Abstract
Statin-induced immune-mediated necrotizing myopathy (IMNM) is a rare subtype of idiopathic inflammatory myopathy associated with statin exposure and characterized by positive anti-hydroxymethylglutaryl coenzyme A reductase antibodies. Here, we describe a case of a 66-year-old male who was admitted with the presumed diagnosis of rhabdomyolysis in the setting of chronic statin use but was later confirmed to have statin-induced IMNM after his symptoms were refractory to treatment. This case highlights the importance of having a high clinical suspicion for statin-induced IMNM when statin-associated muscle symptoms fail to resolve after the discontinuation of statins.
Background
Statins are one of the most frequently prescribed drugs worldwide and are the preferred lipid-lowering agents due to their proven efficacy in reducing risk for atherosclerotic cardiovascular disease (ASCVD). They function by inhibiting an enzyme called hydroxymethylglutaryl coenzyme A reductase (HMGCR), a rate-limiting enzyme in the cholesterol biosynthesis pathway. Their side effect profile is relatively benign and is generally considered to be safe with a small fraction of patients experiencing muscle-related symptoms. The ubiquitous use of statins has unmasked a new and rare subset of inflammatory myopathy that is a distinct entity from statin-induced myalgias called statin-induced immune-mediated necrotizing myopathy (IMNM). This case illustrates the clinical characteristics of statin-induced IMNM and its associated diagnostic and therapeutic challenges.
Case Report
A 66-year-old male with a past medical history of hypertension, hyperlipidemia, class II obesity, prediabetes, and previous stroke complicated by mild right-sided sensory deficits presented to the Emergency Department (ED) with progressive generalized weakness over the past week.
He has been taking atorvastatin 80 mg daily for the past 2 years following his stroke. His primary care physician instructed him to discontinue statin therapy 9 days prior after he was found to have elevated transaminases on routine labs (ALT 368, AST 270) with previously normal baseline. His medication list also included aspirin 81 mg daily. On the day of presentation, the patient experienced worsening weakness and fatigue, leading to gait instability. He denied fever, chills, nausea, vomiting, myalgia, arthralgia, dysphagia, dyspnea, recent sick contact, acute vision, skin, or sensory changes.
Upon presentation, the patient was not in acute distress, afebrile, hemodynamically stable, and saturating well on room air. Physical exam was notable for reduced 4/5+ muscle strength. Physical examination was otherwise noncontributory.
Labs were significant for Cr 0.7 (0.64-1.29 mg/dL), ALT 446 (17-63 IU/L), AST 325 (15-41 IU/L), Alk phos 82 (32-91 IU/L), CPK >13 200 (49-397 IU/L), aldolase 112 (<8.1 U/L), CRP 19.08 (0-7.48 mg/L), ESR 10 (1-25 mm/hour), TSH 4.73 (0.3-6.0 µIU/mL), HbA1c 6.2%, negative urine drug screen (UDS), and negative respiratory viral panel (RVP). Urinalysis was positive for proteinuria and moderate blood with red blood cells within normal limits. In the ED, neurology was consulted due to initial concerns for a recurrent stroke. However, imaging of the head and neck were unremarkable for any acute intracranial abnormalities. The patient was admitted with the initial diagnosis of rhabdomyolysis and aggressive intravenous (IV) fluid resuscitation was initiated.
His symptoms failed to show signs of improvement and the generalized weakness became more localized to the bilateral proximal lower extremities. The CPK level persistently remained elevated (>12 000) despite cessation of statin therapy, aggressive fluid resuscitation, and adequate urine output. Additional labs revealed negative myositis panel (Table 1) but positive ANA with 1/80 speckled pattern and elevated anti-HMGCR IgG antibody of 224 (<20 CU). Magnetic resonance imaging of the lower extremities was significant for multiple areas of increased T2 signal suspicious for infectious or inflammatory myositis (Figure 1a and b) with particularly large areas in the proximal muscles. Biopsy of the left gluteus medius muscle demonstrated numerous degenerating myofibers and necrotic fibers accompanied with small multifocal patches of chronic inflammation. Masson trichrome stain also highlighted endomysial fibrosis.
