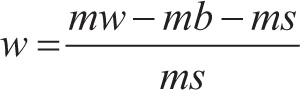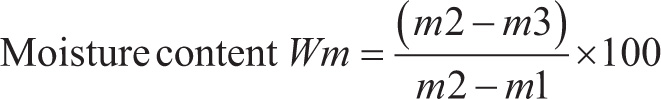Abstract
Aim:
Saliva is a significant hindrance to most dental procedures (e.g., root canal treatment) as the flow of saliva increases in patients undergoing dental treatment due to anxiety. Saliva absorption pads based on superabsorbent polymers can provide a dry oral environment to ease the dental treatment procedure and reduce the swallowing reflexes common during cotton use. This study focused on developing an indigenous saliva absorption pad using biodegradable superabsorbent polymer (BSAP) sponges.
Materials and Methods:
BSAP sponges were synthesized using carboxymethyl cellulose (CMC) as the base matrix. Different crosslinking mechanisms were implemented to prepare BSAP sponges, such as ionic crosslinking using aluminum ammonium sulfate (AlAS) and chemical crosslinking using methylene bisacrylamide. Three different BSAP sponges (Sap-2, SAP-PAA-1, and SAP-PAA-2) were characterized for their free swell capacity and thermal degradation kinetics along with other characterization techniques to optimize the composition for saliva absorption pad. One-way ANOVA was used for statistical evaluation.
Results:
SAP-2, synthesized using 10 wt.% AlAS showed the highest free swell capacity in water and saline (83.21 ± 3.8 g/g and 40.7 ± 3.4 g/g, respectively). However, the moisture content of the particular BSAP sponge was higher (~13%) compared to the standard limit (ISO 17190-4:2001(E)). It was observed that as the crosslinking density increases free swell capacity increases to a threshold point and decreases thereafter. As reported earlier, percentage swelling was controlled by multiple factors including crosslinking that opposes swelling and polymer/water interaction and Donnan pressure that promotes swelling.
Conclusion:
BSAP sponge based on crosslinked CMC matrix is highly advantageous in developing saliva absorption pad. Hydrophobic surface modification is recommended to reduce the moisture content to improve the storage stability of BSAP sponges.
Abbreviations
Aluminum ammonium sulfate
Biodegradable superabsorbent polymer
Carboxymethyl cellulose
Fourier transform infrared spectroscopy
Thermogravimetric analysis
High-pressure liquid chromatography
