Abstract
Aim:
This in vitro study was aimed to evaluate the effect of adding different concentrations of chitosan nanoparticles (NPs) and TiO2 NPs on the shear bond strength (SBS) of an orthodontic adhesive.
Materials and Methods:
In this in vitro study, 72 extracted human premolars were embedded in an acrylic resin and randomly allocated into four groups of 18 specimens. In group 1 (control), brackets were bonded to the tooth with the Transbond XT orthodontic adhesive. In groups 2, 3, and 4, 0.5% chitosan NPs and 0.5% TiO2 NPs, 1% chitosan NPs and 1% TiO2 NPs, and 1.5% chitosan NPs and 1.5% TiO2 NPs were added to Transbond XT, respectively. Then, the brackets were bonded by the modified adhesive. The SBS and adhesive remnant index (ARI) of each group were assessed with a universal testing machine. The SBS test results were analyzed using one-way analysis of variance followed by the posthoc Tukey’s honestly significant difference (HSD) test. The Kruskal–Wallis test was also applied to evaluate the ARI scores.
Results:
The results showed no statistically significant difference between groups 1, 2, and 3, but SBS decreased significantly in group 4. With increasing the concentration of NPs up to 1% chitosan NPs and 1% TiO2 NPs, SBS did not change significantly. However, in 1.5% chitosan NPs and 1.5% TiO2 NPs, SBS decreased compared to the other three groups. No significant differences were found between the groups in terms of ARI scores.
Conclusion:
It is concluded that the orthodontic composite containing 1% chitosan NPs and 1% TiO2 NPs has adequate SBS for use in the clinical setting.
Introduction
As the demand for orthodontic treatments is increasing, researchers make more attempts to develop methods for preventing white spot lesions (WSLs) during fixed orthodontic treatment. 1 WSLs develop around brackets, bands, arch wires, ligatures, and other attachments because these appliances make it difficult to perform oral hygiene completely and lead to prolonged plaque accumulation and caries lesions. 2 Some investigations reported the incidence of WSLs during fixed orthodontic treatment in 73% to 95% cases.3,4 Clinically, WSLs might develop rapidly and can be observed only after a fixed orthodontic appliance is fitted.5,6 WSLs are commonly observed on the buccal aspects of teeth around the brackets, especially in the gingival area. 7 One way to overcome this problem is the active cooperation of orthodontic patients to perform proper oral hygiene that needs patient motivation, clear professional instructions, and adequate tools.8,9 This method is not essentially reliable and requires patient cooperation. 10 It is absolutely necessary to find optimal preventive strategies independent of patient cooperation. 11 The addition of antimicrobial agents into orthodontic adhesives or coatings on brackets/wires or remineralizing agents adjacent to orthodontic appliances is the approach that can be used without patient cooperation.11,12
In recent years, antimicrobial nanoparticles (NPs) have been widely used in various fields of dentistry.13,14 The large surface area and high charge density of NPs help them interact with bacterial cells more effectively and, consequently, increase the antimicrobial activity. 15
TiO2 NPs have recently attracted much attention in various fields of dentistry and medicine.
16
TiO2 nanomaterial has an efficient bactericidal effect and a great potential to reduce microbial adhesion on various dental materials such as prosthesis, acrylic resin, ceramic glass, and dental implants.
17
Alrahlah et al.
18
in their study showed that TiO2 NPs increased the antimicrobial activity of polymethyl methacrylate (PMMA) by significantly reducing bacterial adherence. Elsaka et al.
19
found that conventional glass-ionomer restorative-containing TiO2 NPs possessed a potent antibacterial effect. Salehi et al.
20
reported in their study that nitrogen-doped TiO2-coated orthodontic brackets showed a strong antimicrobial property against
Chitosan is a natural antimicrobial material with a wide range of dental applications. It has many favorable properties such as biocompatibility, hydrophilicity, biodegradability, and a broad antimicrobial activity against Gram-negative and Gram-positive bacteria and fungi.21,22 Chitosan has been studied in various fields of dentistry including the development of toothpastes, enamel repair, modification of dental restorative materials, coating on implants, adhesion, dentin bonding, and acrylic resin.21,23 de Paz et al.
