Abstract
Alveolar adenoma (AA) is a rare type of pulmonary neoplasm showing proliferation of alveolar epithelium and septal mesenchyme. Clinically, AA is asymptomatic, benign, and usually diagnosed incidentally. Surgical excision is curative. The etiology of AA is controversial and the pathogenesis is unknown, but it is thought to stem from type II pneumocytes and fibroblasts or fibroblast-like cells. The stromal cells in AA may show rare staining for CD34. Diffuse CD34 positivity of the interstitial stromal cells has rarely been described and reviewed. Here, we report a case of AA with strong and diffuse CD34 staining in the stromal cells.
Introduction
Alveolar adenoma (AA) is an extremely rare pulmonary neoplasm consisting of cystic spaces lined by a single layer of flat to cuboidal alveolar epithelium overlying spindle-rich stroma. The first 6 cases of AA were described by Yousem and Hochholzer in 1986. 1 It occurs most commonly in adults in their 40s-60s. Patients with AA are usually asymptomatic and diagnosed incidentally. Surgical resection is curative, and no recurrence or metastasis after complete resection has been reported. Although histogenesis is not well characterized, it is well accepted that AA stems from type II pneumocytes and fibroblasts or fibroblast-like cells. As of 2021, AA is a type of adenoma of the lung that has been included in the World Health Organization Classification of Tumors. The stromal cells in AA may show rare staining for CD34.2,3 Although the significance remains unclear, CD34 positivity could signify a true neoplastic nature of the stromal component rather than a secondary reaction. 3 To the best of our knowledge, only 4 cases with stromal cells diffusely positive for CD34 have been reported. In this article, we reported an additional case.
Case Report
A 46-year-old Caucasian woman developed COVID pneumonia in early 2020, during which an incidental left lung lesion was identified on chest x-ray imaging. Follow-up chest computed tomography (CT) showed a solitary, well-circumscribed nodule in the left lower lobe, which had increased in size to 3 cm in early 2022. A CT-guided biopsy of the nodule showed a spindle cell neoplasm. Positron emission tomography/CT scan and brain magnetic resonance imaging excluded regional or distant metastatic disease. The patient also underwent endobronchial ultrasound fine needle aspiration for mediastinal staging purposes, but no suspicious lymph nodes were identified. Left lower lobe lobectomy with mediastinal lymphadenectomy was performed in 2022. The postoperative clinical course was unremarkable.
Gross examination revealed a 3.0 × 2.5 × 2.3 cm tan-pink, well-circumscribed rubbery mass with a smooth and spongy cut surface. Microscopically, the mass was well demarcated (Figure 1(A)) and showed varying sized cystic spaces filled with eosinophilic acellular and granular materials (Figure 1(B)-(D)). The cystic spaces were lined by flat to cuboidal cells and interspersed by septa composed of spindle-shaped fibroblast-like cells. No atypia, mitoses, or necrosis was identified (Figure 1(D)). The cyst lining cells were diffusely positive for cytokeratin 7 (CK7; Figure 1(E)) and thyroid transcription factor-1 (TTF-1; Figure 1(F)). The stromal cells were strongly and diffusely immunoreactive for clusters of differentiation 34 (CD34; Figure 1(G)). Immunostains for S100 (Figure 1(H)), smooth muscle actin (SMA; Figure 1(I)) and Human Melanoma Black 45 (HMB45; Figure 1(J)) were negative for the stromal cells. The histological findings and immunohistochemistry studies confirmed the diagnosis of AA.

Biphasic histologic features in alveolar adenoma. Hematoxylin & eosin histology examination shows a well-circumscribed (A, 10× magnification) multi-cystic lesion with ecstatic spaces (B, 50× magnification; C, 100× magnification) lined by cytological bland flat to cuboidal cells (arrow) and filled with eosinophilic granular materials (star) (D, 400× magnification). The interstitium contains cytological bland spindle cells (arrowhead) and inflammatory cells (D, 400× magnification). Immunohistochemistry staining shows (400× magnification) (E) CK7 is diffusely/strongly positive for cystic lining cells (cytoplasmic staining) and negative for stromal cells; (F) TTF1 is diffusely/strongly positive for cystic lining cells (nuclear staining) and negative for stromal cells; CD34 is diffusely/strongly positive for stromal cells and negative for cyst lining cells (G); S100 (H), smooth muscle actin (I), and Human Melanoma Black 45 (J) are negative for both cyst lining and stromal components.
Discussion
Alveolar adenoma is an extremely rare neoplasm of the lungs that was first described by Yousem and Hochholzer in 1986. 1 In the past, it was considered a form of pulmonary lymphangioma due to unknown histogenesis. The tumor mostly occurs in adults between the 40s and 60s with a mean of 53 and slightly female predominance. Due to its indolent feature, AA is often found accidentally on a routine chest x-ray as a solitary, well-circumscribed, homogeneous, noncalcified mass. 2 It can occur in any lobe of the lungs, although more often seen in the lower left lobe 2 which was the location in our case. In our case, the lesion was discovered incidentally when the patient had a chest x-ray due to COVID infection. The lesion showed a well-circumscribed solitary nodule, which was consistent with the chest x-ray findings described previously. The patient was followed up for about 2 years until the lesion was found increased in size.
