Abstract
We describe a case of an extremely low birthweight infant with a large patent ductus arteriosus and closed foramen ovale resulting in markedly increased pulmonary blood flow, pulmonary venous congestion, and pulmonary hemorrhage.
Keywords
Introduction
Patent foramen ovale (PFO) in the fetus is covered by a one-way “flap valve,” which allows large right to left shunting throughout gestation. After birth with increased blood flow into the left atrium (LA) and reversal of pressure gradient between the left and right atria, the valve of foramen ovale closes with variable valve incompetence allowing left to right shunting during the early neonatal period. Most extremely low birth weight (ELBW; birth weight <1000 g) infants with a large patent ductus arteriosus (PDA) have significant left to right shunting through the PFO with an incompetent valve that prevents excessive volume overloading and left ventricular failure. We report a case of an ELBW infant with a closed foramen ovale (FO) and intact atrial septum in the presence of a large PDA unresponsive to medical management.
Case Presentation
A 725-g African American 26-week gestation male neonate born via emergency cesarean section to a 36-year-old gravida 3 para 2 mother. Pregnancy was complicated by morbid obesity, congestive heart failure, as well as insulin-dependent diabetes, chronic kidney disease, and chronic hypertension with superimposed preeclampsia. Mother did not have prenatal care but received prenatal steroids before delivery. Mother reported a history of daily marijuana use and occasional cocaine use.
Infant’s Apgar scores were 1, 3, and 5 at 1, 5, and 10 minutes, respectively. He was intubated in the delivery room shortly after birth and placed on conventional ventilation with a peak inspiratory pressure of 26, positive end-expiratory pressure of 6, and rate of 30. He received 2 doses of surfactant (Poractant alfa). Initial chest radiograph showed diffuse granular opacities. Initial echocardiogram (ECHO) on second day of life (DOL) revealed a very large PDA with an internal diameter of 2.9 mm (Figure 1), closed FO, enlarged LA with marked bulging of atrial septum into the right atrium (RA) (Figure 2), hyperdynamic left ventricle (LV), and global hypertrophy with low-velocity pulsatile flow profile through the PDA. Infant was started on indomethacin for a 3-day course and due to hypotension was started on dopamine and hydrocortisone. Pink-tinged secretions were aspirated from the endotracheal tube on DOL 5, suggestive of pulmonary hemorrhage. Chest radiograph showed diffuse hazy opacities and increased pulmonary vascular markings consistent with increased pulmonary blood flow and pulmonary edema. A repeat ECHO was performed on DOL 5 with no change in PDA size. Due to pulmonary hemorrhage, and persistent PDA, he was treated with acetaminophen from DOL 5 to 16 with no response. From DOL 2 to 19, he had 5 ECHOs showing persistence of global hypertrophy, PDA internal diameter ranged from 2.8 to 3.2 mm with a low-velocity pulsatile flow profile with pure L-R shunting (Figure 4). LA/aorta ratio ranged from 1.6 to 2.6 with bulging of LA septum into RA, LV shortening fraction ranged from 44% to 59%, and ejection fraction ranged from 78% to 93% (Figure 3). With a closed FO, we were able to estimate pulmonary (Qp) and systemic blood flow (Qs) by Doppler. Qs ranged from 256 to 310 mL/kg/min and Qp 531 to 912 mL/kg/min with an average Qp/Qs of 2.6 to 1.

Suprasternal ductal cut echocardiogram showing PDA measuring 3 mm in external diameter with L-R shunt. Abbreviations: PDA, patent ductus arteriosus; RPA, right pulmonary artery; LPA, left pulmonary artery; DA, ductus arteriosus.

Subcostal coronal view of the right atrium (RA) and left atrium (LA). Note bulging of atrial septum into the right atrium outlined with arrows.

