Abstract
Increasing evidence points to a role for circulating endothelial progenitor cells, including populations of CD34-positive (CD34+) cells, in maintenance of cerebral blood flow. In this study, we investigated the link between the level of circulating CD34+ cells and neovascularization at ischemic brain. Compared with control subjects, a remarkable increase of circulating CD34+ cells was observed in patients with angiographic moyamoya vessels, although no significant change was observed in patients with major cerebral artery occlusion (or severe stenosis) but without moyamoya vessels. Our results suggest that the increased level of CD34+ cells associated with ischemic stress is correlated with neovascularization at human ischemic brain.
Introduction
Increasing evidence points to a role for bone marrow-derived immature cells, such as endothelial progenitor cells, in maintenance of vascular homeostasis and repair. CD34-positive (CD34+) cells comprise a population enriched for endothelial progenitor cells whose contribution to neovasculature includes both direct participation in forming the neovessel and regulatory roles as sources of growth/angiogenesis factors (Majka et al, 2001). Previously, we have shown accelerated neovascularization after administration of CD34+ cells in an experimental model of stroke (Taguchi et al, 2004b) and induced by autologous bone marrow mononuclear cells (rich cell fraction of CD34+ cells) transplanted locally into patients with limb ischemia (Taguchi et al, 2003). In addition, we have observed a positive correlation between the level of circulating CD34+ cells and regional blood flow (Taguchi et al, 2004a), and cognitive function (Taguchi et al, 2007) in patients with chronic cerebral ischemia.
In this study, we have evaluated the level of circulating CD34+ cells in patients with unusually accelerated neovascularization induced by progressive occlusion (or severe stenosis) of the supraclinoid portion of the internal carotid artery, the proximal region of the anterior, and/or middle cerebral artery characterized angiographically by the presence of moyamoya-like vessels (Natori et al, 1997) that supply ischemic brain as collaterals. We have investigated the hypothesis that circulating bone marrow-derived immature cells might be associated with neovascularization at ischemic sites in the human brain.
Patients and methods
