Abstract
Immune-mediated necrotizing myopathy (IMNM) is a subtype of inflammatory myopathy that is characterized by proximal muscle weakness, markedly elevated serum creatine kinase, myopathic electromyographic findings, and muscle biopsies revealing necrosis or regeneration with sparse inflammatory infiltrate. IMNM tends to be idiopathic but has been associated with certain medications. This supports the possibility for other pharmacotherapies to induce IMNM—particularly leflunomide. Leflunomide is used in the treatment for rheumatoid arthritis and has been shown to induce autoimmune diseases—including autoimmune hepatitis and polymyositis. After an extensive review of history and workup of muscle weakness, we conclude that leflunomide induced an IMNM in our patient. As this is the first case of leflunomide-induced IMNM, it is important for clinicians to suspect an inflammatory myopathy in the setting of myositis while on leflunomide.
Keywords
Introduction
Inflammatory myopathies are a heterogeneous group of autoimmune diseases characterized by muscle inflammation and can vary in their associated muscle weakness and extra-muscular manifestations.1-3 A particular subtype includes immune-mediated necrotizing myopathy (IMNM), which is characterized by proximal muscle weakness, markedly elevated serum creatine kinase (CK), myopathic electromyographic (EMG) findings, and muscle biopsies revealing necrosis or regeneration with sparse inflammatory infiltrate.4,5 In fact, approximately two-thirds of IMNM patients will have autoantibodies discerning either signal recognition particle (SRP) or 3-hydroxy-3-methylglutaryl coenzyme-A reductase (HMGCR). 6 IMNM tends to be idiopathic; however, it has demonstrated an association with certain medications. 7 This supports the possibility for other pharmacotherapies to induce IMNM—particularly leflunomide.
Leflunomide is a conventional disease-modifying anti-rheumatic drug typically used for chronic management of rheumatoid arthritis (RA). 8 Although it is well tolerated by many patients, some of the more commonly reported side effects entail diarrhea, nausea, hypertension, transient global hair loss, hepatotoxicity, and peripheral neuropathy. 9 Rarer, more serious adverse effects include pancytopenia, opportunistic infections, Steven-Johnson syndrome, toxic epidermal necrolysis, angioedema, and interstitial lung disease. 10 Leflunomide has also been shown to illicit an autoimmune hepatitis 8 and a polymyositis. 11 Nonetheless, there is minimal literature about the myopathic effects and autoimmunity associated with leflunomide. This case report aims to describe a rare case of leflunomide-induced IMNM in a patient treated for RA.
Case Report
A 66-year-old female with a medical history of rheumatoid arthritis, hypothyroidism, and hypertension presented to the emergency department with the complaint of bilateral lower extremity weakness. Five days prior to presenting to our hospital, the patient experienced an unwitnessed ground-level fall (GLF) devoid of head trauma, loss-of-consciousness, urinary incontinence, and tongue biting. She was found on the ground several hours later fully alert and attributed the incident to her lower extremity weakness. Shortly after being discovered on the ground, she was taken to a freestanding emergency department with a same day discharge after receiving intravenous ketorolac and acetaminophen. There she also received lab work that was reported to be significant for an elevated serum creatinine (1.7 mg/dL), and an elevated serum creatinine kinase (20 000 U/L). After discharge, her symptoms did not improve and significantly worsened—prompting the visit to our hospital.
