Abstract
The association between large pericardial effusion and restrictive cardiomyopathy (RCM) is uncommon and has seldom been described. We describe an uncommon case of a 31-year-old male with RCM who presented with large, recurrent pericardial effusion, heart failure, and echocardiographic findings showing progressive worsening of diastolic function even after total pericardiectomy who was eventually transferred for cardiac transplant evaluation.
Introduction
Restrictive cardiomyopathy (RCM) is seen in approximately 10% to 15% of patients with heart failure with preserved ejection fraction. Early diagnosis and treatment are important to prevent end-organ damage and improve survival. 1 Prognosis of RCM varies widely depending on its cause and ranges from months to years. It usually presents with heart failure, arrythmias, and electrocardiogram (ECG) abnormalities. Pericardial effusion is not usually described as part of its presentation.
RCM can be idiopathic or secondary. Each of its causes can have a distinct clinical presentation. There are a wide range of diseases that can cause restrictive physiology: endomyocardial fibrosis, infiltrative diseases, drugs, radiation, glycogen storage diseases, Friederichs ataxia, amongst others. 1
Large pericardial effusions usually occur due to slow accumulation of pericardial fluid, which allows the pericardium to expand before becoming symptomatic. Half of the time, there is no identifiable cause for the effusion. 2
We present a case of RCM that came to the hospital with 2 months of heart failure and a large pericardial effusion that delayed its diagnosis.
Case
A 31-year-old man with history of hypothyroidism, hypertension, gout, and tobacco abuse, who presented with dyspnea on exertion for approximately 2 months as a transfer from an outside hospital after a computed tomography angiography of the chest showed 3.5 cm pericardial effusion and bi-atrial enlargement (Figure 1). On admission, he was hemodynamically stable. Physical exam was significant for irregular heart rhythm and lower extremity edema. Thyroid function tests were within normal limits (Table 1). An ECG showed atrial fibrillation (AF) with a heart rate of 70 beats per minute and low voltage QRS.

Transverse CT thorax showing severely enlarged right atrium with large pericardial effusion.
Relevant Laboratory Results and Reference Range.

Abbreviations: SPEP, serum protein electrophoresis; UPEP, urine protein electrophoresis; TSH, thyroid-stimulating horomone.
Echocardiogram showed an ejection fraction of 69%, moderately dilated left atrium and severely dilated right atrium, large pericardial effusion without tamponade physiology, dilated inferior vena cava with poor inspiratory collapse, dilated hepatic veins, mitral annular velocity (e’) septal was 7.7 cm/s, e’ lateral 9.39 cm/s, and E deceleration time 132 ms (Figure 2). A right upper-quadrant ultrasound showed cirrhotic changes of the liver and partially reversed hepatic venous waveforms. A liver biopsy showed focal centrilobular congestion and bridging fibrosis with regenerating nodules consistent with cardiac cirrhosis.

Echocardiogram done on first admission: (a) left ventricular inflow and (b and c) tissue doppler of myocardial velocities. (a) E deceleration time 132 ms, (b) e’ lateral velocity 9.39 cm/s, and (c) e’ septal velocity 4.36 cm/s.
The pericardial effusion was thought to be secondary to pericarditis. Ibuprofen and colchicine were started. On serial echocardiograms, the effusion was stable. His CHA2DS2VASc score was 1 due to hypertension, anticoagulation was not started, and he was discharged.
On outpatient follow-up, the pericardial effusion, shortness of breath, and lower extremity edema remained despite adding metolazone and furosemide. A pericardiocentesis was eventually done, and 915 cc of transudative fluid was retrieved. After the procedure, ECG no longer showed low-voltage QRS.
Three months after initial presentation, he was admitted with a left medial cerebral artery stroke which was successfully aborted with tissue plasminogen activator. This was thought to be secondary to AF, he was started on apixaban.
On subsequent echocardiogram (approximately 1 month after drainage), the pericardial effusion reaccumulated and remained large, he was referred to cardiac surgery and was undergoing evaluation for pericardiectomy. However, 6 months after his initial presentation, he came to the emergency department with worsening shortness of breath, lower extremity edema, and chest pain. A pericardial window was done with 1200 cc drained; on postoperative day 2; he developed hemopericardium with tamponade, for which he underwent an emergent total pericardiectomy. His condition improved significantly and was discharged once he was hemodynamically stable.
