Abstract
Introduction:
Type I diabetes mellitus (T1DM) is an autoimmune disorder characterized by absolute insulin deficiency, which is caused by progressive destruction of the β-islet cells of the pancreas. Aminotransferases participate in the interconversion of amino acids and oxalic acids by transfer of amino groups. Organ damage leads to leakage of transaminases into the blood. IgA is the only immunoglobulin that is found in saliva and forms the first line of defense. Alteration in the levels of secretory IgA often indicates a local or systemic disease. The aim of this systematic review is to evaluate the role of salivary aspartate aminotransferase (AST), alanine aminotransferase (ALT), and IgA as biomarkers for T1DM patients.
Materials and Methods:
An electronic literature search was conducted by two independent reviewers using PubMed, Medline and Google scholar without any date filter. The inclusion criteria included case control and cross-sectional studies. Publications in languages other than English, review articles, letters to the editor, and studies done on animal models were excluded from the review. The difference in the levels of salivary AST, ALT, or IgA in T1DM patients and in healthy individuals were compared. Risk of bias assessment was done using the ROBINS-I tool.
Results:
A total of eight relevant articles were assessed, out of which three studies showed elevated salivary AST and ALT levels and five studies showed elevated salivary IgA levels in patients with T1DM.
Conclusion:
Our review reflected that salivary AST, ALT, and IgA were elevated in T1DM patients, and they can be used for monitoring these patients. Further studies are needed to establish the diagnostic accuracy of these parameters for clinical application.
Keywords
Introduction
Diabetes mellitus affects a vast majority of the global population; it is the most common metabolic endocrinal disorder. Type I diabetes mellitus (T1DM) is an autoimmune disorder characterized by absolute insulin deficiency, which is caused by progressive destruction of the β-islet cells of the pancreas. This is clinically manifested as an increase in the blood glucose levels, with an elevated glycosylated haemoglobin level. 1 In the case of T1DM, the earlier the diagnosis is made, the more effective its management. 2 Blood glucose estimation is the standard diagnostic test; however, it has several complications like risk of infections and anxiety due to its invasive nature. 3 The recurrent painful experience may cause profound physical, mental and emotional disturbances and can lead to non-compliance with health care services, commonly called needle phobia. 4 The use of some non-invasive diagnostic means like the use of saliva for monitoring disease progression in such patients would be a boon.
Aminotransferases participate in the interconversion of amino acids and oxalic acids by transfer of amino groups. 5 AST and ALT are enzymes of tissue destruction, and their levels are elevated in blood as well as saliva, following organ damage. 6 They are usually present in renal tissue, brain, skeletal muscle, pancreas, lungs, red blood cells and white blood cells. Damage to the organ parenchyma leads to transaminase leakage into the blood, as they are present in relatively higher concentrations. 5
Secretory IgA (sIgA) is usually present in all the secretions, particularly saliva. sIgA acts as the first line of defense against the pathogens that invade mucosal surfaces. 7 These antibodies promote oral immunity by preventing microbial adherence and neutralizing the toxins. 8 Alteration in the levels of sIgA often indicates a local or systemic disease. Several studies have found an elevated salivary IgA level in diabetes patients. 9
The current systematic review aimed to evaluate the role of salivary AST, ALT, and IgA in the diagnosis and monitoring of T1DM patients.
Materials and Methods
The present systematic review was done in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines, 2020. Ethical clearance and patient’s informed consent were not needed, as the review did not involve any human/animal participants.
Review Questions
Are salivary levels of AST, ALT, and IgA elevated in T1DM patients?
Can salivary AST, ALT, and IgA be used as reliable biomarkers for diagnosing T1DM?
Search Criteria
The search criteria according to the population, intervention, comparator, outcome and study design (PICOS) were as follows:
Population (P): Patients with T1DM and healthy individuals. Intervention (I): Salivary levels of AST, ALT, and IgA in T1DM patients. Comparator (C): Salivary levels of AST, ALT, and IgA in healthy individuals. Outcome (O): Elevation of salivary AST, ALT, and IgA levels in T1DM patients. Study design (S): Case control studies, cross-sectional studies and cohort studies.
