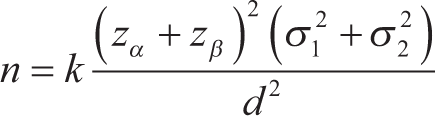Abstract
Aim:
To evaluate the palatal donor site after harvesting connective tissue graft with or without platelet-rich fibrin (PRF).
Material and Methods:
Forty-two patients requiring connective tissue grafting were randomly divided into two groups: the experimental palatal donor site (n = 21) received PRF before suturing, and the control palatal donor site (n = 21) was sutured without any PRF.
Results:
Among 42 enrolled patients requiring connective tissue grafting, 34 patients completed the study. After 10 days, there was an increase in the average thickness of the palatal surgical test site in PRF-treated patients, whereas it decreased in non-PRF-treated patients. Patients’ response and acceptance for the surgical treatment modality in terms of Patient Comfort Score (PCS) and Patient Appearance Score (PAS) showed less discomfort and better/pleasing appearance in the experimental group after treatment as compared with the control group. “Absolute contour match” of the healing lesion to adjacent mucosa was observed in 76.5% of experimental sites, and no “absolute contour match” was observed with the adjacent non-surgical tissue in the control group, whereas, at 3 and 12 weeks, a 100% “absolute match” with the adjacent tissue was observed for all esthetic parameters in both groups.
Conclusion:
PRF usage at the donor site seems to be a simple less-invasive approach of high significance resulting in more accelerated physiological healing phenomenon and better early healing response.
Introduction
Connective tissue grafting is considered the gold standard for root coverage, and it demonstrated the highest success rates with the greatest amount of predictability. For autogenous connective tissue grafting procedures, the palate is usually considered the most preferred donor site. 1 However, these procedures showed few complications after harvesting connective tissue grafts (CTG), for example, pain, inflammation, bleeding, flap necrosis, and infection in the palatal donor site. Zuchelli et al. 2 advocated the use of Equine-derived collagen with compressive sling-anchored sutures to reduce patients’ palatal morbidity.
Platelet-rich fibrin (PRF) has been established in previous studies to be an autologous, easy-to-obtain, economical, non-immunogenic biomaterial with great handling qualities, and its usage at the donor site may be a simple less-invasive approach of high significance that may accelerate physiological healing phenomenon.3–5 Therefore, the prime aim of this study was to evaluate the palatal donor site after harvesting CTG with or without PRF.
Materials and Methods
Setting and Design
The current clinical research prospective randomized interventional study was accomplished in the Periodontology Department from January 2017 to November 2018 (22 months). Non-alcoholic, non-smoker (self-reported) participants with no relevant medical history were recruited from the outpatient Department of Periodontology based on the inclusion and exclusion criteria mentioned under the section sampling criteria.
Intra-observer Reliability
All of the clinical parameters were documented by a single observer who was unaware of the groups. A single observer randomly assessed the palatal thickness in 10 patients at six different points before and after 24 h for intra-class correlation (ICC) analysis to validate intra-observer reliability. Indicative of high intra-observer reliability for the case selection, measurement methods, and surgical procedures, the ICC values revealed a significant (p < .001) and positive correlation for palatal thickness measurements (ICC = 0.967) between the two time intervals.
Sampling Criteria
The participants in this clinical investigation were randomly allocated by probability sampling technique with a simple randomization method to one of the two treatment groups using identical-looking sealed opaque envelopes, thus ensuring that all the participants had an equal chance of being chosen. To prevent heterogeneous sample size, the number of envelopes for each treatment was the same.
Sample Size Determination
The sample size was calculated based on the variation in the thickness of the palatal donor site among the control and experimental groups,
2
using the formula:
where δ1 = 0.47, δ2 = 0.46, the SDs of thickness of thepalatal donor site in the experimental and control groups; d = 0.5, the minimum difference considered to be clinically significant; k = 1.0 the design effect; and type I error α = 5% corresponding to 95% confidence level and type II error β = 20% for detecting results with 80% power of study. So, the required sample size was n = 17 for each group.
Inclusion Criteria
Patients between the ages of 18 and 55 years who are systemically healthy non-smokers, necessitating small connective tissue grafting (thickness < 2 mm) (e.g., periodontal plastic surgical procedure, gingival augmentation for restorative dentistry, orthodontics, and/or tissue build-up around implants); patients with U-shaped or high palatal vault 6 having at least 3 mm of mid-palatal mucosal thickness; 7 and patients well motivated for surgery and maintaining good oral hygiene (full-mouth plaque scores < 20% and full-mouth bleeding scores <15%) were included in the study.
Exclusion Criteria
The exclusion criteria were as follows: pregnant or nursing females; patients with any systemic disease/condition or taking any medication that contraindicates periodontal surgery or may hamper tissue health or healing; no anti-inflammatory drugs or antibiotics for at least 6 months; who were allergic to iodine; wearing any removable or fixed partial denture or Hawley’s appliance in the maxilla; with habit of one or other form tobacco use (self-reported); with history of palate surgery; with stomatological disorders or developmental disabilities in the palate; and with malpositioned teeth.