24
in their study found that chitosan NPs prepared from low-molecular-weight chitosan had a high antimicrobial effect on
The addition of chitosan NPs and TiO2 NPs into orthodontic adhesives for their synergistic antimicrobial effect can be considered as an effective strategy to overcome WSLs and caries. However, it must first be examined whether the modification of adhesives with chitosan NPs and TiO2 NPs has no significant deleterious effect on the shear bond strength (SBS) of orthodontic adhesives. To the best of our knowledge, there have been no studies investigating the effects of this modification on the SBS of orthodontic adhesives. Therefore, the present study aimed to assess the effect of incorporating various weight percentages (w/w) of chitosan NPs and TiO2 NPs on the SBS of an orthodontic adhesive.
Materials and Methods
Setting and Design
This in vitro study was performed in the Dental Materials Laboratory of School of Dentistry of Mashhad University, Mashhad, Iran. This research was conducted from September 2019 to February 2020. After a receipt of approval from the Research Ethics Committee of Mashhad University of Medical Sciences (Protocol No: IR.MUMS.DENTISTRY.REC.1397.009), 72 freshly extracted human premolars were collected from the patients that were referred to the clinic of School of Dentistry in Mashhad University of Medical Sciences. The inclusion criteria for tooth selection were the absence of caries, restorations, cracks, or surface defects. The sample size was estimated according to 80% power analysis at the 95% confidence interval using G power version 3.1.9.7. It was determined to be 18 samples for each group.
Sample Preparation
Chitosan NPs were prepared by ionic gelation with sodium tripolyphosphate (TPP). Chitosan (low molecular weight, Sigma-Aldrich, St. Louis, MO, USA) was dissolved in a 1% acetic acid (Merck, Germany) solution and stirred at room temperature until the solution became clear. Then, the TPP (Sigma-Aldrich, St. Louis, MO, USA) solution was added dropwise into the chitosan solution under magnetic stirring with pH adjusted to 9 using NaOH (Merck, Germany). Finally, the precipitate was lyophilized to obtain chitosan NPs. Moreover, anatase TiO2 NPs (US-NANO, USA) were used in this study.
A total of 72 freshly extracted healthy human premolar teeth were disinfected in a 0.1% thymol solution for one week. The root of each tooth was placed in a cylindrical mold and filled with a cold-cured acrylic resin. All the tooth samples were cleaned with fluoride and oil-free pumice, rinsed for 10 s, air-dried and acid-etched according to the manufacturers’ instructions with 37% phosphoric acid for 30 s. The teeth were randomly divided into four groups (
Group 1 (control): Bonding with the orthodontic adhesive (Transbond XT, 3M Unitek, USA);
Group 2: Bonding with Transbond XT containing 0.5% chitosan NPs and 0.5% TiO2 (anatase) NPs;
Group 3: Bonding with Transbond XT containing 1% chitosan NPs and 1% TiO2 (anatase) NPs;
Group 4: Bonding with Transbond XT containing 1.5% chitosan NPs and 1.5% TiO2 (anatase) NPs.
Study Method
NPs were added to composites in a dark room after being weighed by a digital scale (HR-200, A&D Company Ltd, Japan) with a precision of 0.1 mg. For example, to prepare Transbond XT containing 0.5% chitosan NPs and 0.5% TiO2 NPs, 4 g Transbond XT was mixed with 0.02 g chitosan NPs and 0.02 g TiO2 NPs.
Stainless steel orthodontic brackets (Dentaurum, Discovery, Germany) were bonded to the buccal surface of the teeth according to the manufacturer’s instructions by one operator who was blinded to group allocation. The Transbond XT primer was applied on the acid-etched enamel and a uniform thin layer of the orthodontic adhesive was applied on the bracket base. The bracket was placed on the buccal enamel surface and then was light cured. All the samples were stored in distilled water for 24 hours at 37°C in an incubator and then were thermocycled for 1000 cycles between 5°C and 55°C with a dwell time of 30 s.
Afterward, each specimen was positioned in a universal testing machine (STM20, SANTAM, Tehran, Iran) to test SBS at a crosshead speed of 1 mm/min (Figure 1). The shear force was applied in a direction parallel to the bracket base. SBS was calculated by dividing the shear load with the surface area of the bracket base.
Experimental Setup of the SBS Test. The Force Was Applied Until the Bracket Became Detached From the Tooth

After debonding, the amount of adhesive remaining on the enamel surface was examined under a stereomicroscope at 10× magnification. The adhesive remnant index (ARI) score was determined as follows:
0: no adhesive remained on the enamel surface. 1: less than 50% of the adhesive remained on the enamel surface. 2: more than 50% of the adhesive remained on the enamel surface. 3: all the adhesive remained on the enamel surface.