The diagnosis of AA is based on its histological features. Microscopic examination essentially reveals cystic spaces frequently resembling alveolar spaces, filled with eosinophilic or clear, acellular, granular materials. The cystic spaces are lined by a single layer of small to medium-sized cuboidal epithelial cells resembling type II pneumocytes. The septa contain mesenchymal fibroblast-like cells, which are usually spindled but can also have oval-to-round morphology.2,4,5 There are often mild inflammatory components, such as lymphocytes and plasma cells, presenting in solid areas. Other key features include the absence of nuclear atypia, mitotic activity, or necrosis, which is consistent with the findings in our case (Figure 1(A)-(D)).
The differential diagnoses for AA include lymphangioleiomyomatosis,2,6 papillary adenoma, 6 adenocarcinoma, lymphangioma, hemangioma, emphysema, sclerosing pneumocytoma, 2 and bronchiolar adenoma. 7 These entities can be distinguished by thorough histological examination and confirmed by immunohistochemical analyses. Alveolar adenoma is a well-circumscribed tumor with no cytological atypia or necrosis in the epithelial cells and mesenchymal cells. CK-PAN, CK7, CK18, CK19, EMA, and TTF-1 show strong, diffuse staining of the epithelium while being negative in the mesenchymal cells; surfactant apoprotein B and C are also positive in the epithelial cells because of the resemblance to type II pneumocytes; the mesenchymal cells are variably positive for SMA, rarely positive for CD34 and S100,2,3 and negative for HMB45 (Table 1). Lymphangioleiomyomatosis features thin-walled cystic air spaces and patch clustered to a nodular proliferation of bland spindled cells, which are positive for HMB45, SMA, estrogen receptor, and progesterone receptor. Papilloma adenoma shows papillary proliferation containing fibrovascular cores lined by a single layer of tumor cells that lack atypia, mitoses, or necrosis. Adenocarcinoma demonstrates cytological atypia and infiltrating growth pattern. Lymphangioma and hemangioma are vascular tumors, which are positive for vascular markers such as CD31, CD34, D2-40, and ERG. Emphysema does not form a mass-like lesion and shows no mesenchymal cell proliferation. Sclerosing pneumocytoma is a morphologically heterogeneous tumor with 4 growth patterns (papillary, solid, sclerosing, and hemorrhagic patterns). The tumor cells comprise 2 cell types: cuboidal surface cells and round stromal cells. In some cases, round stromal cells can show spindle morphology, mimicking AA. For sclerosing pneumocytoma, EMA and TTF1 are positive in both surface cell and round cell/spindle cell components. Bronchiolar adenoma is a well-circumscribed lesion with bilayered morphology. There are luminal cells, which may consist of ciliated cells and mucous cells, and subjacent basal cells.
The Implications of the Biomarkers.
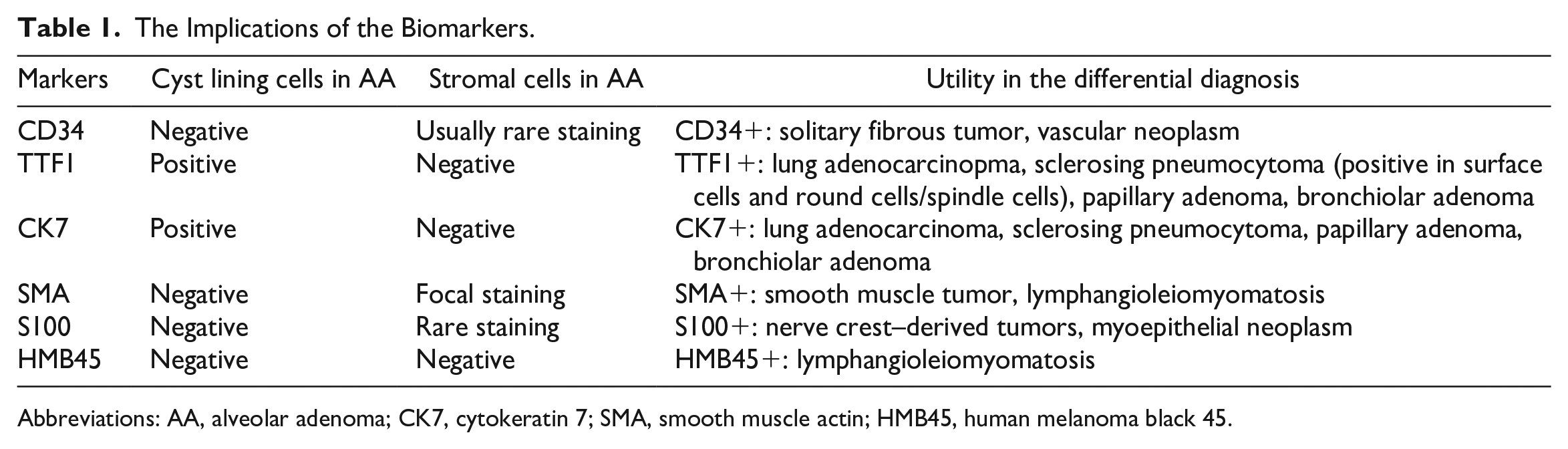
Abbreviations: AA, alveolar adenoma; CK7, cytokeratin 7; SMA, smooth muscle actin; HMB45, human melanoma black 45.
