Abstract
Secondary malignancies including leukemia are an increasing concern in patients with prior primary malignancies treated with alkylating agents or topoisomerase II inhibitors. These can also be referred to as therapy-related leukemia. Therapy-related leukemia most commonly results in myelodysplastic syndrome or acute myeloid leukemia. The alkylating agent can cause chromosomal aberrations typically manifest as deletions in chromosome 11 or loss of part of complete loss of chromosomes 5 and 7. Conversely, acute lymphoblastic leukemia (ALL) has been described following maintenance therapy with immunomodulatory (IMiD) drugs pomalidomide, thalidomide, and lenalidomide. We present a case of a 71-year-old man with a history of multiple myeloma (MM) maintained on lenalidomide after stem cell transplant who presented with treatment-associated ALL. At time of leukemic presentation, chromosomal analysis showed a near-triploid clone consistent with masked double low hyplodiploidy which is associated with a poor prognosis. The patient had a deletion of the long arm of chromosome 5 which has been described in prior case reports with ALL secondary to lenalidomide therapy. There are explicit mechanisms in the literature, which have been attributed to development of ALL after exposure to thalidomide or lenalidomide. At time of submission, there are 20 cases described in the literature linking ALL to IMiD drugs. We describe a case and review the mechanisms of lenalidomide-associated ALL.
Introduction
Secondary malignancies including leukemias are an increasing concern in patients with prior primary malignancies treated with alkylating agents or topoisomerase II inhibitors. 1 These can also be referred to as therapy-related leukemias. Therapy-related leukemias are most often myelodysplastic syndrome (MDS) or acute myeloid leukemia (AML). 1 However, a subset can develop acute lymphoblastic leukemia (ALL) secondary to these therapies. Therapy-related ALL remains elusive to define because there is a lack of large data sets recognizing the defining characteristics of this entity. 2 Acute lymphoblastic leukemia can be due to either typical cytotoxic agents such as alkylating agents and topoisomerase inhibitors or immunomodulatory (IMiD) drugs. 3 Topoisomerase inhibitors act by causing multiple DNA strand breaks which may lead to chromosomal translocation. Alkylating agents such as melphalan usually follow a long period of latency from initial use and present as AML or MDS with unbalanced chromosomal aberrations. 4 The alkylating agent chromosomal aberrations are typically manifested as deletions in chromosome 11 or loss of part of or complete loss of chromosomes 5 and 7. 4 Immunomodulatory drugs such as thalidomide or lenalidomide have differing mechanisms which have not yet been fully eludicated. Ito et al, Kronke et al, and Lu et al demonstrated that lenalidomide exerts its action by binding to cereblon which potentiates the ubiquitination and proteosomal-dependent degradation of 2 transcription factors known as IKZF1 and IKZF3. IKZF1 and IKZF3 are necessary for IMiD treatment effect on myeloma cells.5-7
Case Study
A 71-year-old Caucasian man presented to the hospital with a chief complaint of rib and back pain. He was found to be in acute renal failure with a creatinine of 2.1 mg/dL and hypercalcemia with a level of 13.1 mg/dL. Patient had an elevated intact parathyroid hormone (PTH) of 128.4 ng/L which was suspicious for primary hyperparathyroidism, but sistimibi scan ruled out primary hyperparathyroidism. Further renal workup revealed subnephrotic proteinuria with a 24-hour urine protein excretion of 2117 mg/day. X-ray revealed potential lytic lesions so a positron emission tomography–computed tomography (PET-CT) was obtained which revealed multiple lytic lesions in the axial skeleton. Bone scan also showed focal areas of increased uptake in the left 10th and 11th rib. Kappa-free light chains measured 62.7 mg/dL and lambda-free light chains measured 0.925 mg/dL with a kappa-lambda ratio of 67.8. The urine protein electrophoresis revealed a monoclonal-spike of 55.8 mg/dL. Serum immunofixation revealed a monoclonal kappa population. His β-2-microglobulin measured 4.10 mcg/mL. Bone marrow biopsy revealed plasma cells occupying 25% of bone marrow cellularity. The myeloma cells were monotypic kappa expressing CD38 and CD138. IgG measured 672 mg/dL and IgM measured 125 mg/dL. IgA was within normal limits. The patient was diagnosed with International Staging System (ISS) stage II kappa light chain multiple myeloma (MM) and underwent treatment with lenalidomide, dexamethasone, and bortezomib for 4 months. He achieved a very good partial remission and proceeded with autologous hematopoietic stem cell transplant. Clinically, he responded as evidenced by normalization of his creatinine, normal kappa-lambda light chain ratio, resolution of M-spike, and normal calcium level. Hematopoietic stem cells were harvested with granulocyte-colony stimulating factor (GCSF) and plerixafor. After receiving melphalan 200 mg/m2 conditioning on T_1, stem cells were infused on T+0. He had expected toxicities of high-dose melphalan therapy including cytopenia. He received red blood cell and platelet transfusions as needed and GCSF was administered per protocol beginning T+5 until engraftment occurred. After recovery from transplant, he initiated on lenalidomide, maintenance therapy. The patient remained stable on this regimen with close follow-up. During each toxicity evaluation, he was found to be in clinical remission with normal lab values including complete blood counts, protein electrophoresis, and kappa-lambda light chain ratio.