Superabsorbent polymer
Attenuated total reflectance
Introduction
Saliva is a major hindrance to most dental procedures (e.g., root canal treatment) as the flow of saliva increases in patients undergoing dental treatment due to anxiety and stimulations, and so on .1,2 Small absorption pads or cotton pieces are usually used to absorb saliva from the mouth of the patient during the dental procedure. These pads are typically positioned between the teeth and cheek or between the teeth or tongue and replaced when saturated. 3 Saliva absorption pads based on a superabsorbent polymer (SAP) sponge can provide a dry oral environment to ease the dental treatment procedure. They can reduce the swallowing reflexes commonly seen during cotton roll isolation.4–7 Commercially available SAPs are often synthetic resins that are mainly polymerized using acrylic/vinyl monomers such as acrylic acid, acrylamide, vinyl alcohol, and acrylonitrile.8–12
Even though synthetic SAP currently available in the markets are biocompatible and have no direct threat to human life, disposal of such non-degradable material waste is a source of various environmental problems.13–15 Therefore, natural polymers-based SAPs have received significant attention as they are degradable via multiple mechanisms of action, including enzymatic reactions, microbial attack, and hydrolysis.15–19 SAP based on natural polymers (e.g., cellulose,20–22 chitosan,23–26 starch,27–29 and alginate)30–32 are renewable, biodegradable, and non-toxic. However, the main challenge in the area of biodegradable SAP is to synthesize fully degradable SAP that would rapidly and reversibly absorb water and have good mechanical properties.33–37
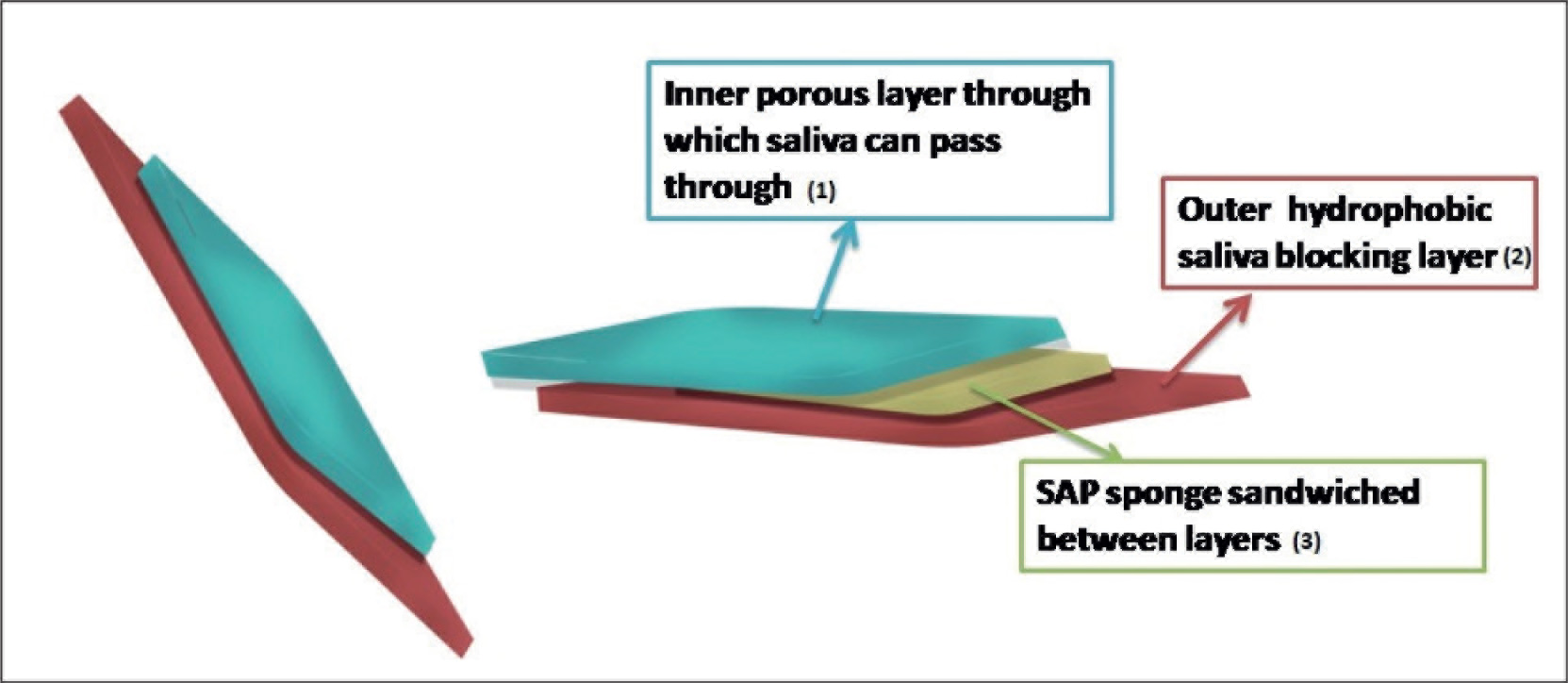
This study focused on developing an indigenous saliva absorption pad using biodegradable superabsorbent polymer (BSAP) sponges. Figure 1 represents the design of the saliva absorption pad used in the study. BSAP sponges were synthesized using different polysaccharides including carboxymethyl cellulose (CMC) as the base matrix. CMC is anionic water-soluble cellulose commonly known as cellulose gum, which is approved by the FDA as a food additive (
Schematic Representation of 3-D View of Saliva Absorption Pad.
