Abstract
Aim:
To examine the effects of fluoride, hydroxyapatite, and bromelain-containing toothpaste types on the surface roughness of nanohybrid, nanofilled, flowable, and bulk-fill composites; conventional, light-curing, and low-viscosity glass ionomers; and compomers.
Materials and Methods:
A total of 160 specimens were fabricated using nanohybrid, nanofilled, flowable, and bulk-fill composites; conventional, light-curing, and low-viscosity glass ionomers; and compomers. Each material group was divided into four subgroups (n = 5) as follows: Control (C), fluoride toothpaste (FT), bromelain toothpaste (BT), and hydroxyapatite toothpaste (HT). Surface roughness values of all specimens were examined using an optical profilometer and a scanning electron microscope at 500, 1,000, and 3,500 magnifications.
Results:
The highest average roughness value (Ra; µm) was obtained from the conventional glass ionomer material (P < .001). While the mean Ra value was 0.155±0.116 µm at baseline, it was 0.262±0.203 µm in the fluoride group, 0.237±0.192 µm in the hydroxyapatite group, 0.260±0.293 µm in the bromelain group, and 0.198± 0.187 µm in the control group (P < .001).
Conclusion:
We have found that brushing with various types of toothpaste containing fluoride, bromelain, and hydroxyapatite may not result in obtaining different surface roughness values from different types of restorative materials. Differences in surface roughness values of restorative materials may be found at different magnifications under scanning electron microscope.
Introduction
In today’s modern dentistry practice, advances in adhesive techniques have brought the preservation of healthy dental tissues to the fore. 1 Composite systems have undergone considerable transformation and development since they were first introduced, becoming an indispensable part of daily dentistry practice along with the advances in adhesive systems. 2 The most important change has been recently introduced through the addition of small-size strong filler particles in the material composition. The addition of inorganic particles in the material content allowed for obtaining restorations with higher wear resistance and improved polishability.2–4
Glass ionomer cements are used in routine clinical practice as a permanent direct restorative material type. 5 Their anticariogenic potential associated with fluoride release and their biocompatibility and adaptive capacity to dental chemistry have made glass ionomer cement types a specialized material group. However, their weak mechanical characteristics including low fracture strength, hardness, and wear resistance curtail their use as restorative material types in the posterior regions of the dental arch exposed to intense stress.5,6
Tooth brushing is the most important way of maintaining oral hygiene. Among all dental products, toothpaste is one of the most effective and a widely used cosmetic and therapeutic agents used routinely by consumers. 7 Anticaries, dentin desensitizing, whitening, and remineralizing agents are added to the content of toothpaste for product variety and to introduce various types into the markets. 8 Such agents include fluoride, casein phosphopeptide-amorphous calcium phosphate complex, bromelain, and hydroxyapatite.
Fluoride toothpaste has a crucial role in individual and social anticaries programs and has been in use for many years. 9 Active fluoride ions in fluoride toothpaste are introduced to the enamel surface to promote remineralization via the formation of fluorapatite. Furthermore, these ions prevent bacterial adhesion to tooth enamel and thus bacterial acid production. 10 Hydroxyapatite particles have high affinity to the tooth surface and they are easily absorbed from the enamel surface. 8 Therefore, hydroxyapatite has been used as a desensitizing agent in the treatment of dentin sensitivity, the remineralization of white spot lesions in initial caries, and the prevention and remineralization of dental erosion.8,11,12 Bromelain is a protein-digesting enzyme and is obtained from the fruit or stem of pineapple. Bromelain is composed of a mixture of different thiol endopeptidases and phosphatases, glucosidases, peroxidases, cellulases, escharases, and protease inhibitors. In vitro and in vivo studies have reported fibrinolytic, antiedematous, antithrombotic, and anti-inflammatory activities of bromelain. 13
Restorative materials should essentially have smooth surfaces for the well-being of teeth and surrounding tissues and the maintenance of the aesthetic appearance appropriately. A rough restoration surface can cause discoloration, high wear rate, plaque accumulation, reduced brightness of the restoration, and gingival irritation.14,15 Studies in the literature have shown that tooth brushing with abrasive-containing toothpaste can lead to roughness on the restoration surface.16–19
A literature review reveals that surface roughness is usually evaluated on hybrid, microfilled, or microhybrid composite resins.17,18,20 Moreover, no studies have investigated the effect of bromelain and hydroxyapatite-containing toothpastes on the surface roughness of restorative dental filling materials. This study included four different types of composite materials, namely, nanohybrid, nanofilled, flowable, and bulk-fill types. Additionally, it included conventional glass ionomers, light-cured glass ionomers, low-viscosity glass-ionomer fissure sealants, and compomers (CM). Thus, we aimed to evaluate the impact of fluoride, bromelain, and hydroxyapatite-containing toothpastes on the surface roughness of eight different aesthetic restorative material types included in the study. A blind hypothesis was formed assuming that different types of toothpastes would not result in different surface roughness values of different composite restorative materials.
