Abstract
Aim:
The aim of this study was to evaluate the effect of the ozonated water on corrosion of a cobalt–chromium (Co-Cr)-based alloy, which is applied for the fabrication of metal frameworks of removable partial dentures.
Materials and Methods:
In this in vitro study, a total of 30 disk-shaped samples of a Co-Cr alloy were papered and randomly divided into two groups of 15 specimens. In group 1 (control), the specimens were stored in distilled water (DW), and in group 2, the specimens were stored in ozonated water. Around 90 immersions were performed, and the weight change of each specimen was determined. The ion release was analyzed using an inductively coupled plasma-optical emission spectrophotometer. The potentiodynamic polarization test was performed for each group to assess the corrosion resistance of the Co-Cr alloy. The statistical analysis was performed using SPSS version 22. Data were analyzed by independent samples’ t-test.
Results:
The results showed no significant difference between the weight changes of the two groups. The test using an inductively coupled plasma-optical emission spectrophotometer demonstrated no significant difference between the groups in Co and Cr ions release. In the potentiodynamic polarization test, both groups present similar corrosion behavior, and ozonated water has no deleterious effect on the corrosion resistance and passive range of the Co-Cr alloy compared to DW.
Conclusion:
As compared to DW, ozonated water has no significant deleterious effect on the corrosion resistance of the Co-Cr frameworks and can be used for cleaning the removable partial dentures.
Introduction
Despite the reduced rate of tooth loss, the need for removable partial dentures (RPDs) remains high and will continue to rise in the future. 1 RPDs provide simple and cost-effective treatment to restore missing teeth in millions of partially edentulous individuals worldwide. 2 Accumulation of microorganisms on acrylic resins is one of the important challenges in using these materials. 3 Proper cleaning of RPDs is one of the important issues that should be considered to keep a healthy oral mucosa. Biofilm accumulation and microbial colonization on RPDs can increase the incidence of oral and periodontal diseases and caries among RPD wearers. 4 Several methods are recommended for cleaning the RPDs, including chemical, mechanical, or a combination of these methods.
Brushing is the most common mechanical cleansing method for RPDs, but it is difficult for some patients who have motor incoordination and low dexterity. 5 In addition, brushing the acrylic resin denture bases may increase the surface roughness, and consequently increase the formation and adhesion of the biofilm. 6
Alkaline peroxides, alkaline hypochlorite, acids, disinfectants, and enzymes are cleaners, which are used in chemical methods. The chemical method can dislodge food debris, biofilm, and tobacco stains from the RPD surface effectively. This method has some disadvantages and limitations. For example, sodium hypochlorite is capable of whitening acrylic resins and removing corrosion of the metal components of RPDs. In addition, several studies have demonstrated the short-term and long-term effects of denture cleaners, such as Efferdent (Pfizer Consumer Health Care, Scarborough, Ontario, Canada), chlorhexidine, peroxide, and other chemical cleaners, on the color stability of dentures. It has been reported that this negative effect is increased during the time by repeated uses of these cleaners. 7
It must be considered that a disinfection method should be effective without deleterious effects on the properties of materials used in RPDs. 8 The surface changes of the acrylic resin have been extensively studied with various cleaning methods; however, concerning the metallic frameworks of RPDs, the literature is scarce. 9
Cobalt–chromium (Co-Cr) alloys are among the best-known base metal alloys in dentistry applied for the fabrication of metal frameworks of RPDs. 10 Co improves the mechanical features of the alloy, and Cr and other elements increase the corrosion resistance, biocompatibility, and mechanical properties of the Co-Cr alloy. 11 The biocompatibility of the Co-Cr alloy is related to the corrosion resistance and ion release in the oral environment. Metal ions released in the oral cavity from dental prosthesis alloys may damage the cellular metabolism or proliferation and lead to hypersensitivity or allergies. 12 It has been shown that the chemical disinfection method can have negative effects on the corrosion resistance of the Co-Cr frameworks.13,14
The application of ozone for various aspects of dentistry has been proposed because of its antimicrobial, disinfectant, biocompatibility, and healing properties.15,16 In this regard, ozone therapy has been shown to be effective for the acceleration of wound healing, treatment of dental caries, oral lichen planus, periodontal diseases, halitosis (bad breath), osteonecrosis of the jaw, postoperative pain, teeth whitening, plaque and biofilm inhibition, disinfection of the root canal, dentin hypersensitivity, and temporomandibular joint disorders. 16 Ozone has powerful antimicrobial activity against bacteria and fungi, such as Candida albicans, Streptococcus mutans, and Pseudomonas aeruginosa.17–19 A recent study 20 demonstrated that an immersion of the denture base with acrylic resins in ozonated water that was prepared by a home ozone generator could inhibit Candida growth and colonization on the denture base acrylic resins. However, it must be investigated to ensure that ozonated water has no significant negative effect on the corrosion resistance of the Co-Cr frameworks. In line with the mentioned study, 20 we evaluated the effect of the ozonated water on corrosion of a Co-Cr-based alloy, which is used for the production of metallic frameworks of RPDs. The null hypothesis was that the ozonated water does not influence the corrosion behavior of the studied Co-Cr.