Materials and Methods
Materials
Acrylic acid, sodium hydroxide, N,N'-methylene-bis-acrylamide, and ammonium persulfate were purchased from Sigma-Aldrich, Bangalore, India, and used without further purification. CMC (1200–1600 cps) and sodium chloride were purchased from Nice Chemical Private Limited, Cochin, India. AlAS dodecahydrate and aluminum sulfate were purchased from Merck Life Sciences, Bangalore, India. All other reagents were of analytical grade and used as such without any further purification.
Preparation of SAP Sponges
Different batches of BSAP sponges were prepared using CMC as the starting material. In the first batch, BSAPs were synthesized using CMC, and acrylic acid at different molar ratios (i.e., 80/20, 90/10) via free radical polymerization using ammonium persulfate (2 wt.%) as the initiator and 0.2 mol% methylene bisacrylamide as a crosslinker. The reaction mixture was stirred at 70oC for 2 hours using a magnetic stirrer equipped with a hot plate (SCOTT, D-55122 mainz). After 2 hours, hydrogel formed was transferred to a petri dish and freeze-dried using a lyophilizer (CHRIST, Alpha 1-4 LD) for 48 hours. The corresponding sponges were denoted as SAP-PAA-1 and SAP-2. In the second batch, BSAP sponges were prepared using CMC and different weight ratios of AlAS (i.e., 2 wt.%, 5 wt.%, 10 wt.%, and 15 wt.%). Briefly, 2% CMC solution (100 mL) was prepared in distilled (DI) water and stirred mechanically at 1000 rpm for 30 min at room temperature. To the homogenous CMC solution, 100 mL of AlAS solution at a concentration of 50 mg/mL in DI water was added at a stretch. Continued the stirring for 2 hours at 1000 rpm, and the reaction product (hydrogel) was transferred to a petri dish and freeze-dried using a lyophilizer (CHRIST, Alpha 1-4 LD) for 48 hours. The corresponding sponge was denoted as SAP-2.
Evaluation of Residual Monomer Content
Residual acrylic acid was extracted from 1 g of BSAP sponge using 200 mL of 0.9% saline. The solution was stirred at 500 rpm for 60 min and allowed to settle for 5 min. The supernatant was filtered through 0.45 µm filter and analyzed using HPLC (HPLC; Shimadzu, Spincotech Pvt. Ltd., Tamil Nadu). Mobile phase: 1% acetonitrile–phosphoric acid solution (10:90) was used as the mobile phase at a flow rate of 0.8 mL/min using a UV detector associated with Shimadzu HPLC at 210 nm.
FTIR Spectroscopy Studies
Infrared qualitative analysis was carried out and transmission spectra were collected in the range of 4000–400 cm–1 to confirm the complete conception of acrylic acid during polymerization using Nicolet 5700 FTIR Spectrometer in attenuated total reflectance (ATR) mode.
Thermogravimetric Analysis
The TGA of the dried BSAP sponge was carried out on an SDT Q600 (simultaneous TGA-DTA, TA Instruments) in the temperature range of RT to 800°C at a scan rate of 10°C/min under a nitrogen atmosphere.
Free Swelling Capacity
About 200 mg of BSAP sponges was weighed and transferred to a heat-sealable bag. The sponge is positioned horizontally in the bag and laid on the surface of the saline solution or DI water along with two blank bags. Allowed each bag to wet for 1 min and pushed it under the liquid surface. Bags were removed from the saline solution after 30 min and hung diagonally on a line from one of the double-sealed corners for 10 ± 1 min. Weighed each bag using a microbalance (Kern Analytical Balance, ABT 120-4NM) and recorded the mass. Experiments were repeated quadruple. Free swell capacity is expressed in (g/g) mass fraction
where ms, mass of dry SAP sponge; mb, average mass of the wet blank bag; and mw, mass of the wet bag containing SAP sponge.
Determination of Moisture Content by Mass Loss upon Heating of SAP Sponge
Before starting the experiment, four petri dishes and lids were placed into the oven at 105°C for 3 hours, according to the standard. After cooling for 30 min, weighed the empty lidded petri dishes in grams (m1). Removed the lid and added approximately 0.2 g of SAP sponge powder to each petri dish. Replaced the lid and weighed the lidded dish containing the sample immediately (m2). Placed open dishes with SAP sponge and lids together in a hot air oven at 105°C. After 3 hours, immediately lidded the dish and placed in a desiccator, and allowed to cool for 30 min. Removed and weighed immediately (m3). Experiments were done in quadruplicate.