Participant Allocation
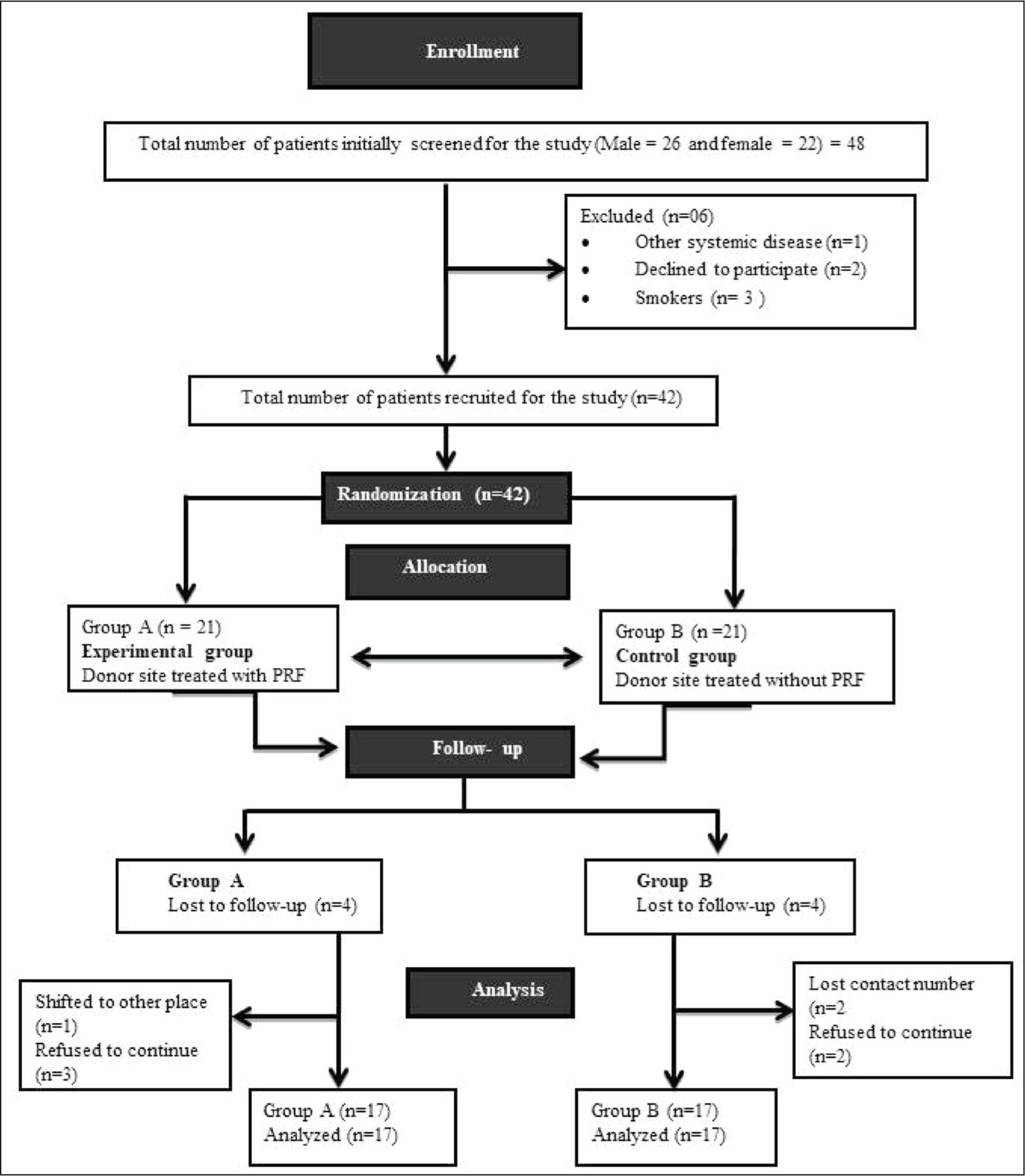
The groups were allocated as follows: Group A (PRF): palatal donor site after harvesting CTG treated with PRF and Group B (Control): palatal donor site after harvesting CTG without PRF (Figure 1).
Flow Chart of the Study Design.
Variables Outcome
The primary outcome of the study was palatal thickness and wound healing as explained in the previous study. 2 The secondary outcomes of the study included patient satisfaction analysis, postoperative pain, burning sensation, Patient Comfort Score (PCS), and Patient Appearance Score (PAS). The study might be confounded by variations in gingival thickness based on ethnicity, type of palate vault, and non-split mouth design.
Grouping and Allocation
Biased in the study was tackled by using a simple randomization method via sealed envelopes. While considering attrition in samples during follow-up, a total of 42 patients in need of soft tissue augmentation, who met the inclusion criteria, were enrolled to participate. They were randomly 1:1 allocated to experimental [with PRF (n = 21)] and control [without PRF (n = 21)] groups, via the sealed envelope method. Eight of the patients refused to participate, resulting in a final sample of 34 individuals (n = 17 PRF, n = 17 control). During the 12-week follow-up period, there were no subjects withdrawing from the study.
