Abstract
Aim:
The present study aimed to assess the efficacy of combining microneedling (MN) and injectable platelet-rich fibrin (i-PRF) administration with i-PRF alone in augmenting the thickness of gingiva to manage thin periodontal phenotype.
Methodology:
The clinical trial involved 36 patients who were diagnosed with thin periodontal phenotype. They were randomized under two groups: MN + i-PRF versus i-PRF alone, carried out as a split-mouth trial. One quadrant was treated only with i-PRF delivery through the gingival sulcus, while the contralateral side was treated with MN and i-PRF in 3 sessions at 10-day intervals. The primary parameter measured was the thickness of the gingiva, at baseline and 3 months following therapy. Statistical analysis was performed using the Mann-Whitney U test. Wilcoxon’s signed ranking test for inter-group and intra-group analysis, respectively.
Results:
The study results showed a significantly greater increase in gingival thickness (GT) in sites where both MN and i-PRF injection was done, compared to i-PRF alone, with a p value of .04.
Conclusion:
Both treatment modalities showed improvement in GT, being a minimally invasive alternative for mucogingival surgery. Adjunctive MN was found to be more beneficial than i-PRF alone. However, further research should be carried out to address the drawbacks of the study and for a better understanding of this novel approach.
Abbreviations
Gingival thickness
Injectable platelet-rich fibrin
Microneedling
Cone beam computed tomography
Patient-related outcome measures
Introduction
The gingival biotype has gained significant importance in periodontal literature, with regard to esthetics and function. The term periodontal phenotype describes changes in both the width and thickness of the keratinized tissue.1,2 Apart from the soft tissues, it also includes the thickness of the buccal cortical plate. 3 This term was later replaced by “Periodontal biotype,” which was introduced by Lindhe and Siebert, and describes the buccolingual thickness of gingiva. 4 According to a classification system given by Ochsenbien and Ross, the gingival biotypes can be categorized as scalloped and thin gingiva or flat and thick gingiva.5,6 Gingival thickness (GT) of 2 mm or more is accounted as thick biotype, whereas GT of 1.5 mm is considered as thin biotype. 7 With regard to gingival recession coverage procedures, GT plays a major role in the extent of root coverage, where in thickness of > 0.8 mm brings about complete coverage and thin gingiva results in partial recession coverage. 8
Gingival biotype can be evaluated by direct visual assessment, or by assessing the transparency using a periodontal probe through the gingival sulcus, or by transgingival probing with an endodontic reamer. 9 Less invasive methods include ultrasonics, or cone beam computed tomography (CBCT). Gingival biotype is considered “thick,” in the presence of adequate width of keratinized tissue, with a flat marginal gingiva. Whereas, it is considered thin, when the width of keratinized tissue is inadequate, with a scalloped marginal gingiva. Due to the significantly good bone architecture, the thick gingival biotype is considered to be less prone to the spread of inflammation compared to the thin gingival biotype. 10 Gingival biotype is a major risk factor for the development of gingival recession. Gingival recession poses both cosmetic and functional issues, and augmentation of GT is vital in the prevention of gingival recession. 11 Although soft tissue grafting is accepted as the gold standard in increasing the GT, a non-surgical option for augmentation of thin gingival biotype is not in clinical practice. Minimally invasive treatment techniques have recently taken precedence over surgical procedures field of periodontics. 12
Platelet concentrations have the ability to release various growth factors.10,11 Injectable platelet-rich fibrin (i-PRF) can be obtained by subjecting the blood sample to centrifugation for 3 min in a plastic tube at a rate of 700 rpm. 12 Due to the slow rate of centrifugation. i-PRF offers various advantages over PRF, including a higher concentration of leukocytes, slower and prolonged release of growth factors.13–17 From the current literature review and meta-analysis, it is evident that PRF has the capacity to enhance the thickness of the gingiva in patients with thin periodontal phenotype. 18
Microneedling (MN) is a therapeutic procedure which involves the induction of percutaneous collagen production. This causes bleeding which initiates the wound-healing mechanism and triggers the release of numerous growth factors. In response to MN, the tissue responds with the proliferation of fibroblasts and the production of collagen, thus conserving the integrity of the skin.19,20 This process of formation of collagen and elastic fibers occurs through a process known as neocollagenesis, over a period of about 5 to 8 weeks, thus increasing the thickness of the tissue.21,22 This process is followed by the formation of new blood vessels by endothelial cells (neoangiogenesis). Tissue remodeling occurs from 8 weeks to almost 1 year following the MN procedure. 23
Considering the beneficial regenerative effects of i-PRF and MN, the study aimed to compare the efficacy of i-PRF versus i-PRF in conjunction with MN in the treatment of thin gingival phenotype. The rationale behind this study is that the thin gingival phenotype is more prone to gingival recession, and altering the phenotype using minimally invasive techniques should be considered prior to periodontal and orthodontic therapy. However, if this non-surgical procedure proves to be beneficial in augmenting the GT, it can be considered as an alternative to conventional surgical technique.