After approximately 6 years of maintenance therapy, he was discovered to have worsening cytopenia. Lenalidomide was discontinued. A bone marrow biopsy was performed and revealed hypercellular marrow with 95% involvement by B-lymphoblastic leukemia. He was referred to a tertiary care center for further treatment. Bone marrow flow cytometry revealed the B-cell lymphoblasts to be positive for CD20, CD79a, PAX-5, CD10, TdT, and CD34. The chromosomal analysis revealed a near-triploid clone which masked a double low hyplodiploidy. The patient had deletion of the long arm of chromosome 5q. There was no MYC rearrangement detections or BCR-ABL rearrangement. Fluorescence in situ hybridization (FISH) leukemia panel demonstrated positive trisomy and tetrasomy 6 (20.5% and 10% of cells, respectively) and positive for trisomy and tetrasomy 21 (17% and 17.5% of cells, respectively). There was no rearrangement of KMT2A (MLL). These results were consistent with a near-triploid/near-tetraploid clone with some cells having loss of chromosome 9/9q. Cytogenetics revealed 2 of the 22 mitotic cells were near-triploid and characterized by 2 copies of a chromosome 5 with additional material on the long arm resulting in deletion 5q; a gain onto triploidy of 1 copy of chromosomes 1, 6, 11, and 21; and loss of 1 copy of chromosomes 2, 3, 4, 7, 9, 13, 14, 16, 17, and 20. Cytogenetics, chromosomal analysis and FISH analysis are summarized in Table 1. He was initiated on mini-hyper CVD which consists of cyclophosphamide, vincristine, and dexamethasone with rituximab and inotuzumab. He received 2 cycles before this was discontinued due to toxicity from cytopenia and infections. He transitioned to best supportive care and required multiple packed red blood cell and platelet transfusions. He subsequently succumbed to his leukemia soon after treatment was discontinued. Treatment and outcomes are summarized in Table 2.
Cytogenetics and FISH.

Abbreviations: FISH, Fluorescence in situ hybridization; ALL, acute lymphoblastic leukemia.
Treatment and Outcomes.

Abbreviations: LBD, lenalidomide, bortezomib, dexamethasone; ASCT, autologous stem cell transplant; CVDRI, cyclophosphamide, vincristine, dexamethasone, rituximab, inotuzumab-ozogamicin; M, melphalan; L2, lenalidomide; ALL, acute lymphoblastic leukemia.
Discussion
This patient had deletion of the long arm of chromosome 5q karyotype (Figure 1). This is typical of therapy-related leukemias which tend to be associated with alkylating agents in AML. 8 The other studies revealed 2 cases of ALL after lenalidomide exposure to be positive for CD34 and CD79a.9-11 The most consistent genetic abnormality found across studies is the 11q23 (KMT2A) rearrangement, followed by monosomies of chromosomes 5, 7, and/or 17, hypodiploidy. 12 This patient lacked the 11q23 (KMT2A) rearrangement and the monosomy of chromosome 5. The patient was, however, positive for monosomy 7 and 17 along with being hypodiploid (Figure 1). Immunohistochemical studies show the blasts to be positive for CD20, CD79a, PAX-5, CD10, TdT, and CD34, all consistent with ALL (Figure 2). Bone marrow hematoxylin-eosin (H&E) stain was also shown to be hypercellular (Figure 3).