Methodology
Sample Preparation
Platelet-rich Fibrin
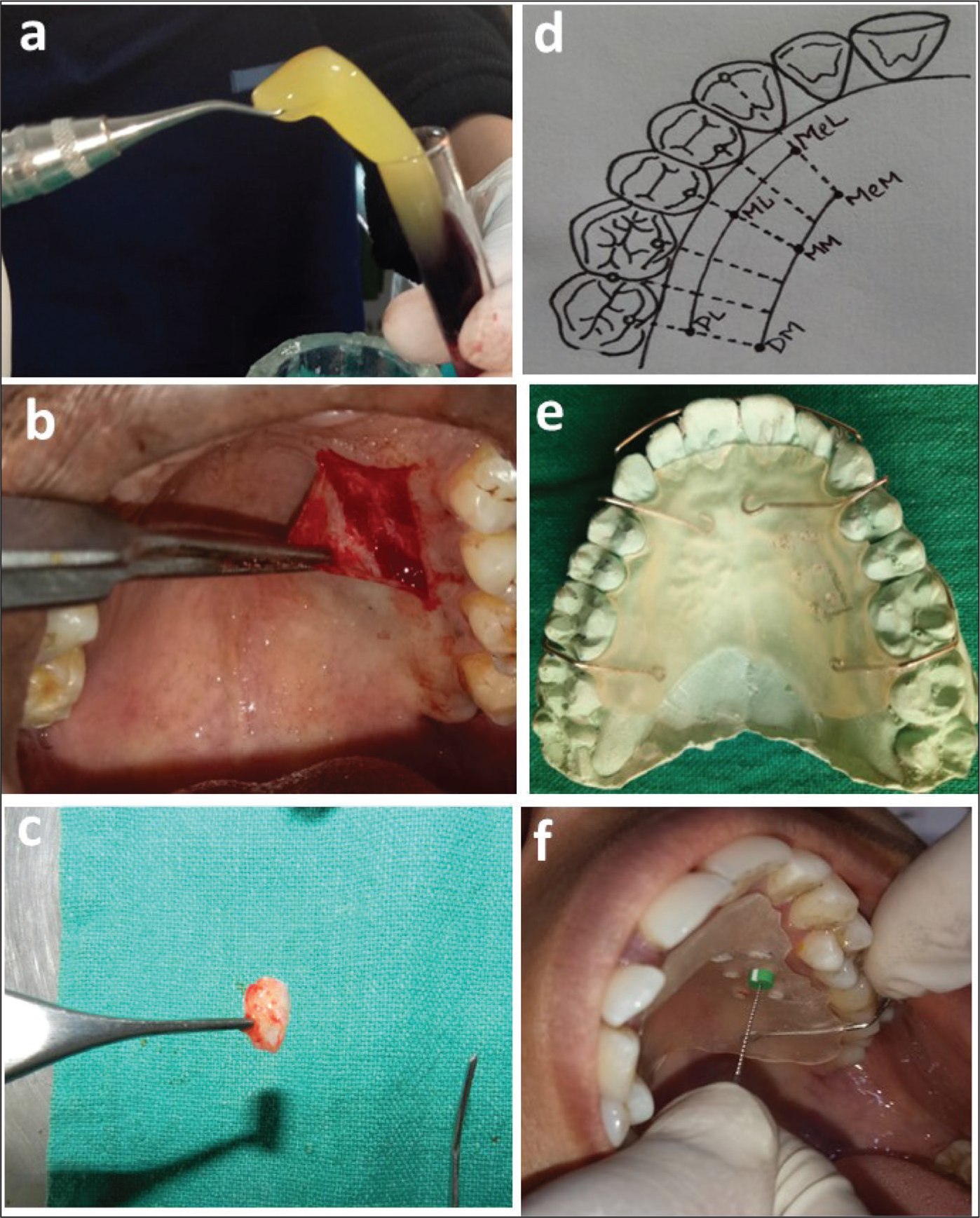
In this study, PRF was prepared from 10 mL of blood taken from the patient’s median cubital vein in a sterile tube without anticoagulant and centrifuged (REMI, Laboratories, India) immediately for 12 min at 3,000 rpm according to the protocol devised by Choukroun et al. 8 The PRF clot thus attained was then used at the surgical donor site (Figure 2a).
Connective Tissue Graft
According to Liu & Weisgold’s Class III Type A incision design description, the CTG for the current study was harvested from the palate vault of the same side of the teeth being treated. 9 The anatomic area between the mesial line angle of the palatal root of the first molar and the distal line angle of the canine ensures optimum thickness for CTG harvesting.10–12 Between the distal side of the canine and the palatal root of the first maxillary molar, approximately 2–3 mm away from the gingiva margin, a superficial horizontal incision was made [not extending to the bone, approximately equivalent to the bevel of the blade (about 1–1.5 mm)]. For this study, similar landmarks were utilized as guidelines for CTG procurement and a uniform thickness of 1.5–2 mm CTG was harvested (Figure 2b and c). A layer of fatty and glandular tissue, if present, was carefully removed. Until placed in the recipient area, CTG was well-maintained in normal saline. Vicryl 4-0 sutures were used to fix the palatal graft over the recipient site. The surgical donor palatal site was covered by repositioning the partial thickness flap, and 4-0 black braided silk interrupted sutures were placed to obtain primary closure.
Initial Therapy
The study procedure was thoroughly explained to each of the selected patients, and they were all admonished to adhere to strict plaque control practices and follow them to the letter. Phase I therapy was administered to each participant in the research.
Fabrication of Stent
A study model was created, and a uniform-thickness self-cured clear acrylic stent covering the palate was fabricated which was used as a fixed reference for measuring gingival thickness only, using transgingival probing as explained below. Six measure points were defined keeping mid-palatine raphe and gingival margin as reference points. Starting at the mid-palatal aspect of the canine and ending over the palatal root of the first molar, two distinct lines a and b were formed running parallel to the gingival margin. The distances between lines a (laterally) and b (medially) from the gingival margin were 3 and 12 mm, respectively. On each of the lines, three points were specified, corresponding to three positions: mid-palatal aspect of canine, mid-palatal aspect of second premolar, and mid-palatal aspect of the first molar. On line a, point MeL denoted the canine’s mesio-lateral aspect, point ML the second premolar’s mid-lateral aspect, and point DL the first molar’s disto-lateral aspect. On line b, point MeM denoted the canine’s mesio-medial aspect, point MM the second premolar’s mid-medial aspect, and point DM the first molar’s disto-medial aspect (Figure 2d). These six points were labeled for assessing the palatal mucosal thickness. Bold markings were made on the cast to represent these six markings which were transferred to the clear acrylic stent (Figure 2e). Fissured diamond burs were used to create holes at the marked six measurement points on the stent. Masticatory mucosal thickness was measured in the hard palate on these six markings by bone sounding using a sterile Endodontic K-file (no. 20) (Dentsply Maillefer, USA) with a silicon disk stopper after 20 min of anesthetizing the palatal mucosa with block anesthesia using 2% lignocaine HCL with 1:2,00,000 adrenaline solution (Indoco Warren Pharmaceuticals, India). The file was gently introduced into the soft tissues through the palatal mucosal surface until a sturdy surface was encountered, and care was taken to avoid its bending by strong resistance from bone contact. The extent of penetration of the Endodontic k-file was measured by using a caliper to the nearest millimeter (Figure 2f).
