Abstract
Aim:
To evaluate the effects of papain (Brix 3000), bromelain, sodium hypochlorite (NaOCl), and chlorine dioxide (ClO2) application to the deep dentin surface on shear bond strength (SBS), microleakage, and dentin surface properties.
Materials and Methods:
Deep dentin surface (n = 100) for evaluating SBS, class V preparation at the buccal surface for testing microleakage (n = 100), and deep dentin slices (n = 20) for evaluating surface properties were conducted on the 220 molar teeth. Four different deproteinizing agents (Brix 3000, 40% bromelain, 5.25% NaOCl (Chloraxid), and 0.12% ClO2) were applied to the dentin, and then the universal adhesive was used in self-etch (SE) and etch&rinse (E&R). Deproteinizing agents were not applied to the control group. All of the samples were subjected to 5000 cycles of thermal aging at 5ºC–55ºC. SBS (MPa) was tested by a universal testing machine. The microleakage of resin composite bonded with different adhesive modes was evaluated under a stereomicroscope. The changes in the surface morphology were examined with scanning electron microscopy (SEM) and attenuated total reflection – fourier transform infrared spectroscopy (ATR-FTIR).
Results:
ClO2 exhibited the highest bond strength among deproteinizing agents. Compared to the SE mode, E&R mode significantly showed higher bond strength (p < .05). In gingival margin, bromelain SE exhibited the highest marginal leakage, while Brix 3000 SE had the lowest mean microleakage score.
Conclusion:
Deproteinizing with ClO2 was effective in improving the SBS of universal adhesive in the E&R mode to deep dentin. Deproteinization with bromelain before universal adhesive in SE mode showed more microleakage on both the occlusal and gingival surfaces.
Introduction
In dentistry, both dental materials and treatment techniques have shown significant improvement over time. Nowadays, the widespread adoption of the minimally invasive treatment approach by dentists has accelerated the developments in adhesive systems.1, 2
Universal adhesives are widely preferred in dental practice because of their simplified bonding procedures and superior bonding performance to tooth surfaces. 3 Also, universal adhesives can be used for different adhesion strategies, with or without previous acid etching, according to each specific clinical situation. However, these adhesives without previous acid etching cannot completely remove the smear layer, which is composed of disorganized organic debris binding mineral particles, due to their mild acidity.3, 4 The hybridized smear layer inhibits resin monomer infiltration into the underlying dentin and causes a weak link at the resin–dentin interface. 3 Self-etch (SE) adhesives can dissolve the mineral phase in the dentin smear layer, but not in the organic phase. 5 The deproteinization with deproteinizing agents dissolves the organic phase of the dentin smear layer, 6 increases the mineral/organic ratio on the dentin surface, 7 and increases the promotion of the infiltration of resin monomer into the underlying dentin.7, 8 As a result of acid application, dentin is demineralized and collagen fibrils appear. Afterward, the adhesive application penetrates the entire demineralized dentin and network of collagen fibrils.9, 10 Insufficient resin infiltration into the unsupported collagen network causes hydrolytic degradation.9, 11, 12 This problem can be overcome by applying a deproteinizing agent to the etched dentin surface. The application of deproteinizing agent changes the surface energy of dentin by increasing the hydroxyapatite ratio on the dentin surface.9, 11
Recently, the proteolytic enzymes, papain and bromelain, have been introduced for dental practice as deproteinizing agent.13, 14 Papain is an endoprotein similar to human pepsin (present in gastric juices), which has bactericidal, bacteriostatic, and anti-inflammatory activities.15–18 It also partially dissolves intact unmineralized type I collagen fibrils. 19 The most recent modification made on papain-based gels was the introduction of Brix 3000 from Argentina in 2016. The concentration of papain in Brix 3000 increased to 3000 U/mg in each 10%. 20 To date, there are no studies evaluating Brix 3000 as a deproteinizing agent.
Bromelain is yet another proteolytic enzyme isolated from the tropical fruit pineapple (Ananas comosus L., family Bromeliacease). 21 In addition to using bromelain as a medicinal product for many years,21–23 it has been used in dentistry since 1960 for its anti-inflammatory effects after minor oral surgeries. 24
Sodium hypochlorite (NaOCl) suggested to be used for smear layer deproteinization is known as oxidizing agent. 25 NaOCl can effectively remove the organic phase of the smear layer. 7 In particular, when applied for a longer time (e.g., 30 sec), NaOCl may lead to compromised dentin bonding due to the residual radicals on the dentin surface.26, 27
Chlorine dioxide (ClO2) is used in dental unit waterlines, 28 some commercial mouth rinses, 29 and endodontic irrigants.30, 31 ClO2 is less cytotoxic than NaOCl. 30 It also can dissolve inorganic and organic tissues and remove the smear layer. 32 A more recent study has demonstrated that ClO2 solution can be preferred as a deproteinizing agent to enhance the dentin bond strength of hypocalcified amelogenesis imperfecta-affected primary teeth. 33 However, to the best of our knowledge, there is no information available regarding how deproteinization on dentin bond strength in permanent teeth is affected by ClO2.
