.6 kg/m². On physical examination, he was alert and oriented without any neurological deficits. There was an S3 with a new-onset 3/6 holosystolic murmur and scattered crackles on auscultation of the lung fields. There was mild peripheral edema. Coronavirus 2019 polymerase chain reaction (PCR) testing was negative upon admission to the emergency department. A 12-lead electrocardiogram indicated borderline sinus tachycardia without any acute dynamic changes consistent with ischemia and/or infarction. A portable chest radiograph indicated pulmonary edema without cardiomegaly.
His initial laboratory investigations revealed leukocytosis and thrombocytosis with normal hemoglobin. Renal function tests reflected mild acute kidney injury. Hepatic function panel and coagulation studies were normal (Table 1). Cardiac troponin, NT(N-terminal-prohormone-brain natriuretic peptide, and D-dimer were all elevated. He was subsequently hospitalized for a tentative diagnosis of suspected IE and acute heart failure. The patient was shifted to the intensive care unit and empirically instituted on renally dosed meropenem, linezolid, levofloxacin, and fluconazole after blood cultures were obtained. In addition, he was administered low-dose, continuous nitroglycerin and furosemide infusions for his acute decompensation. An urgent 2-dimensional transthoracic echocardiogram demonstrated a large vegetation on the anterior mitral valve leaflet with perforation, ruptured chordae tendinae of the mitral valve apparatus and torrential mitral regurgitation with Coandă effect. It also revealed a large vegetation on the aortic valve with torrential aortic regurgitation (Image 1).
Patient’s Diagnostic Testing and Laboratory Investigations.

Abbreviations: ANA, antinuclear factor; ALP, alkaline phosphatase; ALT, alanine aminotransferase; AST, aspartate aminotransferase; BUN, blood urea nitrogen; CCP, cyclic citrullinated peptide; CRP, C-reactive protein; ds-DNA, double-stranded deoxyribonucleic acid; ELISA, enzyme-linked immunosorbent assay; ESR, erythrocyte sedimentation rate; Hb, hemoglobin; HIV, human immunodeficiency virus; IgG, immunoglobulin G; IgM, immunoglobulin M; RF, rheumatoid factor; VDRL, venereal disease research laboratory; WCC, white cell count.
Bold text indicates clinically significant and pertinent results.
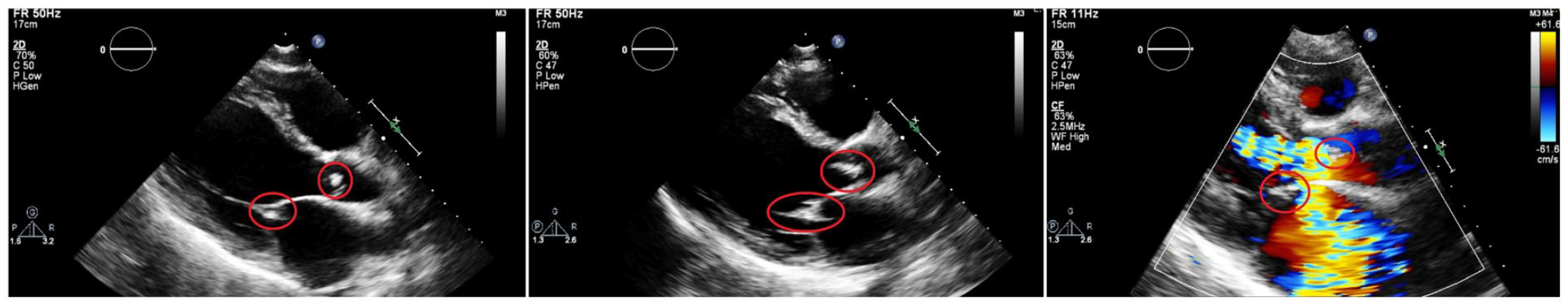
The patient’s 2-dimensional transthoracic echocardiogram series: (a) Parasternal long-axis view: The mitral and aortic vegetations are encircled in red; (b) parasternal long-axis view (magnified): The mitral and aortic vegetations are encircled in red; and (c) parasternal long-axis view (magnified) with color-flow Doppler: The mitral and aortic vegetations are encircled in red, with torrential mitral and aortic regurgitation, respectively.
Repeat transthoracic echocardiography 3 days later indicated both interval enlargement of the vegetations and deteriorating valve function. The infectious diseases team was consulted due to the complexity of the case—CNE and concern for drug-drug interactions for antibiotics and renal transplant status. Serology testing eventually returned positive for immunoglobulin (Ig) M titers for both Bartonella henselae and Coxiella burnetii, for which he was immediately transitioned to intravenous doxycycline and gentamicin. He also underwent transesophageal echocardiography, which revealed similar findings except with the possibility of a newly imaged bicuspid aortic valve (BAV) (Image 2).