Surgical Procedure
Compliant patients who maintained good oral hygiene were scheduled for surgery. On the day of the surgery, baseline clinical values were obtained. As mentioned earlier, right before surgery, selected sites were randomly allocated to one of the two treatment approaches. The patient’s extraoral surface was swabbed with betadine prior to the procedure. A volume of 10 mL of undiluted 0.2% chlorhexidine digluconate solution was used for achieving oral antisepsis. The surgical area was anesthetized using lignocaine HCL with 1:2,00,000 adrenaline solution.
Blood was collected from the patient in Group A (PRF) to prepare PRF, as aforementioned. After obtaining adequate anesthesia, the recipient site was prepared. Subsequently from the palatal donor site, CTG was obtained as described by Liu and Weisgold 9 as explained earlier.
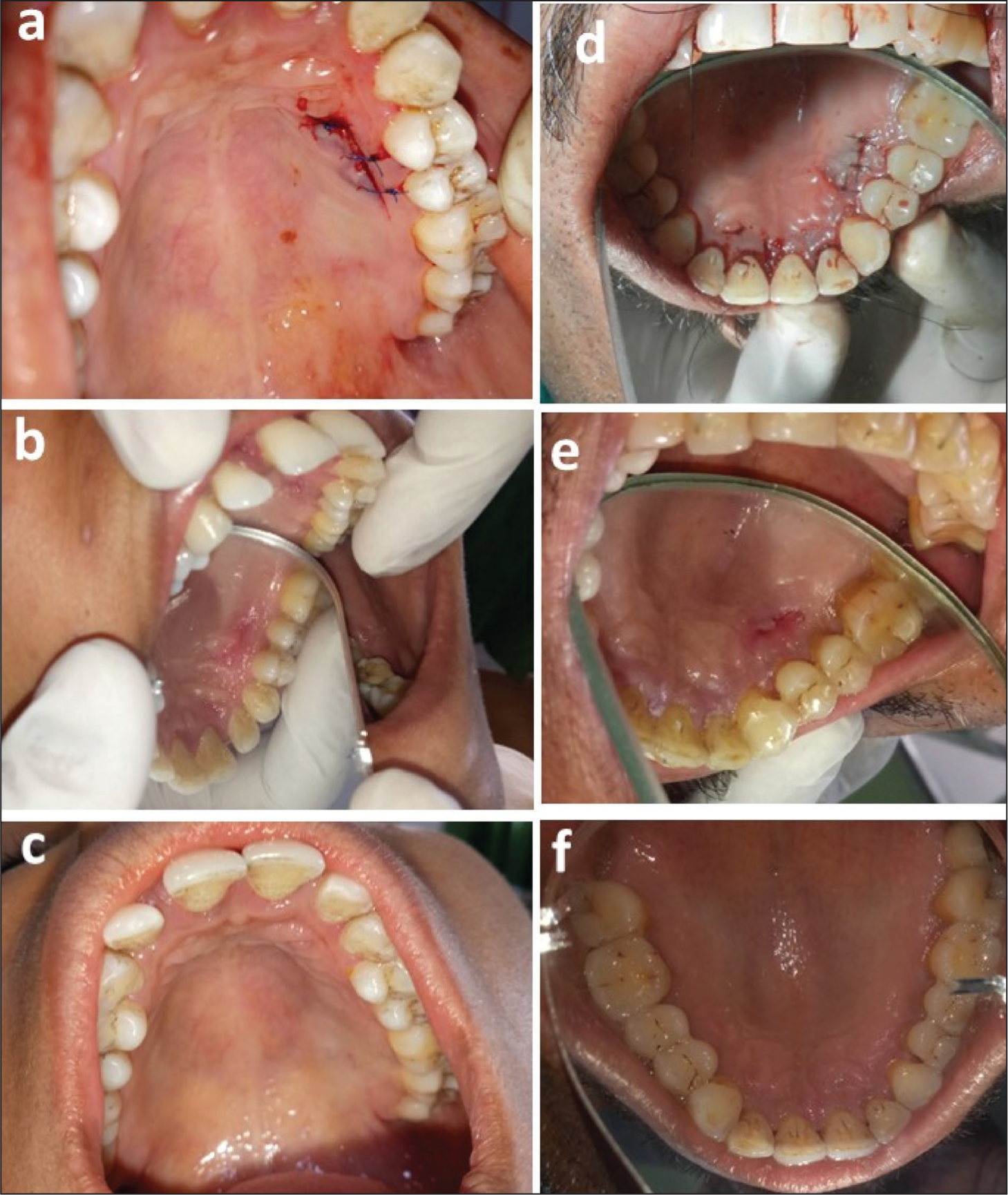
In the experimental site [Group A (PRF)] after harvesting CTG, PRF was positioned at the donor surgical palatal site and squeezed using a wet gauze piece, thus limiting the fluid attained to the test surgical site. The PRF membrane and blood clot were stabilized by applying pressure to the donor site with wet gauze. PRF placed on the palatal donor site was covered by repositioning the partial thickness flap, secured in place by interrupted sutures using 4-0 black braided silk sutures to obtain primary closure (Figure 3a).