Two of the 22 mitotic cells examined were near-triploid and characterized by 2 copies of a chromosome 5 with additional material on the long arm resulting in deletion 5q, a gain onto triploidy of 1 copy of chromosomes 1, 6, 11, and 21; loss of 1 copy of chromosomes 2, 3, 4, 7, 9, 13, 14, 16, 17, and 20; and a range of 1-3 marker chromosomes. The karyotype description is a composite, listing only the consistent clonal changes. No abnormalities were evident in the remaining 20 cells. These results suggest a near-triploid clone which is masked double low hypodiploidy. Low hypodiploidy (30-39 chromosomes) appears to be a distinct entity of ALL where particular chromosomes are gained into a hyploid karyotype. Gains include chromosomes 1, 5, 6, 8, 12, 18, 21, and 22, while chromosomes 3, 4, 7, 13, 15, 16, and 17 are almost always monosomic. Structural abnormalities might also be present. The loss of terminal 5q is also associated with ALL.

The core bone marrow is hypercellular for age (90%). There is a diffuse proliferation with mononuclear blast-like cells. Immunohistochemical studies show the blasts to be positive for CD20, CD79a, PAX-5, CD10, TdT, and CD34 (subset).
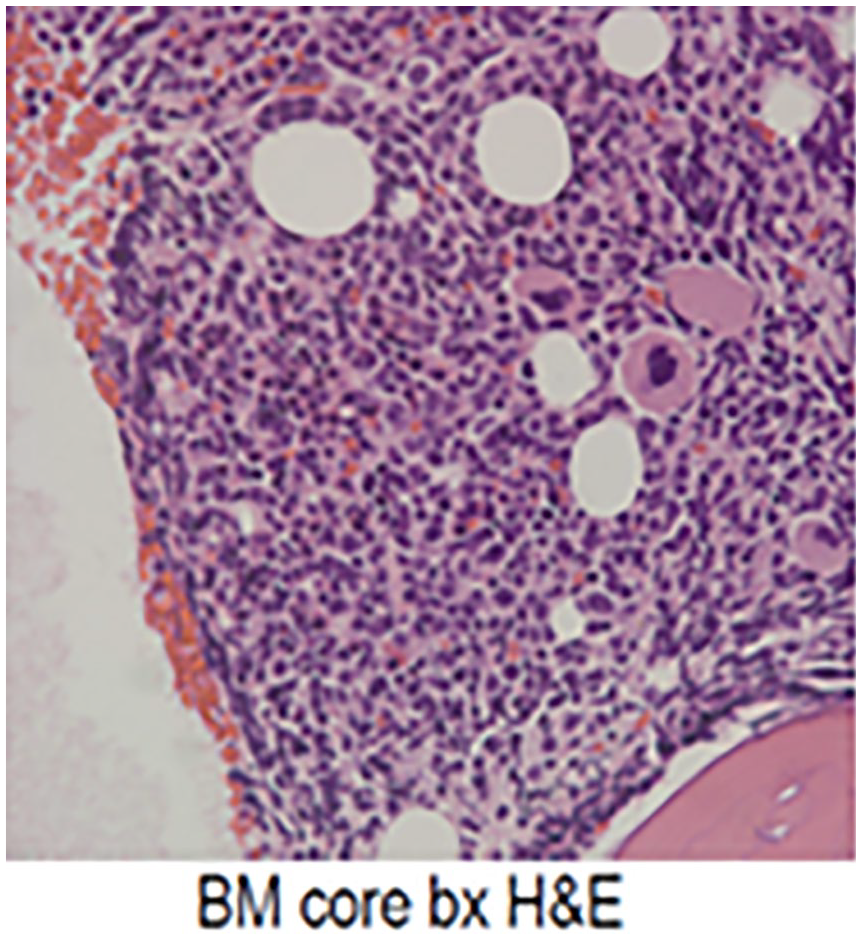
Hypercellularity seen on bone marrow H&E stain.