Inclusion and Exclusion Criteria
Case control and cross-sectional studies that compared the salivary levels of AST, ALT, or IgA in T1DM patients and in healthy individuals were included in the review.
Studies that assessed these biomarkers in other tissue fluids like blood or gingival crevicular fluid were excluded. Publications in languages other than English, reviews, letters to the editor, studies assessing these parameters in other diseases and studies done on animal models were also excluded.
Search Strategy
An extensive literature search was done using PubMed, Google scholar and Medline databases without any date filter. A manual search was also done. MeSH terms like ‘Type I Diabetes Mellitus’, ‘Aspartate Aminotransferase’, ‘Alanine Aminotransferase’, ‘Secretory IgA’ and ‘Salivary biomarker’ were used independently and in several combinations.
During data extraction, the following parameters were assessed: author and year of publication, biomarker analysis, statistical analysis and significance (p value), mean salivary value of the biomarkers and whether the salivary levels are elevated or not. The data extracted were tabulated (Table 1).
Risk of Bias Assessment
We used the ROBINS-I risk of bias assessment tool to understand and appraise strengths and weaknesses in the included non-randomized studies. Two investigators independently assessed the risk of bias of each study. Factors assessing the methodological quality such as confounding, participant selection, deviations from intended interventions, classification of interventions, missing data, selection of reported results and measurement of outcomes were evaluated. Low risk of bias was assigned to a study characteristic with complete available data, while moderate risk was assigned to a study characteristic with few missing data. In contrast, a higher risk of bias was awarded to a study characteristic with limited or insufficient data. In cases where the study characteristics were uncertain or not explained completely, the risk of bias was entered as uncertain. Any disagree-ments between the two investigators were resolved by consensus.
Results
Search Selection
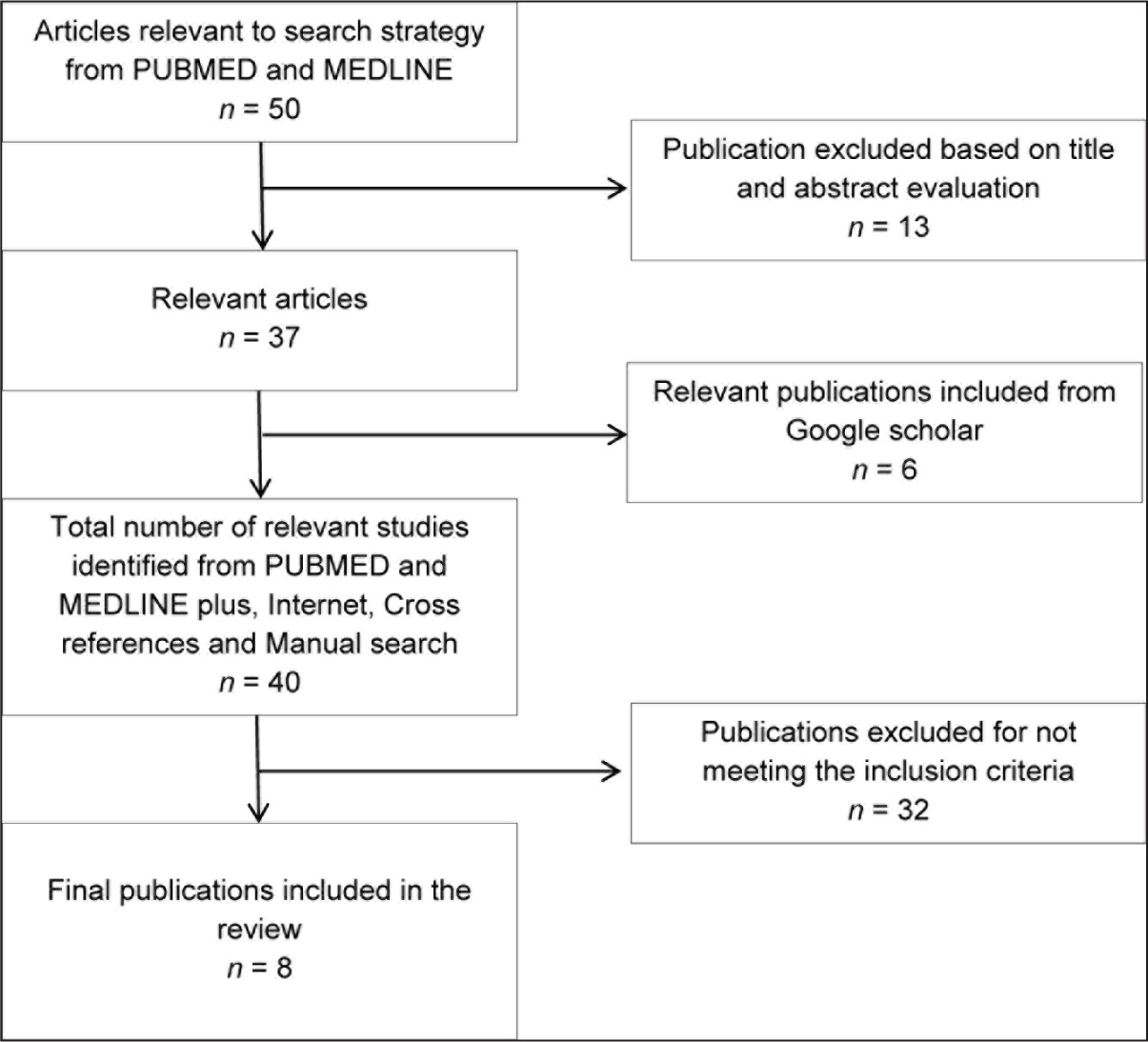
A total of 50 articles were identified using the predetermined MeSH terms, of which seven were duplicates. The PRISMA flowchart (Figure 1) was developed to summarize the selection of the included studies. Forty-three titles and abstracts were identified as the related subjects of inclusion criteria. After analyzing 43 abstracts, 40 publications were finalized for full article screening. Finally, eight articles were acknowledged to meet the review criteria. Table 1 signifies an overview of data extraction.
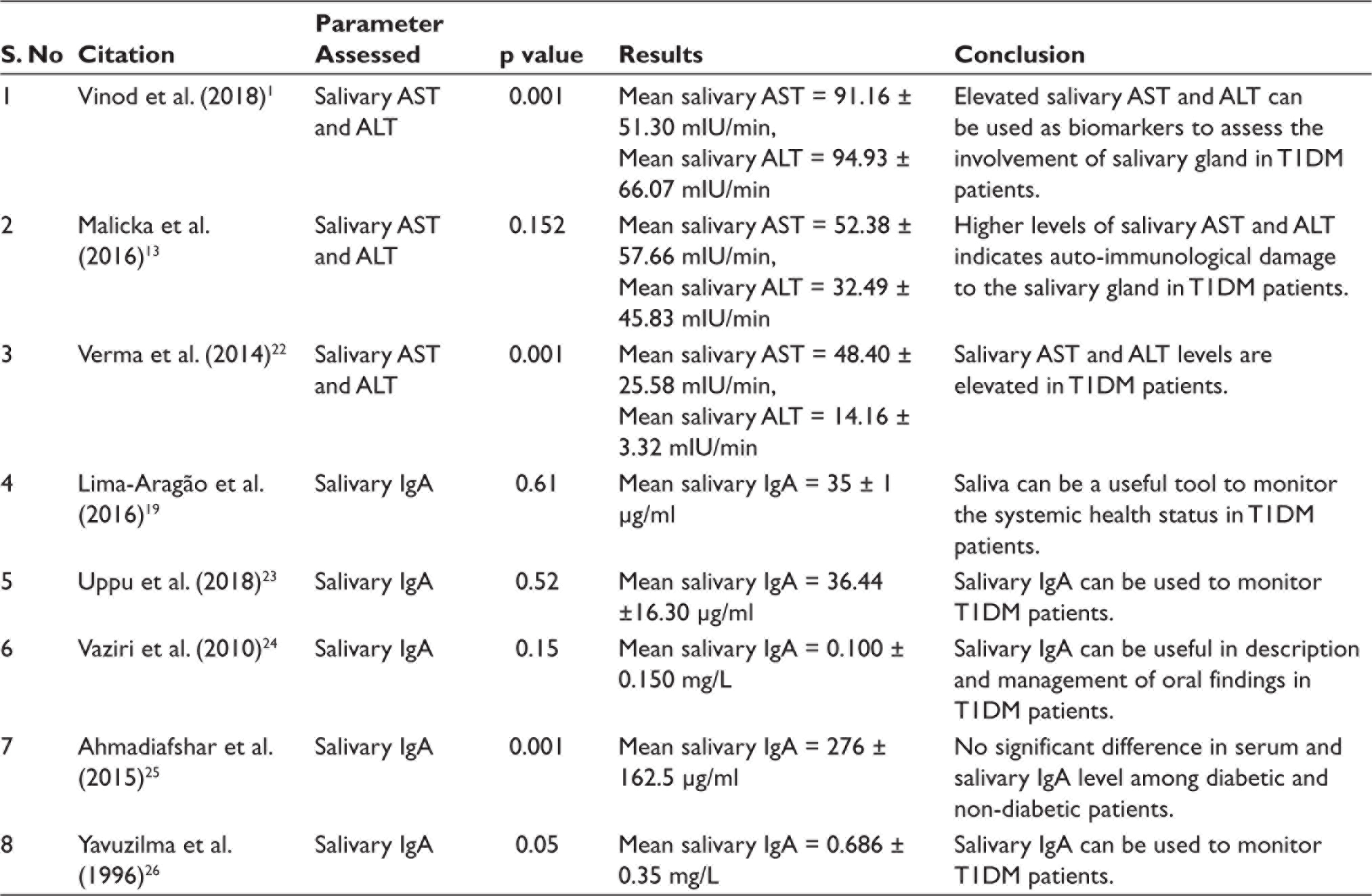
Table of Included Studies.
PRISMA Flowchart.
Description of Included Studies