Materials and Methods
This study was designed as an in vitro research and was performed in the Dental Materials Laboratory of School of Dentistry of Mashhad University and Central Laboratory of Ferdowsi University of Mashhad, Mashhad, Iran. This research was conducted from May 2020 to October 2020. The necessary ethics committee approval for the study was obtained from the Research Ethics Committee of Mashhad University of Medical Sciences (Protocol No: IR.MUMS.DENTISTRY.REC.1398.063). The sample size was estimated according to an 80% power analysis at the 95% confidence interval based on a pilot study. The simple random sampling method was used for sample selection by using a free internet-based program. In this in vitro study, the corrosion behavior of Co-Cr in the ozonated water was studied using three different methods, including weight loss, ion release, and potentiodynamic polarization curves. A total of 30 disk-shaped samples of Flexicast (American Dent-All, Glendale, CA, USA) Co-Cr alloy (Table 1) were papered with a diameter of 12 mm and a thickness of 3 mm.
Chemical Composition of the Investigated Co-Cr-Mo Alloy
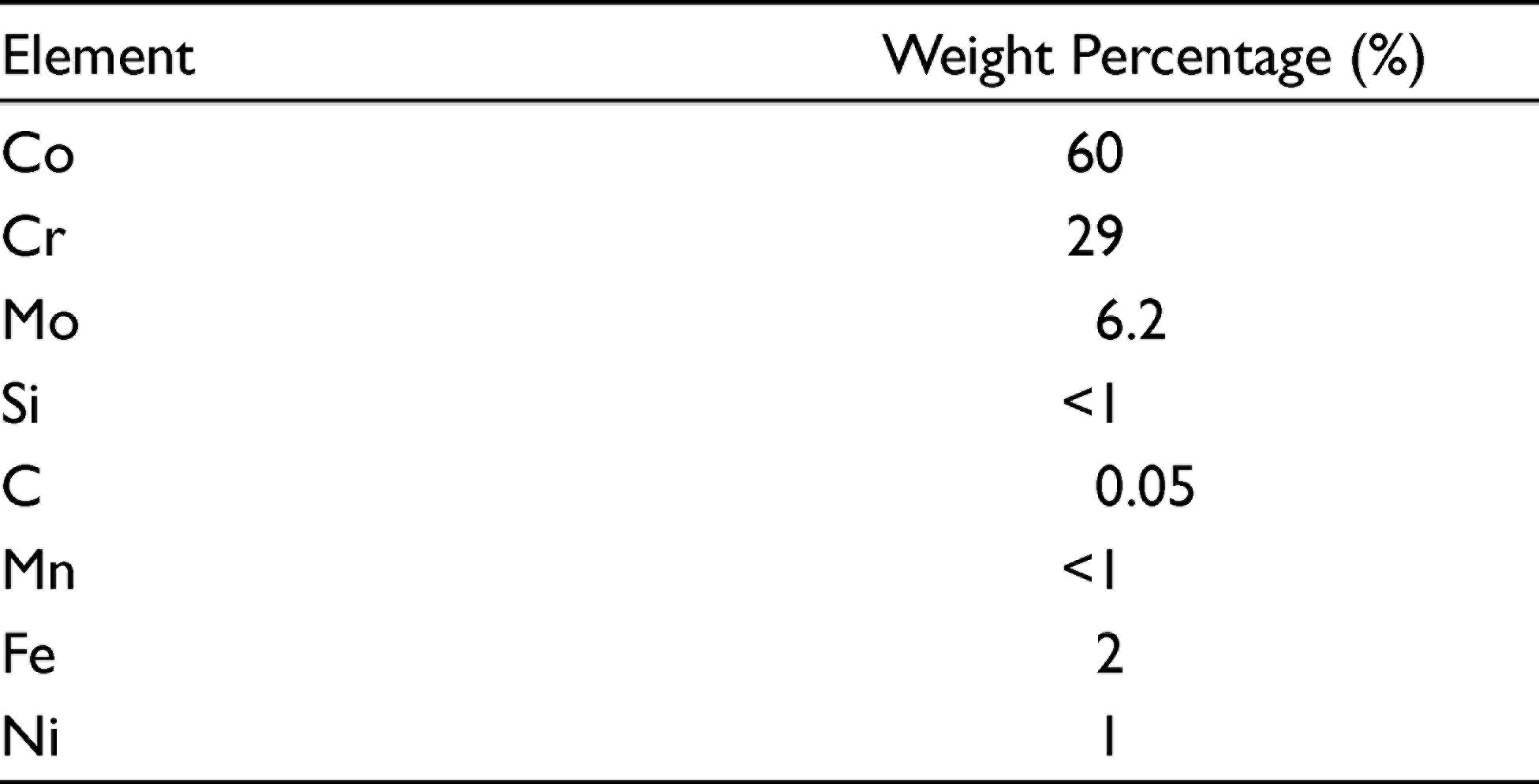
After casting, the specimens were polished with a 1500 grit sandpaper. The samples were cleaned using ethanol and an ultrasonic device, dried with an absorbent paper, and randomly divided into two groups of 15 specimens. In both groups, each specimen was weighed in an analytical balance (HR-200, A&D Company Ltd, Japan) with a precision of 0.1 mg, which was calibrated before each measurement.
In group 1 (control), samples were immersed in distilled water (DW) at 37ºC ± 1ºC for 8 hours, and in group 2, the specimens were immersed in ozonated water (1 ppm) produced by a home ozone generator (ARDA, MHP1H, Iran) for 8 hours (Figure 1).