Materials and Methods
Study Design
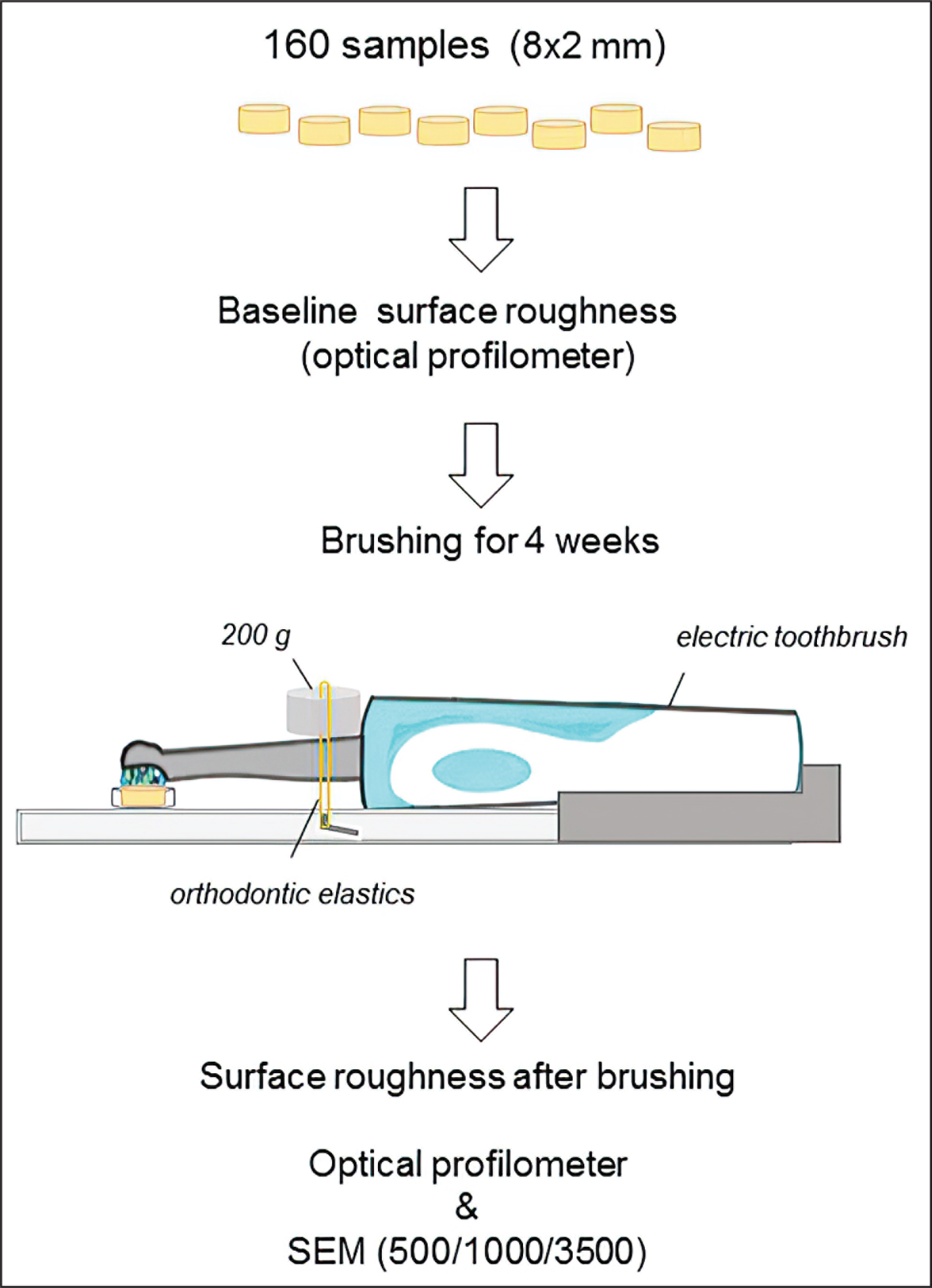
This randomized, single-blinded, controlled in vitro study was conducted to compare the surface roughness of different restorative materials after the use of different types of toothpastes containing fluoride, bromelain, and hydroxyapatite (Figure 1). The G*Power software package (G*Power Ver. 3.0.10, Franz Faul, Üniversität Kiel, Germany,
Specimen Preparation
The eight types of restorative materials and three types of toothpastes used in the study are listed in Table 1. Using a cylindrical metal mold, a disc-shaped specimen was prepared for each type of the restorative material, making a total of 160 disc-shaped specimens (n = 20). Twenty specimens were prepared for each of the composite and glass ionomer materials. Each material group was equally divided into four groups as follows: the control group (C) that was brushed with distilled water, the FT group brushed with fluoride toothpaste, the HT group brushed with hydroxyapatite toothpaste, and the BT group that was brushed with bromelain toothpaste.
Materials Used in the Study

Each disc-shaped specimen was 2 mm thick and had a diameter of 8 mm. The specimens were covered with a Mylar strip and both sides of each specimen were covered with glass microscope slides. The restorative materials used in the study were polymerized using a light emitting diode light source (VALO Cordless, Ultradent, South Jordan, Utah, USA; 1100 mW/cm²), applying the energy for 40 s over the glass microscope slides on both sides of the brass molds. The power of the light source was checked regularly with a radiometer (TR-P004, TREE International Trading Company, China) before the polymerization procedures in each group.
After the specimens were kept in distilled water at 37°C for 24 h, dental polishing was performed by a single investigator using medium, fine, and superfine aluminum oxide discs (Sof-Lex system, 3M ESPE, St. Paul, MN, USA). A handpiece was used to perform the procedure at 8,000 rpm to apply light pressure and rotational action over the entire sample surface for 20 s. Then, the surface roughness of the polished specimens was determined before the brushing process.
Each specimen was brushed with an electric toothbrush (Oral B Vitality, Braun, Germany; 31,000 strokes per min) at a medium pressure value of 200 g. The samples were brushed for 30 s twice daily every day for four weeks. 21 The battery of the brushing device was charged after 30 min, in accordance with the manufacturer’s instructions. The electric toothbrush was replaced every week with a new one. The control group specimens were brushed using distilled water. Each type of toothpaste was applied on the specimen surfaces as a mixture of 40 mL of deionized water and 25 g of toothpaste. 17 At the end of the brushing procedure, each specimen was rinsed under running water and the cleaning process was completed by sonication in deionized water for 10 min.