The review included studies that mainly focused on the evaluation of potential of salivary AST, ALT and IgA in the diagnosis of T1DM. Out of eight studies that were included in the review, three studies (37.5%) assessed the salivary levels of AST and ALT, while five studies (62.5%) evaluated the levels of salivary IgA. The sample size of the selected 8 articles consisted of a total of 456 cases and 325 healthy controls, in which salivary levels of AST, ALT and IgA were assessed.
Two studies by Vinod et al. 1 and Verma et al. 22 noted a significant elevation of salivary AST and ALT levels in T1DM patients, when compared to healthy individuals, and concluded that these can be used as diagnostic biomarkers. The study by Malicka et al. 13 demonstrated elevated levels of salivary AST and ALT in T1DM patients, but the difference was not statistically significant.
Salivary IgA levels were reported to be significantly overexpressed among T1DM patients in two studies by Lima-Aragão et al. 19 and Yavuzilma et al. 26 Additionally, Lima-Aragão et al., 19 Uppu et al. 23 and Vaziri et al. also noted an increase in the expression of salivary IgA in T1DM patients compared to healthy volunteers, but this difference was not statistically significant.
Saliva collection methods were not uniform across the included studies. Unstimulated whole saliva was collected in five studies, the passive drool method was used in two studies and the Navazesh method was used in one study for saliva collection. The method employed to quantify the salivary levels of the biomarkers also varied, including the Nephelometric method, enzyme-linked immunosorbent assay, radial immuno-diffusion method, immuno-enzymatic colorimetric method and reflectance photometer.
