Abstract
A 43-year-old man, with severe obesity (43 kg/m2) and diabetes (presumed as type 2 diabetes [T2D]), underwent vertical sleeve gastrectomy in 2009 and Roux-en-Y gastric bypass in 2013. Recently, whole exome sequencing (conducted to search for monogenic obesity) serendipitously revealed that the individual harbored a heterozygous glucokinase (GCK) variant p.(Arg422Leu) that was bioinformatically strongly predicted to be likely pathogenic. Therefore, he is likely to have concomitant maturity-onset diabetes of the young (MODY) type 2 (GCK-MODY). A retrospective evaluation of the clinical data showed that the subject was diagnosed with T2D (given his severe obesity) in 2005 and was treated with oral antidiabetic monotherapy. His hyperglycemia was mostly mild (HbA1c [hemoglobin] < 8.1%), consistent with that of MODY2, despite severe obesity. After vertical sleeve gastrectomy, complete diabetes remission (HbA1c <6.0% and fasting plasma glucose <5.6 mmol/L without use of antidiabetic medication) was achieved. The percentage of maximum body weight loss attained after surgery was 23.6%. Euglycemia was maintained during the subsequent decade, up to the last follow-up in 2019, without any sign of hypoglycemia. In conclusion, we report a decade-long clinical experience of a man with severe obesity and diabetes likely due to the coexistence of GCK-MODY and T2D, serendipitously treated with metabolic surgery. Interestingly, metabolic surgery was effective and safe for him.
Introduction
Maturity-onset diabetes of the young (MODY) is a genetically heterogeneous group of hereditable monogenic diabetes that affects 1% to 2% of all diabetes. 1 In contradistinction with type 1 diabetes (T1D) and type 2 diabetes (T2D), MODY is a non-autoimmune, non-insulin-dependent and non-adiposity-driven form of diabetes. MODY is primarily caused by single genetic defects in autosomal-dominant genes, resulting in reduced production of insulin in the pancreatic β-cells that often leads to early-onset diabetes in adolescence or young adults (age <35 years). To date, 14 MODY-associated genes have been identified including the glucokinase (GCK) gene. 1 Accurate diagnosis of MODY can inform antidiabetic treatment precisely. However, majority of MODY cases are misclassified as either T1D (36%) or T2D (51%), partly due to substantial overlap in clinical features. 2 Only 6% of MODY individuals are accurately identified clinically. Therefore, misassigned diabetes subtype is common, especially in the previous decades when genetic test for MODY has been largely unavailable in the clinics.
In our current obesogenic environment, adiposity-driven T2D is increasing globally. It is therefore not inconceivable that the pathogenic processes, which lead to T2D, may occur in an individual with propensity for other forms of diabetes (eg, T1D or monogenic diabetes). In other words, individual who has one form of diabetes is not protected from developing another form at a different time space. Therefore, it is recognized that more than one form of diabetes can coexist in a person. 3
Mounting evidence has established metabolic surgery as the most effective intervention for rapid weight loss and amelioration of T2D. 4 Currently, no study has investigated the effect of metabolic surgery on MODY. Recently, we identified an adult who harbored a GCK variant serendipitously treated with metabolic surgery for severe obesity and T2D. Individuals with GCK-MODY typically display sustained mild fasting hyperglycemia and low risk for vascular complications. 1 Nonetheless, people with GCK-MODY are also susceptible to T2D, if the predispositions are present, such as exposure to obesogenic environment and ageing.1,5 Accurate diagnosis of GCK-MODY is clinically actionable, because antidiabetic pharmacotherapeutics (eg, multiple daily insulin injections) are not recommended, given the anticipated long-term stable glycemic trajectory and relative nonresponsiveness to most glucose-lowering agents. 6 In this report, we describe the long-term clinical experience of a man with severe obesity and coexistence of GCK-MODY and T2D.
