Abstract
Aim:
Oral candidiasis is a fungal infection of the oral cavity caused by Candida albicans. Currently used antifungal drugs fail to treat oropharyngeal candidiasis. Herbal remedies are found to be safe and effective to treat bleeding gums, halitosis, mouth ulcers, and reduce tooth decay. Cardamom is known to possess antimicrobial, antioxidant, and immunomodulatory activity. In an in vitro study, we aim to evaluate the antifungal potential of cardamom-based DenteezTM mouthwash against opportunistic fungal infections in the oral cavity.
Materials and Methods:
Five Candida species were included in the study: namely C. albicans, C. tropicalis, C. krusei, C. parapsilosis, and C. glabrata. Additionally, 54 clinical isolates obtained from oral cavity of healthy individuals and patients suffering from oral candidiasis were randomly selected and included. Various antifungal agents tested included fluconazole, itraconazole, ketoconazole, miconazole, and amphotericin B. Minimum inhibitory concentration (MIC) and time-kill assay were performed as per standard guidelines.
Results:
The MIC obtained for fluconazole against different Candida species was 1.25 µg/ml. The MIC of azole drugs was in the range of 0.312–0.62 µg/ml. The MIC of DenteezTM mouthwash was determined as 1.25%. The clinical isolates of Candida showed major resistance to fluconazole (75.9%) and absolute susceptibility to amphotericin B. DenteezTM was found effective in inhibiting 98.2% of the clinical isolates. DenteezTM mouthwash showed time-dependent inhibition of all Candida species.
Conclusion:
The mouthwash has comparable efficacy to that of other routinely used antifungal agents. DenteezTM can be effectively used to deal with oral candidiasis and antifungal resistance.
Introduction
Oral candidiasis is a fungal infection of the oral cavity caused by Candida albicans. Invasive candidiasis is the leading cause of mycosis-associated mortality. Immuno-logical responses in healthy individuals often do prevent further damage and growth of C. albicans. Factors such as improper oral hygiene, immunosuppression, poorly adapted dentures, and consistent use of certain oral medications can lead to higher incidences of candidiasis. 1 C. albicans and other fungi associated with candidiasis are prevalent in those who use daily inhalers for long duration. Clinical signs of oral thrush are white patches that surround the surfaces of the buccal mucosa, throat, tongue, and gum linings. 2 Proper oral hygiene is recommended that include brushing teeth and flossing. The conventional treatment plan for dealing with oral Candidiasis includes the use of antifungal agents such as azole and polyenes. Frequently, Candida biofilm antifungal resistance in these long-term steroid users is due to exopolymeric matrix, increased cell density, metabolically inactive cells, and up-regulation of efflux pumps.3-5 Such biofilms are mostly resistant to azoles and amphotericin B. 6 Often prevalent in the oral cavity, Candida species including C. glabrata and C. krusei are also discovered to be resistant to azole medications. 7 Although itraconazole and fluconazole are frequently used, particularly for individuals with underlying systemic illnesses, infection resurgence is a typical clinical concern. 8 Thus, antifungal resistance is becoming an increasing problem with the Candida species making them very difficult to treat. This has resulted in widespread attention among researchers and professionals in oral health to conduct research on the possible antifungal properties of plant-derived material.
Mouthwashes can be beneficial having antimicrobial, antibacterial, and antifungal activity. Commercial mouthwashes containing chlorhexidine digluconate and cetylpyridinium chloride decrease the ability of C. albicans to spread in the oral cavity and provide positive balance for oral health.
But, commercial mouthwashes have certain negative effects, including tooth discoloration, altered taste, and increased supragingival calculus formation.9-11 Herbal medicines with their naturally occurring ingredients can be a potential and secure way to maintain the oral fitness and also address the concern of antifungal resistance. With oral health being the most important factor for maintaining the overall wellbeing, it certainly becomes necessary to restore the health by the most safer and reliable way.
Scientific studies have shown that natural remedies including Triphala, Tulsipatra, Jyestiamadh, Neem, Clove oil, Pudina, Ajwain, and many more are both safe and effective for treating a variety of oral health issues such as bleeding gums, halitosis, mouth ulcers, and reducing tooth decay. 12
Cardamom can be explored for its possible antifungal efficacy and provide health benefit over other commonly used antifungal agents. Oil extracted from cardamom seeds contains cineole as the active component that has strong antimicrobial, antioxidant, and also immunomodulatory activity.13,14 However, only few studies have been conducted to test for antifungal effects of a cardamom wash against oral thrush.