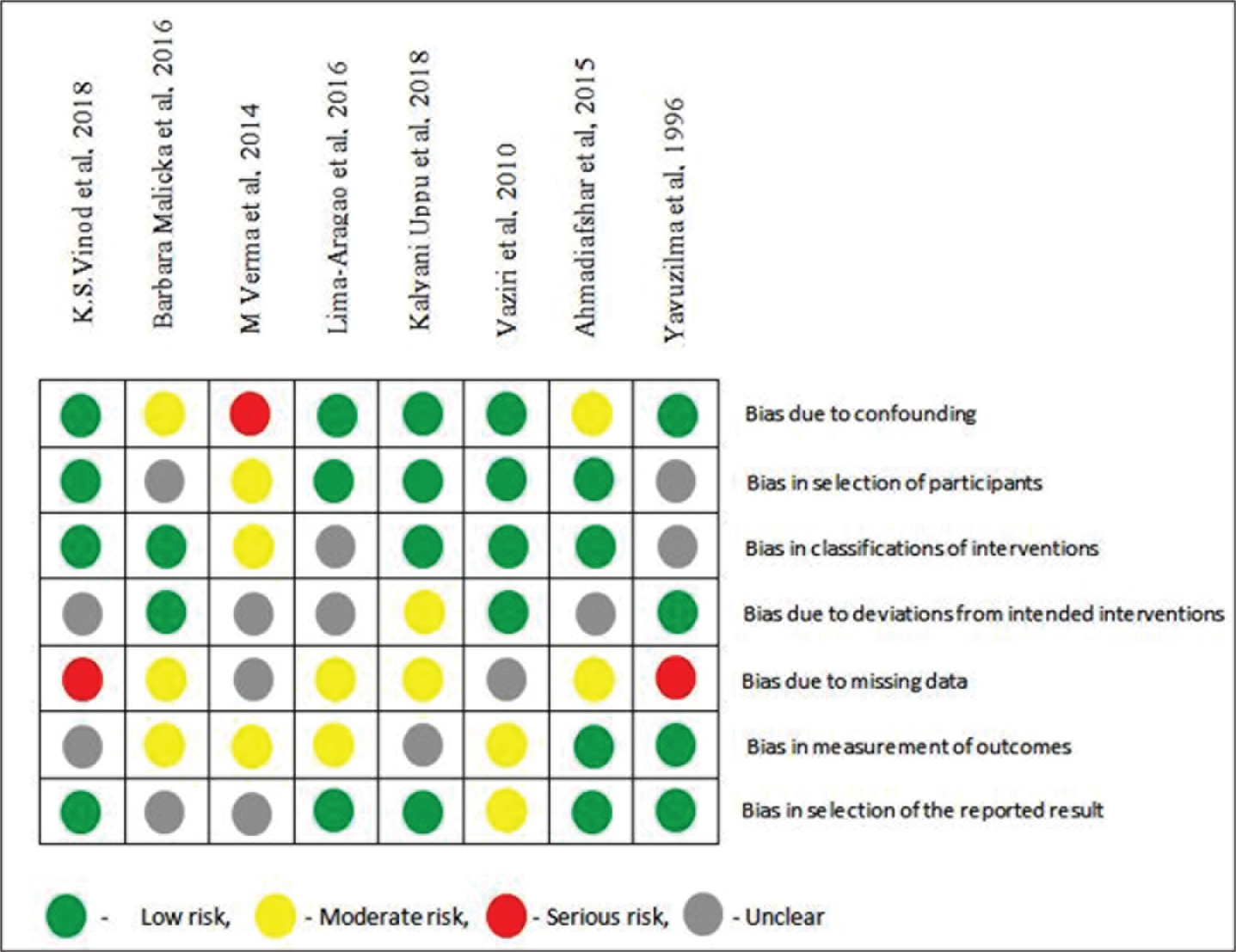
Risk of Bias Assessment
Figure 2 depicts the risk of bias assessment for non-randomized studies. Three studies had a high risk of bias, while six studies had a moderate risk of bias. One study had a high risk of bias due to confounding, while two studies had a high risk of bias due to missing data. All eight studies had an unclear risk of bias in multiple categories.
Risk of Bias Assessment.
Discussion
The current review represents a comprehensive analysis of all the studies published on the evaluation of salivary levels of AST, ALT and IgA in T1DM patients.