The aim of this study was to evaluate, for the first time in the literature, the shear bond strength (SBS) of universal adhesive in both etch & rinse (E&R) and SE modes to deep dentin with four different deproteinizing agents (Brix 3000, bromelain, NaOCl, and ClO2) and to assess their effects of marginal microleakage in class V cavities. Additionally, the deep dentin deproteinization abilities of these agents were evaluated using attenuated total reflection – fourier transform infrared spectroscopy (ATR-FTIR) spectroscopy and scanning electron microscopy (SEM). The null hypotheses tested were as follows: (1) no difference in SBS after using neither deproteinizing agents nor different adhesive modes, and (2) no difference in the microleakage to class V cavities.
Methods
Specimen Preparation
The effect size (f = 1.01) was calculated by online calculator (
Following ethical approval by the ethics committee of Recep Tayyip Erdogan University under protocol number 2020/170, 220 extracted human molars were collected and stored in distilled water containing 0.1% thymol solution at room temperature for 6 months prior to the experiments. For the SBS testing, 100 teeth were embedded in acrylic resin (Integra, Birlesik Group Dental Co, Ankara, Turkey) 1–2 mm below the cement–enamel junction of the teeth. Then, the occlusal surface was removed with a trimmer using a low-speed water-cooled diamond saw (Micracut 152, Metkon Co, Bursa, Turkey) until all the enamel was completely removed and until flat dentin surfaces were achieved. The dentin level after the removal of cusp tips was accepted as superficial dentin. One millimeter below the superficial dentin level was accepted as deep dentin.34, 35 The specimens were excluded from the test when the dentin level and the highest point of the pulp horn were not at least 1 mm. This condition was checked by taking radiograph. Subsequently, the surface of deep dentin was polished with 600-grit SiC paper under 30 sec flowing water to standardize the smear layer.7, 26
For the microleakage test, class V cavities were prepared at the buccal surface of the 100 teeth with 4-mm mesiodistal width, 3-mm buccolingual width, and 2-mm depth using a water-cooled high-speed cylindrical flat-ended fine diamond bur (837L-012; Hicare Medical Equipment and Materials Co. Guangzhou, China). A new bur was used for each group. The gingival wall of the cavities was located 1 mm below the cement–enamel junction. The dimensions were checked using a digital micrometer.
SBS Testing
The specimens (n = 100) were randomly divided into two groups such as SE (n = 50) and E&R (n = 50) groups, and equally divided into five subgroups: (1) control, (2) Brix 3000, (3) 40% bromelain, (4) 5.25% NaOCl, and (5) 0.12% ClO2.
In E&R groups, dentin was first etched with 37% phosphoric acid for 15 sec, washed with water for 10 sec, and air-dried for 5 sec, then deproteinizing agents (Brix 3000, 120 sec; bromelain, 60 sec, NaOCl, 15 sec; ClO2, 60 sec) were applied separately according to the subgroups. In SE groups, dentin was deproteinized with Brix 3000 (120 sec), bromelain (60 sec), NaOCl (15 sec), and ClO2 (60 sec) without acid etching. Deproteinizing agents were not applied to the control group. Afterward, deproteinizing agents were washed with water for 10 sec and air-dried for 5 sec and universal adhesive (Single Bond Universal, 3M Deutschland GmnH, Neuss, Germany) was applied according to manufacturer’s instructions. The application of materials was performed as indicated in Table 1.
Products Used in this Study.