This study intends to add further knowledge on this by testing the effects of a cardamom-based DenteezTM mouthwash on oral thrush in vitro. DenteezTM mouthwash (Developed by Zum Heilen Diagnostic and Therapeutics, Thrissur, Kerala, India) contains 5% cardamom oil, glycerine, preservatives, sweetening, and flavoring agents. We aim to investigate the anti-fungal effect of DenteezTM mouthwash and assess its possible use in treating opportunistic fungal infection causing oral thrush in comparison to commonly used antifungal agents.
Materials and Methods
Fungal Strains and Growth Conditions
Five Candida species were included in the study, namely C. albicans ATCC 10231, C. tropicalis ATCC 13803, C. krusei ATCC 14243, C. parapsilosis ATCC 22019, and C. glabrata ATCC 15126. The strains were procured from IMTECH, Chandigarh, India. The strains were propagated on Sabouraud’s dextrose agar and incubated at 37°C in a bacteriological incubator. The isolated colonies were gram stained and further identified phenotypically by the combination of germ tube test, pigment produced on chromogenic medium, and carbohydrate assimilation test with various sugars. The concentration of confirmed colonies was adjusted by using 0.5 McFarland’s turbidity standard.
In addition, 54 clinical strains isolated from oral cavity of healthy as well as from patients suffering from oral candidiasis were randomly chosen and included in the study. Since the current study only includes stored clinical isolates, ethical approval as well as patient consent is not applicable to the study. The sample size of 54 was estimated based on the normal distribution with acceptable margin of error being 5% and confidence level at 95%.
Antifungal Agents
Along with cardamom mouthwash, the various antifungal agents tested included fluconazole, itraconazole, ketoconazole, miconazole, and amphotericin B. All antibiotics were procured from Sigma Aldrich-Merck, Bangalore, India. Amphotericin B was in injectable form. The stock solution of rest of the antifungal agents were prepared in their respective solvents and stored at -20°C in aliquots. The working concentration for fluconazole was 512 µg/ml and for all other antifungal agents was 160 µg/ml.
Minimum Inhibitory Concentration
Minimum inhibitory concentration (MIC) was carried out as per Clinical and Laboratory Standard Institute (CLSI) guidelines. 15 The test was performed in sterile U-bottom 96-well microtiter plate. To all the wells, 100 µl of RPMI 1640 medium with MOPS was added. The respective antifungal agent was added in a volume of 100 µl to the first well and then serially diluted to well number 2. After proper mixing, this procedure was repeated up to well number 10. This is followed by the addition of 10 µl of Candida suspension to the first 11 wells. Well number 11 served as growth control as it contained broth and organism. Well number 12 served as sterile control as it contained only medium. The microtiter plate was covered with lid and incubated at 37°C overnight. After the incubation, the fungal growth was recorded visually.
As per the guidelines for in-vitro susceptibility testing of Candida spp. and selected azoles, for fluconazole MIC of >8 µg/ml was considered as resistant, whereas for all other drugs MIC value of >1 µg/ml was considered as resistant. 15 Keeping this in mind, the concentration range for fluconazole was tested from 256 to 0.5 µg/ml, and for other drugs, it was tested from 80 to 0.15 µg/ml. The cardamom mouthwash (DenteezTM) was tested in the range of 5%–0.0075%.
Time-kill Curve
Time-kill assay was carried out by the broth microdilution method following the CLSI guideline. 15 Based on the MIC values obtained for DenteezTM that originally contains 5% cardamom, we further diluted the mouthwash and tested three different concentrations of the Cardamom mouthwash (2.5%, 1.25%, and 0.625%) against 5 Candida ATCC strains. The different time periods tested in the study were baseline (1 min), 5, 10, 20, and 30 min. In a set of three 1.5-ml eppendorf tubes, 250 µl of RPMI 1640 medium with MOPS was added to all the three tubes. To the first tube, 250 µl of 5% cardamom mouthwash was added and then serially diluted till tube number 3. Now, the tubes had 2.5%, 1.25%, and 0.625% of cardamom mouthwash, respectively. To each of these, 50 µl of standard Candida species was added. At each specified time interval, the suspension was mixed and plated onto Sabouraud’s agar plate. With each set, a broth control and a growth control were set up. The plates were incubated at 37°C in a bacteriological incubator. The plates were checked for growth and number of colonies from each plate was recorded and expressed as colony-forming units per ml (CFU/ml).