The patient’s 2-dimensional transesophageal echocardiogram series: (a) Mid-esophageal 2-chamber view: The mitral and aortic vegetations are encircled in red; (b) mid-esophageal 2-chamber (magnified) with color-flow Doppler: The mitral and aortic vegetations are encircled in red, with torrential mitral and aortic regurgitation, respectively; and (c) mid-esophageal aortic valve short-axis view with color-flow Doppler: The aortic vegetation is encircled in red. The yellow triangles delineate the BAV.
The cardiothoracic surgery team was consulted, and he underwent dual valve replacement of the mitral and aortic valves (On-X; Artivion, Inc., Kennesaw, Georgia) 1-week into his hospitalization. His postoperative course was uneventful, and he was started on aspirin and warfarin to maintain an international normalized ratio of 2.5 for his dual mechanical valves, in addition to his pre-existing immunosuppressive regimen. He returned to multidisciplinary Cardiology, Cardiothoracic, and Infectious Diseases outpatient clinics 10 days and 1 month postoperation without any attendant complications. He was also evaluated at 3 months post-discharge and was deemed clinically well with no evidence of prosthetic valve endocarditis or valve dysfunction. Thus far, he has complied with his 18-month course of hydroxychloroquine therapy.6 -8
Discussion
Culture-negative endocarditis presents a huge diagnostic dilemma as the causative organism, and its most appropriate treatment cannot be identified. Generally, these negative blood cultures are due to 1 of the 4 possibilities: (1) Most commonly, it is due to the administration of antibiotics prior to the sample being taken, in which case the typical causative organisms staphylococcus, streptococcus, and enterococcus are the likely culprits; (2) it can be due to fastidious organisms which require special culture techniques and prolonged incubation such as the HACEK (Hemophilus, Aggregatibacter [previously Actinobacillus], Cardiobacterium, Eikenella, Kingella) organisms, nutritionally deficient streptococcus or candida species; (3) it can be non-IE such as marantic endocarditis or Libman-Sacks endocarditis; and (4) it can be due to intracellular pathogens that cannot be detected by blood cultures such as Bartonella species and Coxiella burnetii.9,10 These organisms are usually identified by serology, PCR testing, or histopathological examination.
Bartonella henselae is the etiological agent implicated in “cat scratch disease,” characterized by a papule at the portal of entry, lymphadenopathy, and fever. This infection is usually benign and self-limiting in an immunocompetent host, although adverse sequelae can infrequently occur, such as encephalopathy, oculoglandular Parinaud syndrome, and retinitis. 11 This pathogen can also lead to bacillary angiomatosis, peliosis hepatis, and osteomyelitis. 12 Fulminant infection can occur in an immunocompromised host, such as solid-organ transplant recipients, with resultant thrombotic microangiopathy, recurrent hemophagocytosis, and endocarditis.13,14 Bartonella was initially described as a cause of endocarditis in a patient with human immunodeficiency virus (HIV) in 1993. 15 There is male preponderance and predilection for the aortic valve, although it can afflict both. 16 It is most commonly due to either Bartonella henselae or Bartonella quintana and rarely involves other species. 17 Alcoholism and being displaced (homeless) are associated with Bartonella quintana, while exposure to cats and pre-existing valvular heart disease are associated with Bartonella henselae infection. 18 There is a broad spectrum of clinical presentations, including acute kidney injury leading to renal failure, systemic embolic with cerebrovascular events, heart failure, and multi-organ failure. Most cases require valvular surgery due to the severity of damage at index diagnosis. 19 Documented Bartonella culture–positive endocarditis should be treated with oral doxycycline at 100 mg for 6 weeks and intravenous gentamicin at 3 mg/kg daily for 14 days. 20
The microorganism causing “Q fever” is Coxiella burnetii, a gram-negative pleomorphic intracellular coccobacillus, phylogenetically related to Legionella. It can survive in the low pH of the host cell, which is also essential for its survival. 21 Animals such as dogs, cats, cattle, sheep, and goats are reservoirs for this pathogen, and humans can become infected by inhaling the aerosolized organism. Clinically, it typically presents as a benign self-limiting “flu-like” illness, atypical pneumonia; however, in more disseminated disease, it can result in endocarditis, embolic phenomenon, worsening renal or hepatic dysfunction, and hematologic abnormalities. Vegetations are usually small and nodular and often overlooked in imaging. Furthermore, this organism is difficult to culture; thus, the diagnosis is primarily clinched with serological testing. 22 The optimal treatment for acute “Q fever” is doxycycline 100 mg orally twice daily for 14 days. 23 In cases of refractory disease or intolerance to doxycycline, minocycline 100 mg twice daily, trimethoprim-sulfamethoxazole 160/800 mg twice daily, or clarithromycin 500 mg twice daily are pharmacotherapeutic alternatives. 24