After discharge he developed bilateral large pleural effusions and was readmitted and drained approximately 2 L of transudative fluid. Echocardiogram showed e’ septal 4.36 cm/s, e’ lateral 6.92 cm/s, E deceleration time of 60 ms, and E/A ratio was 4.23 (Figure 3). Cardiac magnetic resonance images (CMRs) (Figure 4) showed bi-atrial enlargement, but signs of infiltrative disease could not be completely evaluated due to poor visualization of the myocardium on delayed post-contrast images. Pericardial tissue, liver tissue from previous biopsies, SPEP/UPEP, and immunofixation were used to investigate for amyloidosis (Table 1); however, no signs of it were found. Hemochromatosis and glycogen storage diseases were also ruled out.
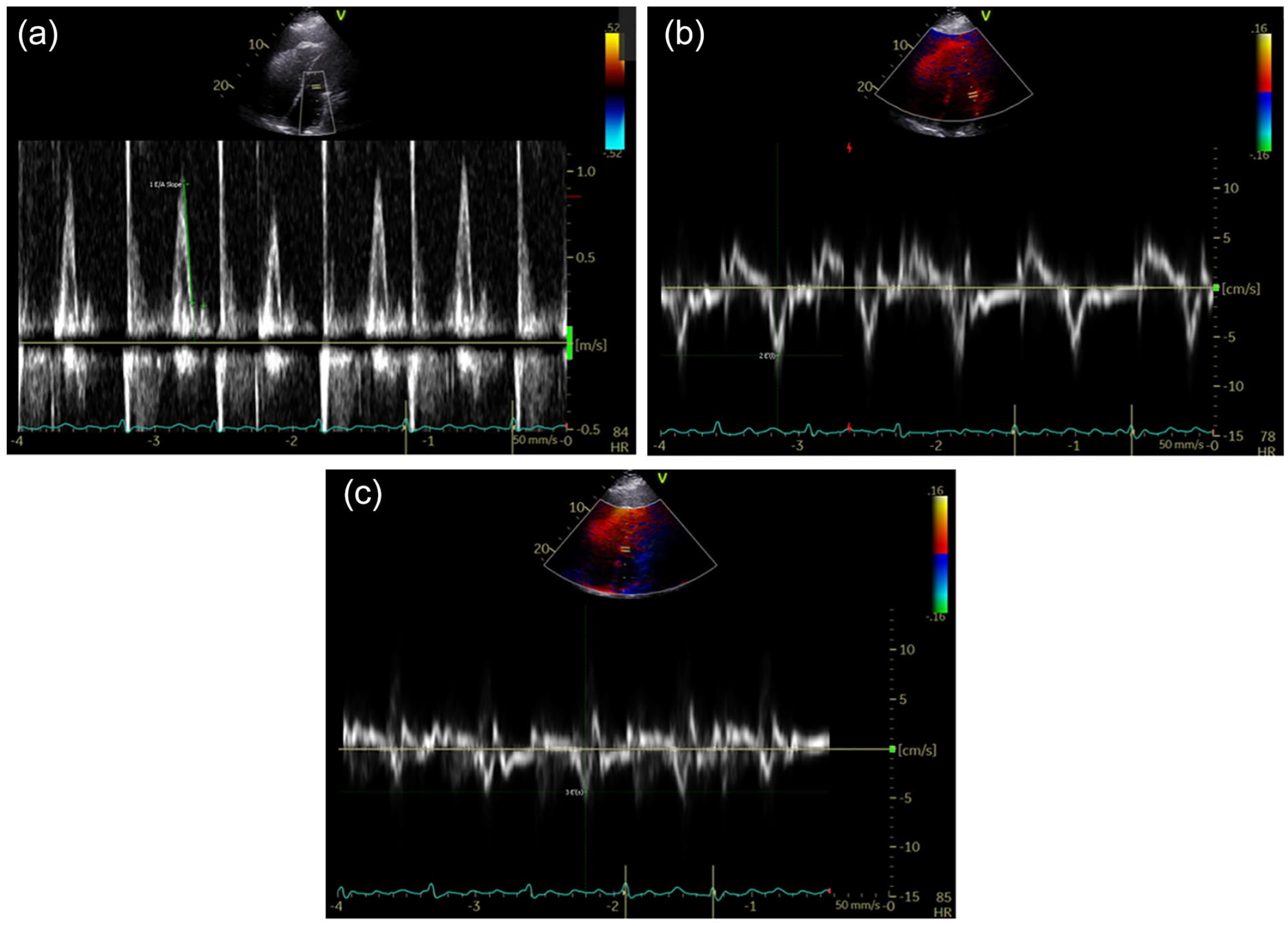
Echocardiogram done after pericardiectomy: (a) left ventricular inflow and (b and c) tissue doppler of myocardial velocities. (a) E deceleration time 60 ms, (b) e’ lateral velocity 6.92 cm/s, and (c) e’ septal velocity 4.36 cm/s.