Case Presentation
A man with severe obesity was referred for vertical sleeve gastrectomy (VSG) at a hospital in June 2009. A retrospective evaluation of his clinical data showed that he was first diagnosed with T2D in August 2005 when he was 39 years old (HbA1c = 8.1%; Figure 1). His fasting plasma glucose (FPG) ranged from 6.1 to 6.5 mmol/L over 3 visits within 2005 (Figure 1). Between November 2005 and September 2006, his HbA1c (hemoglobin A1c) showed a constant reading of 6.7% over 4 clinic visits. However, in 2007, his glycemic control deteriorated, with HbA1c and random glucose levels peaking at 8.6% and 18.7 mmol/L, respectively, but they were gradually returned to normal levels of HbA1c <6.0% and FPG <5.6 mmol/L with oral diabetes monotherapy. Optimal glycemic control persisted for a year up to his scheduled VSG. The highest recorded body mass index (BMI) of the subject was 43 kg/m2 in January 2007 (Figure 1). He succeeded in reducing his BMI to 36.8 kg/m2 before VSG that may be partly attributable to lifestyle modification and a very low-calorie diet. His other medical conditions included dyslipidemia, hypertension, microalbuminuria, and mild obstructive sleep apnea. The patient had a presurgical fasting C-peptide level of 0.67 nmol/L. His insulin resistance index of 1.46 (calculated by Homeostasis Model Assessment; HOMA2) was considered normal. After VSG, the oral hypoglycemic agent was stopped within 2 weeks, and his HbA1c (5.1% to 5.9%) and FPG (4.3-5.1 mmol/L) concentrations were maintained within normal ranges, although HbA1c fluctuations were observed (Figure 1). His hemoglobin concentrations decreased to below normal range (reference range = 13-17 g/dL) after surgery, ranging from 11.1 to 12.2 g/dL based on results obtained between July 2009 and June 2011, indicating mild anemia. In January 2013, the man underwent Roux-en-Y gastric bypass (RYGB) to resolve symptoms of gastroesophageal reflux disease and weight regain. After RYGB, HbA1c fluctuations appeared mitigated. His hemoglobin level restored to normal range 6 months after RYGB (13.6 g/dL); and his last hemoglobin record was 15.8 g/dL in January 2016. Complete diabetes remission (defined as an HbA1c of <6.0% and a FPG of <5.6 mmol/L without use of antidiabetic medication) was sustained for more than 10 years after the first surgery. No hypoglycemic events were reported.

Graph of HbA1c (hemoglobin A1c), FPG (fasting plasma glucose), and BMI (body mass index) measured during 2005 to 2019, 2005 to 2014, and 2007 to 2018, respectively. Actual data points are represented by markers. T2D (type 2 diabetes) was diagnosed in 2005. VSG (vertical sleeve gastrectomy) and RYGB (Roux-en-Y gastric bypass) were performed in 2009 and 2013, respectively.
In 2019, whole exome sequencing analysis for research revealed a heterozygous missense variant in the C-terminal coding region of the GCK gene (NM_000162.5 (GCK):c.1265G>T p.(Arg422Leu)) of the patient, which leads to an amino acid substitution at a highly conserved residue across species. The variant was confirmed by bidirectional Sanger sequencing (Figure 2). The allele frequency from all populations in the Genome Aggregation Database (gnomAD) showed 0%. Computational tools (SIFT, PolyPhen-2, and Mutation Taster) predicted the variant to be functionally deleterious. According to the American College of Medical Genetics and Genomics classification, the variant was interpreted as likely pathogenic (PM1, PM2, PP2, PP3, and PP5).
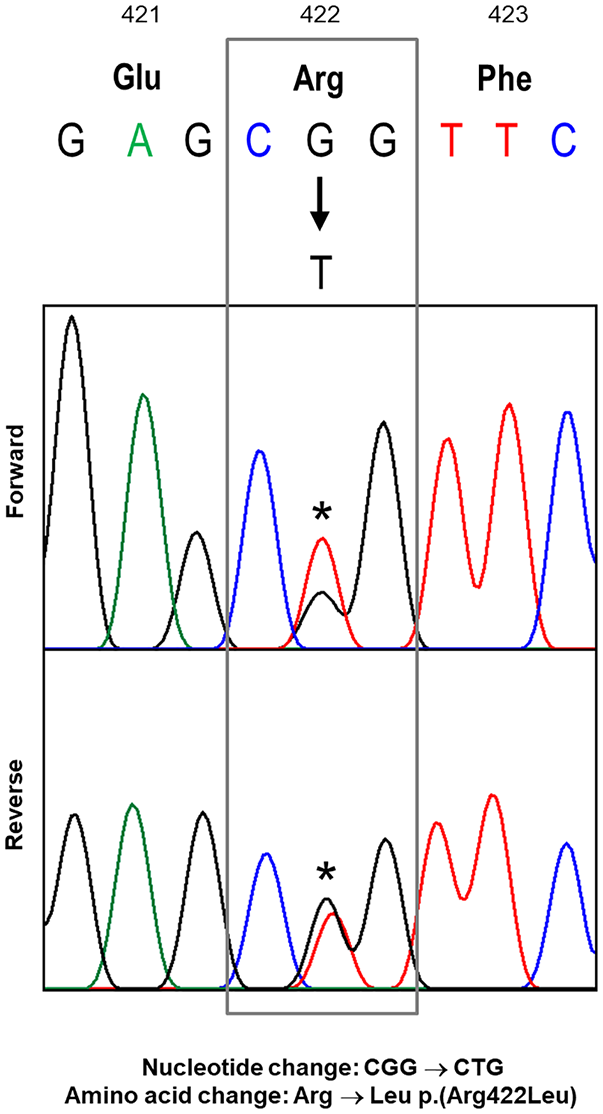
Bidirectional Sanger sequencing of the GCK gene. A nucleotide change from G to T resulted in an amino acid change from arginine to leucine.