On admission to our hospital, the patient reported bilateral proximal lower extremity pain with associated weakness, gait inability, and decreased range of motion of the bilateral lower extremities with an onset of a few months prior to her GLF. She denied any other ground-level falls since the initial episode as she became bedbound secondary to the rapidly progressive lower extremity weakness. She also denied any fever, chills, headache, blurred vision, lymphadenopathy, chest pain, palpitations, shortness-of-breath, cough, abdominal pain, nausea, vomiting, diarrhea, hematochezia, melena, hematuria, dysuria, lower extremity swelling, numbness, tingling, joint swelling, and joint pain. Surgical history was significant only for cesarean section and bilateral tubal ligation. Family history was noncontributory, and she denied any social history including the use of alcohol, tobacco, and illicit drugs. Her home medications included leflunomide, levothyroxine, and valsartan-hydrochlorothiazide. On further inquiry, the patient mentioned that she began taking leflunomide 6 months prior to this hospitalization for the management of her RA. Her admitting vitals showed a temperature of 36.6 °C, blood pressure of 168/72 mm Hg, pulse of 81 beats per minute, respiratory rate of 18 breaths per minute, and an oxygen saturation of 92% on room air. A neurological examination revealed a decreased range of motion in the bilateral lower extremities, decreased strength of 2/5 in the proximal muscle groups of the bilateral lower extremities, and normal muscle strength of 5/5 in the distal muscle groups. Sensory exam and reflexes were normal bilaterally in both upper and lower extremities. Her heart sounds were normal with a regular rate and normal rhythm with no gallops or rubs. Her lung sounds were clear to auscultation bilaterally with no increase in respiratory effort.
Laboratory studies revealed a leukocytosis (26.41 × 103/UL) with neutrophil predominance (23.67 × 103/UL), hyponatremia (125 mmol/L), hyperkalemia (6.7 mmol/L), albumin-corrected high anion gap metabolic acidosis (23.8 mEq/L), elevated serum creatine (9 mg/dL), hypocalcemia (corrected to 7.6 mg/dL), hyperphosphatemia (13.9 mg/dL), elevated transaminases (aspartate transaminase 804 IU/L, alanine transaminase 590 IU/L), elevated erythrocyte sedimentation rate (126 mm/h), elevated C-reactive protein (>18 mg/dL), elevated serum creatine kinase (74 988 U/L), and an elevated serum myoglobin (20 000 ng/mL). Lactic acid and aldolase were within normal limits. A thyroid panel was within normal limits. A urinalysis with urine microscopy was consistent with rhabdomyolysis. Workup for infectious etiology was negative for human immunodeficiency virus (HIV), Borrelia burgdorferi, human T-cell lymphotropic virus (HTLV) type I and II, Group A and B coxsackievirus, hepatitis panel, two sets of blood cultures, and a urine culture. In addition, a respiratory panel was negative for etiologies such as influenza and coronavirus. Autoimmune workup was negative for anti-nuclear antibodies (ANA), anti-3-hydroxy-3-methylglutaryl-coenzyme A reductase (Anti-HMGCR), and Anti-Jo-1. However, a line blot Myositis Specific 11 Autoantibody Panel revealed positivity for anti-Signal Recognition Particle (anti-SRP) at 31 SI, anti-alanyl-tRNA synthetase (anti-PL12) at 16 SI, and anti-nucleosome remodeling deacetylase beta (anti-Mi-2β) antibody at 13 SI.
Computed tomography (CT) of the head was negative for any acute pathologies, and a CT angiogram of the head and neck did not reveal intracranial arterial occlusions or aneurysms. Magnetic resonance imaging (MRI) of the lumbar spine showed neural foraminal narrowing at L4-L5 level with some impingement on the traversing nerve root; however, this did not correlate with the patient’s clinical examination and symptomatology. EMG showed abnormal spontaneous activity including complex repetitive discharges and positive sharp waves with fibrillations and small polyphasic myopathic units in the bilateral vastus lateralis and the left biceps muscle. Muscle biopsy of the right vastus lateralis muscle was obtained and showed scattered necrotic fibers, myophagocytosis, regenerative myofibers, and a paucity of inflammatory cells (Figure 1). The patient’s rhabdomyolysis, electrolyte imbalance, and acute renal failure were managed medically with aggressive hydration and diuresis; however, her symptoms did not begin to improve until administration of intravenous methylprednisolone 500 mg twice daily for 3 days followed by the transition to 60 mg prednisone orally daily. Six months status-post discharge, the prednisone dose was gradually tapered by alternating 2.5 mg and 5 mg every other day with a concurrent use of daily methotrexate sodium 2.5 mg (Figure 2). At that time, she denied any proximal muscle weakness and reported effective ambulation with assistance from a front-wheeled walker.
