Abstract
Hyperemesis gravidarum is a common disease. Most patients are effectively treated with conservative measures, but gastric feeding and, rarely, post-pyloric feeding can be necessary. A 27-year-old woman, G3P2002, with a history of refractory hyperemesis in previous pregnancies, required placement of a nasojejunal tube but was removed due to an oropharyngeal ulcer. Endoscopic placement of a percutaneous endoscopic transgastric-jejunostomy (PEG-J) tube caused resolution of her symptoms. Twelve days after placement, the distal tube became dislodged and was endoscopically replaced with hemoclip anchoring in the jejunum. PEG-J tube placement is a safe and effective option for nutritional support in refractory hyperemesis gravidarum.
Introduction
Hyperemesis gravidarum (HG) occurs in up to 2% of pregnancies nationwide and up to 20% can expect symptoms to persist throughout the pregnancy, often leading to severe complications. 1
Clinically, patients develop nausea and vomiting, associated hypovolemia, and ultimately maternal and fetal compromise. Persistent HG can lead to maternal complications such as dehydration, metabolic abnormalities, weight loss, muscle weakness, Boerhaave’s syndrome, and encephalopathy due to vitamin deficiencies. 2 The pathophysiology is complex and multifactorial; however, it can be summarized as being a combination of hormonal changes, hepatic dysfunction, metabolic derangements, lipid alterations, and psychological/sociocultural factors. 3 Symptom management and initial aggressive intravenous fluid resuscitation is paramount. However, medical management can be inadequate in controlling severe symptoms, occasionally requiring enteral feeding either via nasoenteric or percutaneous enteric tube feeds. We present a rare case of a pregnant female with severe HG that ultimately required a percutaneous endoscopic transgastric-jejunostomy (PEG-J) due to the necessity of post-pyloric feeding.
Case Report
A 27-year-old female, G3P2002, with a past medical history of HG in all prior pregnancies presented complaining of persistent nausea and worsening pain in the posterior oropharynx. The patient had a nasojejunal tube (NJT) placed with lead shielding of the fetus 8 weeks prior when she was 6 weeks pregnant (Figure 1). The NJT was placed because the patient had persistent HG that was not adequately managed with medications. The patient either had side effects or found no relief from ondansetron, metoclopramide, prochlorperazine, ginger, diphenhydramine, meclizine, or vitamin B6/dicyclomine. She was having at least 30 episodes of emesis daily and was losing weight. The NJT could no longer be continued due to a posterior oropharyngeal ulcer and the discussion of a percutaneous endoscopic gastrostomy (PEG) tube versus PEG-J was explored. Due to persistent nausea and vomiting, bypassing intragastric feeding by placement of a PEG-J tube was determined to be the best option by the obstetric and gastroenterology team. The patient was reassured that propofol, an anesthetic agent without risks of fetal compromise, would be chosen and that fetal heart tones would be monitored closely for proof of viability. Upper endoscopy revealed normal esophagus and stomach. A 24-Fr PEG was placed, and a 12-Fr jejunostomy catheter was passed through the PEG into the jejunum (Figure 2). The large-caliber catheters were chosen to reduce the risk of jejunostomy tube occlusion. In the recovery room, the patient experienced nausea with multiple dry heaves. The patient was discharged on postprocedure day 4 after her symptoms subsided. Unfortunately, she returned as an outpatient 12 days later with worsening nausea and vomiting. Abdominal radiograph revealed retrograde migration of the jejunal portion of the tube in the distal stomach (Figure 3).
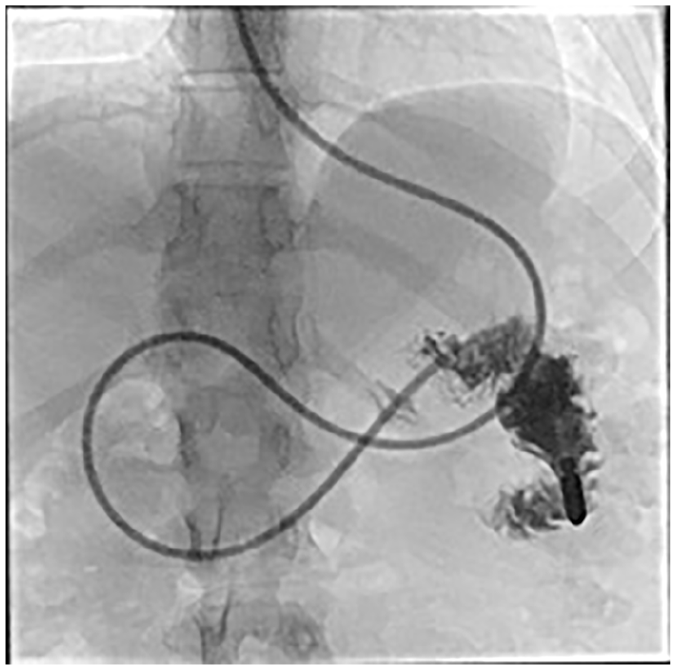
Nasojejunal tube placement confirmed with contrast.