Cardiac MRI with and without contrast showing small ventricles with severe right atrial enlargement (a, b,c) and left atrial enlargement (d).
At this point, we hypothesized that the patient likely had idiopathic RCM and was transferred to another facility for transplant evaluation.
Discussion
The diagnosis of RCM is based on echocardiography, CMR, hemodynamic studies, and endomyocardial biopsy.
The classic finding of RCM is severe atrial dilation, although this is not specific. Other echocardiographic findings are reduced early diastolic mitral annular velocity (e’), impaired LV relaxation (E/A ratio < 1), and short E deceleration time. 1 Cardiac magnetic resonance imaging is a useful diagnostic tool, which can identify cardiac function, anatomy, and detect inflammation, fibrosis, as well as signs of infiltrative diseases. 1
Pericardial effusion can be seen as a result of elevated right atrial pressures, given that the venous drainage of the pericardium comes from tributaries of the brachiocephalic veins. 3 Such is the case of transudative effusions seen on heart failure and pulmonary hypertension. 2 Fluid evaluation has a 7% diagnostic yield. 4
When pericardial effusion is not associated with inflammation, there are no specific therapies. Nonsteroidal anti-inflammatories and colchicine are usually not helpful. In these cases, removal of fluid with pericardiocentesis, pericardial window, or pericardiectomy should be considered. 2
There is little information about pericardial effusion in RCM. We found one case report of RCM presenting with in-utero pericardial effusion at 22 weeks’ gestation that eventually required a cardiac transplant at 2 years old. In this case, the diagnosis was suspected by frequent monitoring with serial echocardiography, demonstrating reduced LV function, mitral inflow, and pulmonary vein doppler data consistent with RCM; the diagnosis was confirmed with cardiac catheterization. 5
Although the main differential for RCM is constrictive pericarditis, in the presence of pericardial effusion, effusive-constrictive pericarditis should be part of the differential. In our case, the previously mentioned echocardiographic findings and the fact that his symptoms persisted after total pericardiectomy point toward RCM.
In our patient, echocardiogram showed bi-atrial enlargement, normal ventricular size, decreased e’ velocities, and short E deceleration time both before and after pericardiectomy, these findings on their own would normally point toward RCM. It is important to note that the echocardiographic values were significantly worse after the pericardiectomy, this can be explained by movement of the heart on a bed of large pericardial fluid during the first exam impairing the ability to obtain precise measure or progression of the underlying condition. However, in the presence of large pericardial effusion, his symptoms were initially attributed to idiopathic pericardial effusion.
Conclusions
Restrictive cardiomyopathy can present with large, recurrent pericardial effusion, and its presence should not dissuade us from pursuing its diagnosis, especially when associated with bi-atrial enlargement and characteristic echocardiographic findings.
Footnotes
Acknowledgements
A.C.S. contributed by conceptualizing and writing the original draft. Z.N., A.A., S.B.,a nd MB took part in review and editing. H.K. participated in supervision and review.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our Institution does not require ethical approval for reporting individual cases.
Informed Consent
Informed consent for patient anonymized information to be published in this article was not obtained from the patient because our institution does not require informed consent for individual case reports with information anonymized.