Immersion of the Specimen in Distilled Water

To simulate the daily hygiene of RPDs for three months, 90 immersions were performed in each group, and all solutions were renewed every 8 hours. An immersion of 24 hours was equaled with three immersions of 8 hours per day. Therefore, the samples were immersed for 30 continuous days. In each group, five specimens were selected randomly and their solutions were collected and stored in a container during the study. After 30 days, the second weight measurements were done, and the difference between the first and second weighings of each specimen was calculated.
The solution of each container was analyzed by an inductively coupled plasma-optical emission spectrophotometer (Spectro Arcos 7600/CRMA, Germany) to recognize the Co and Cr ions.
The potentiodynamic polarization is probably the most commonly used electrochemical technique for measuring the corrosion resistance. 21 The potentiodynamic polarization test for each group was done using the three-electrode electrochemical cells—calomel reference electrode, platinum electrode, and the working electrode (Co-Co alloy) attached to the potentiostat/galvanostat (WonATech, ZIVE SP1). Polarization curves were recorded between −250 and 600 mV per second with a scan rate of 1 mV/s.
Results
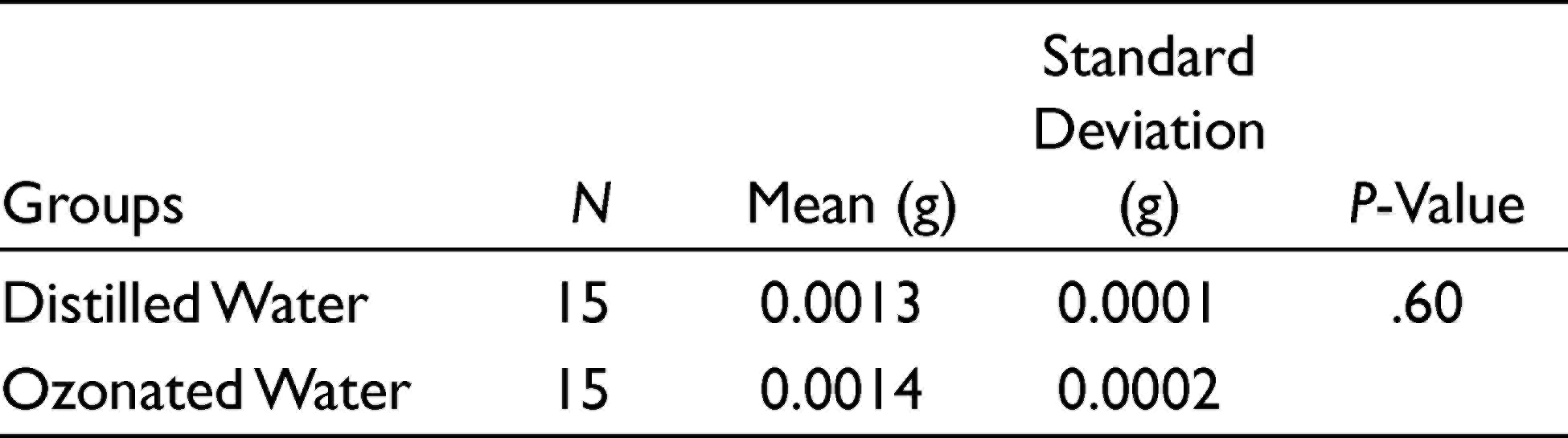
Descriptive statistics of the weight changes of the two groups are tabulated in Table 2. Since the data had a normal distribution, an independent t-test was used. The independent t-test demonstrated that there was no significant difference (P = .60) between the weight changes of the two groups.