Results
In the present study, antifungal activity of 5% cardamom mouthwash (DenteezTM) was tested against five Candida species and 54 clinical isolates of Candida. The results were compared with those of 5 anti-fungal agents that included amphotericin B, fluconazole, itraconazole, ketoconazole, and miconazole.
MIC obtained for tested antibiotic agents and DenteezTM was as shown in Table 1. Most of the standard Candida species were inhibited by fluconazole at 1.25 µg/ml concentration. The MIC for other azole drugs was found in the range of 0.312–0.62 µg/ml. The MIC of DenteezTM was found to be 1.25% at which most of the standard Candida species were inhibited (Table 1).
MIC of Tested Antifungal Agents and DenteezTM Against Standard Strains of Candida Species.
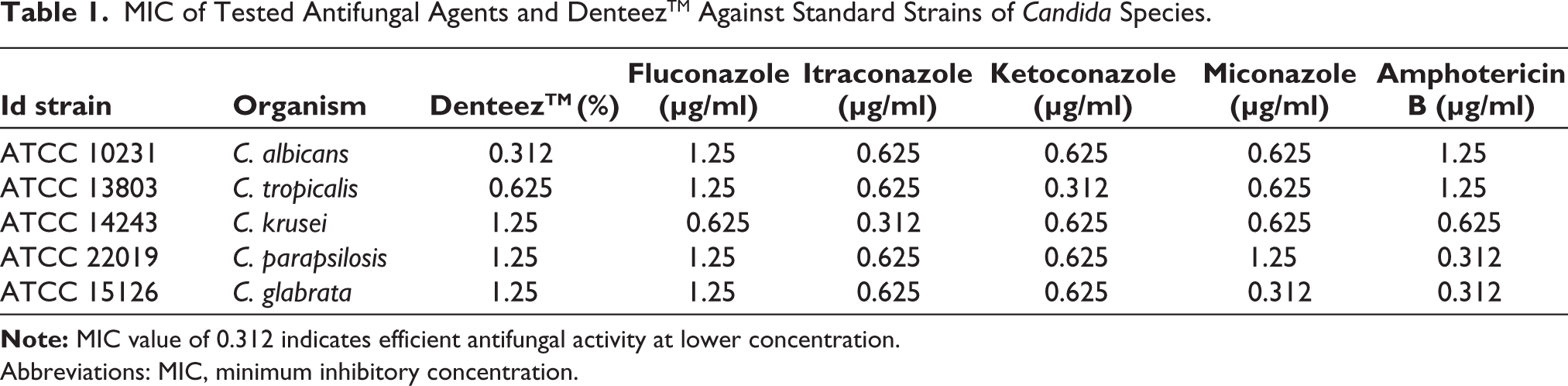
Abbreviations: MIC, minimum inhibitory concentration.
Among 54 oral Candida isolates, C. albicans was isolated in maximum frequency (29.62%) followed by C. krusei (25.92%), C. guilliermondii (12.96%), C. tropicalis (12.96%), C. kefyr (9.25%), C. glabrata (7.40%), and C. parapsilosis (1.85%). When DenteezTM was tested against these oral Candida isolates, it was seen that across the species the mouthwash showed inhibition at 0.312% or 0.625%. Only a few strains were inhibited at concentration of 1.25%. Our results show that among the Candida isolates, all were susceptible to amphotericin B and displayed a high level of resistance to fluconazole. There were 41 (75.9%) oral isolates that were found resistant to fluconazole. The clinical isolates were further found to be resistant to itraconazole (59.2%), ketoconazole (46.2%), and miconazole (42.5%) displaying the multidrug resistant pattern. Interestingly, almost all Candida species (98.2%) isolated from oral cavity were found to be susceptible to DenteezTM. A single oral isolate of C. krusei was found to be resistant to DenteezTM, but there were other 13 (92.9%) isolates of C. krusei, which were found susceptible to DenteezTM (Table 2). Among the clinical strains, total nine isolates showed multidrug resistant pattern that included C. albicans (4), C. krusei (3), C. parapsilosis (1), and C. kefyr (1).
The Data Show the Number and Percentage of Candida Strains Resistant and Susceptible to Various Antifungal Agents and 5% Cardamom Mouthwash (DenteezTM) Against 54 Oral Clinical Isolates of Candida Species.