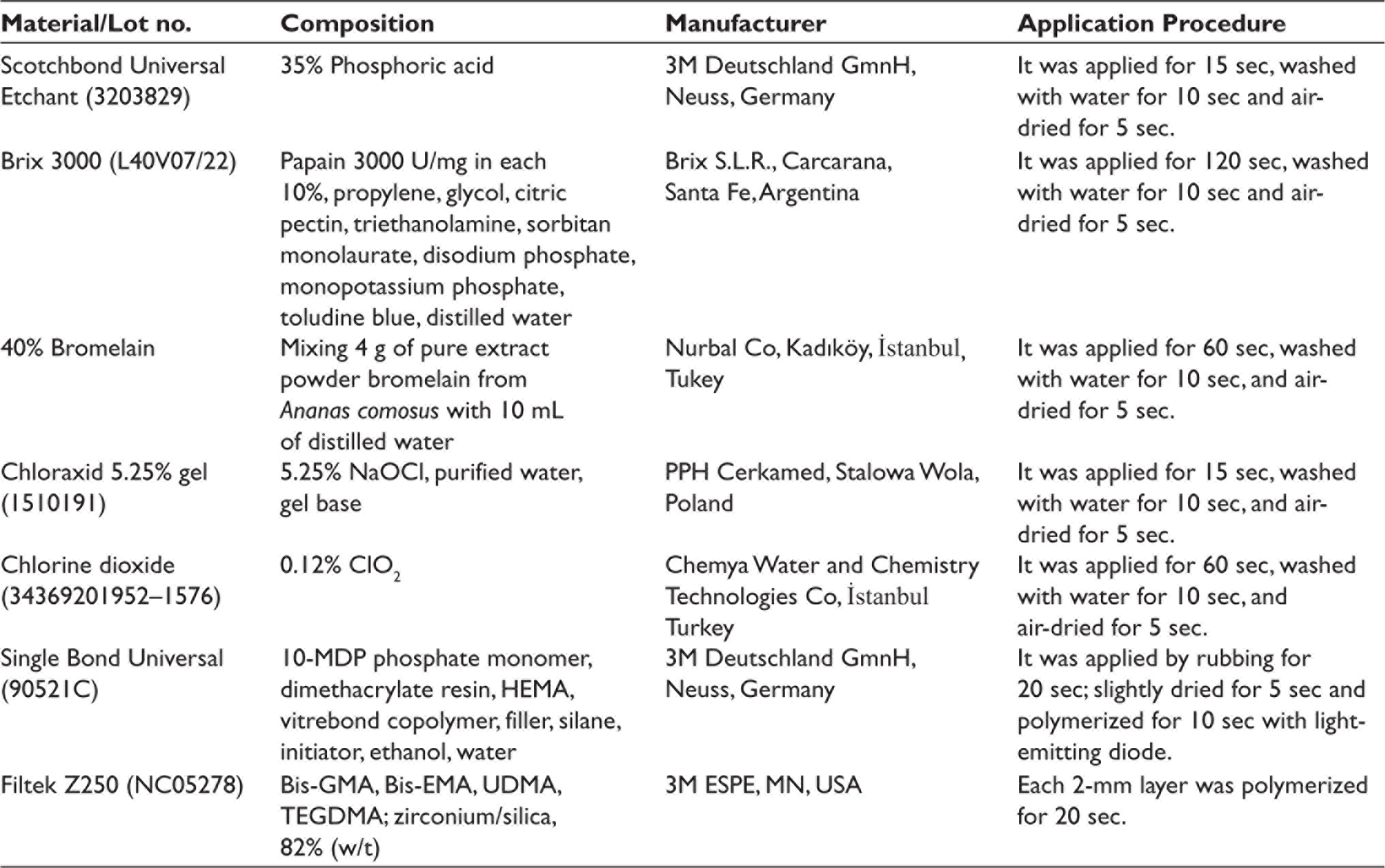
The microhybrid composite (Filtek Z250; 3M ESPE, St. Paul, USA) was placed in two increments of 2 mm thickness using plastic molds (3 mm diameter and 4 mm height) on the deep dentin surface. Each increment was cured for 20 sec using a light-emitting diode unit (VALO Cordless; Ultradent, South Jordan, UT) with an irradiance of 1000 mW/cm2, keeping it as close as possible. Specimens were stored in distilled water at 37°C for 24 hr to minimize the amount of residual monomer. Teeth were submitted to 5000 thermal cycles between 5°C and 55°C in distilled water, with 15 sec dwell time in each bath before SBS testing to simulate approximately 6 months of aging.36, 37 The specimens were examined in a universal testing machine (Model 3344; Instron Corporation, MA, USA) at a crosshead speed of 1 mm/min.13, 38 The maximum loading failure was divided by the bonding surface area (пr2 = 6.75 mm2), and the SBS results were recorded in megapascals (MPa). After testing, fractured specimens were detected with a stereomicroscope (Stemi 305; Carl Zeiss Microscopy GmbH, Göttingen, Germany) at 25× magnification. Failures were classified as adhesive (failure between the dentin and composite), cohesive (failure within the dentin or composite), and mixed (partially adhesive and partially cohesive failure).