Results
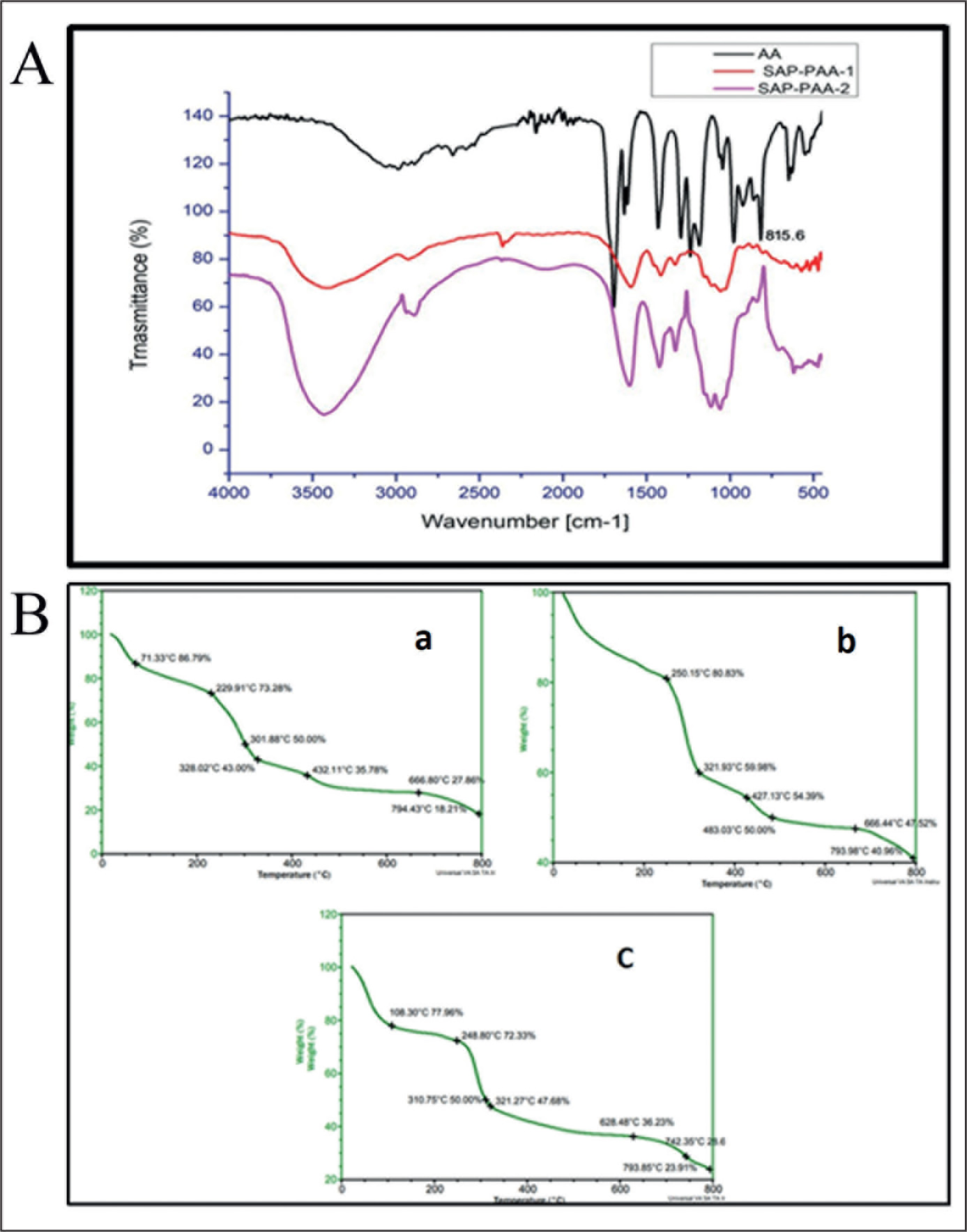
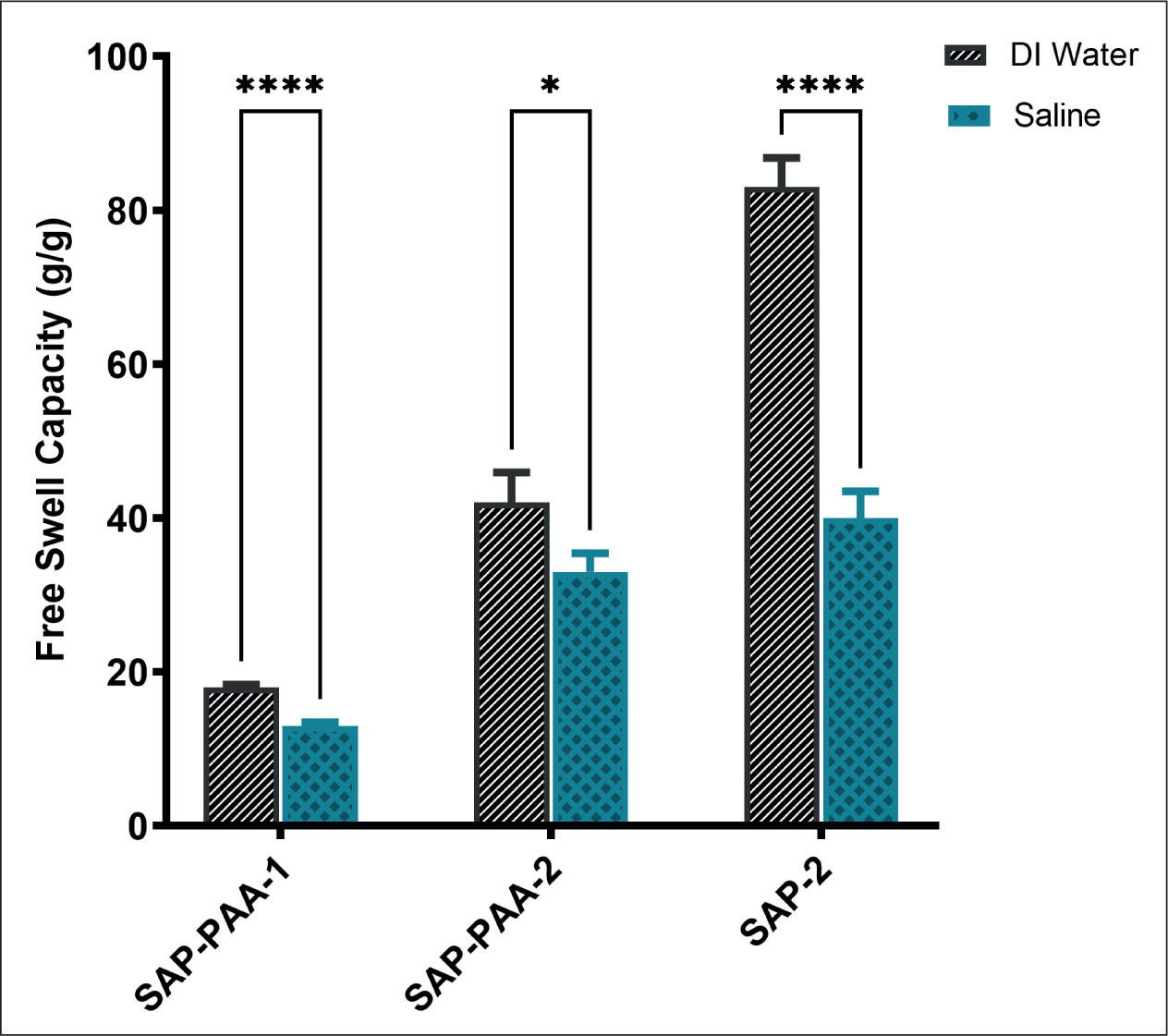
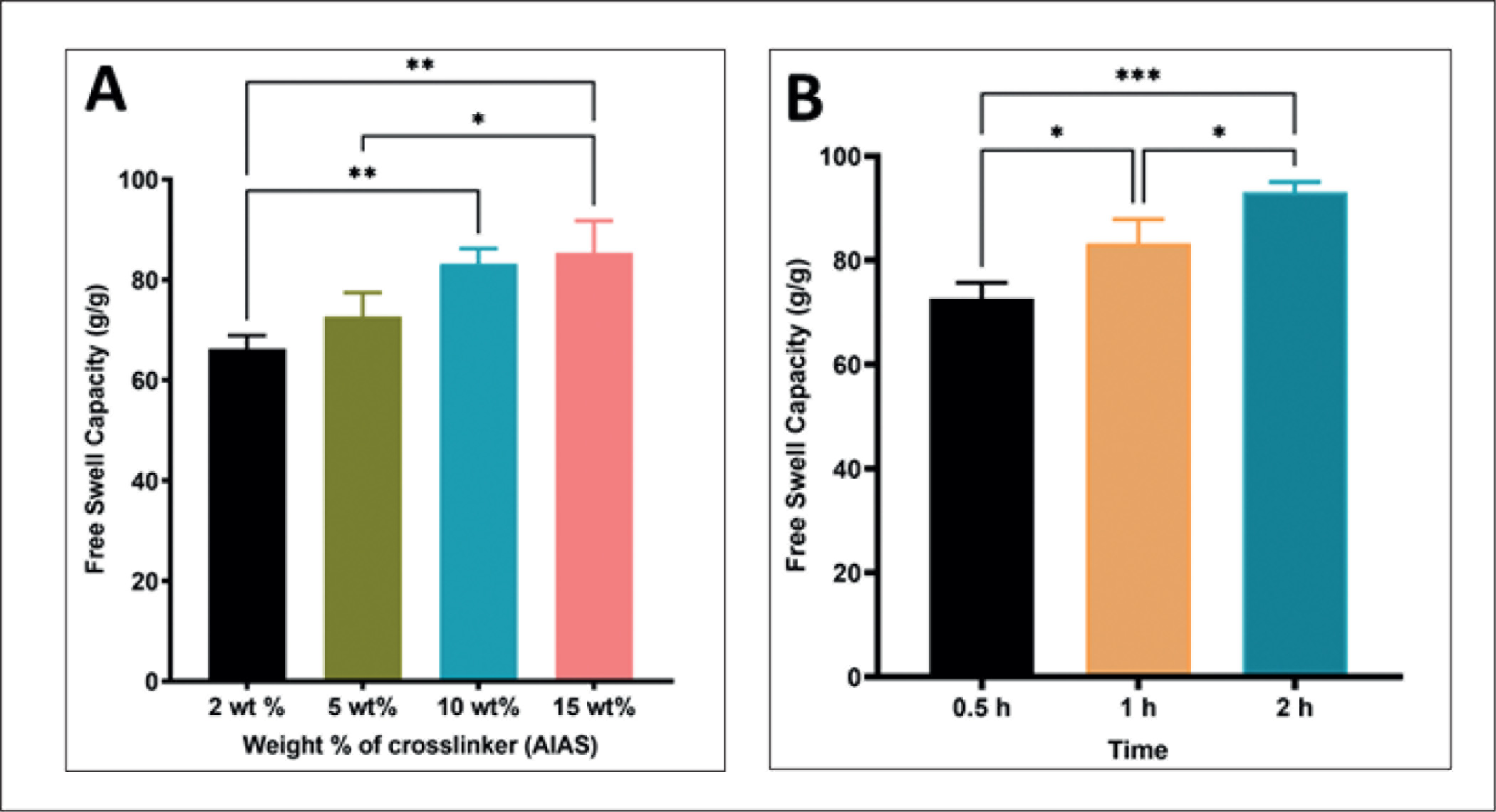
The BSAP sponges were prepared and analyzed using various physiochemical characterization techniques. The residual monomer content of SAP sponges was analyzed using HPLC. The mass fraction of residual acrylic acid was 2.3 ± 0.05 g/kg and 3.05 ± 0.03 g/kg, respectively, for SAP synthesized using 10 mol% and 20 mol% acrylic acid. SAP synthesized using 10 mol% and 20 mol% were designated as SAP-PAA-2 and SAP-PAA-1, respectively. The FTIR spectra showed a broad absorption peak at 3200–3600 cm–1. The peak was attributed to the vast amount of carboxyl groups present in SAP. The absorption peaks at 1721 cm−1 were assigned to the -C=O stretching vibration due to the presence of carboxylate groups of acrylic acid (AA). An intense peak of =CH bending of acrylic acid was present