Statistical Analysis
The statistical analysis was conducted using the SPSS software version 22 (SPSS Inc., Chicago, IL, USA). The SBS test results were analyzed using one-way analysis of variance (ANOVA) followed by the posthoc Tukey’s HSD test. The Kruskal–Wallis test was also applied to evaluate the ARI scores.
Results
The descriptive statistics of SBS values for the four groups are tabulated in Table 1. The lowest SBS was found in the control group. One-way ANOVA demonstrated a significant difference concerning SBS among the groups (
Descriptive Statistics of SBS Values for the Four Groups
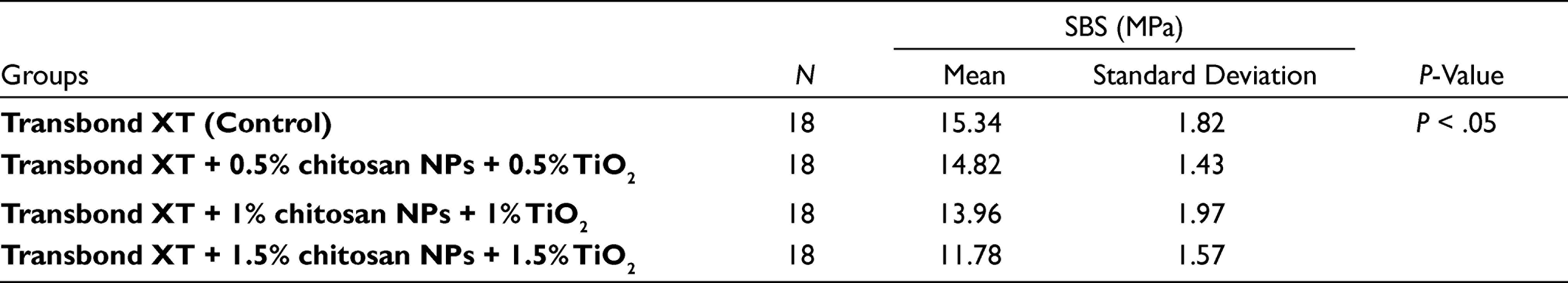
Abbreviations: SBS, shear bond strength;
Posthoc Tukey’s Multiple Comparison Test Between Groups for SBS
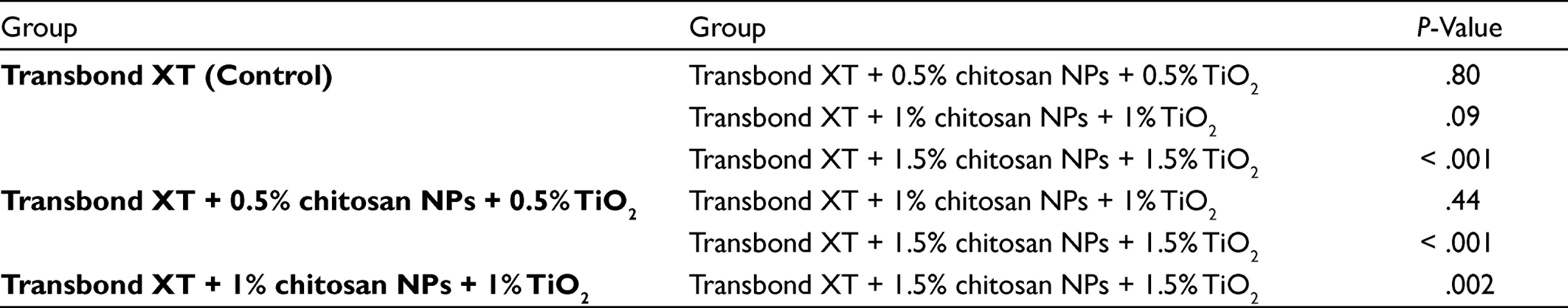
Abbreviation: SBS, shear bond strength.