Myositis Panel.
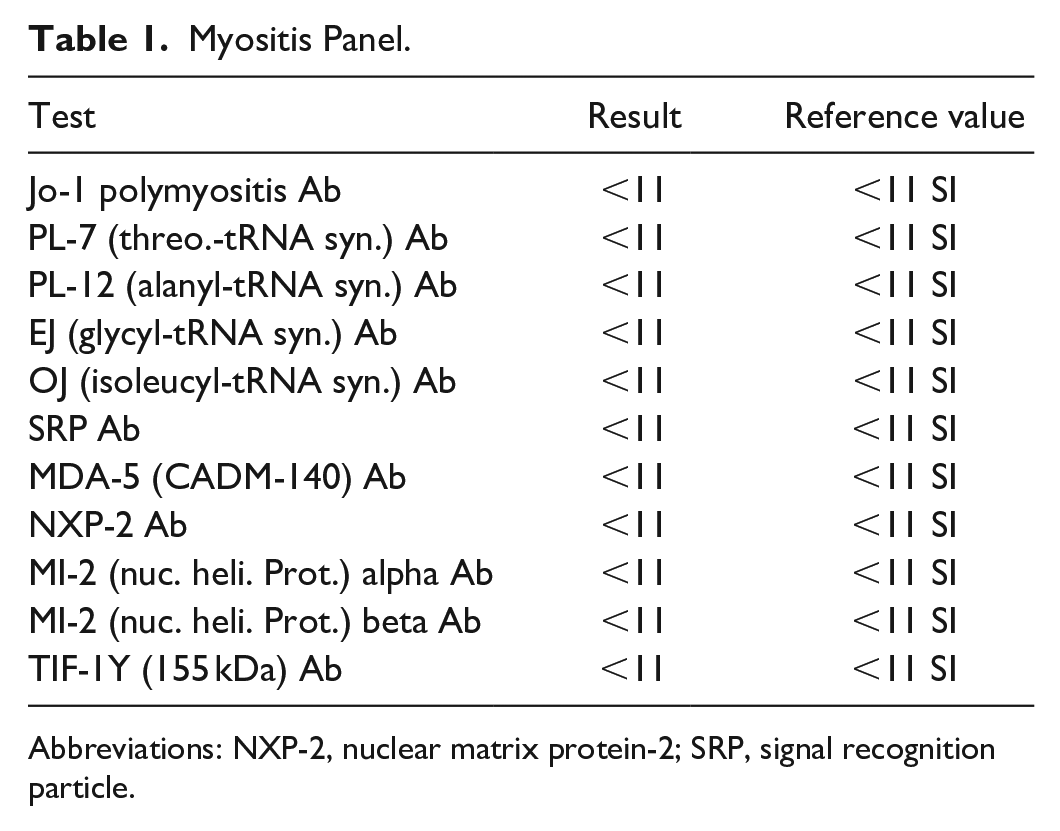
Abbreviations: NXP-2, nuclear matrix protein-2; SRP, signal recognition particle.

(a) Axial STIR (Short Tau Inversion Recovery) MRI sequence. (b) Axial T2 FS sequence. MRI, magnetic resonance imaging. (blue arrows; areas of increased signaling).
The patient was subsequently started on prednisone 30 mg daily and intravenous immunoglobulin (IVIG) 140 mg divided over 2 consecutive days every 4 weeks. Immunosuppressive therapy was deferred at this time as the patient’s hospital course was complicated by bacteremia for which IV antibiotics were initiated. The patient responded well to steroid and IVIG therapy demonstrated by downtrending CPK levels and improvement in strength. The patient was discharged on a tapering course of oral prednisone and IVIG infusions with close rheumatology follow-up.