The institutional review board of the National Cardiovascular Center approved this study. All subjects provided informed consent. A total of 50 individuals, including 24 patients with occlusion or severe stenosis (>90%) at the C1 portion of the internal carotid artery or the M1 portion of the middle cerebral artery, and 26 age-matched healthy volunteers with cardiovascular risk factors, but without history of vascular disease, were enrolled. The diagnosis of cerebral artery occlusion or stenosis was made angiographically and four patients were found to have classical angiographic evidence of moyamoya-like vessels, including one with right C1 occlusion, one with right M1 occlusion, and two with bilateral C1 severe stenosis. All patients with cerebral artery occlusion or stenosis had a history of cerebral infarction. Individuals excluded from the study included patients who experienced a vascular event within 30 days of measurements, premenopausal women, and those with evidence of infection and/or malignant disease. The number of circulating CD34+ cells was quantified as described (Taguchi et al, 2007). In brief, blood samples (200 μl) were incubated with phycoerythrin-labeled anti-CD34 antibody, fluorescein isothiocyanate-labeled anti-CD45 antibody, 7-aminoactinomycin-D (7-AAD), and internal control (all of these reagents are in the Stem-Kit; BeckmanCoulter, Marseille, France). After incubation, samples were centrifuged, and supernatant was removed to obtain concentrated cell suspensions. 7-Aminoactinomycin-D-positive dead cells and CD45-negative cells were excluded, and the number of cells forming clusters characteristic of CD34+ cells (i.e., low side scatter and low-to-intermediate CD45 staining) was counted. The absolute number of CD34+ cells was calculated using the internal control. Mean cell number of duplicate measurements was used for quantitative analysis. Statistical comparisons among groups were determined using analysis of variance or χ2 test. Individual comparisons were performed using a two-tailed unpaired Students’
Results
Enrolled individuals were divided into three groups: control subjects, patients with cerebral occlusion or severe stenosis, but without the presence of vessels with angiographic characteristics of moyamoya disease, and patients with angiographic evidence of moyamoya-like vessels. Baseline characteristics of the groups are shown in Table 1. The modified Rankin scale evaluation of patients with and without moyamoya-like vessels was 0.5±0.5 and 1.3±0.2, respectively (
Baseline characteristics
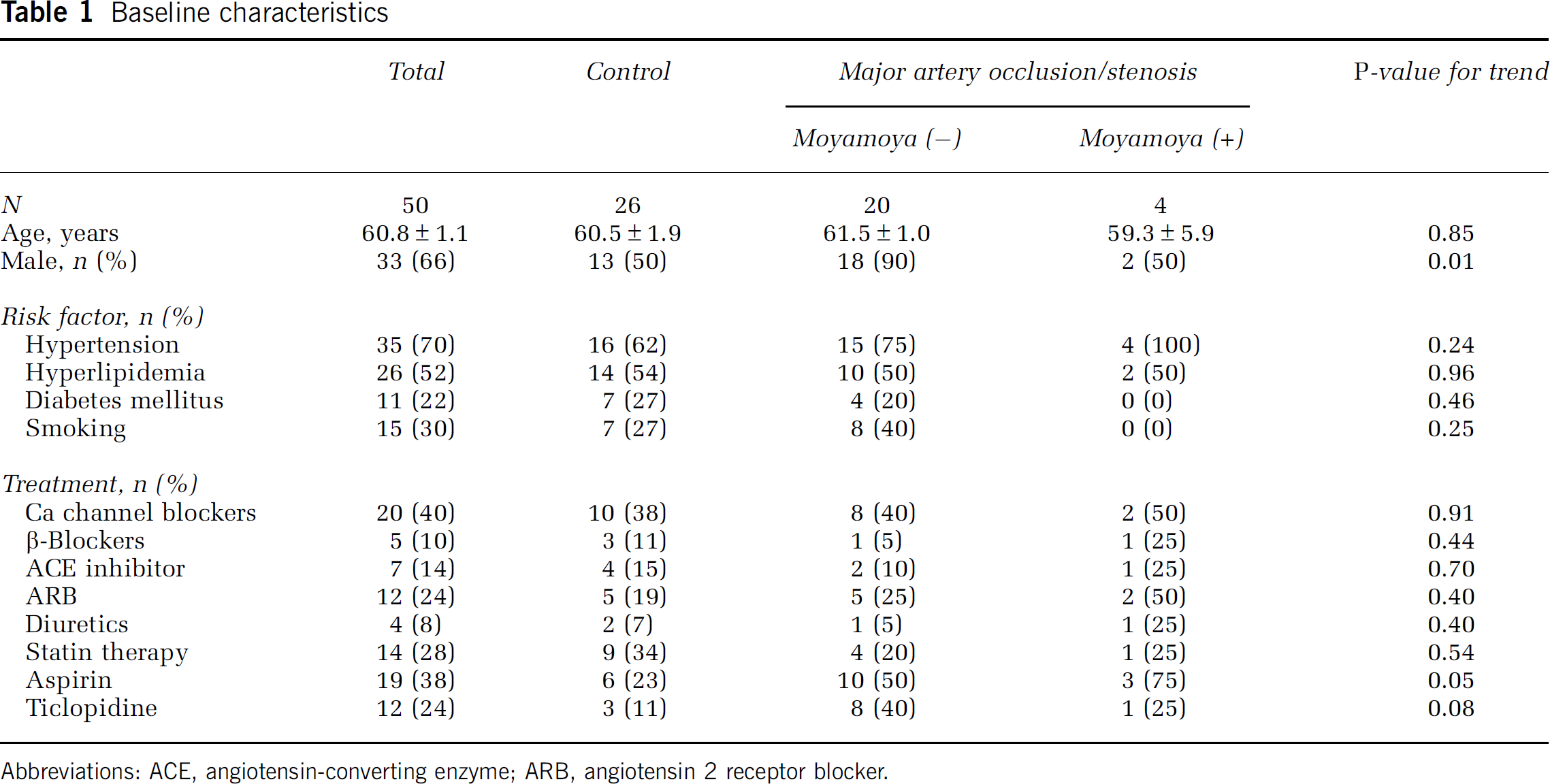
Abbreviations: ACE, angiotensin-converting enzyme; ARB, angiotensin 2 receptor blocker.