In the control site [Group B (Control)], hemostasis was achieved by applying wet pressure gauze to the donor area, and the mucosal flap was sutured (Figure 3d). To preserve the underlying tissue, a periodontal dressing was applied to both the donor and recipient sites. The prescribed medications were Amoxicillin 500 mg thrice a day for 5 days and a combination of Paracetamol (325 mg) and Diclofenac (50 mg) [tab. Diclomol] three times daily for 3 days. Postoperative appointments were conducted for any complications, and patients were evaluated on a subjective and objective basis.
Patient Follow-up
The dressing and sutures were cautiously removed after 10 days without encroaching on soft tissue healing, and the surgical site was cleansed with normal saline. After 10 days (Figure 3b and e), 3 weeks, and 12 weeks (Figure 3c and f), the patient was scheduled for a recall appointment. Oral hygiene instructions were reiterated at every visit. If necessary, supragingival scaling was performed.
(a) Palatal Experimental Donor Site at Baseline. (b) Palatal Experimental Donor Site at 10 Days. (c) Palatal Experimental Donor Site at 12 Weeks. (d) Palatal Control Donor Site at Baseline. (e) Palatal Control Donor Site at 10 Days. (f) Palatal Control Donor Site at 12 Weeks.
Observational Parameters
Clinical parameters and patient satisfaction scores were analyzed by a single trained observer who was unaware of the material and methodology of the study. These observational parameters were recorded using visual examination and subjective verbal communication which were rated using the registration of visual analog scale (VAS) score (0–10) during the follow-up period.
Clinical Parameters
VAS was used to assess pain and burning sensation assessment.
The tissue color of the palatal mucosa was evaluated by comparing it to the adjacent and opposite sides. Scores of 0 meant no color match, 1 meant slight match, and 2 meant absolute match.
Contour of the surgical area was compared with the adjacent tissue.
Consistency of palatal mucosa as soft edematous/firm and resilient on palpation was determined with a blunt instrument after 10 days, 3 weeks, and 12 weeks.
Surface texture of the surgical area was compared with the adjacent tissue.
Thickness of the palate was measured using a sterile Endodontic k-file with a silicon disk stopper.
Patient Satisfaction Analysis
At baseline, 10 days, 3 weeks, and 12 weeks, patient satisfaction concerning comfort, hypersensitivity, and esthetic appearance was assessed using VAS.
Patient Comfort Score: Patients were inquired about their experiences with pain, edema, and other conditions related to the technique and instruments used during surgery to assess PCS. A VAS scale was used to rate the reported discomfort, with the two extremes being “unbearable discomfort” (score 10) and “no discomfort” (score 0). It was reported at baseline within 24 h of the treatment. 13
Patient Appearance Score: Patients’ responses were scored as “score 10” for unpleasing look to “score 0” for pleasing appearance to assess the PAS concerning the appearance, color, and contour-form of the selected site.
Statistics
Descriptive statistics were used to assess the results and make comparisons between the groups. Quantitative data were summarized as mean ± SD, while discrete data were expressed as proportions and percentages (%). The intra-class coefficient (ICC) analysis was used to determine the intra-observer agreement. Analysis was done using the SPSS software (Windows version 17.0). Shapiro–Wilk’s test of normality was used for normality testing of the data for all the parameters. The arithmetic mean, the standard deviation (δ), the Chi-square test, and the Student’s unpaired t-test were used to compare the equality of means of the same parameter between two independent groups. The null hypothesis of the test was that no significant difference existed between the means, whereas the alternative hypothesis was that there is a significant difference (two tails) or that one group has a higher mean than the other (one tail test). A two-sided (α = 2) p < .05 was considered statistically significant.
Results
For this study, 42 patients (23 males and 19 females) agreed to participate and finish the study after undergoing phase I therapy. Among the 21 patients (12 males and 9 females) recruited in Group A (with PRF), four patients (one patient shifted to another place and three were refused for follow-up) failed to complete the study, and among 21 patients (14 males and 7 females) recruited in Group B (without PRF), four patients lost follow-up and refused to come. Henceforth, a total of 34 patients (17 patients in each group) completed the study uneventfully and were finally analyzed.
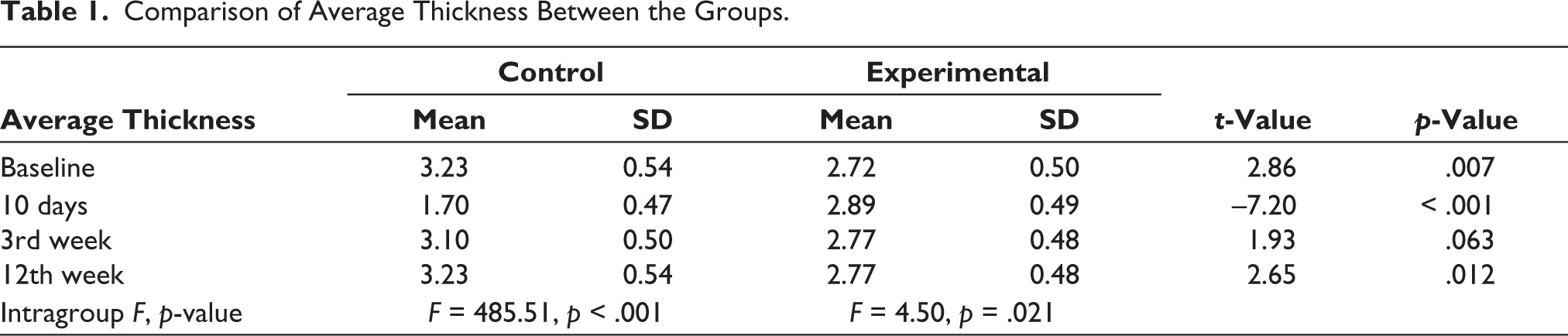
At baseline, there was a significant difference in mean thickness between the groups (p = .007), 10 days (p < .001), and 12 weeks (p = .012), whereas no significant difference was found at 3 weeks (p = .063) (Table 1). Intergroup comparisons were done by unpaired t-test. The intragroup comparison of average thickness using repeated-measures ANOVA revealed that significant changes occurred in both the control and experimental groups (p < .001 and p = .021, respectively). However, more changes were seen in the control group (with an F value of 485.51) in comparison with the experimental group (F = 4.50) (Table 1).