Surface Roughness Measurement
Before and after simulated tooth brushing, the surface roughness of the specimens was evaluated by a blinded independent investigator. A surface testing machine (MarSurf/M 300, Mahr GmbH, Germany) with a radius of 0.35 µm was used at a constant speed of 0.5 mm/s to apply a force of 0.75 mm. The device was calibrated before the measurement procedures in each group, setting a cut-off value of 25 µm. Measurements were performed in five different regions of each specimen and the average surface roughness values (Ra, µm) were calculated using these five values obtained from each specimen.
Surface Evaluation
Following the tooth brushing procedure, the specimens were gold coated and evaluated under an scanning electron microscopy (SEM) (JSM-6510, JEOL Ltd, Tokyo, Japan). Three images were collected from each specimen at 500, 1,000, and 3,500 magnifications.
The SEM examination of the specimens was carried out by two investigators. Cohen’s kappa statistic was used to test the interexaminer agreement (κ = .9). The surface roughness scores were determined based on the results reported by previous studies investigating the same subject matter in the literature as follows.22,23
Criteria for the evaluation of glass particles in the restorative material:
0: particles appear intact with no visible etched surface 1: moderate degradation with pitting or slight cracking 2: severe cracking and pitting
Criteria for matrix evaluation:
0: particles totally embedded in an undisturbed matrix 1: minimal surface changes in the composite matrix. The glass ionomer matrix remained intact but with a large number of degradation and surface irregularities with particles partially protruding. A limited number of voids are present. 2: moderate changes in the composite matrix but severe degradation in the glass ionomer matrix.
Statistical Analysis
The data were analyzed using the IBM SPSS program version 23. The conformity of the Ra values to a normal distribution was analyzed with the Shapiro-Wilk test. The results showed that the data exhibited a normal distribution. The changes in the Ra values by the material and toothpaste types were examined by two-way repeated-measures analysis of variance. The Bonferroni test was used for post-hoc analyses. The score comparisons of the particle and matrix of restorative materials through SEM photographs at 3,500, 1,000, and 500 magnifications were examined with the Wilcoxon test. A P value of < .05 was accepted to indicate statistical significance.
Results
Profilometer
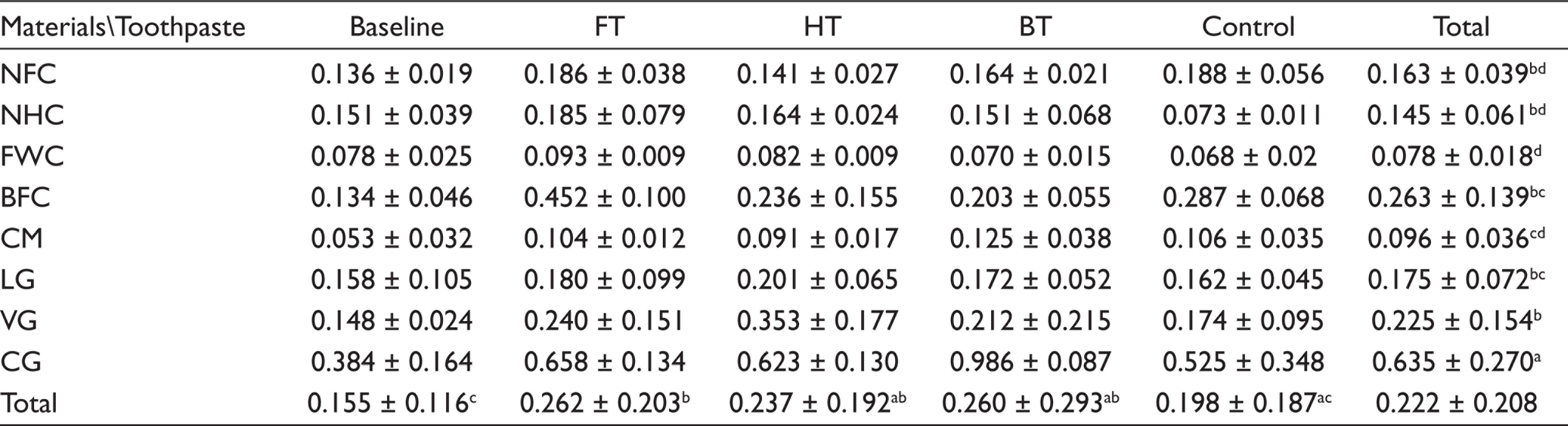
The surface roughness values of the restorative materials obtained after being brushed with different types of toothpastes are shown in Table 2. The effect of the type of restorative material on the mean Ra value was found out to be statistically significant in our study (P < .001). While the mean Ra value of the flowable composite (FWC) was 0.078 µm, the mean Ra values were found to be 0.096 µm for the CM, 0.145 µm for the nanohybrid composite (NHC), 0.163 µm for the nanofilled composite (NFC), 0.175 µm for the light-cured glass ionomer (LG), 0.225 µm for the low-viscosity glass ionomer (VG), 0.263 µm for the bulk-fill composite (BFC), and 0.635 µm for the conventional glass ionomer (CG).