A 24-Fr percutaneous endoscopic gastrostomy (PEG) was placed, and a 12-Fr jejunostomy catheter was passed through the PEG into the jejunum.
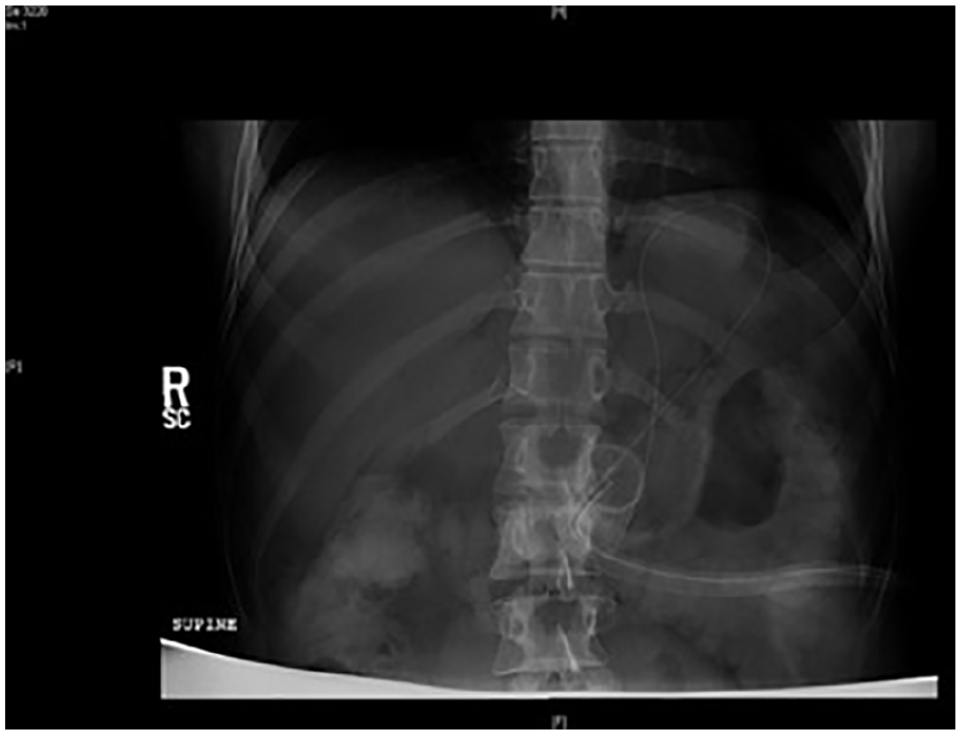
Abdominal radiograph revealed the jejunal portion of the tube to be in the distal stomach.
The patient underwent a repeat upper endoscopy, and PEG-J dislodgement was confirmed. The PEG-J was removed under endoscopic vision with help of a wire. A measurement device was used to determine the size of low-profile PEG-J required. An externally removable 22-Fr AVANOS MIC-KEY low-profile PEG-J tube was placed using the existing gastrostomy stoma. Proper position of the tube was confirmed by relook endoscopy, the distal tip was secured to the jejunal wall with 3 Boston Scientific Resolution hemoclips (Figure 4 and 5). The patient was subsequently asymptomatic for the following 18 weeks of pregnancy, and the PEG-J was removed with manual traction at bedside on postpartum day 1 without any complications. There were no identifiable findings of fetal malnutrition.

Proper position of the tube was confirmed by relook endoscopy, the distal tip was secured to the jejunal wall with 3 Boston Scientific Resolution hemoclips.