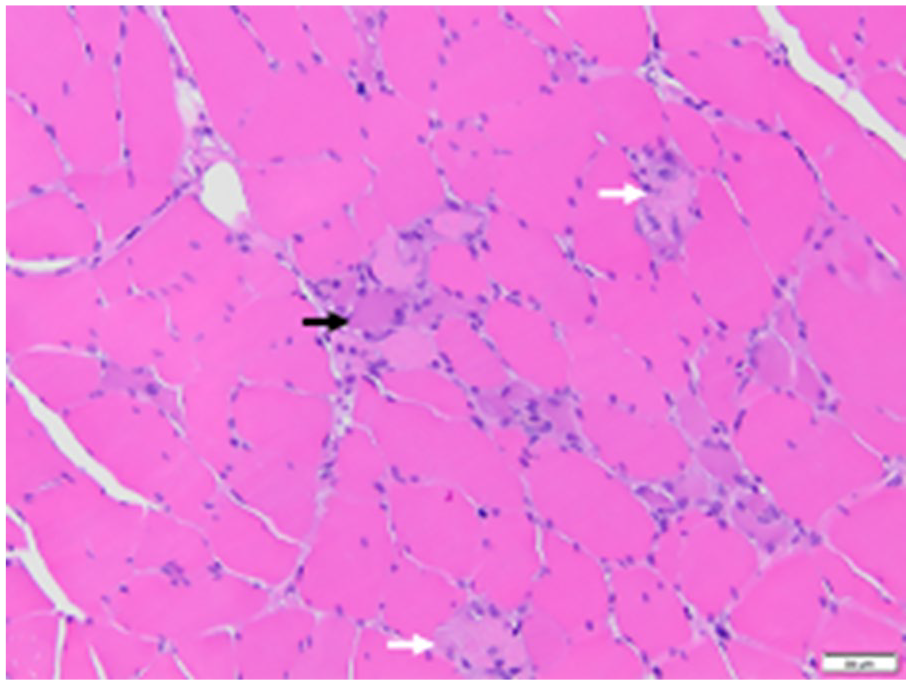
Histologic section of the skeletal muscle biopsy from the right vastus lateralis muscle at 100x power. Findings include scattered necrotic fibers and myophagocytosis (white arrows), regenerative myofibers (black arrow), and a paucity of inflammatory cells. It is important to note that the necrotic myofibrils are in different stages of myonecrosis, myophagocytosis, and regeneration.

Creatine kinase (U/L) levels over time. It is important to note that the patient’s creatine kinase levels eventually normalized, indicating successful remission of the immune-mediated necrotizing myopathy. Of note, the patient was hospitalized and treated with 500 mg methylprednisolone intravenous piggyback every 12 hours and discharged with 60 mg prednisone orally daily with gradual taper. Methotrexate was initiated at the 4-month follow-up at 2.5 mg (4 tablets per week) that was subsequently increased to 6 tablets per week.
Discussion
Inflammatory myopathies can be distinguished from one another with the use of myositis-specific autoantibodies (MSAs). 12 Particularly, anti-SRP is an MSA specific for IMNM; however, anti-PL12 is specific for antisynthetase syndrome (ASyS), 13 and anti-mi-2β is specific for dermatomyositis. 3 Interestingly, our patient tested positive for all three of these antibodies using a line blot Myositis Specific 11 Autoantibody Panel. This is an exceedingly rare circumstance as MSAs are generally considered to be mutually exclusive. In fact, the prevalence of detecting multiple MSAs on the gold standard radiolabeled immunoprecipitation (IP) is less than 0.2%. As IP modalities are restricted to select centers, the advent of easy-to-use commercially available assays such as line and dot immunoassays have allowed an increase in prevalence of multiple MSAs being detected. Depending on the type of commercial assay, the prevalence of multiple MSAs can range from 3.4% to 8.4%. 14 Interpretation of multiple MSAs can be challenging for the clinician as this finding may likely be attributed to the specificity discordance between various types of assays. Unfortunately, there is no specific recommendation on which combination of assays to employ. In our case, we deduced that anti-SRP was the implicated antibody among the multiple MSAs detected in light of its relatively higher titer and closer correlation to the clinical and histopathological findings. Nevertheless, despite the intuitiveness of considering the highest titer of MSA to be the true positive, we acknowledge the fact that this is not always the case.14,15 After extensive discussion with the patient regarding the risks and benefits of obtaining a repeat Myositis Specific 11 Autoantibody Panel for reassessment, she did decline due to financial circumstances.