Comparison of Weight Loss of the Co-Cr Alloy into the Distilled Water and Ozonated Water
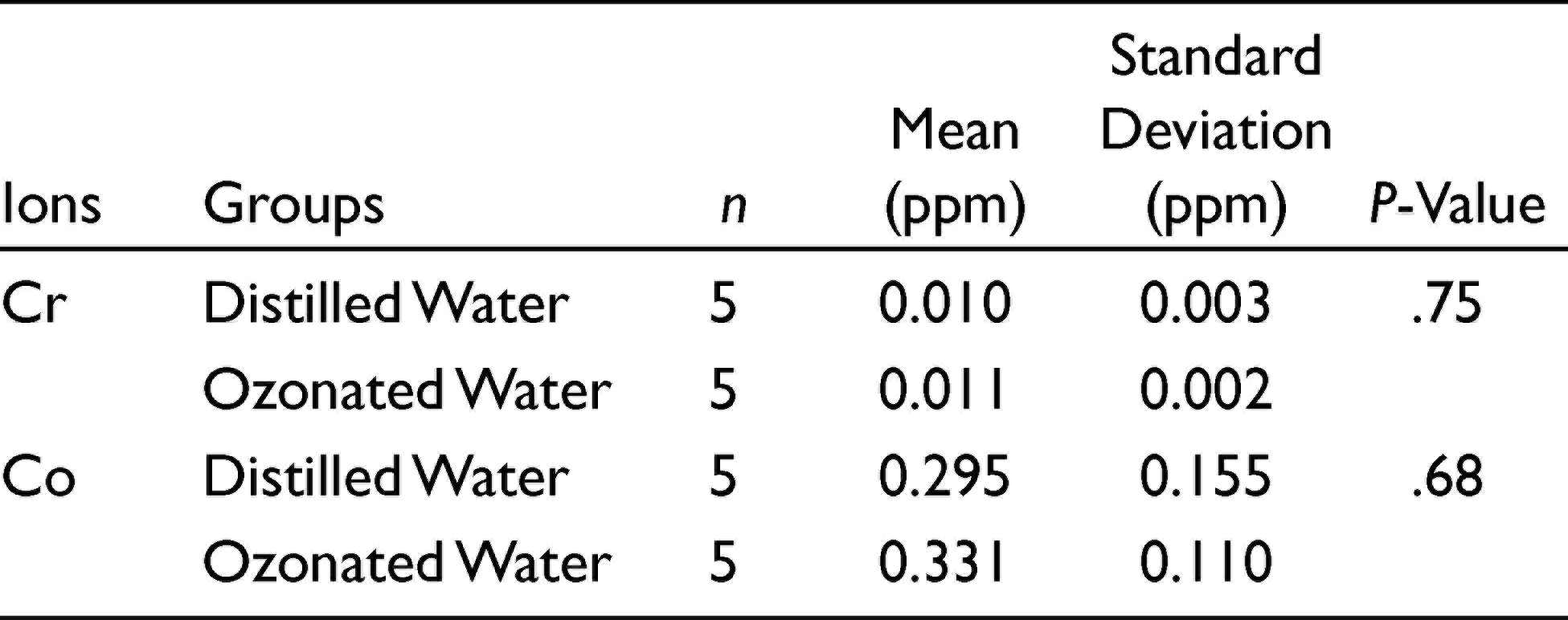
The values of the release of Co and Cr ions are presented in Table 3. The independent t-test demonstrated no significant difference between the groups in Co and Cr ion release (P > .05). As can be seen, Cr ion release was much lower than Co ion release.