Investigation of the Effect of Material and Toothpaste on Ra
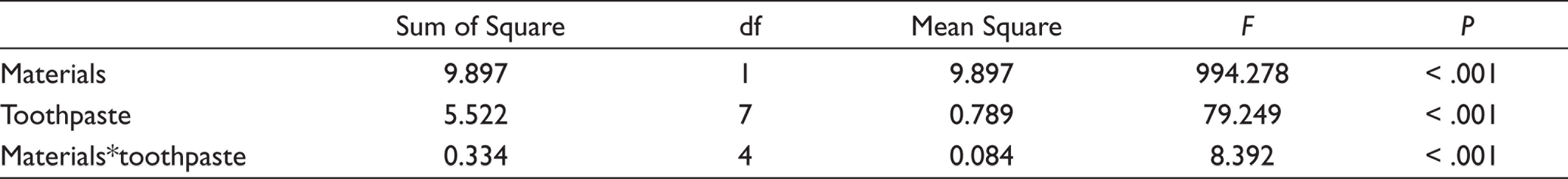
The highest mean Ra value was obtained from the CG group. The FWC and CM groups had statistically lower surface roughness values compared to the BFC, LG, VG, and CG groups. However, the roughness of the FWC, CM, NFC, and NHC groups were similar. No statistical differences in surface roughness were observed across the NFC, NHC, LG, VG, and BFC groups (Table 3).
The effects of dental material and toothpaste interactions on surface roughness were statistically significantly different (P <.001). While the mean value was 0.155 µm at the baseline, it was 0.198 µm in the control group, 0.237 µm in the hydroxyapatite group, 0.260 µm in the bromelain group, and 0.262 µm in the fluoride group. There was not a difference between the baseline value and the value obtained from the control group. Compared to the baseline and control group values, the fluoride group had the highest roughness value. However, there was not a difference across the FT, HT, and BT groups. In addition, the roughness values were similar in the HT and BT groups and in the control group (Table 3, Figure 1).