This case report is informative as there are several interesting facets of its presentation and management. As aforementioned, it is exceedingly rare to have CNE related to either Bartonella or Coxiella, and co-infection has not been previously reported to our knowledge. Although in an immunocompromised patient, a fastidious organism may be the culprit, and it would be highly unexpected to encounter 2 being implicated in CNE as most clinicians may discontinue the diagnostic work-up after discovering an initial organism, obfuscating the presence of another pathogen. This alludes to the critical point of completing comprehensive and exhaustive testing in high-risk patients. Cases of co-infection have been described in blood culture-positive IE due to typical causative organisms (staphylococcus, streptococcus, or enterococcus) and other pathogens such as Coxiella, COVID-19, or fungi.25 -27 While a blood culture may be time-consuming and yield false-negative results, it remains the gold standard for diagnosis. Culture-negative endocarditis, which accounts for up to 20% of IE cases, poses a clinical challenge with increasing mortality. Serology tests work best in limited pathogens, such as Coxiella burnetii and Bartonella infections. 28 This patient had an intrinsically accentuated risk of IE due to his BAV, and this “perfect storm” scenario may have precipitated his CNE-related sepsis and concomitant heart failure. It is also integral to consider his outpatient antibiotic stewardship, renal transplant, and immunosuppressive regimen as significant drug-drug interactions (hydroxychloroquine and tacrolimus) can lead to serious adverse events.29,30 Bicuspid aortic valve is the most common congenital cardiac malformation, affecting 1% to 2% of the population. The incidence of IE in the BAV population ranges from 10% to 30%, and conversely, 25% of IE cases occur in BAV.31 -33 The main indications for early surgery in patients with IE are primarily heart failure, severe, refractory infection, and prevention of embolism. It is critical to have a multidisciplinary collaboration among cardiologists, cardiac surgeons, and infectious disease specialists concerning the indication and timing of surgical intervention, which should be predicated on individual risk-benefit analysis.34,35
Elevated Bartonella species antibodies in patients with chronic “Q fever” due to serological cross-reactivity have been well documented. 36 La Scola and Raoult 37 used cross-adsorption studies and protein immunoblotting to confirm the cross-reactivity. Graham et al highlighted a case of chronic “Q fever” endocarditis with extensive serological cross-reactivity, which included cross-reactive antibodies to Bartonella species. They also highlighted the difficulty of making a definitive diagnosis of IE in the setting of positive serology but negative cultures, given the cross-reactivity noted. This challenge is further highlighted by the difficulty isolating these organisms on culture and handling Coxiella burnetti specimens, which are potentially dangerous to laboratory personnel and require specific biosafety measures. 36 Polymerase chain reaction on blood or valvular tissue, where available, can be very useful in these patients to assist in definitive diagnosis. Culture-negative endocarditis patients are often investigated with several serological tests as well as blood PCR or valvular tissue PCR. Co-infections, though rare, can be seen. 38 There have been reported cases of “Q fever” and culture-positive IE, as well as acute “Q fever” and scrub typhus. 19 The possibility of “Q fever” and Bartonella co-infection, or perhaps consecutive infection, was considered in this immunocompromised host with positive serological tests. He had serologically significant Bartonella henselae antibodies and serology in keeping with “Q fever” endocarditis. Bartonella henselae immunoglobulin G (IgG) antibodies in our patient were greater than those seen in the study by Scola et al. Our patient also had positive Bartonella henselae immunoglobulin M (IgM) antibodies. Neither blood PCR nor valvular tissue PCR was available in our setting. Given that our patient was an immunocompromised host with significantly elevated “Q fever” and Bartonella henselae antibodies, in a setting where blood and tissue PCR was not available, we treated him for both “Q fever” endocarditis and Bartonella endocarditis, and there was significant clinical improvement in his condition.
Conclusion
We present the case of a 23-year-old man with a previous deceased-donor renal transplant maintained on tacrolimus and prednisone who developed CNE of the mitral and aortic valves. He was suspected of being co-infected with Bartonella henselae and Coxiella burnetii, confirmed with serology testing. He was successfully managed with appropriate antibiotics and dual valve replacement. Clinicians should have a high index of clinical suspicion of co-infection with rare, atypical organisms in immunocompromised patients presenting with CNE.
Footnotes
Author Contributions
All authors contributed equally to writing the article, and all authors read and approved the final article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed Consent
The patient has provided verbal and written informed consent to have the details and images of his case published; institutional approval was not required for publication.
Data Availability Statement
All available data can be obtained by contacting the corresponding author.