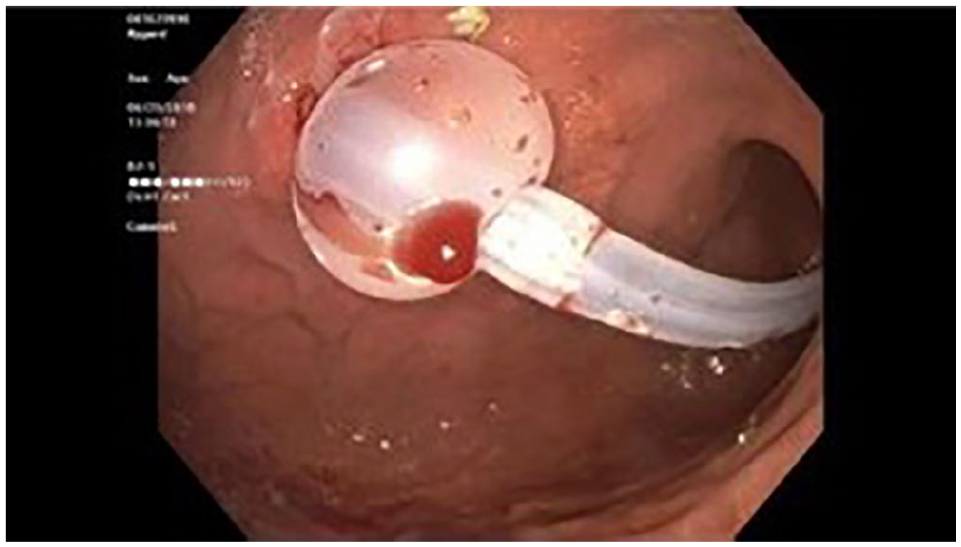
An externally removable 22-Fr AVANOS MIC-KEY low-profile percutaneous endoscopic transgastric–jejunostomy tube was placed using the existing gastrostomy stoma.
Discussion
Recent literature discussing refractory HG cases requiring PEG-J tube placement is scarce. The first case of PEG-J tube placement for enteral feeding in the setting of refractory HG was described in 1997. 4 Another report soon followed of 2 cases performed at the same institution and then a subsequent case series analyzing 5 patients from 1998 to 2005.5,6 All of which deemed the procedure a safe and effective advanced treatment for refractory HG. Our patient developed severe, refractory HG that required multiple procedures to adequately control her emesis and provide nutritional support. Intravenous antiemetics and NJT placement were ineffective, which led to significant weight loss and dehydration. Out of concern for fetal and maternal nutritional deficiencies, PEG-J placement was the most appropriate and least invasive intervention. After initial PEG-J tube placement, she had ongoing nausea and vomiting, presumably from PEG-J migration, but other causes such as adverse effects from propofol must be considered. There is no evidence to suggest that HG patients develop worsening nausea and vomiting after propofol administration, but the literature is limited to a handful of case studies describing propofol as being well tolerated in HG patients. 7 Furthermore, there are data to suggest that refractory HG patients may have persistent symptoms due to Helicobacter pylori infection, which was not tested in this patient. 8 Complications of tube dislodgement are attributed to the dynamic maternal/fetal anatomy throughout pregnancy in addition to the increased intragastric pressure during vomiting and retching. One retrospective study analyzed tube migration after using hemoclips in PEG-J placement and found that only 7% experienced tube migration.9,10 There is substantial evidence in the literature that attaching a nonabsorbable (eg, nylon) loop at the end of the J-tube, and clipping it to the surrounding mucosa with an over-the-scope clip is a great way to prevent tube migration. 9 Perhaps placing hemoclips during the initial procedure would have prevented the need for a second procedure. The low-profile PEG-J has been shown to have fewer tube dislodgement complications as well as shorter duration of hospital stays in the pediatric population. 11 More studies need to be performed in the adult population. The disadvantages to low-profile PEG-J tube placement are that there are lower reported success rates as defined as increased risk of infection and peristomal leakage, which our patient did not have. 12 Alternative management options include parenteral nutrition, which has been found to have the highest complication rates. 13 These complications are well characterized and widely recognized, but include catheter-associated infections, liver disease, gallbladder dysmotility, intrahepatic cholestasis, and metabolic abnormalities. In conclusion, this case highlights the indication for a PEG-J feeding tube, reviews potential complications of the procedure in pregnancy, but, most important, demonstrates how jejunostomy feeding tubes can be placed safely endoscopically by gastroenterologists and is well tolerated by the mother and fetus.
Footnotes
Authors’ Note
This abstract has been presented at the following conferences: (1) ACG Annual Scientific Meeting 2019; San Antonio, TX; October 2019.
Author Contributions
David Kruchko: Primary author; image acquisition.
Natasha Shah: Writer of manuscript, image acquisition.
Charles Broy: Reviewer of manuscript.
Dean Silas: Primary endoscopist; reviewed/edited manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Verbal informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