The Study Protocol and Schematic Diagram of the Brushing
Descriptive Statistics for Ra Measurements of Materials and Toothpastes
Surface Evaluation
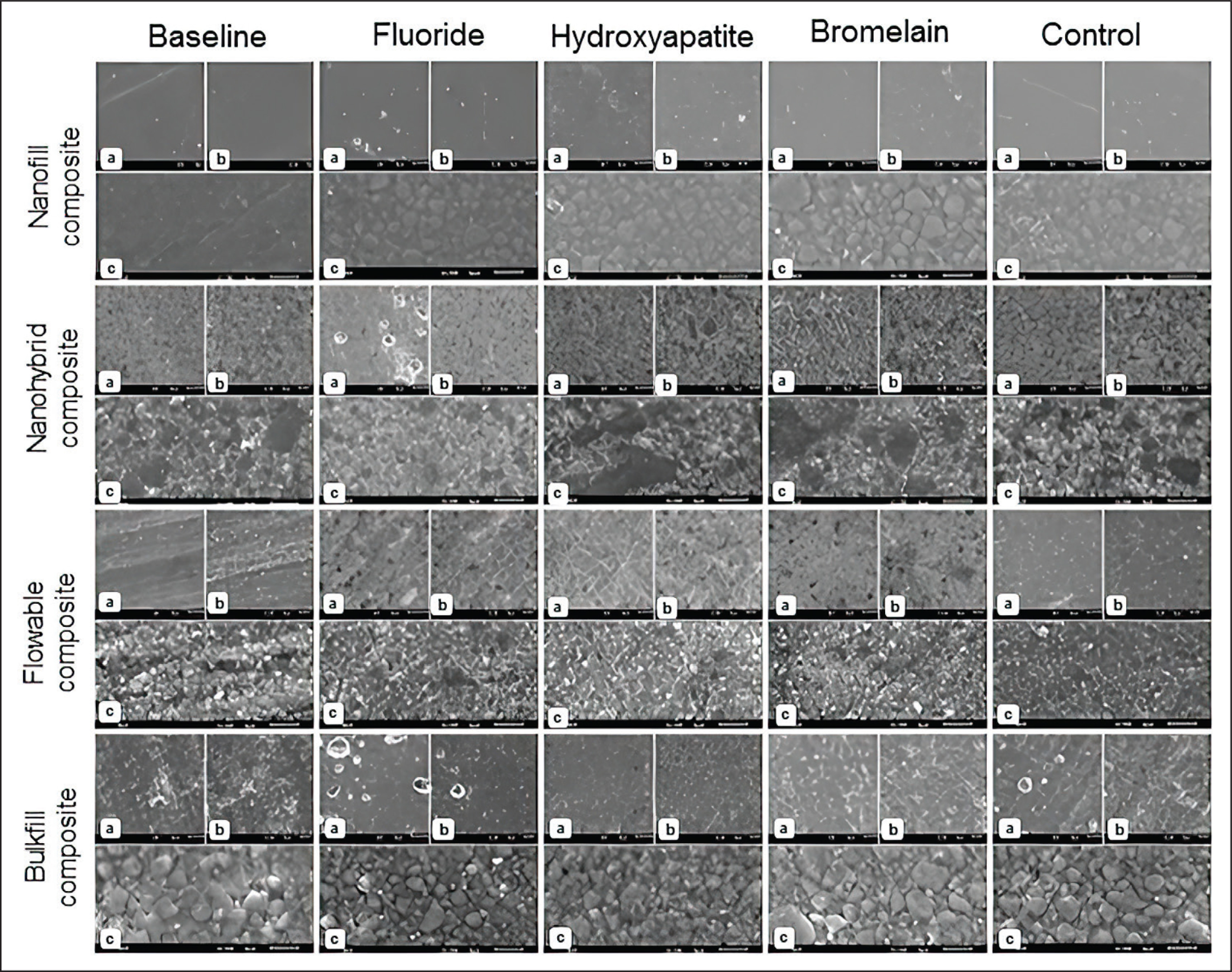
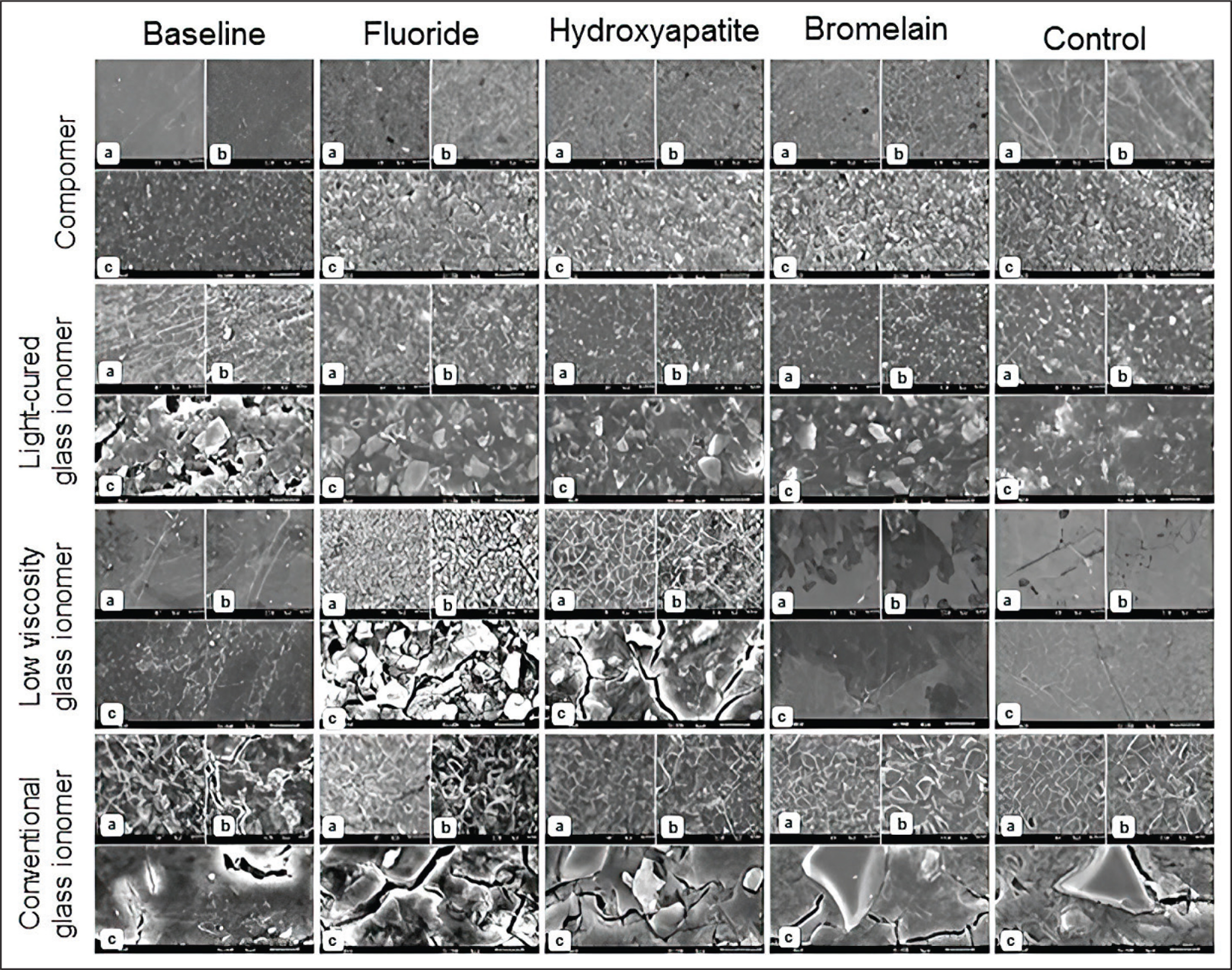
The particle and matrix changes of the specimens were examined by SEM at 3,500, 1,000, and 500 magnifications (Figures 2 and 3). Each obtained image was compared with the particle and matrix images of the control group. The statistical analysis results have been presented in Table 4.