Materials and Methods
Setting and Design
The present study was 3 months follow-up, split mouth, single blind, prospective randomized controlled trial, at the Department of Periodontology, Saveetha Dental College. The research was performed between October 2020 and February 2021.
Sampling Criteria
The sample size was estimated with the data from an earlier study using G power 3.0 software, with a power set at 80%. 22 The sample size was calculated to be 38 and was rounded up to 40. A total of 40 patients were recruited in the trial.
Inclusion Criteria
Age: 20–45 years. Pregnant and lactating women. GT of mandibular teeth less than 0.8 mm.
Bone dehiscence in relation to lower front teeth, identified by CBCT.
Exclusion Criteria
Past history of periodontal surgery. Medication causing gingival enlargement. Mucogingival discrepancies. Bruxism.
Patients who reported to the Department of Periodontology, and were examined under CBCT and found to have thin phenotype and buccal bone dehiscence were included in the study. Four patients who missed their appointments were excluded from the study. The study thus included 216 lower front teeth in 36 patients (23 females and 13 males), with a mean age of 32.4 years.
Study Methodology
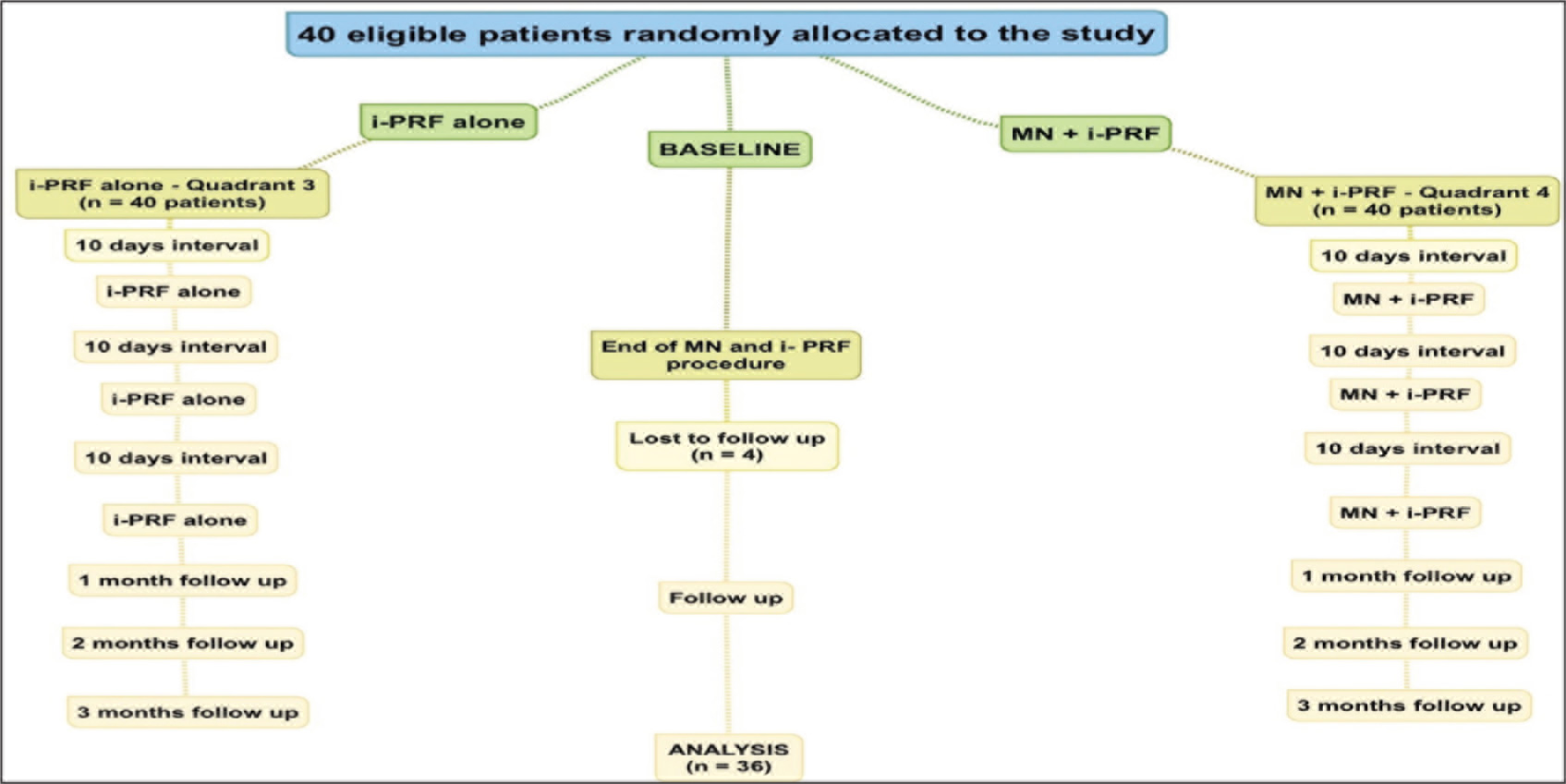
All the study participants underwent Phase 1 periodontal therapy and received oral hygiene maintenance instructions. Patients with GT lesser than 0.8 mm and buccal bone dehiscence at the level of 1/2 to 1/3 of the apices of the lower front teeth were included in the study. 8 The study protocol is described in Figure 1.
Study Protocol Depicted as CONSORT Flow Chart. 28
Assessment of GT
Measurement of the GT was done at 1.5 mm from the marginal gingiva using a No.15 endodontic spreader along with a stopper. The spreader was introduced perpendicularly through the thickness of the gingiva, about 2 mm apical to the marginal gingiva under topical anesthetic. After that, the silicone stopper is placed in close proximity to the soft tissue. A digital vernier caliper was used to measure the penetration depth starting from the spreader’s tip to the silicone stopper. 25
Clinical Procedure (Figure 2)
Assessment of Gingival Phenotype and MN and i-PRF Injection. 28
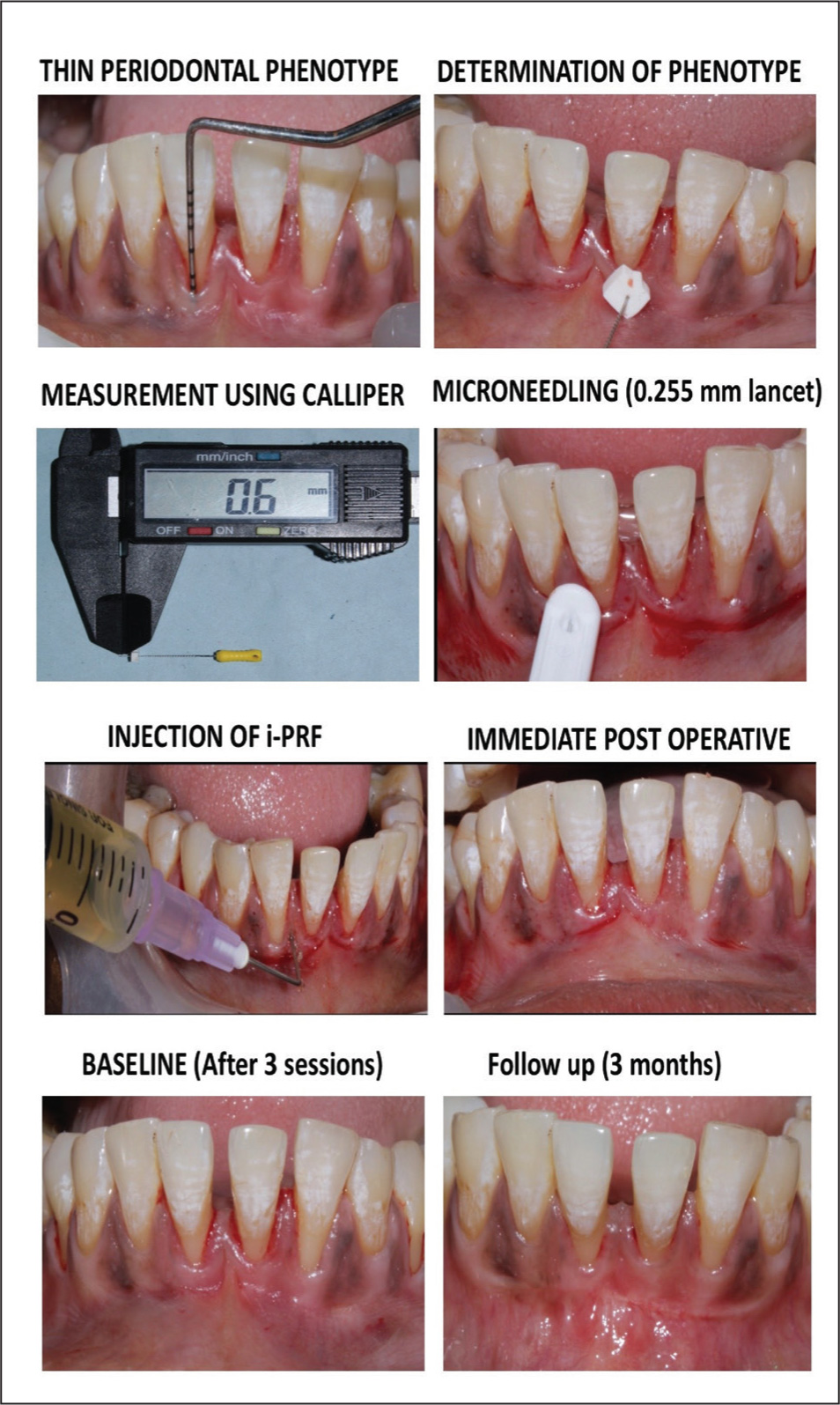
Preparation of i-PRF
Twenty milliters of venous blood was withdrawn from each patient and was transferred to 2 i-PRF tubes (measuring 10 ml each). To obtain i-PRF, the collected blood sample was subjected to centrifugation for 3 min at the rate of 700 rpm. i-PRF was then injected into the tissues using a syringe having a 27-gauge needle.
Microneedling
A 30-gauge lancet was inserted perpendicularly into the soft tissue, till it comes in contact with the underlying bone, multiple times, thus performing MN on the keratinized gingiva. The number of microchannels was calculated to be 250 in each quadrant, involving the central, lateral incisor and canine. 26
Administration Protocol
Local anesthesia was administered to anesthetize the sextant 5 region. On one side, i-PRF was directly administered through the crevice of the gingiva, whereas on the contralateral side, i-PRF was injected through the microchannels created by MN. The same protocol was carried out for 3 sessions with a time interval of 10 days. Measurement of the GT was done at baseline and for up to 3 months, in all the monthly follow-up visits. 27
Statistical Analysis
All the statistical analysis was performed using SPSS software version 23.0. The inter-group and intra-group comparison was done using the Mann-Whitney U test and Wilcoxon’s signed ranking test.