at 815.6 cm–1, whereas no significant peak of =CH bonding was present in both SAP-PAA-1 and SAP-PAA-2 (Figure 2A). TGA of SAP-PAA-2, and SAP-2 showed a percentage weight loss of ~82% at 800°C, whereas in SAP-PAA-1, only 60% weight loss was observed at 800°C. In all three BSAP sponges, ~20% weight loss was observed around 100°C and a drastic weight loss after 300°C (Figure 2B). The free swell capacities of BSAP sponges were studied both in distilled water and 0.9% saline (Figure 3). It was observed that the free swell capacities of BSAP sponges in DI water were higher than that measured in saline. SAP-2 showed a free swell capacity of 40.7 ± 3.44 g/g in saline. Likewise, SAP synthesized via crosslinking CMC with acrylic acid and methylene bisacrylamide, SAP-PAA-2 and SAP-PAA-1 showed a free swell capacity of 13.48 ± 0.43 g/g and 33.78 ± 2.42 g/g, respectively, in saline. The free swell capacity of SAP-2 was reduced >50% in saline compared to that in DI water. The free swell capacity of SAP-2 in DI water was 83.21 ± 3.8 g/g, whereas a reduced free swell capacity was observed for both SAP-PAA-2 and SAP-PAA-1 in water, 42.4 ± 3.9 and 18.15 ± 0.35, respectively. The free swell capacity of SAP-2 increases with an increase in the weight percentage of AlAS (Figure 4A). However, no significant increase in swelling was observed beyond 10 wt.% AlAS. The SAP-2 at 10 wt.% AlAS was further characterized for its saturation swelling time in the water. An increase in free swell capacity was observed with time (Figure 4B). The saturation swelling was detected at 2 hours of incubation without damaging the integrity of BSAP sponges. The mass loss upon dehydration was measured after heating BSAP sponges at 105 ± 2°C for 3 hours. The average moisture content observed was 13.33 ± 2.35%.
(A) FTIR Spectra of Acrylic Acid (black line), SAP-PAA-1 (red line), and SAP-PAA-2 (Pink line). There is an Intense Peak of =CH Bending at 815.6 cm-1 in the Acrylic Acid Spectrum. Whereas No Significant Peak of =CH Bending was Present Both in SAP-PAA-1 and SAP-PAA-2 Indicating Almost Complete Consumption of Acrylic Acid During the Reaction. (B) TGA Curves of (a) SAP-PAA-2, (b) SAP-PAA-1, and (c) SAP-2 Indicating the Percentage Weight Loss at Different Temperatures up to 800oC.
Free Swelling Capacity Expressed in Mass Fraction g/g. Data Between Two Groups Saline and DI Water were Compared; P < .05.
(A) Free Swell Capacity of Carboxymethyl Cellulose-Based SAP Crosslinked Using Different Weight Ratio of AlAS (SAP-2) Expressed in Mass Fraction (g/g). (B) Free Swell Capacity of SAP-2 Cross-Linked Using 10 wt.% AlAS at the Different Swelling Times Expressed in Mass Fraction (g/g). Data Between Groups were Compared; P < .05.