Microleakage Evaluation
The specimens were divided into groups as in the SBS test. The microhybrid composite was placed in a single step and polymerized. Afterward, the samples were stored in distilled water at 37°C for 24 hours. Then, specimens were exposed to thermal cycles (5000 cycles, 5°C–55°C, 15 sec dwell time). After thermocycling, the external surface of the entire tooth was coated with two layers of nail varnish, leaving 1 mm wide varnish-free margins around the restoration. The samples were immersed into a 0.5% basic fuchsin solution for 24 hours at 37°C. At the end of the immersed period, samples were rinsed under running water for 1 min. Then, they were perpendicularly sectioned across their centers in the buccolingual direction using a water-cooled slow-speed disc. The samples were examined for both occlusal margin and cervical margin under a stereomicroscope at a magnification of 25× to determine dye penetration: 0, no dye penetration; 1, dye penetration up to half depth of the cavity; 2, dye penetration deeper than half depth of the restoration, but not reaching the axial wall; and 3, dye penetration reaching the axial wall.
Morphological Observation by Scanning Electron Microscope
One tooth was chosen from each group for SEM observation. The sample was sectioned using a water-cooled slow-speed diamond disc to obtain horizontal deep dentin specimens of 1 mm thickness. Pretreatment was applied to the samples as indicated before. Then, the dentin surfaces were rinsed off with water for 20 sec and dehydrated with 25%, 50% and 75% ethanol for 20 min each, 95% ethanol for 30 min, and 100% ethanol for 60 min. After the final ethanol step, the specimens were immersed in hexamethyldisilazane for 10 min and dried on a filter paper for 24 hours at room temperature. 39 The specimens were then manually fractured into two parts to obtain both an occlusal and a cross-sectional view of each specimen. After gold sputter-coating, the specimens were examined using the SEM (Evo LS 10, Carl Zeiss NTS, Germany) at 2000× magnification.
ATR-FTIR Spectroscopy
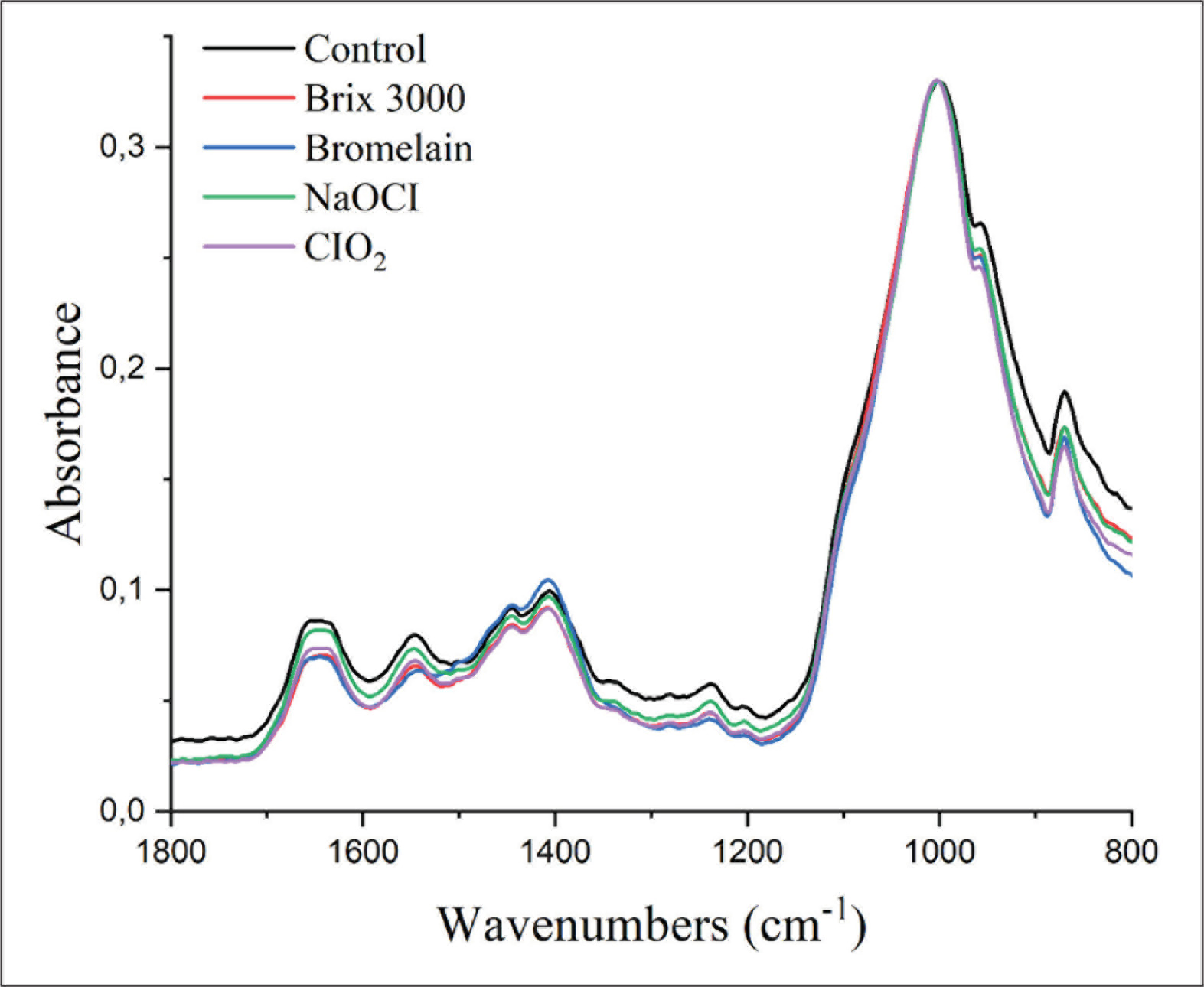