Parasternal short axis M-mode of left ventricle. Note hypertrophy of anterior right ventricular wall, interventricular septum, and posterior wall of left ventricle.
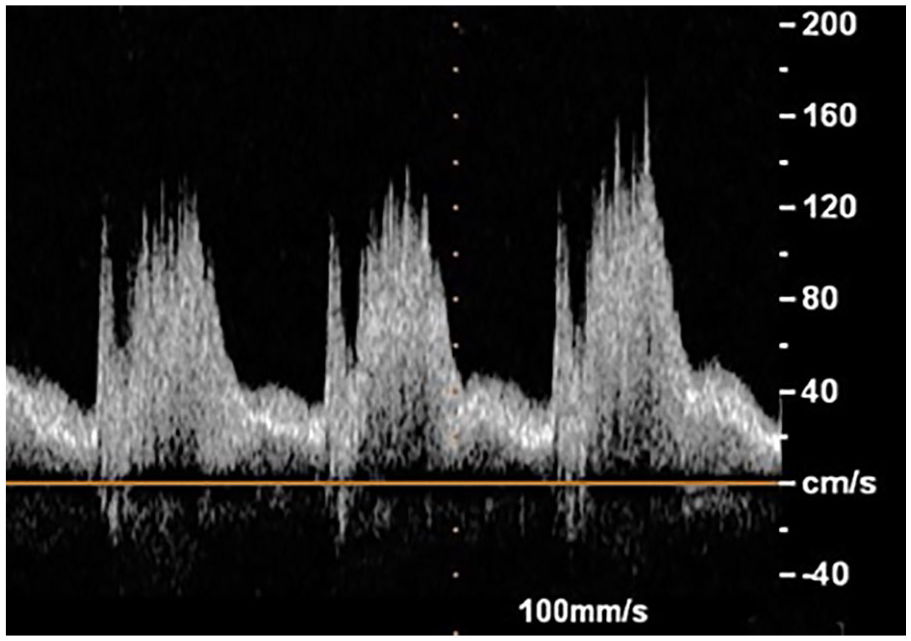
Doppler flow pattern of the patent ductus arteriosus, showing low-velocity pulsatile flow with left to right shunting.
He subsequently developed further difficulty with ventilation and was placed on high-frequency oscillatory ventilation on DOL 21 with a mean airway pressure of 13.5. Due to the continued hemodynamic instability and worsening lung disease, patient underwent ligation of the PDA on DOL 23. He was quickly weaned off of dopamine and hydrocortisone in the week after ligation. He was extubated from high-frequency oscillatory ventilation to nasal intermittent positive-pressure ventilation on DOL 35, 12 days after the surgery. ECHO on DOL 33 showed a closed PDA, normal cardiac function with resolution of the atrial enlargement with LA/aorta ratio of 0.89, and no septal bowing; however, global hypertrophy was still present on DOL 111. He required supplemental oxygen at 36 weeks postmenstrual age with a diagnosis of moderately severe bronchopulmonary dysplasia.
Discussion and Conclusion
The foramen ovale is a communication between the LA and RA located between the septum primum and septum secundum to provide right to left shunting of blood to bypass the lungs and supply systemic circulation during fetal life. 1 The upper portion of septum primum that overlaps the septum secundum on the left side is known as flap valve, which acts as a 1-way valve, shunting blood in utero from the RA to the LA. After birth, the sudden decrease in pulmonary vascular resistance allows the left atrial pressure to rise above the right, eliminating the right to left shunting of blood leading to closure of the FO. 2 There is minor valve incompetence in most neonates allowing left to right atrial shunting shortly after birth, especially in the presence of a PDA. 3
There is a high incidence of left to right interatrial shunting through PFO of various degrees in premature neonates during early neonatal period. 4 There is also a high incidence of PDA of various sizes in premature neonates. 5 In most cases, up to 85% of ELBW infants will experience spontaneous PDA closure prior to discharge. 6 In the presence of a large shunt through the PDA, a large left to right shunting through PFO typically leads to distribution of the shunt flow to both ventricles, preventing excessive volume overloading of the LV. Therefore, infants without significant interatrial shunt in the presence of a large PDA are at risk of pulmonary hemorrhage secondary to left atrial overload and pulmonary venous congestion.
Isolated restriction or closure of FO in fetuses has been reported, which in all cases will lead to enlargement of the RA and the right ventricle in the fetus and, if severe, fetal hydrops and fetal demise. 7 In our case, however, ECHO obtained at 2 days of life had normal right atrial and ventricular sizes, which indicates that closure of the PFO was probably a postnatal event. In the presence of a massive left to right shunt through a very large PDA, marked overloading of the LV occurred. Although this was partially compensated by the hypercontractile state of the LV with a shortening fraction ranging from 44% to 59%, increasing left atrial pressure led to pulmonary venous congestion, pulmonary edema, and pulmonary hemorrhage. This occurred despite mechanical ventilation with high positive end-expiratory pressure.
This clinical case brings up an important consideration in the highly controversial approach to the treatment of PDA in premature neonates. Although surgical ligation is being reserved as the last resort, in this case, it should probably have been among the first option. This patient sustained significant barotrauma being on mechanical ventilation for the first 3 weeks of life, while there was no change in the size of PDA with medical management. We are not aware of other reported cases of ELBW infants with large PDA in association with closed PFO. Although this is a rare case, ELBW infants with a large PDA and restrictive PFO may also be at similar risk of developing pulmonary hemorrhage and need for prolonged mechanical ventilation.
It was also noted that this infant had global ventricular hypertrophy, which was still present on the ECHO obtained at 111 days of life. The patient was an infant of diabetic mother who had a history of preeclampsia. Although the ventricular hypertrophy of infants of diabetic mothers resolves over the first 1 to 2 months of life, hypertrophy related to preeclampsia may be the result of concentric remodeling of the ventricles that can persist until adolescence and beyond. 8 Further echocardiographic follow-up is necessary to determine whether this was a transient or persistent left ventricular hypertrophy.
We suggest ELBW neonates with a large persistent PDA unresponsive to medical management and closed or severely restricted PFO with a diameter of <1.5 mm may be exposed to marked LV volume overload and pulmonary venous congestion. In these cases, early surgical ligation may be warranted.
Footnotes
Author Contributions
Dr. Jaclyn Kappico contributed to the implementation and analysis of the measurements as well as drafting the manuscript. Mahmood Ebrahimi, RDCS, contributed to the echocardiogram analysis. Dr. Bijan Siassi performed the echocardiographic measurements and planed, supervised, and drafted the manuscript. Dr. Rangasamy Ramanathan contributed to the design and editing of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Verbal informed consent was obtained from the patient’s parent for their anonymized information to be published in this article.