Discussion
BSAP sponges were prepared using CMC by different crosslinking mechanisms, such as ionic crosslinking using AlAS and chemical crosslinking using N,N'-methylene-bis acrylamide. BSAP sponges having optimum characteristics (e.g., swelling capacity) and ease of production will be used for the development of a saliva absorption pad. Sponges were evaluated for their residual monomer content and chemical composition to know more about their stability and swelling characteristics. The residual monomer content was a key to the measurement of percentage polymerization and the reaction efficacy. As evident from the results, ~99.7% polymerization was detected in both SAP-PAA-2 and SAP-PAA-1. The results were confirmed using FTIR studies. No significant peak of =CH bending was present for both SAP-PAA-1 and SAP-PAA-2 indicating almost complete consumption of acrylic acid during the reaction.
Thermal decomposition studies were carried out to confirm the thermal stability of BSAP sponges. The reduced percentage weight loss of SAP-PAA-1 compared to SAP-PAA-2, and SAP-2 was mainly attributed to the presence of a higher percentage of synthetic counterpart, where 20 mol% of acrylic acid was copolymerized with CMC. In all these sponges, the weight loss at 100°C can be mainly due to absorbed moisture as BSAP sponges were highly hygroscopic. The pyrolysis followed by chain scission leads to a drastic weight loss beyond 300°C.
The free swell capacity of BSAP sponges was higher in distilled water compared to that in saline. High ionic concentration of saline reduces the osmotic pressure inside the crosslinked network (gel) and thereby less saline intake compared to that of distilled water. SAP-2 showed a higher free swell capacity among the group. In SAP-2, CMC was ionically crosslinked using 10 wt.% AlAS. The free swell capacity of SAP-2 was reduced >50% in saline compared to that in DI water. A simple reaction procedure that involved high-speed stirring of CMC and crosslinker solution at room temperature followed by freeze-drying leads to the formation of SAP-2. Considering the high swelling and simple preparation procedure, SAP-2 was studied further to optimize the crosslinker content and saturation swelling time. The free swell capacity of SAP-2 increased with an increase in the weight percentage of AlAS up to 10 wt.%. As reported earlier, swelling characteristics were controlled by multiple factors such as crosslinking that opposes swelling, and polymer/water interaction and Donnan pressure that promote swelling. 41 An optimum crosslinking would prevent the dissolution of hydrophilic polymer chains in an aqueous solution. The SAP-2 at 10 wt.% crosslinker content was further characterized for its saturation swelling time. An increase in the free swell capacity of BSAP sponges was detected with time. The swelling was saturated at 2 hours of incubation in DI water. Integrity of these BSAP sponges was retained even at its saturated swelling. High moisture content of SAP-2 sponges (≥5%) demand chemical modifications to improve their storage stability. Even though simple ionic crosslinking mechanism improved the free swell capacity of these sponges in saline, further modifications are still preferred to improve its free swell capacity in saliva which is more viscous and contains multiple components, such as water (>99%), electrolytes, mucus, enzymes, and antimicrobial agents, compared to saline.
Conclusion
BSAP sponges were prepared using CMC as the main polymer matrix to develop an indigenous saliva absorption pad. The free swell capacity of SAP sponges in DI water and saline was evaluated. Data showed a high absorption capacity for SAP-2 sponges (83.21 ± 3.8 in DI water and 40.7 ± 3.4 in saline) in which CMC is ionically crosslinked using AlAS. As the moisture content of these SAP sponges was high (~13%) compared to the standard limit, surface modifications were recommended to improve the storage stability of these BSAP sponges.
Footnotes
Acknowledgements
The authors gratefully acknowledge Dr. Deepu D.R., Division of Dental Products, SCTIMST for HPLC analysis and Dr. Renjith S, Central analytical facility, SCTIMST for FTIR analysis.
Authors’ Contributions
MS conceived the idea, planned experiments, and contributed in writing and reviewing the manuscript. PS did experiments and contributed in writing the first draft of the manuscript. SL did swelling studies and helps in drawing schemes and graphs for the manuscript.
Data Availability Statement
All the raw data are available with the authors for any further clarification.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The study involved no cell/tissue culture studies, no animal studies, and no human subject/tissue/material/data.
Funding
The manuscript was a part of the M.Tech. thesis of (hidden for anonymity). Authors received no specific financial support for the research or publication of this article.
Informed Consent
Not applicable.