One tooth in each experimental group was used for ATR-FTIR analysis to evaluate the chemical changes on the dentin surface. Deep dentin slices and pretreatment of the dentin surface were prepared the same way as for the SEM observation. The control spectra were collected from all the specimens prior to the pretreatment using an ATR-FTIR spectrometer (Vertex 70v, Bruker, USA) with the co-addition of 64 scans in the range of 750–4000/cm−1 at 4/cm−1. The spectra were collected again under the same conditions after the application of deproteinizing agents as described in the Specimen Preparation section. The deproteinizing effect was evaluated using the amide/phosphate ratio, that is, the ratio of the amide I band stretching around 1643 cm–1 to ν3 PO43- vibrations around 1026 cm–1. Given that the amide band is representative for collagen and the phosphate band for hydroxyapatite, a decrease in the amide/phosphate ratio indicated collagen removal from the dentin surface.7, 40
Statistical Analysis
The normality of the data and the homogeneity of the variance were performed using the Shapiro–Wilk test. Since the SBS values were normally distributed, the two factors (application mode of adhesive, different deproteinization agent application) were analyzed using two-way analysis of variance (ANOVA) and Tukey’s post-hoc test. The Wilcoxon test was used to compare the gingival and occlusal microleakage of the same sample. The scores of gingival and occlusal margins for each group were analyzed using the Kruskal–Wallis test. The statistical significance level was accepted as p < .05. All analyses were performed using the IBM software (version 23.0; SPSS Inc., IL, USA).
Results
Shear Bond Stength
The SBS test results and the percentage of failure modes in each group are shown in Table 2. The two-way ANOVA showed that the effect of E&R and SE universal adhesive on the bond strength (p = .034) and the difference between deproteinizing agents (p = .005) were statistically significant. However, there was no common effect of deproteinization agents and adhesive modes on the SBS (p = .059).
Mean and Standard Deviations of SBS (MPa) and Failure Mode.
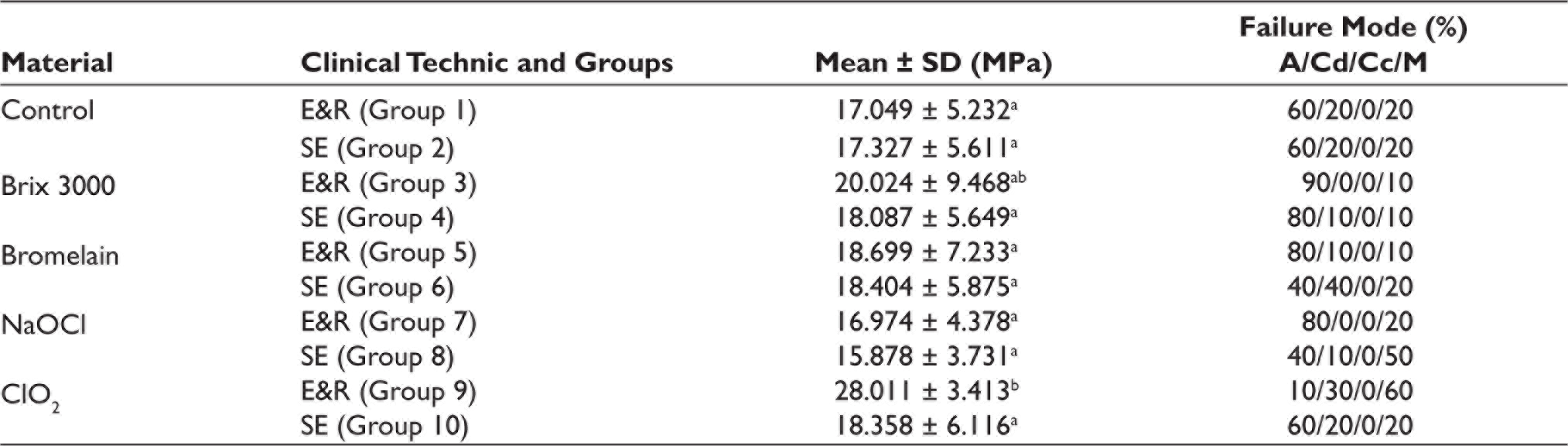
Tukey’s test found that the E&R group of ClO2 showed significantly highest bond strength than all other groups (p = .015). A significant difference was found only between the E&R group and SE group of ClO2 among all groups. However, statistically significant differences were not noted in Brix 3000, bromelain, and NaOCl treated groups with the E&R and SE modes. The most adhesive failure was seen in the Brix 3000 E&R group (n = 9). In bromelain, SE group presented a high number of dentin cohesive failures (n = 4). The most mixed failure was seen in the ClO2 E&R group (n = 6). Cohesive failure in composite was not observed in any of the groups.