It is reasonable to suspect an underlying causal relationship between our patient’s autoimmune conditions—RA and hypothyroidism—and the development of IMNM; though, this is unlikely the case. There is minimal literature regarding any causal relationship between preexisting autoimmune diseases and IMNM; however, we cannot rule out a potential association between the diseases. To our knowledge, there is only one reported case of hypothyroidism in the setting of IMNM, 16 and no reported cases regarding an association between RA and IMNM. Regarding other autoimmune conditions in the setting of IMNM, we learned of one reported case of IMNM in the setting of systemic lupus erythematosus (SLE). Jackson et al 17 supports this association with the mention of nearly 21% IMNM patients have SLE, implicating a possible autoimmune association. We also learned of a case report entailing IMNM in the setting of multiple sclerosis, which further underscores the possibility of an autoimmune association with IMNM. 18 To reiterate, a causal relationship between RA or hypothyroidism and IMNM cannot be demonstrated, but there may be some association between the diseases. With autoimmune conditions, there is a suggested immunologic dysfunction secondary to genetic susceptibility and environmental factors. 18 In our case, the likely triggering environmental factor is the initiation of leflunomide.
To confirm the diagnosis of IMNM secondary to leflunomide, it was first important to differentiate IMNM from other myopathies that cause muscle necrosis. Some non-immune necrotizing myopathies (NIMNM) include toxins, medications, infections, and metabolic derangements. The distinguishing factor between IMNM and NIMNM is the fact that removal of NIMNM etiologies result in clinical improvement without the need for immunotherapy. 19 In this case, our patient required immunotherapy for the improvement of her symptoms, thereby enabling us to rule out NIMNM etiologies. In addition, after receiving the muscle biopsy and MSA titer results, it was evident that this was a case of IMNM. Clinical features of IMNM entail a subacute, moderate-to-severe, symmetrical proximal muscle weakness associated with a markedly elevated serum CK—all of which our patient experienced.4,19,20 Muscle biopsy results for IMNM tend to display scattered necrotic myofibrils in different stages of myonecrosis, myophagocytosis, and regeneration with an associated paucity of lymphocytic infiltrate—consistent with our patient’s biopsy results.19,20 Of note, no special stains were employed for our patient’s biopsy results, such as immunohistochemical staining, due to inadequate laboratory resources. Finally, the presence of a MSA specific for IMNM—either anti-SRP or anti-HMGCR—helps solidify the diagnosis. It is worth mentioning that our patient did experience frank rhabdomyolysis concurrent with IMNM likely secondary to her GLF; however, her CK level did remain elevated upon discharge from her hospital stay in spite of aggressive medical management (Figure 2). Unfortunately, there is no optimal treatment regimen for IMNM; however, it is recommended to initially begin high-dose corticosteroids with the addition of methotrexate within 1 month of diagnosis. 19 Nearly half of all patients treated for IMNM with anti-SRP positivity will recover in terms of muscle strength; however, relapses tend to occur with treatment tapering. 5 In fact, various demographics that our patient exhibited is associated with serum CK level improvement—such as steroid treatment, female sex, and older age. 21
Conclusion
This report highlights the first case of leflunomide-induced IMNM. Leflunomide is a drug that acts by inhibiting the mitochondrial enzyme dihydroorotate dehydrogenase and is increasingly being used to treat RA. Although its adverse effect profile has yet to be fully elucidated, it has been shown to induce an autoimmune hepatitis 8 and a polymyositis. 11 Because of these rare adverse effects, it is crucial to remain cognizant of leflunomide’s potential to induce an autoimmune myopathy. This awareness should prompt clinicians to suspect a possible inflammatory myopathy in the setting of myositis while on leflunomide.
Footnotes
Authors’ Note
This case was presented as an abstract at the Southern Regional Meeting, New Orleans, Louisiana, on February 11, 2022.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Verbal informed consent was obtained from the patient for their anonymized information to be published in this article.