Results
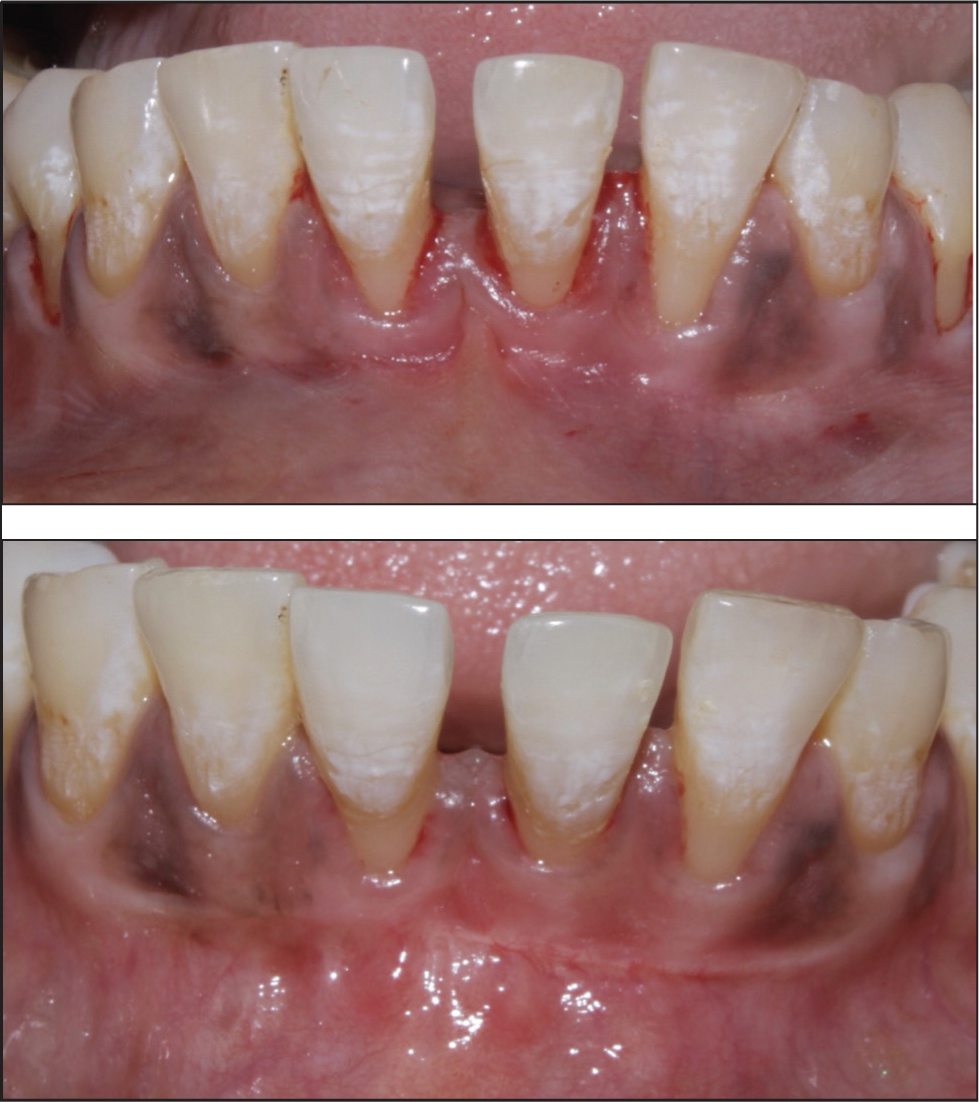
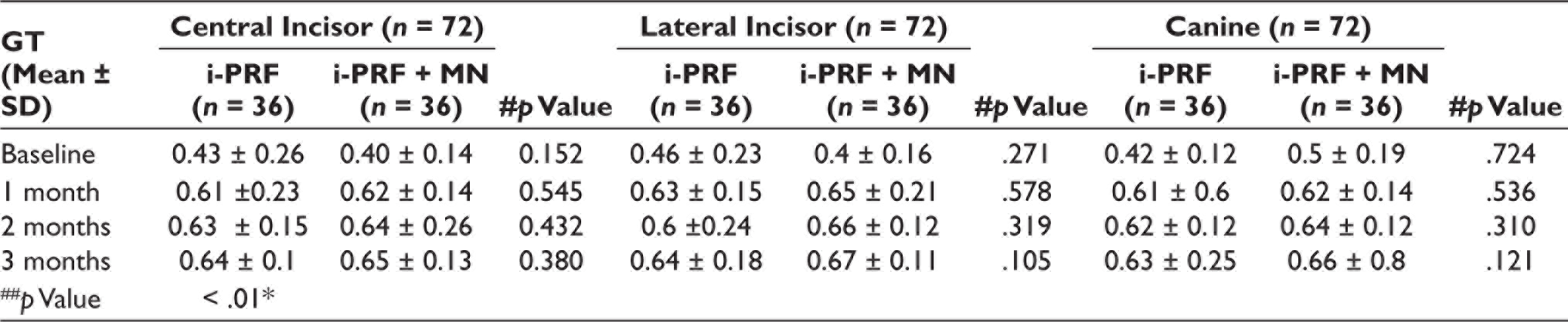
From the data obtained, it is evident the there is a statistically significant difference was found between groups with a greater increase in GT favoring the combination of the MN and i-PRF in all the lower anterior with a p value of .04 as shown in Table 1 and Figure 3. Tables 1, 2 and Figure 4 depicts the statistically significant difference in GT with respect to all lower anterior teeth.
Comparison Between Baseline and Follow-Up. (a) Baseline gingival thickness (GT) before micro-needling; (b) Improved gingival thickness (GT) at 3 months follow up.