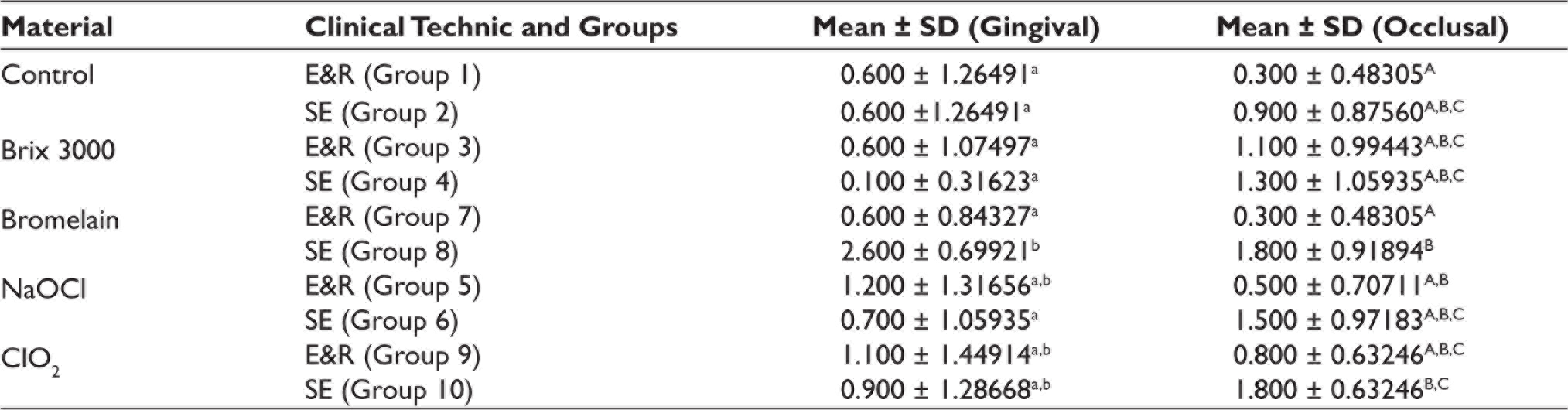
Microleakage
The marginal leakage was evaluated according to the scores between 0 and 3. According to the Wilcoxon test, the mean microleakage in the gingival and occlusal margins was statistically different only in the Brix 3000 SE group (p = .020). The mean microleakage in occlusal margins was statistically different only between bromelain E&R and SE groups. In gingival margin, bromelain SE exhibited the highest marginal leakage, while Brix 3000 SE had the lowest mean microleakage score (Table 3).
Statistical Analysis using the Kruskal–Wallis Test for Microleakage Scores.
Morphological Assessment
SEM images of the deproteinizing agent and control groups applied to the dentin surfaces at a magnification of 2000× are shown in Figure 1. In the acid-treated control group, the orifices of some dentinal tubules on the deep dentin surface were completely open. The samples in the E&R showed a similar appearance to the control group.

A homogeneous smear layer was formed on the deep dentin surface and the dentinal tubules were completely clogged in the control SE group. No change was observed in the smear layer after NaOCl and ClO2 application to the smear layer–covered dentin surface. However, after deproteinization with Brix 3000 and bromelain, there were gaps observed at some dentinal tubule orifices.