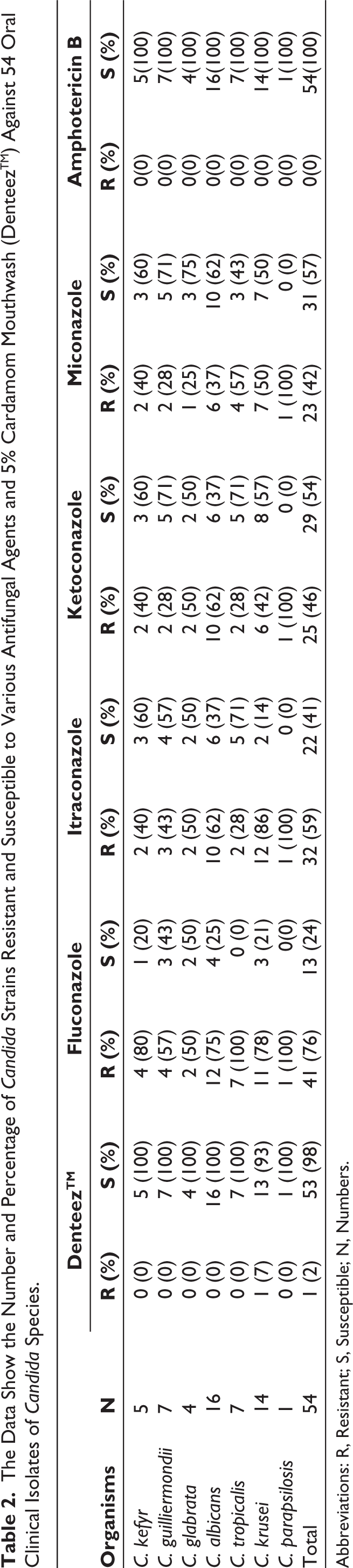
Abbreviations: R, Resistant; S, Susceptible; N, Numbers.
Time-kill assay was performed to determine the rate at which concentration of an anti-microbial agent kills a microbial isolate. There has to be a reduction of >30% in the number of colonies grown at any given concentration at a specified time period to infer that the product is effective. In the present study, it could be seen that when compared to the baseline, there is a drastic reduction in the growth at all time intervals with all the three concentrations of the cardamom mouthwash.
There has to be a reduction of organism growth by 3 log10 compared to growth control if the drug is effective at a particular concentration. 16 However, our results show that as the concentration of the mouthwash decreased, there was a slight increase in the number of colonies grown (mean number of colonies at all time intervals 11.4, 15.25, and 17.3 at 2.5, 1.25, and 0.625%, respectively) indicating efficacy of mouthwash was reduced as there was reduction in the concentration (Table 3).
Time-kill Assay Performed with DenteezTM 2.5%, 1.25%, and 0.625% Against Standard Candida Strains at Different Time Intervals.
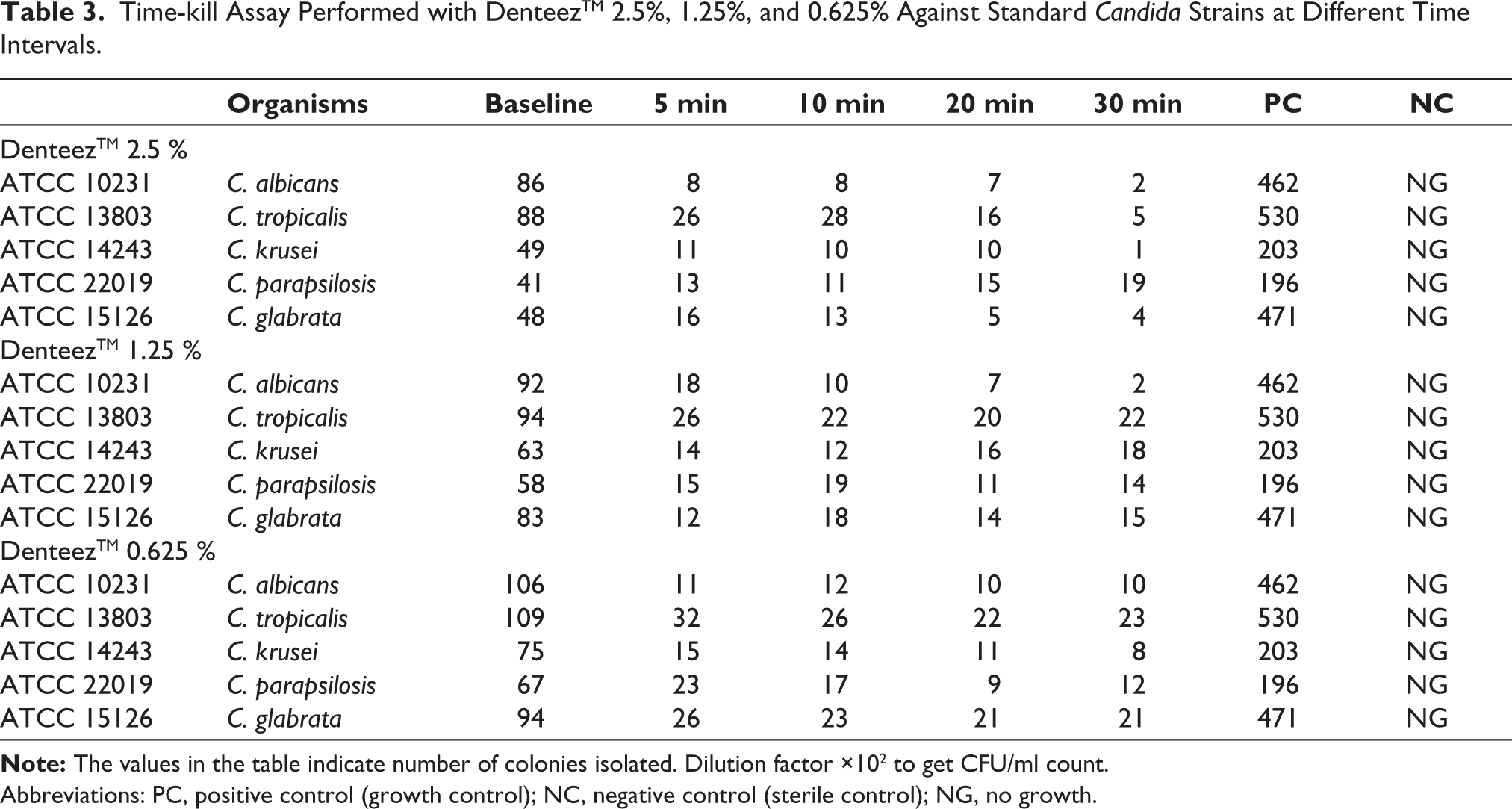
Abbreviations: PC, positive control (growth control); NC, negative control (sterile control); NG, no growth.
Discussion
Human fungi specifically identified as Candida species pose a multitude of recurring superficial mycoses mostly in the vaginal and oral mucosae. 