Comparison of Average Thickness Between the Groups.
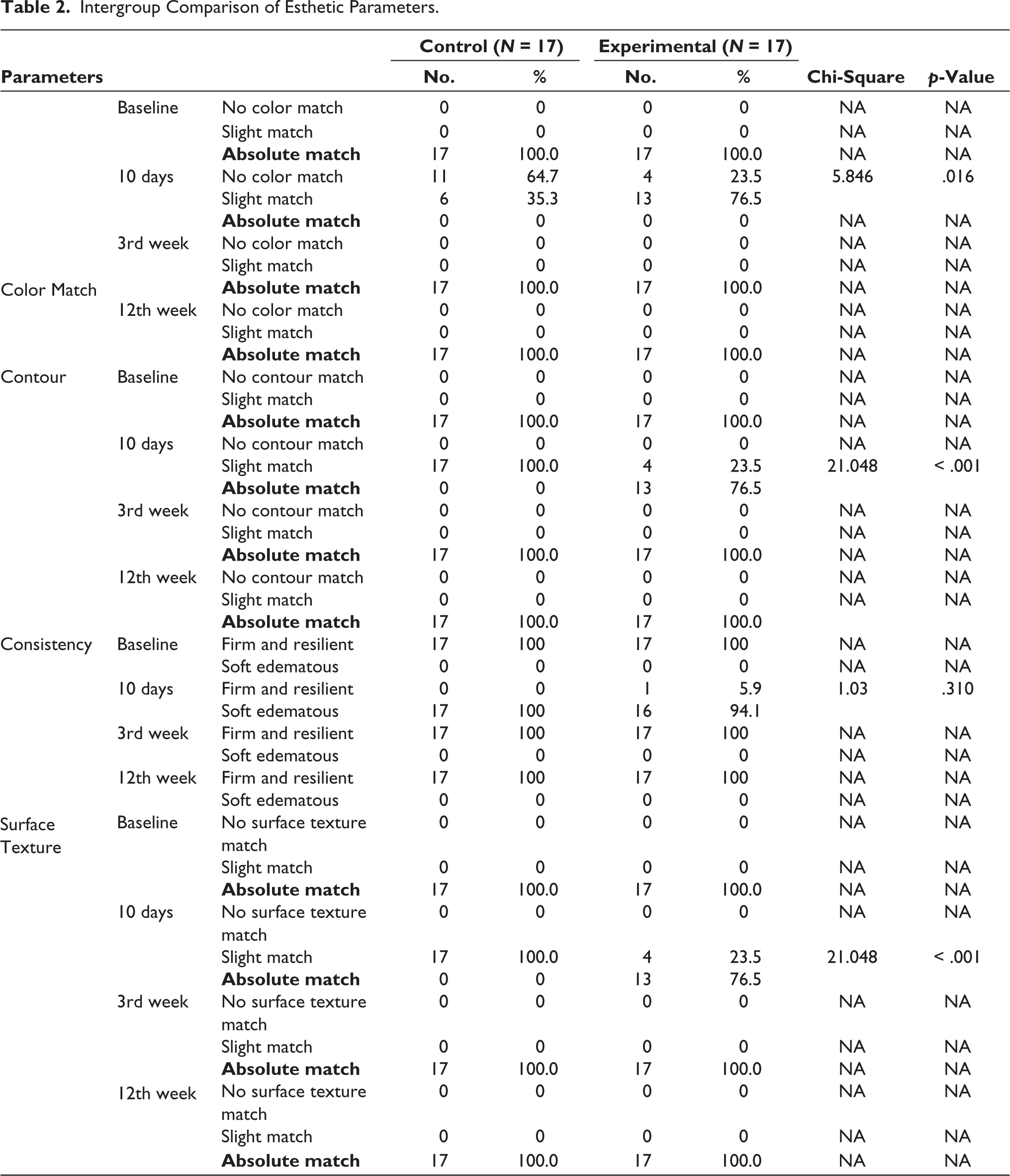
At baseline, the intergroup comparison revealed an absolute (100%) match for all esthetic parameters (consistency, contour, surface texture, and color) in both groups. At 10 days, 64.7% in the control group and 23.5% in the experimental group showed no color match. Notably, 76.5% of contour and surface texture showed absolute match and 5.9% showed firm and resilient consistency in the experimental group (Table 2). A total of 35.3% of subjects showed slight color match, and all showed slight contour and surface texture match and soft and edematous consistency in the control group, whereas 76.5% color, 23.5% contour, and surface texture showed slight match and 94.1% showed soft and edematous consistency in the experimental group (Table 2). The difference in the proportion of color, contour, and surface texture between the control and experimental groups was statistically significant in all esthetic parameters, except consistency, in which it was insignificant (p = .310) (Table 2). At 3 and 12 weeks, both groups showed 100% absolute match in all esthetic parameters.
Intergroup Comparison of Esthetic Parameters.