A representative angiogram showing characteristics of moyamoya-like vessels is shown in Figures 1A and 1B. Angiographic moyamoya-like vessels were observed around the M1 portion of an occluded middle cerebral artery. Compared with a normal subject (Figure 1C) and patients without angiographic evidence of moyamoya-like vessels (Figure 1D), a remarkable increase in levels of peripheral CD34+ cells was observed in patients with moyamoya-like vessels (Figure 1E) based on fluorescence-activated cell sorting. To confirm this impression, levels of circulating CD34+ cells were quantified (control, CD34+ cells=0.89±0.07/μL; moyamoya (−), CD34+ cells=0.98±0.13/μL; moyamoya (+), CD34+ cells=2.28±0.53/μL) and found to be significantly increased in patients with moyamoya-like vessels more than two-fold higher than in controls (Figure 1F,
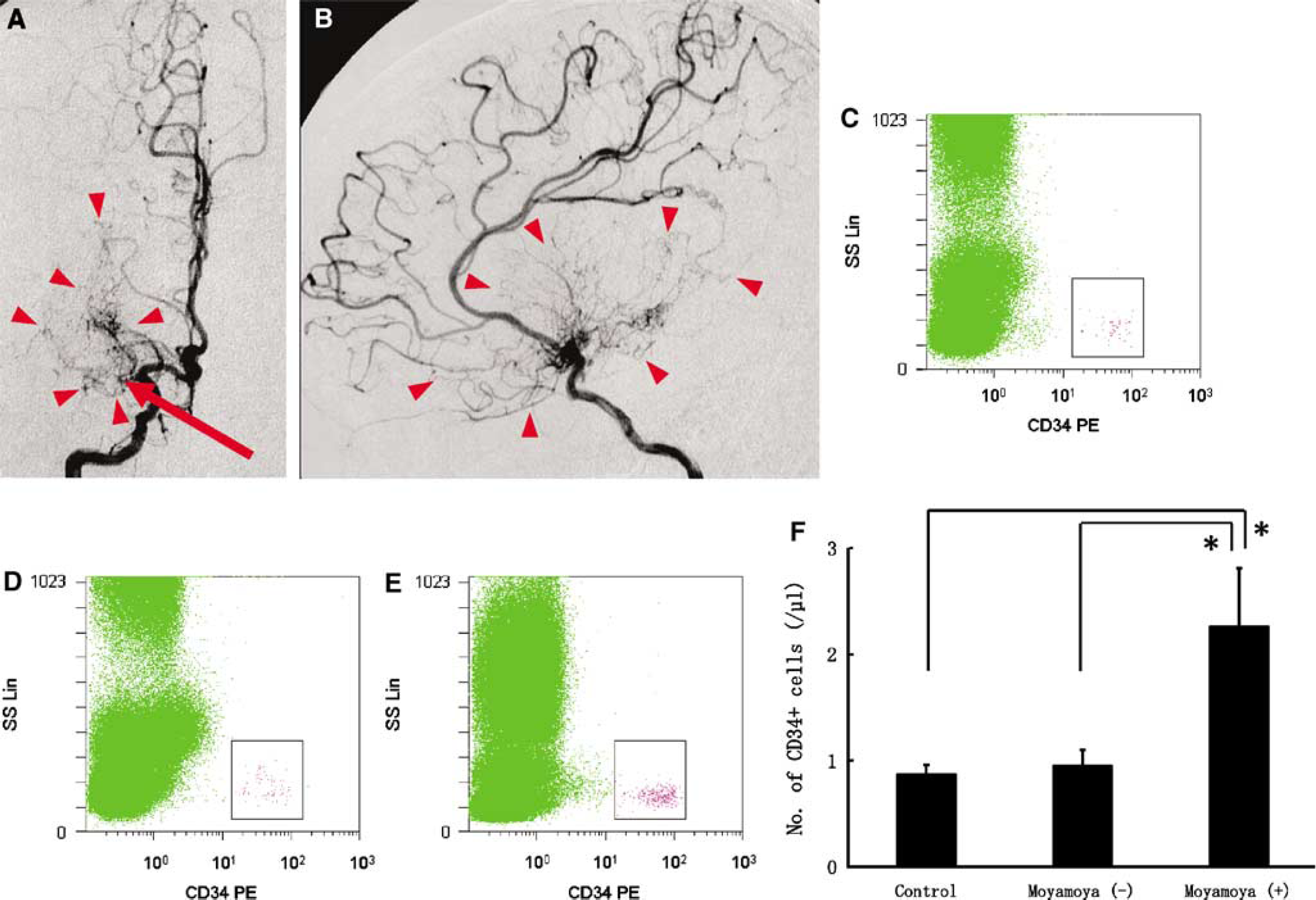
Increased levels of circulating CD34+ cells in patients with angiographic evidence of moyamoya-like vessels. (
Discussion
In this study, we have found that a feature of unusually accelerated neovascularization, evidence of moyamoya-like vessels in the immediate locale of an occluded major cerebral artery, can be correlated with a robust increase in the level of circulating CD34+ cells. The latter was determined using a newly developed method that enables quantification of few CD34+ cells in peripheral blood in a highly reproducible manner.
After acute cerebral ischemia, mobilization of CD34+ cells from bone marrow has been shown in stroke patients (Taguchi et al, 2004a). Furthermore, transplantation of CD34+ cells (Taguchi et al, 2004b) and bone marrow cells (Borlongan et al, 2004a, 2004b) has been shown to restore cerebral blood flow in experimental models of stroke. In chronic ischemia, transplantation of CD34+ cells has also been shown to accelerate neovascularization, including formation of collateral vessels, in patients with chronic ischemic heart disease (Boyle et al, 2006) and limb ischemia (Kudo et al, 2003). In addition, there is a report regarding the correlation between inadequate coronary collateral development and reduced numbers of circulating endothelial progenitor cells in patients with myocardial ischemia (Lambiase et al, 2004). In this study, we show, for the first time, a correlation between neovascularization of the cerebral arterial circulation and increased levels of circulating CD34+ cells. Our results support the hypothesis that circulating CD34+ cells potentially contribute to neovascularization at sites of ischemic brain injury.
Footnotes
Acknowledgements
We thank K Obata and Y Okinaka for technical assistance.
The authors state no conflict of interest.