Intra-Group and Inter-Group Comparison of GT for all Lower Anteriors. 24
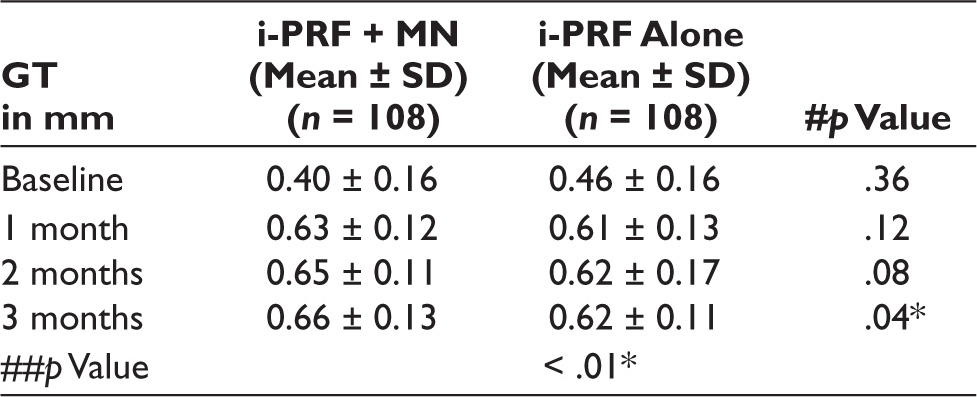
Comparison of Mean Gingival Thickness between Baseline and Follow-Up, Represented as Bar Graphs. The Teeth Assessed was Represented as X-axis, Pre-operative and Post-perative i-PRF Alone was Depicted as Blue, and Green Bar Graphs, respectively. Pre-operative and Post-operative MN and i-PRF was Represented as Orange and Purple, respectively. Both the Inter-Group and Intra-Group Comparisons Showed Significant Difference with p Value < .05.