17 The current antifungal drugs typically fail to treat oral or oropharyngeal candidiasis, especially when it affects immunocompromised patients. This has imposed a thrust for research to identify and develop new antifungal agents that possess less side effects, more safety, and easy accessibility. 18 Cardamom (Elettaria cardamomum) often referred as the “Queen of Spices,” a member of the Zingiberaceae family, has been shown to have a number of pharmacological activities.19–23 In an in vitro study, we aimed to evaluate the antifungal efficacy of cardamom-based DenteezTM mouthwash by means of MIC and time-kill curve assay.
MIC is the lowest antimicrobial agent concentration that, following overnight incubation, prevents the observable development of microbial cells. 15 In the present project, five species of Candida ATCC strains and 54 clinically isolated Candida species were included. These clinical isolates were C. albicans, C. kefyr, C. guilliermondii, C. krusei, C. tropicalis, C. glabrata, and C. parapsilosis. These strains were tested against various concentrations of cardamom mouthwash and the results were compared with those of five antifungal agents that included amphotericin B, fluconazole, itraconazole, ketoconazole, and miconazole.
The oral Candida isolates showed major resistance to fluconazole (75.9%) and considerable susceptibility to amphotericin B (100%). DenteezTM was found effective in inhibiting 98.2% of the clinical isolates and becomes potential agent to deal with oral candidiasis. The mouthwash showed promising results with majority of the Candida species showing inhibition at 0.312% and 0.625% concentration of the compound, though we suggest that a concentration of 1.25% would be comprehensive in inhibiting the growth of most oral Candida species.