Therapy-related ALL is secondary to cytotoxic chemotherapy and radiation therapy. 12 In therapy-related ALL by Ribera, breast cancer with higher utilization of alkylating agents and topoisomerase II inhibitors was the most common solid tumor associated with treatment-related ALL. 12 In Aldoss et al, 2 MM (11%-12%) was one of the original diagnoses in which patients received cytotoxic chemotherapy and subsequently progressed to ALL. This cannot be completely attributed to therapy-related ALL secondary to lenalidomide as melphalan is commonly used as the conditioning regimen prior to autologous stem cell transplant (ASCT). 13 This patient received treatment with lenolidomide for 72 months and subsequently developing ALL. In other case reports, the median latency time seen until secondary hematologic malignancy was 35.5 months (median, 35.5 months; range, 23-96). 3 The latency period between developing ALL secondary to cytotoxic chemotherapy/radiation was 6.8 years. 2 This might suggest cytotoxic chemotherapy-related ALL in this case more closely as related to duration (6.8 vs 3 years), but this does not take into account the cytogenetic information. This case does have some cytogenetics associated with therapy-related ALL after exposure to thalidomide and lenalidomide. To date, there are 20 cases reported in the literature with 13 cases being present in a 2018 case report and another 7 added to the literature since the initial case report was published.3,9,10,14,15 One study was a clinical trial evaluating lenalidomide maintenance therapy in relapsed/refractory chronic lymphocytic leukemia (CLL) in which the trial had to be discontinued due to development of 3 cases of ALL. 15 A purported mechanism is loss of IKZF1 and IKZF3 as described in above references and in the clinical trial. 5 -7,15 In a recent review, IKZF1 and IKZF3 are critical for T-cell and B-cell differentiation and deletion of IKZF1 and IKZF3 are associated with B-cell progenitor ALL suggesting a tumor suppressor function. 16 Lenalidomide suppresses IKZF1 levels in human CD34 almost equivalently to a genetic loss of IKZF1 while also protecting self renewal. 15 Lenalidomide also induces the expansion of CD34 progenitor cell pool which taken together has led to the hypothesis that lenalidomide induces this expansion with increased cycling of CD34+ stem along with a deregulated metabolic state contributes additional DNA damage which can lead to leukemogenesis.15,17 The deregulated metabolic state is secondary to the loss of IKZF1. 15 Lenalidomide has a dose-dependent reduction in IKZF1 and IKZF3 which could possibly mean reduction in dose could decrease these effects if current dosages are enough to cause near-equivalent genetic loss of IKZF1.5,15 IKZF1 is critical in B-cell development as it induces rag1 and rag2 which mediates chromatin accessibility during immunoglobulin rearrangement. 18 In pre-B-cell differentiation, IKZF1 regulates transcription of genes important in pre-B-cell receptor signaling, cell survival, stromal cell adhesion, and B-cell commitment which suggests a deficit in IKZF1 is associated with deregulated lymphoblasts as stated above. 18
In addition, IKZF2 is resistant to the effects of lenalidomide and acts as a tumor suppressor in hypodiploid ALL which may explain the latency period and rarity of ALL in patients treated with lenalidomide.6,19 Another question that needs to be answered is whether lenalidomide has an association with masked hypodiploid ALL as this is associated with an extremely poor prognosis.14,20 Masked hypodiploidy is a described phenomenon where hypodiploid cells (<43 chromosomes) appear hyperdiploid but are actually from the cells doubling the chromosomal content of their previously hypodiploid cells. 20 In addition, masked hypodiploidy is associated with a high rate of TP53 mutations which are associated with a poor response to therapy.20,21
Melphalan is an alkylating agent which has been implicated in therapy-related ALL but is most commonly implicated with KMT2A rearrangements (formerly known as MLL) or hypodiploidy with loss of chromosomes 5, 7, and 17 which were more common in patients with prior exposure to alkylating agents and topoisomerase inhibitors. 22 KMT2A rearrangement was more common with t-ALL as a malignancy occurring specifically after alkylating agents.23,24 Therapy-related ALL (t-ALL) secondary to alkylating agents carries a similar profile to therapy-related myeloid neoplasms. 22 Whether t-ALL is due to alkylating therapy, transplant, myeloma itself, or IMiDs is an active area of investigation. 22 It is interesting to consider that lenalidomide can induce a deregulated metabolic state due to loss of IKZF1 with additional DNA damage accumulated such as DNA damage seen with alkylators which can accelerate leukemogenesis. The only aspect of this case as compared with other t-ALL secondary to lenalidomide is appearance of pancytopenia after several years of remission with maintenance lenalidomide. 10 It is important to note that approximately 4% of patients following autologous hematopoietic stem cell transplantation and lenolidomide maintenance may develop a secondary hematologic malignancy. 25
Conclusion
Awareness of secondary primary malignancies is crucial to treatment with certain agents such as the immunomodulator lenalidomide in MM; we suggest increased awareness of possible ALL which has been rarely described in the literature when treating with IMiDs, notably lenalidomide, which may present with new onset pancytopenia. Thus, new onset pancytopenia in a patient with MM who has been stably in remission should have low threshold for bone marrow biopsy.
Footnotes
Acknowledgements
We want to thank the patient for allowing us to share this with the medical literature.
Authors’ Note
This case was submitted as a poster to Florida ACP.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Verbal informed consent was obtained from the patient for their anonymized information to be published in the article.