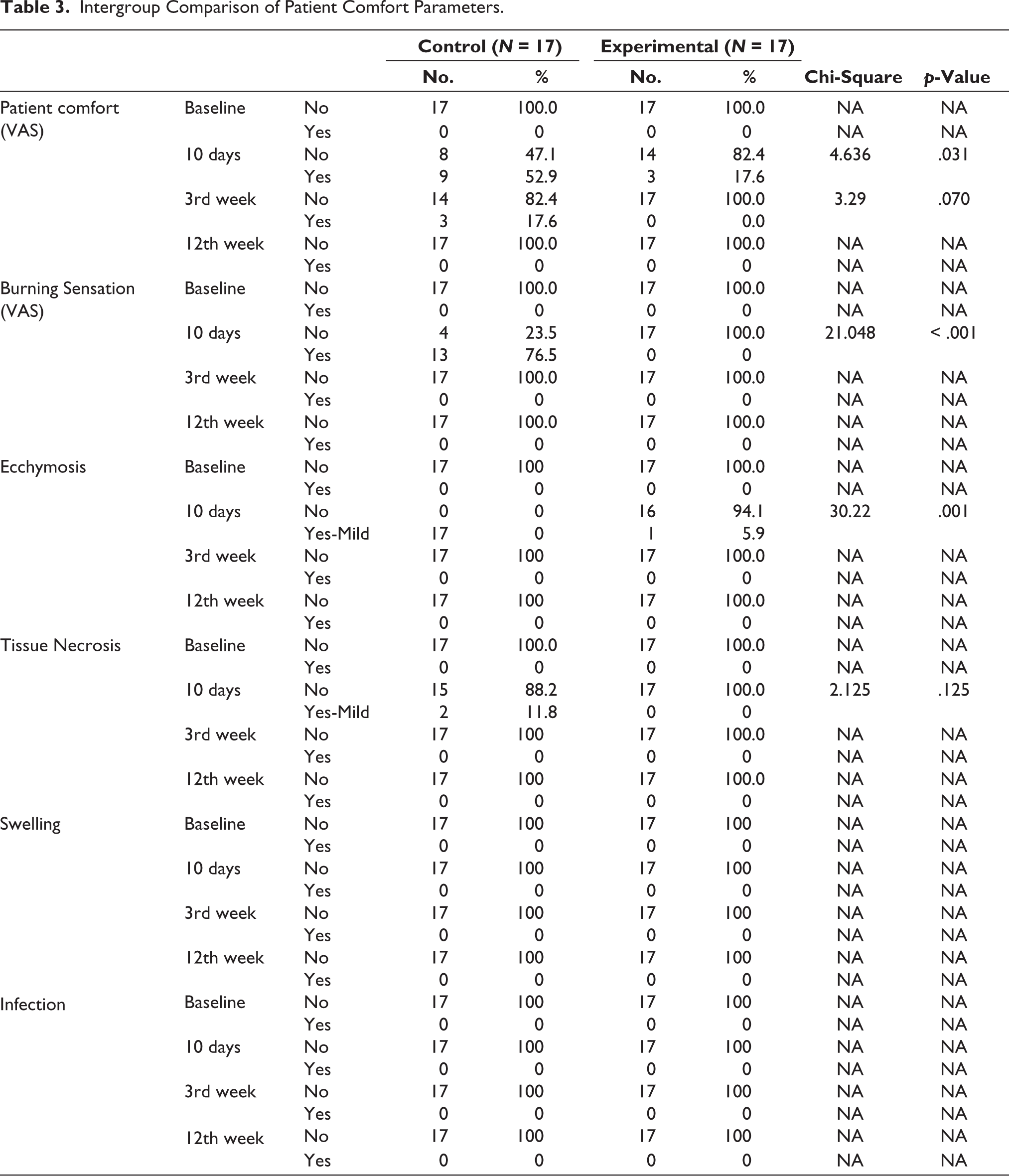
The intergroup comparison of patient comfort between the control and experimental groups revealed 100% patient comfort, no burning sensation, no ecchymosis, no tissue necrosis, and no swelling and infection in all subjects of both groups at baseline. At 10 days, 52.9% of subjects had discomfort, 76.5% had a burning sensation, 100% showed mild ecchymosis, and 11.8% showed mild tissue necrosis in the control group, while 17.6% of subjects had discomfort, none had burning sensation or tissue necrosis, and 5.9% showed mild ecchymosis in the experimental group. The difference in the proportion of patient comfort between the control and experimental groups was statistically significant (p < .001) in all these parameters except for patient comfort at 3 weeks (p = .070) and tissue necrosis at 10 days (p = .125) (Table 3). At 3 and 12 weeks, both groups showed no burning sensation, no sign of ecchymosis, and no tissue necrosis, whereas 17.6% showed discomfort in the control group at 3 weeks and none at 12 weeks. In intergroup comparison, no sign of swelling and infection was observed in both groups in any subject at baseline, 10 days, 3 weeks, and 12 weeks.
Intergroup Comparison of Patient Comfort Parameters.
Discussion
Primarily, the outcome of this study revealed that PRF when used at the surgical site after harvesting CTG resulted in faster and complete wound closure as compared with the non-PRF (control)-treated sites which, in turn, results in less patient discomfort and pain. Results of this study revealed that early healing with less inflammation could be seen in Group A (PRF) as compared with Group B (control) resulting in a significantly larger proportion (76.5% versus 35.3%) of “slight-color matched” than in Group B (Control). Similarly, Yen et al. 1 also reported that platelet concentrate-treated sites showed faster healing in terms of color match. However, at 3 and 12 weeks, 100% “absolute color match” with the adjacent tissue was observed among the participants of both groups, revealing almost complete healing of the palatal donor site after 3 weeks regardless of experimental materials.