One of the most common complications of T1DM is tissue destruction. 10 Injury to cells with disruption of the plasma membrane leads to elevation of aminotransferases. 11 The elevation of serum AST and ALT indicates cell damage, especially in the case of liver and cardiac tissues.5,12 There is a significant overlap in protein content between saliva and plasma. Since saliva is easily available and cheap, it can be used as a non-invasive diagnostic tool. 1
In the present review, among the three studies that analyzed the salivary levels of AST and ALT, studies by Verma et al. and Malicka et al. 13 found a threefold to fourfold increase in their expression in T1DM patients when compared to healthy individuals.1, 13 Another study noted a positive association between the glycosylated haemoglobin level and salivary ALT. 13 In their study, Musumeci et al. 15 hypothesized that elevated salivary AST and ALT expression in T1DM patients could be a result of salivary gland cell damage by lymphocytic infiltration, which has been proven histopathologically.14, 15 This was supported by Burrack et al., 16 who stated that salivary glands were subjected to the activity of autoimmune cells, either through action of circulating antibodies directed against beta cells of pancreatic islets or through specific antibodies against salivary gland antigens. 16 However, none of the included studies assessed the sensitivity, specificity or diagnostic accuracy of salivary AST and ALT.
Among the five studies that assessed the level of salivary IgA, two studies by Lima-Aragao et al. 19 and Yavuzilma et al. showed a significant increase in its expression among T1DM patients. In their study, Bachrach et al. 17 suggested that alterations in the composition of whole saliva can be utilized to detect possible variations and impaired host immune responses, including the secretion of IgA. Local factors such as increased plaque accumulation and calculus deposition could also be responsible for elevated IgA in these patients.18, 19 In their study, Mestdagh et al. 20 postulated that overexpression of salivary IgA in T1DM patients could be due to the presence of specific anti-insulin and anti-Streptococcus mutans antibodies. However, none of the included studies assessed the sensitivity, specificity and diagnostic accuracy of salivary IgA in T1DM patients.
Saliva collection methods among the studies included unstimulated whole saliva collection in five studies, the passive drool method in two studies and the Navazesh method in one study. In their study, Granger et al. 21 suggested that using the passive drooling method, large volume of saliva could be collected in a short duration, with minimal potential sources of error. The method of saliva storage was not elaborated in any of the included studies.
Clinical Significance
Although their diagnostic accuracy remains to be assessed completely, this review highlights that salivary levels of AST, ALT and IgA are significantly elevated in T1DM patients when compared to healthy individuals. Thus, these salivary biomarkers can be used as a non-invasive alternative for the early diagnosis of T1DM patients.
Limitations
Although a systematic approach was adopted, this review has some limitations. The limitations include different saliva sample collection methods, variability in the detection method used for quantification of the biomarkers, and the different units of measurement used across the studies. Studies have proved that there could be an inconsistency in results due to the difference in saliva collection and detection methods. 19 Due to the heterogeneous nature of included studies, a meta-analysis could not be done. Hence, comments on sensitivity, specificity and diagnostic accuracy cannot be made.
Future Scope
Further high-quality studies with an adequately large sample size and uniformity of assessment are essential to investigate the sensitivity, specificity and diagnostic accuracy of the salivary biomarkers.
Conclusion
Our review reflects that salivary AST, ALT and IgA are elevated in T1DM patients, and they can be used for diagnosis and monitoring of these patients. Further studies are needed to establish the diagnostic accuracy of these parameters for clinical application.
Footnotes
Authors’ Contribution
Rahul Mohandas: Conceptualization, Literature search, Analysis and interpretation of data, Manuscript writing.
Pratibha Ramani: Study supervision, Manuscript review, Validation.
Subhashree Mohapatra: Literature search, Analysis and interpretation of data, Manuscript editing.
Data Availability Statement
The data that supports the findings of this study are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval Institutional Statement
The institutional review board deemed that ethical approval was not necessary, as this was a systematic review where electronically available data was retrieved and evaluated retrospectively.
Funding
The author received no financial support for the research, authorship and/or publication of this article.
Abbreviations
ALT: alanine aminotransferase
AST: aspartate aminotransferase
T1DM: Type 1 diabetes mellitus