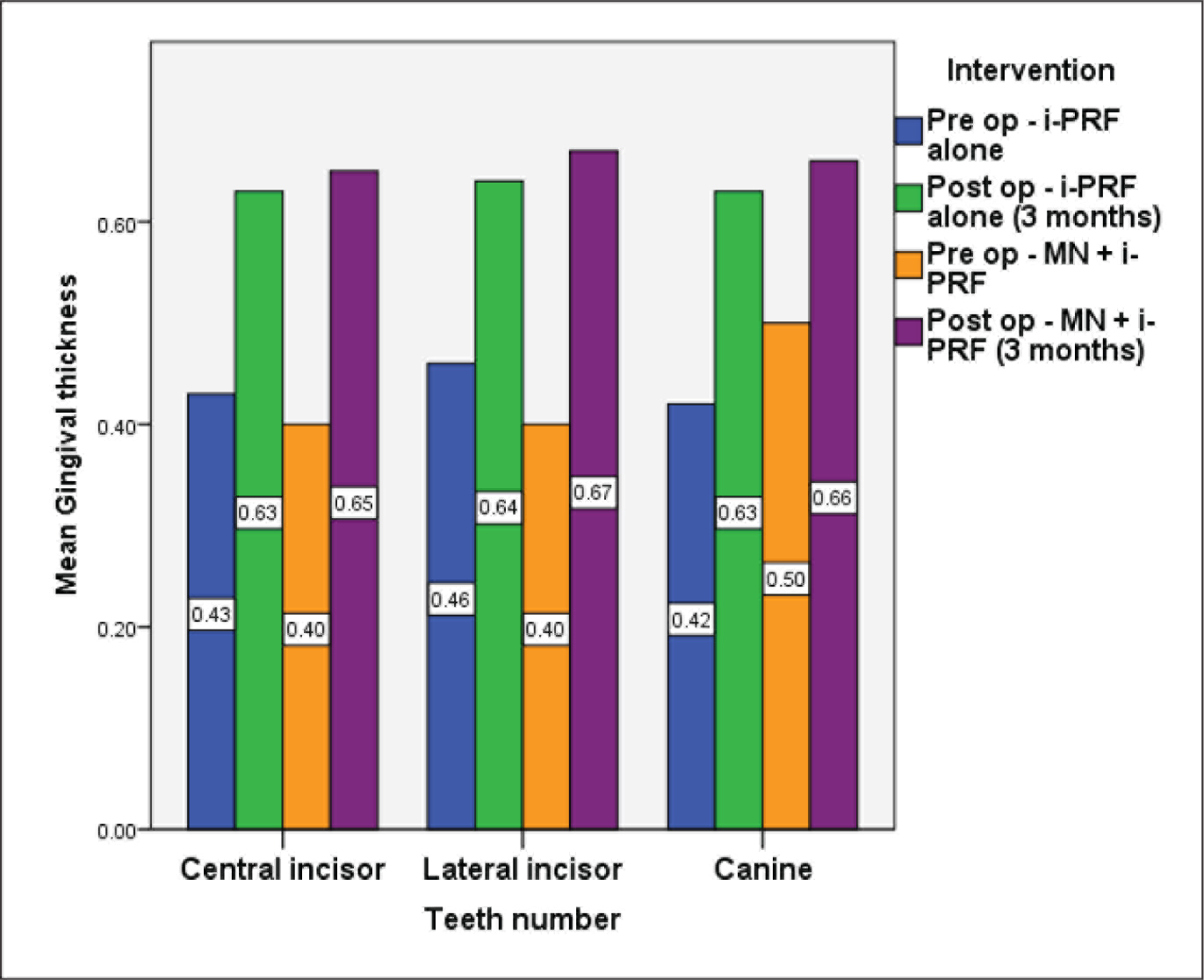
Intra-Group and Inter-Group Comparison of GT of Each Tooth. 24
Discussion
At 3 months follow-up, the results revealed that the GT improved significantly in both the study protocols. However, the sites which underwent both MN and administration of i-PRF showed better results than the i-PRF alone. There are no previous studies focusing on the effect of non-surgical, atraumatic procedures to enhance gingival phenotype. 28 Since GT was more strongly associated with periodontal phenotype when compared to other parameters like the width of keratinized tissue and height of the papilla. Hence, the GT was chosen as the primary parameter of the current study.29,30 Among the various methods for measurement of GT, transgingival probing and ultrasonography-assisted measurement are considered to be accurate. 31 In this study, transgingival probing technique was opted due to its cost-effectiveness and reproducibility. The topical anesthetic was preferred over the injection of local anesthetic solution as it might interfere with the accuracy of measurement of GT and also, the vasoconstrictor can negatively impact the distribution of i-PRF into the study sites. 29
The association of GT and gender is considered to show controversial results. One study reported that thin phenotype showed a higher incidence among females, compared to males. 32 However, Kolte et al. concluded that gender had no effect on the GT. 33
Repeated MN sessions are required to obtain the desired outcomes in the GT. Studies carried out in dermatology have reported that 3 to 5 sessions of MN cause a reduction of acne scars to about 50%–70%. 34 Additionally, it was found that there was maximum collagen formation between 7 to 14 days. 34 The time interval between the resorption of PRF and the release of growth factors from i-PRF was estimated to be around 7 to 11 days. 12 Three sessions of MN were performed in the present study with an interval of 10 days.
The peak level of GT at 3 months is attributed to neoangiogenesis and neocollagenesis, which is favoring the results of combining MN and i-PRF in the current study. This is in accordance with the results reported in a similar study where the gain in GT in the i-PRF group was 44.19% and the combination of MN and i-PRF was 65%. 28 Furthermore, it is theorized that the neocollagenesis and neoangiogenesis can be prolonged by MN. 12