We also performed time-kill assay by micro-broth dilution method by following the CLSI guidelines. 15 The results show that >30% of the colonies have been inhibited by DenteezTM at all tested concentrations and specified times indicating effectiveness of the product. There was time-dependent inhibition of all Candida species at 2.5%, 1.25%, and 0.625% of the cardamom mouthwash. The reduction of colonies at 0.625% is limited compared at 2.5% and 1.25% without any significant difference. Hence, it is relevant to assume that the mouthwash can be effectively used at a concentration of 1.25%.
There are very few studies that have reported the antifungal property of cardamom as a mouthwash. However, it was seen that cardamom is extremely beneficial in preserving oral health. 24 Cardamom improves periodontal health by anti-inflammatory and anti-bacterial activity. The NF-kB signaling pathway maintains chronic inflammation that was inhibited by cardamom preventing an increase in the levels of inflammatory markers. 25 The anti-microbial activity of cardamom extract was evaluated against dental caries causing organisms. 26 It was seen that cardamom is effective in inhibiting oral pathogens like Streptococcus mutans and C. albicans. The ethanolic extract of cardamom was found to possess antibacterial activity against S. mutans and Lactobacillus casei and reduces microbial viability levels of biofilm produced from saliva samples of pediatric patients. 27
According to a report, resistance to fluconazole causes cross-resistance to other azoles or there could be a pathogen switch from C. albicans to less susceptible species like C. glabrata and C. krusei. 28 It was found that fluconazole resistance in C. glabrata is about eight times higher than in C. albicans, and it can quickly increase with continued use of the medication. 29 In addition, different isolates of the same species from different sources have been identified to have dissimilar fluconazole susceptibilities. 30 In the clinical setting, this has primarily led to complex diagnosis and treatment. Therefore, precise species-level identification and susceptibility testing are essential for the clinical management.
The results from our study show that cardamom-based DenteezTM mouthwash is effective in treating fungal infections and is as effective as other routinely used antifungal agents. This has also provided safer and more potential alternative to maintain the oral health and manage the concern of antifungal resistance in oral candidiasis.
Overtime, our understanding of how the opportunistic pathogen Candida colonizes humans has grown, and it is now widely known that Candida is frequently found in respiratory specimens. 31 There are also evidences of oral thrush being majorly detected in patients with asthma and chronic obstructive pulmonary disease (COPD). C. albicans, the main fungus responsible for oral thrush, appears to be more prevalent in such communities of people who use inhalers for long period of time. 32 Cardamom mouthwash has a substantial antifungal effect, which suggests that people who frequently use inhalers can be benefitted from its use. Additional investigation in this area is required using oral samples from these patients, which can mostly support our findings.
Current study involved 54 clinical isolates collected from a local population. Larger study design with clinical isolates collected from wide geographical location needs to be studied. Also, the comparison of DenteezTM mouthwash with other commercially available mouthwashes is recommended. Furthermore, our research was conducted exclusively in vitro, and to determine the efficacy and acceptability of this mouthwash in vivo, suitable clinical trials are required.
Conclusion
Our study demonstrates that Cardamom-based mouthwash, DenteezTM, possesses potent anti-fungal property and there is time-dependent inhibition of Candida species at all tested concentrations of mouthwash. The mouthwash has comparable efficacy to that of other routinely used antifungal agents. Concentration of 1.25% found to be effective in inhibiting the standard Candida species and all oral isolates of Candida. Cardamom mouthwash (DenteezTM) can be effectively used to deal with oral candidiasis and antifungal resistance. As the research was conducted exclusively in vitro, the results may differ in clinical settings and require suitable in vivo studies to confirm the outcomes.
Footnotes
Acknowledgements
The authors would like to thank Dr. Ramakant Nayak, Principal, Maratha Mandal’s NGH institute of Dental Sciences and Research Centre, Belagavi, Karnataka, India for allowing us to conduct this study.
Author Contributions
Conception: MT-MK; Design: KB; Supervision: KB-PV; Data collection and/or processing: MJ-BN; Analysis and/or interpretation: MK; Literature review: MT-MK; Writer: MK-KB; Critical review: PV.
Data Availability Statement
The data of the study are available for access upon request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
The current study only includes stored clinical isolates, hence ethical approval as well as patient consent is not applicable to the study.
Funding
This research work has been funded by Zum Heilen Diagnostic & Therapeutics Pvt. Ltd, Thrissur, Kerala, India.