ATR-FTIR Analysis
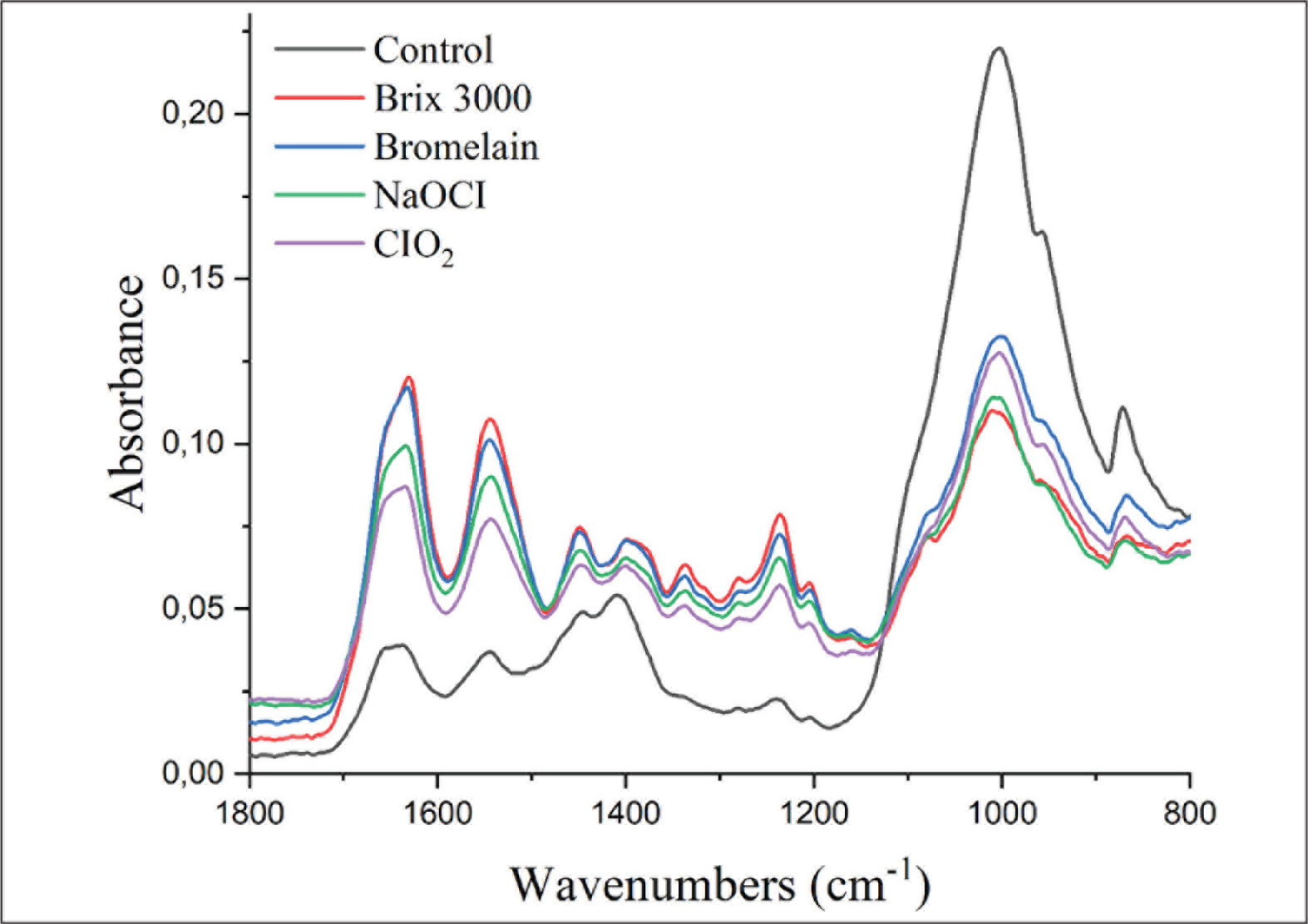
The spectra of SE groups were normalized to the phosphate stretching vibration at 1026/cm–1 before amide/phosphate ratio calculation. There were no significant changes observed in the amide/phosphate ratio when NaOCI, ClO2-treated spectra, and control spectra were compared. However, the amide/phosphate ratio decreased after pretreatment with Brix 3000 and bromelain (Figure 2). After the application of acid and deproteinizing agent, there was a decrease in phosphate peak while an increase in amide I peak was observed (Figure 3).
Representative FTIR Spectra of Smear Layer–Covered Deep Dentin in Each Pretreatment. The Spectra were Normalized to Phosphate v3 (peak at 1026/cm–1).
Representative FTIR Spectra of Acid-Etched Deep Dentin in each Pretreatment. The Spectra was Not Normalized.
Discussion
According to the present results, using universal adhesive in E&R mode and the application of ClO2 after the use of phosphoric-acid etching could significantly improve shear bond strength. Bromelain SE group showed more microleakage on both the occlusal and gingival surfaces. Thus, the null hypotheses can be rejected.
SBS tests more successfully reflect the load distribution in the oral environment. 41 Cardoso et al., 42 in their study comparing shear tests and tensile tests, stated that shear tests gave higher bonding results and better imitated clinical conditions. Therefore, SBS test was used in our study.
The aim of in vitro microleakage tests is to investigate the infiltration of the dye solution into the resin–dentin interface. 43 Heintze showed no correlation between laboratory studies evaluating microleakage and clinical outcomes. 44 However, Mali et al. 45 reported that laboratory studies on microleakage are an acceptable method to investigate the sealing of adhesive and restorative materials.