The mean ARI scores of the four groups after debonding are shown in Table 3. The Kruskal–Wallis test demonstrated no significant difference (
Frequency Distribution of ARI Scores for Four Groups
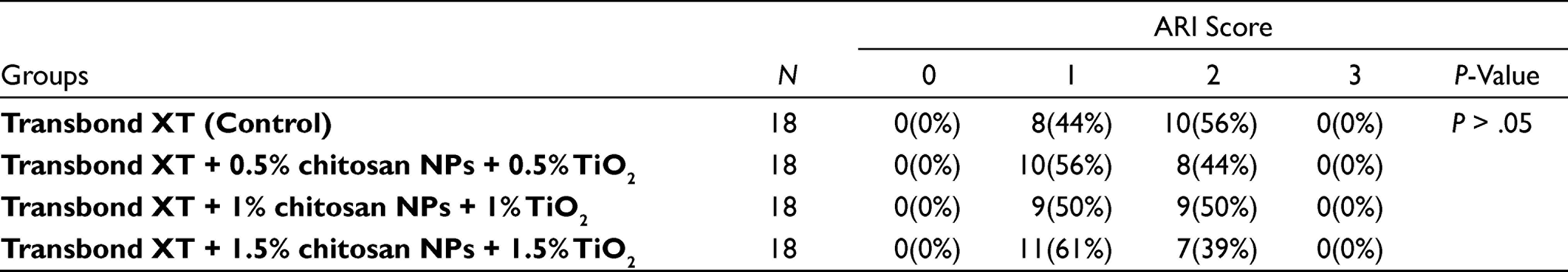
Abbreviations: ARI, adhesive remnant index;
Discussion
The bond strength of the bracket bonding system has a major role in withstanding against orthodontic forces and other mechanical forces in the oral cavity to achieve the precise control of tooth movement. 28 In the present study, we evaluated the SBS of an orthodontic adhesive containing different concentrations of chitosan NPs and TiO2 NPs. Our results showed that SBS did not change significantly with increasing the concentration of NPs up to 1% chitosan NPs and 1% TiO2 NPs, but decreased in 1.5% chitosan NPs and 1.5% TiO2 NPs. In other words, the combination of 1% chitosan NPs and 1% TiO2 NPs was the maximum amount with no significant negative effect on SBS. The dispersion of particles in the polymer matrix, the NPs–matrix interfacial adhesion, and the agglomeration of NPs are significant factors that could affect the mechanical properties of composite resins (adhesives). Chitosan NPs and TiO2 NPs in adhesives may have impurities in the polymer matrix that may decrease SBS in composite resins. The addition of NPs may have adverse effects on the degree of conversion in the polymerization of composite resins. Chitosan NPs and TiO2 NPs may agglomerate, and agglomerated particles act as stress-concentrating centers in the polymer matrix. These factors may cause a decrease in SBS.
However, a study by Poosti et al. 29 reported that the addition of 1% TiO2 NPs to the Transbond XT adhesive had no significant effect on SBS. Mirhashemi et al. 30 demonstrated that a mixture of chitosan NPs and ZnO NPs has induced an antibacterial activity in orthodontic composite resins (Transbond XT). Akhavan et al. 31 observed that the incorporation of TiO2 NPs into composite resins confers antibacterial properties to adhesives, while the SBS of composites containing 1% and 5% NPs is still in an acceptable range.
Assery et al. 32 found that the addition of 1% TiO2 NPs to the experimental composite resin demonstrated a promising flow and antibacterial effect without compromising the adhesion strength or chemical properties. Felemban and Ebrahim 33 observed that the incorporation of 1% ZrO2–TiO2 NPs into the Transbond XT orthodontic adhesive improved SBS.
ARI is one of the most commonly used parameters for evaluating the quality of adhesion between the adhesive and the tooth and also between the adhesive and the bracket. 34 It is desirable that after debonding some adhesive is left on the tooth surface, thereby minimizing the damage of enamel surface during bracket removal procedures. The present study results showed that there were no significant differences concerning ARI at all the concentrations of chitosan NPs and TiO2 NPs. Poosti et al. 29 found that the ARI scores of Transbond XT alone and Transbond XT containing 1% TiO2 were not significantly different after debonding.
In continuation to our research, a study by Niazi 35 demonstrated that the incorporation of 1% chitosan NPs and 1% TiO2 NPs into orthodontic composites leads to the inducement of a superior antibacterial property in the adhesive (Transbond XT) of fixed orthodontic treatment. One limitation in this study was that the study was performed in vitro, and thus did not represent complete oral conditions. Future studies can more closely mirror the in vivo situation. Moreover, other aspects such as antimicrobial and physical properties must be evaluated in future studies.
Conclusion
It is concluded that the orthodontic composite containing 1% chitosan NPs and 1% TiO2 NPs has adequate SBS for use in the clinical setting.
Footnotes
Acknowledgements
We would like to thank the Research Vice Chancellor of Mashhad University of Medical Sciences for financial support (grant no. 961679) to conduct this study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by the Research Vice Chancellor of Mashhad University of Medical Sciences for financial support (grant no. 961679).