Four cases of AA with diffuse CD34 positivity in the stromal cells have been reported (Table 2). Alveolar adenoma patients with diffuse CD34 positivity, including our case, are relatively younger (39 vs 53 years old). There has been no recurrence after 7 to 132 months post-surgery follow-up. The immunohistochemical CD34 positivity of the interstitial spindle cells has been proposed as evidence of the primitive mesenchymal cell of the alveolar septum adjacent to the capillary differentiated into specific lineages,3,8 which is against stromal reaction secondary to the epithelial proliferation.9,10 Diffuse CD34 positivity in the stromal cells, although rare, further supports the neoplastic origin of AA. Alveolar adenoma with CD34-positive stromal cells should be differentiated from solitary fibrous tumor on a biopsy specimen. The presence of epithelial cells and the lack of staghorn-like vasculature and STAT6 immunoreactivity preclude the diagnosis of solidary fibrous tumor on a biopsy. In addition, there are limited data on molecular alterations in AA. Only one case reported shows der (16)t(10;16)(q23; q24) in fluorescence in situ hybridization study. 9
Clinical Features of Reported Cases of CD34-Positive AA.
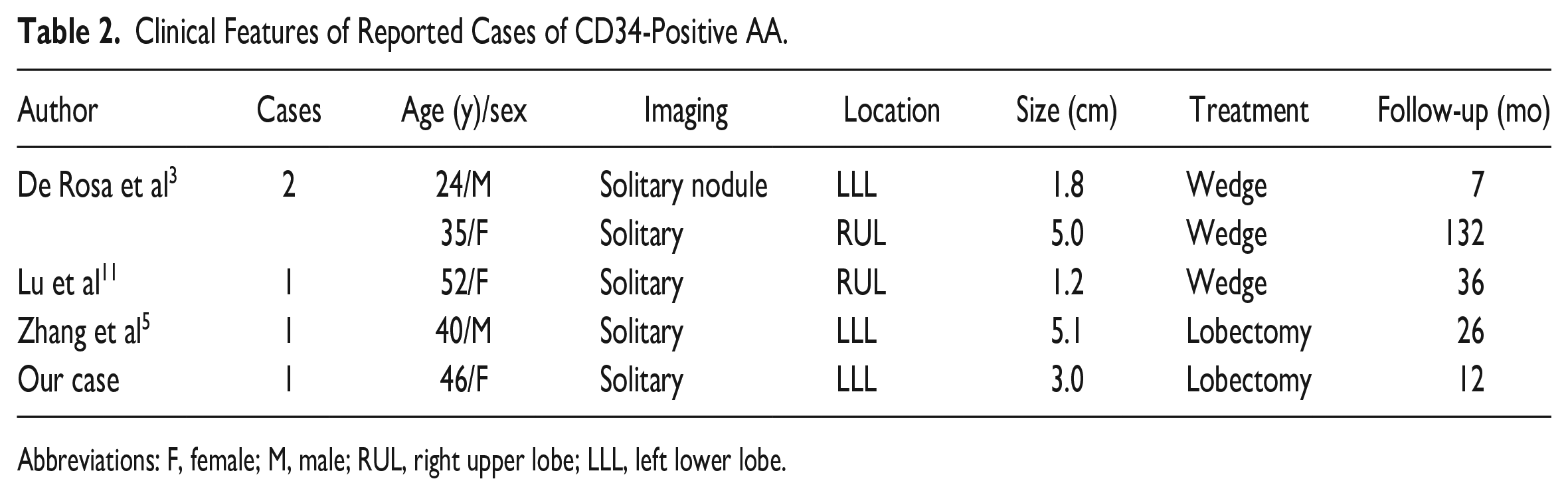
Abbreviations: F, female; M, male; RUL, right upper lobe; LLL, left lower lobe.
In conclusion, AA should be considered when dealing with a solitary pulmonary nodule. Recognition of its characteristic morphology and immunohistochemical pattern is the key to distinguishing it from other differential diagnoses. Especially, AA with CD34 diffusely positive stromal cells should be differentiated from solitary fibrous tumor on a biopsy specimen. CD34 positivity may help decipher its histogenesis. More molecular studies and case reports are needed to further characterize this rare neoplasm.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Informed consent for patient information to be published in this article was not obtained because of case report.