Brix 3000 has not been previously evaluated as a deproteinizing agent. In the present study, both the Brix 3000 SE and Brix 3000 E&R groups showed similar SBS and microleakage values compared with the control groups. This finding is similar to the results of Alshahli et al. in bovine incisors and Hasija et al. in primary tooth enamel.14, 46 However, Sharefeddin et al. 13 indicated that the deproteinization of smear layer–covered superficial dentin with 10% papain solution significantly increased the SBS value. In this study, deep dentin was used as the adherent. This is because deep dentin has wider dentin channels and less intertubular dentin, and moisture control is difficult than superficial dentin. Differences in findings between studies can be explained by papain-based products and the adhesive used. In the Brix 3000 SE group, the microleakage on the occlusal surface was more than the microleakage on the gingival surface. This finding may be due to Brix 3000 being more difficult to rinse from the tooth surface due to its higher viscosity compared with other deproteinizing agents. In addition, the occlusal area is wider than the gingival area may have made it difficult to completely remove a more viscous gel from the surface.
Pretreatment with bromelain has been reported to effectively remove the unsupported collagen matrix of the etched dentin surface. 47 Hasija et al. 46 in their study on the enamel of primary molar teeth stated that applying a 60% concentration of bromelain solution to the enamel surface for 60 sec increased the SBS. Sharafeddin et al. 13 applied a 6% bromelain solution to the superficial dentin surface for 60 sec, but they did not observe an increase in SBS. Therefore, in this study, we used a concentration of 40% bromelain solution because it was applied to deep dentin. According to the SEM and ATR-FTIR spectroscopy results, partial dissolution occurred by applying bromelain to the smear layer–covered deep dentin surface. However, no significant difference was found between the control and bromelain groups. This indicates that the 40% bromelain solution have adversely affected the polymerization. This estimation may support the fact that the bromelain SE group showed the most microleakage.
NaOCl is frequently used in dentistry for both its disinfecting and nonspecific deproteinizing effects, however, there are different results regarding the effect of NaOCl pretreatment on dentin bond strength.33, 48 The diversity in results may be explained by differences in test methods and adhesive composition.25, 33, 46 Similar to our study, previous studies stated that the NaOCl treatment for 5 sec resulted in a bond strength similar to that of the flat dentin surface.26, 27, 40 In addition, in this study, no difference was observed in SEM and ATR-FTIR examinations with the application of NaOCl for 15 sec to the etched and nonetched dentin surfaces. A previous study also revealed that NaOCl treatment for 15 sec could partially remove the hybridized smear layer, and more extended application of NaOCl for 30 sec could altogether remove it. 7 Short-term application of NaOCl is recommended because long-term application of NaOCl causes incomplete polymerization due to free-radical formation.26, 27 However, the application of 5.25% NaOCl for 15 sec did not show any effect on the bond strength, microleakage and surface morphology.
This study indicated, for the first time that, deproteini- zation of etched dentin with ClO2 increased the bond strength. This finding is similar to the results of Bayrak et al., who showed deproteinization with ClO2 can be considered effective in enhancing dentin bonding of hypocalcified primary teeth. 33 Moreover, consistent with SBS, the most mixed failure was observed in the ClO2 E&R group. These results might show that the deproteinizing effect with the ClO2 solution extended to deeper regions of the dentin substrate. 14 Besides, ClO2 deproteinization did not increase microlea- kage in both SE and E&R clinical techniques and did not show any alteration on the surface of the etched and non- etched deep dentin according to SEM and ATR-FTIR spectro- scopy. However, these results could not be validated as no previous reports have been in the literature about the effects of ClO2 on the surface feature of dentin and microleakage.
The limitations of the current study were that the Brix 3000 might not be completely removed by washing and drying because of its high viscous pattern. Also, different tissues of the tooth, such as the enamel were not used in this study. If the effects of deproteinization agents on enamel containing highest percentage of hydroxyapatite had been evaluated, we could have been able to obtain beneficial results regarding the retention of restorative dental materials.
Conclusions
In conclusion, this study demonstrates for the first time that the deproteinization of etched dentin with ClO2 increased the bond strength, but did not have a beneficial effect on microleakage. In addition, E&R technique showed higher bond strength compared with the SE technique. The bond strength of composite to deep dentin was not affected significantly using papain, bromelain, or NaOCl agents. Deproteinization with bromelain before universal adhesive in SE mode showed more microleakage on both the occlusal and gingival surfaces.
Footnotes
Ethical Approval
The study was approved by the ethics committee of Recep Tayyip Erdogan University under protocol number 2020/170.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by Recep Tayyip Erdogan University Research Fund under grant number (2020/1144).
Informed Consent
Not applicable