At baseline, 10 days, 3 weeks, and 12 weeks, the contour of the palatal donor site was compared with that of the surrounding tissue and the opposite side of the non-surgical area. Results showed that, at 10 days, there was a “slight contour match” in 100% of subjects of Group B (Control) and 23.5% of subjects of Group A (PRF). However, an “absolute contour match” of the healing lesion to adjacent mucosa was observed in 76.5% of Group A (PRF), and in contrast to it, no absolute contour match was harmonized well with the adjacent non-surgical tissue in Group B (Control) at 10 days. The results of this study were as per the study done by Cheung and Griffin 13 who after 8 months found that palatal wound healing showed better tissue contour when platelet concentrate graft (PCG) was used.
Consistency match at baseline showed 100% firm and resilient match in both the groups on intergroup comparison, and 10 days results revealed “soft and edematous” consistency in 100% subjects of Group B (Control) and 94.1% subjects of Group A (PRF), whereas none of the patients in Group B (Control) and 5.9% in Group A (PRF) showed “firm and resilient” consistency of healing lesion when compared with adjacent mucosa. In the 3rd and 12th weeks, both the groups showed absolute consistency match. Similar results were shown by Shanmugam et al. 14
For surface texture of the palatal donor site, the results of this study revealed that, at 10 days, there was a “slight match” of surface texture in 100% of subjects of Group B (Control) and 23.5% of subjects of Group A (PRF). However, the absolute match of the surface texture of healing lesion to adjacent mucosa was 76.5% in Group A (PRF), and in contrast, no absolute match was found in Group B (Control). In concurrence with this study, Cheung and Griffin 12 also demonstrated that PCG resulted in more natural appearance in surface texture and this might be due to accelerated early wound healing.
In this study, posterior palatal mucosal thickness was assessed at six points, similar to Wara-aswapati et al. 15 and Khatri et al., 16 such as, MeL, ML, DL, MeM, MM, and DM. A prepared clear acrylic stent was fabricated to ensure consistent locations for the repeated assessments of mucosal thickness by bone sounding following the administration of local anesthesia using a sterile Endodontic K-file (No. 20) with a silicon disk stopper. The recordings were rounded off to the nearest millimeter and averaged. A similar gingival thickness evaluation of posterior palatal mucosa was done in previous studies.16,17 Other studies have used different methods to measure the gingival thickness of posterior palatal mucosa, Zucchelli et al. 2 used a short needle for anesthesia and a 3-mm-diameter silicon disk stop, whereas Schacher et al. 18 used cannulas and probes for the same.
Results of this study revealed mean higher values of mucosal thickness at the healing site after 10 days in Group A (PRF) as compared with Group B (Control) sites. This may be related to the fact that PRF produces large amounts of primary growth factors during the first 7 days. Lektemur-Alpan and Torumtay-Cin, 19 Gusman et al., 20 and Basma et al. 21 also observed better healing properties with PRF. Within the group also, there was a statistically significant higher mucosal thickness in Group A (PRF) as compared with statistical decrease in Group B (Control) after 10 days. After harvesting CTG, Yen et al. 22 investigated the effect of platelet concentrate on palate donor site healing at clinical and histological levels and observed that platelet concentrate may expedite soft tissue healing and palatal tissue thickness regeneration at 6-week intervals. According to Donn, 23 the palatal donor site will not experience any depression following the healing of the palate after 1 month, making it possible to approach the area once more to harvest more connective tissue.
To evaluate patients’ comfort (responses to pain and burning sensations), the VAS scale was used. To the utmost surprise, no participants in Group A (PRF) reported a burning sensation after 10 days as compared with 76.5% of participants in Group B (Control). The results observed may be attributed to early healing and anti-inflammatory properties of PRF used as healing gel in Group A (PRF). The continuous polymerization mechanism of coagulation in PRF promotes the assimilation of circulating cytokines into fibrin mesh (intrinsic cytokines), which aids wound healing by reducing inflammation. 24 After 3 months, none of the participants reported a sensation of pain at the surgical site in both groups. Results revealed that only 5.9% of participants reported signs of ecchymosis at the surgical donor site as compared with 100% of participants in Group B (Control) after 10 days. No sign of ecchymosis was observed after the 3rd and 12th weeks in either of the groups.
This study has limitations, including small sample size, short follow-up, and no histological examination of the palatal donor site healing. Furthermore, partial and complete survival of the graft could not be assessed because of the presence of periodontal dressing at both donor and recipient sites during the first 10 days.
Conclusion
Considering the constraints of this randomized clinical study for the palatal donor site, it can be concluded that, after 10 days, esthetic parameters, such as, color, contour, consistency, and surface texture revealed that a “slight-color match” was found higher in 76.5% of participants of Group A (PRF) as compared with 35.3% of participants of Group B (Control) with the adjacent tissue of the healing lesion. The mean thickness of palatal mucosa at the surgical site was increased after 10 days in Group A (PRF) as compared with the decrease in Group B (Control), and the mean difference was significantly higher for Group A (PRF) as compared with Group B (Control). Patients’ response and acceptance of the surgical treatment modality in terms of PCS and PAS showed less discomfort and pleasing appearance in Group A (PRF) after treatment as compared with Group B (Control).
Footnotes
Authors Contributions
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consents
After approval from the Institutional Research & Development Committee and Institutional Human Ethics Committee (IHEC) (SDC/IHEC/2016/MDS/P-26 dt.20/01/2017), the study procedure was executed following the ethical guidelines outlined in the Declaration of Helsinki 1998, as revised in 2013. All acquiescent patients received a verbal explanation of the study’s methodology and signed written informed consent before proceeding with the trial.
Funding
Infrastructure support for the research was provided by the Saraswati Dental College & Hospital, Lucknow, India.