Comparison of Co and Cr Ion Release From the Co-Cr Alloy into the Distilled Water and Ozonated Water
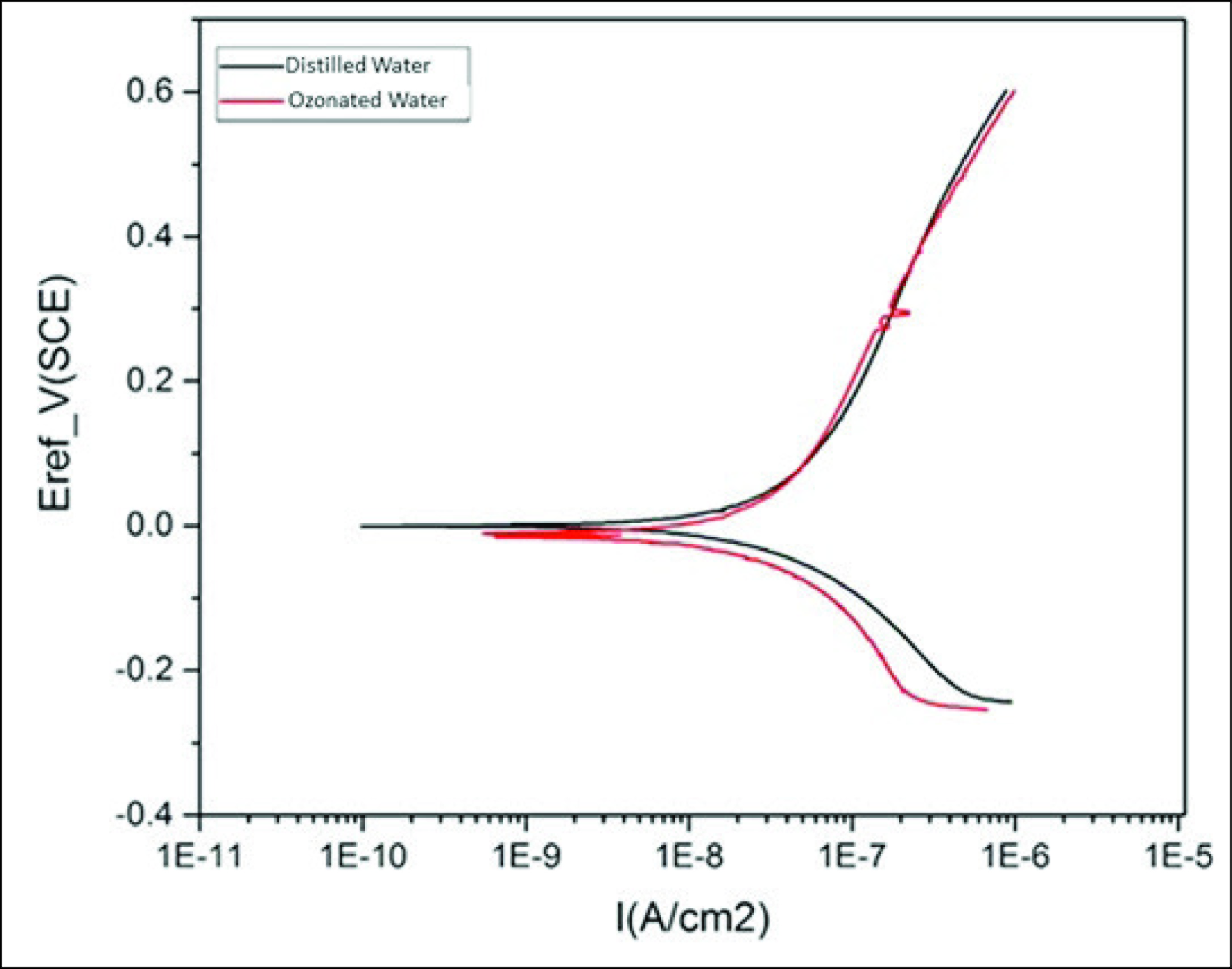
The potentiodynamic polarization curves for the Co-Cr alloy, immersed in DW and ozonated water, are presented in Figure 2. As can be seen, both groups present similar corrosion behavior, and ozonated water has no deleterious effect on the corrosion resistance and passive range of the Co-Cr alloy compared to DW.