By the end of 1 month, it is believed that both groups showed improvement in GT when compared to baseline. But this is considered to be unstable as the maturation of the gingival fibers is not complete in 30 days. As there is not much evidence on MN in dentistry, it is only possible to compare the study results with dermatological studies. About a 40% increase in stratum corneum layer, with improved collagen content, has been reported in dermatology following MN at around 6 months.18–20
From the recent literature evidence, it was reported when micro-needling was performed along with the administration of topical vitamin, the increase in thickness of the epidermis was about 140%, compared to 22% in topical vitamin treatment alone. 21 Whereas in an eight-month follow-up study by Fabbrocini et al., it was reported that employing two sessions of micro-needling improved the tissue thickness by 0.45 mm. 35 This was consistent with the findings of GT in the current study.
The most common etiology of gingival recession is traumatic tooth brushing, especially in the case of thin gingiva which is more prone to traumatic changes. Procedures which aim in increasing the thickness of the gingiva reduce the impact of tooth brushing on the gingiva, thus making it more trauma-resistant. 36
Baldi et al. reported that for every 0.1 mm increase in flap thickness, gingival recession can be receded by 0.2 mm. 8 Therefore, augmentation of GT can be considered as a promising procedure prior to carrying out periodontal surgery. Literature evidence has stated that buccal bone dehiscence is common in patients with thin periodontal phenotype, especially after orthodontic therapy. Hence, MN and injection of i-PRF can be considered beneficial in such cases.37–40
The major drawback of the study is the lack of assessment of the impact of this procedure on variables pertaining to patient outcomes including patient-related outcome measures (PROM). Other disadvantages include short-term follow-up and a small sample size. Thus the long-term beneficial effects of this protocol are still unclear.
Conclusion
The administration of i-PRF and a combination of MN and i-PRF in patients with thin periodontal phenotype, improved the GT even without surgical periodontal procedures, according to the findings of the research. MN is believed to be imparting a beneficial effect on enhancing GT. In order to address the drawbacks of the study and for a better understanding of this novel approach, further research should be carried out.
Footnotes
Ethical Approval
The study protocol was based on the Declaration of Helsinki and was performed after obtaining clearance and approval from the Institutional Review Board (IEHC/PERIO-18/2020/32).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
The participant has consented to the submission of the article to the journal.