Discussion
To our knowledge, this is the first report describing the impact of metabolic surgery on an adult with a genetic diagnosis of MODY and concomitant clinical diagnosis of T2D. The classic GCK-MODY phenotype is lean (BMI < 25 kg/m2), mild nonprogressive hyperglycemia, which is present soon after birth and persists lifelong, and no metabolic syndrome (unless exposed to an obesogenic environment). 5 Acknowledgedly, the clinical features of the case were atypical of classical GCK-MODY. The patient was diagnosed with diabetes at the age of 39 years, and had history of morbid obesity, hypertension, dyslipidemia, and microalbuminuria. The relatively late age of diabetes diagnosis may be attributed to the subject’s mostly mild and asymptomatic hyperglycemia that could have missed earlier detection. Conceivably, the presence of gross obesity in GCK-MODY may accentuate certain features of metabolic derangements that are more typical of T2D. In a longitudinal analysis of MODY individuals, it was found that GCK-MODY subjects with unexpected deterioration in glucose intolerance had higher body weight gain during follow-up, although the difference compared with those with stable glucose tolerance was not statistically significant. 7 Separately, a case study of a family with heterozygous variant of GCK p.(Arg36Trp) reported coexistence of clinical features overlapping GCK-MODY and T2D. 5 In contrast to the proband who had phenotype consistent with GCK-MODY, his older brother and father who were carriers of the same GCK variant displayed features of metabolic syndrome that are characteristics of T2D including overweight/obesity, hypertension, dyslipidemia, and raised hyperglycemia. Hence, people with GCK-MODY are similarly susceptible to pathophysiological changes (ie, weight gain and insulin resistance) associated with ageing and obesogenic environment.
Glucokinase serves as a glucose sensor that regulates insulin release from pancreatic β-cells, and a rate-limiting step in glucose metabolism that allows storage of excess glucose as glycogen in the liver to maintain glucose homeostasis.1,8 Therefore, variants in the GCK gene can cause chronic (albeit often mild) hyperglycemia or hypoglycemia due to altered “glucose-stat” threshold. To date, more than 600 such variants have been reported, with majority being missense (65%). 8 Here, we identified a heterozygous missense GCK variant p.(Arg422Leu) that was previously identified in people with GCK-related hyperglycaemia.2,9 However, the clinical and functional characteristics of the variant remain undefined. Neighboring variants found to be linked to GCK-MODY include p.(Lys420Glu), p.(Phe423Ser), p.(Phe423Tyr), and p.(His424Tyr). 9
People carrying GCK variants rarely need any pharmacological treatment as dietary management alone is often sufficient as an effective therapeutic option.1,2 However, given that weight gain and deterioration of HbA1c can also occur over time among people with GCK-MODY, the sustainability in exemption from anti-hyperglycemic treatment (by medication or bariatric surgery) for GCK-MODY has become controversial.7,10 Our study subject was treated with oral diabetes monotherapy before VSG that may have helped lower his hyperglycemia, that is, HbA1c decreased from the highest of 8.6% to 5.2% prior to surgery. His medication was stopped within 2 weeks after surgery. The case was successful in keeping his HbA1c and FPG at <6% and <5.6 mmol/L, respectively, without showing any sign of hypoglycemia for more than 10 years postsurgery. Despite some weight regain, euglycemia was maintained, possibly attributable to the weight-independent, surgery-specific effect on islet function, hepatic glucose production, and insulin clearance.11,12 Hence, metabolic surgery seems to be able to induce the remission of hyperglycemia and sustain its remission over a decade in such a hybrid situation. Taken together, the findings suggest that metabolic surgery is a safe and effective diabetes treatment intervention for the subject.
Several study limitations need to be acknowledged. We could not accurately ascertain the age of onset of diabetes, which is often the case in GCK-MODY or mild T2D. The information on the patient’s lifestyle and pharmacological treatment prior to VSG was not recorded. However, metformin (a weight-friendly antidiabetic agent) was likely prescribed because our clinical practice guidelines have recommended it as first-line therapy for diabetes in the last decade. The family history of diabetes is unavailable. Detailed glycemic profiling (eg, oral glucose tolerance test) is unavailable. As the subject has ceased to be under our follow-up, we could not invite his family members for genetic testing of GCK-MODY.
Conclusion
We have identified a rare case with severe obesity and coexisting phenotype of GCK-MODY and T2D. The individual has benefited from metabolic surgery in terms of weight loss and glycemic control in the long-term.
Footnotes
Acknowledgements
The authors thank the patient for his participation in this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by the Alexandra Health Fund Ltd through the Small Innovative Grant, SIGII/14034, and the Science – Translational & Applied Research (STAR) Grant, STAR20202. SC Lim is supported by the Singapore Ministry of Health’s National Medical Research Council Clinical Scientist Award (NMRC/CSA-INV/0020/2017).
Ethics Approval
Clinical data and sample from the patient were obtained with informed consent under a protocol approved by the National Healthcare Group Domain Specific Review Board (2014/01234).
Informed Consent
Informed consent for patient information to be published in this article was not obtained because the patient data and sample are de-identified.