The Potentiodynamic Polarization Curves for Co-Cr Alloy, Immersed in Distilled Water and Ozonated Water
Discussion
Our data showed that there were no significant differences in weight loss, ion release, corrosion resistance, and passive range of the Co-Cr alloy between ozonated water and DW (control). Therefore, the null hypothesis was accepted. In addition, it was observed that Cr ion release was much lower than Co ion release, which is because of the very low release rate of the passive layer formed on Cr. 22
Felipucci et al. 13 investigated the effect of different cleansers on the weight and ion release of RPDs. Their results showed that Corega Tabs (Stafford-Miller Ind., Rio de Janeiro, RJ, Brazil) and Medical Interporous (MST Laboratories AG, Vaduz, Liechtenstein) had higher values of weight loss with higher changes in VeraPDI (Aalba Dent. Inc., Cordelia, CA, USA) alloy compared to DeguDent (Dentsply Ind. e Com. Ltda, Sao Paulo, SP, Brazil). The solutions that caused more ion release were NaOCl (Q-Boa, Anhembi S/A, Osasco, SP, Brazil) and Medical Interporous. Curylofo et al. 9 studied the effect of different denture cleansers—effervescent tablets (NitrAdine, Bonyf AG, Vaduz, Liechtenstein), cetylpyridinium chloride (Cepacol, Sanofi-Aventis Farmaceutica Ltda, Suzano, SP, Brazil), and experimental solutions of Ricinus communis (Instituto de Química de Sao Carlos, Sao Carlos, Brazil)—on the surface of the Co-Cr alloy for a period of five years. Their results showed no change in the surface roughness of the Co-Cr alloy immersed in the solutions for a five-year simulated period of use.
Keyf and Güngör 23 observed that Corega Tabs, Axion (Colgate Palmolive Co., Turkey), and tap water have no significant effect on the weight of the Co-Cr alloy. Kiesow et al. 24 evaluated the denture materials and antimicrobial activity of consumer products commonly used to clean the dentures. They observed that the bleach was highly antimicrobial but incompatible with metal dental prosthesis components. Their results showed that isopropyl alcohol and mouthwash were antimicrobial but damaged the acrylic resin. In addition, specialist denture cleanser tablets (Polident Triplemint) gave a good combination of antimicrobial efficacy and reasonable material compatibility. In another study, Garcia et al. 25 found that commercial cleaners containing hydrogen peroxide could remove the pellicle without affecting the surface hardness and roughness of either resin or the Co-Cr alloy.
In other studies, the effect of the ozonated water on other components of the RPDs was investigated. Mirmortazavi et al. 20 evaluated the antifungal effects of ozonated water on the adhesion of the C. albicans to the surface of the acrylic resin. Their results showed that the ozonated water could inhibit Candida growth and colonization on the acrylic resin. Estrela et al. 26 observed that the addition of ozone to an ultrasonic cleaning system containing different experimental solutions resulted in antibacterial activity against Staphylococcus aureus. Nakhaei et al. 27 investigated the effect of ozonated water and two common denture cleaners on the tensile bond strength and hardness of a silicone soft liner. They observed that ozonated water and cleansing tablets (Corega) have a lower effect on the hardness of soft denture liners than that of 0.5% NaOCl.
Our research gives important knowledge about the effect of the ozonated water on corrosion of a Co-Cr-based alloy, which is applied for the fabrication of metal frameworks of RPDs. The present study did not consider all materials used in RPDs, such as acrylic resin, so this can be considered as the limitation of the study. Further research has to be focused on the effect of ozone on the surface properties of the acrylic resin (e.g., roughness and hardness) used in the RPD frameworks.
Conclusion
Despite the limitations of this study, it can be concluded that as compared to DW, ozonated water has no significant deleterious effect on the corrosion resistance of the Co-Cr frameworks and can be used for cleaning the RPDs.
Footnotes
Acknowledgements
This study was the result of Reyhaneh Imani thesis (Thesis no. 3103). We would like to thank the Vice Chancellor for Research, Mashhad University of Medical Sciences, for financial support (grant no. 971723) to conduct this study.
Funding
This study was supported by the Vice Chancellor for Research of Mashhad University of Medical Sciences for financial support (grant no. 971723).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