Discussion
Statin associated musculoskeletal symptoms refers to all muscle symptoms temporally related to statin use but without regard to causality. 1 They represent a wide spectrum of clinical presentations ranging from nonspecific muscle aches to debilitating severe necrotizing myopathy. 2 Statin-induced IMNM is a relatively newer entity that is reported to occur in about 2 to 3 patients for every 100 000 individuals exposed to statins. 3 Although rare, with the increasing use of statins, it is becoming gradually more recognized as a cause of proximal muscle weakness. IMNM is a distinct subgroup of idiopathic inflammatory myopathy (IIM) that can be distinguished from other more well-known IIM such as polymyositis, dermatomyositis, and inclusion body myositis by extramuscular involvement, autoantibodies, and histological findings. 4
The pathogenesis of statin-induced IMNM is not fully understood. Studies suggest that statin exposure plays a role in upregulating HMGCR in genetically susceptible patients. 5 HMGCR expression is upregulated not only in muscle tissues exposed to statins but also in regenerating muscle cells. The immune system becomes sensitized to the increased levels of HMGCR that are upregulated in response to statins and triggers an autoimmune response resulting in a necrotizing immune-mediated injury to muscle tissue. 6 This immune response is perpetuated long after the discontinuation of statin therapy due to the persistently increased HMGCR expression in regenerating muscle fibers. 7 Among the different statins, atorvastatin is reported to be most strongly associated with the development of IMNM compared to simvastatin or rosuvastatin. 8 No clear dose-dependent effect has conclusively been determined and even low-dose statins have been found to cause IMNM. 9
Clinically, statin-induced IMNM presents with subacute or chronic progressive symmetric proximal muscle weakness. Other symptoms include myalgia and dysphagia, which are present in approximately one-third of patients. 10 Skeletal muscles in the respiratory system can also be affected and case reports have described mortality due to respiratory complications. 11 Initially, this condition may be indistinguishable from common statin-induced toxic myopathy. As forementioned, the degree of disease severity can vary widely, ranging from simple myalgia, to myositis, to rhabdomyolysis, to life-threatening IMNM. What makes matters more challenging is that patients’ symptoms don’t need to present in this continuum. The key clinical clue that may help differentiate statin-induced IMNM is the muscle weakness that persists or worsens after the cessation of statins.
The presence of anti-HMGCR antibodies is a highly specific biomarker for the diagnosis of statin-induced IMNM. 12 Studies have shown that positive anti-HMGCR antibodies has a sensitivity of 94.4% and specificity of 99.3%. 13 The disease is also associated with increased levels of CK. Although CK is a nonspecific marker for muscle injury, it can be used as a surrogate marker for disease activity. 14 In addition to positive anti-HMGCR antibodies, the diagnosis of statin-induced IMNM can be made by muscle biopsy characterized by myofiber degeneration and necrosis out of proportion to the minimal lymphocytic infiltration. 8 The inflammatory infiltrate primarily consists of macrophages with characteristic distribution throughout the endomysium.
Treatment strategies for statin-induced IMNM are not well established nor are there widely accepted treat-to-target goals. The mainstay of treatment is discontinuation of statins and immunosuppression with steroids being first line. The combination of glucocorticoid with IVIG is suggested for initial therapy, but other adjuvant agents include methotrexate, azathioprine, mycophenolate mofetil, plasma exchange, cyclophosphamide, and cyclosporine. Despite treatment, many patients experience clinical relapse with initial tapering and may end up requiring escalation of immunosuppressive therapy.
Postdiagnosis consideration includes pulmonary function testing particularly in patients with any associated respiratory symptoms for assessment of restrictive lung disease. Although statin-induced IMNM is not strongly associated with malignancy, it is recommended that patients undergo age and sex appropriate screening. Statins should not be reintroduced and is an absolute contraindication in patients with known statin-induced IMNM given the known risk of disease flare. 10 In these patients, other FDA-approved nonstatin agents such as PCSK9 inhibitors and ezetimibe should be considered to help facilitate the reduction in low-density lipoprotein (LDL) and prevention of ASCVD.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Considerations
Our institution does not require ethical approval for reporting individual cases or case series.
Consent to Participate
Both verbal and written informed consent to participate were obtained from the patient.
Consent for Publication
Both verbal and written informed consent to publish were obtained from the patient.
Prior Presentation of Abstract Statement
The abstract was previously presented at the 2024 ACP Southern California scientific meeting in Orange, California on 10/5/2024.