SEM Images of the Nanofill, Nanohybrid, Flowable, and Bulk-Fill Composite Brushed with Fluoride, Bromelain, and Hydroxyapatite-Containing Toothpaste (a) 500 Magnification, (b) 1,000 Magnification, (c) 3,500 Magnification
SEM Images of the Conventional Glass Ionomer, Low-Viscosity Glass-Ionomer Fissure Sealant, Light-Cured Glass Ionomer, and Compomer Brushed with Fluoride, Bromelain and Hydroxyapatite-Containing Toothpaste (a) 500 Magnification, (b) 1,000 Magnification, (c) 3,500 Magnification
Comparison of Particle and Matrix of Restorative Materials Through SEM Photographs at 3,500, 1,000, and 500 Magnifications
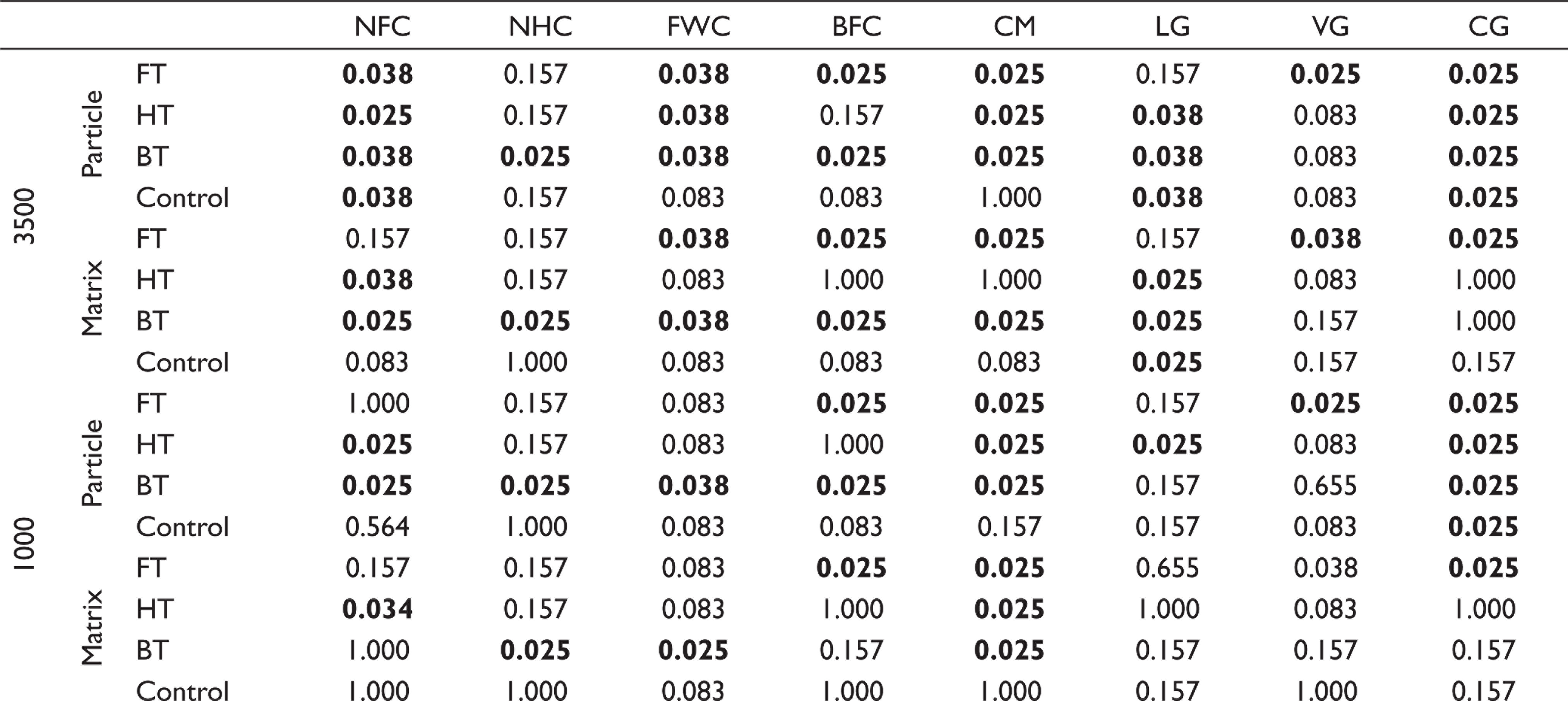
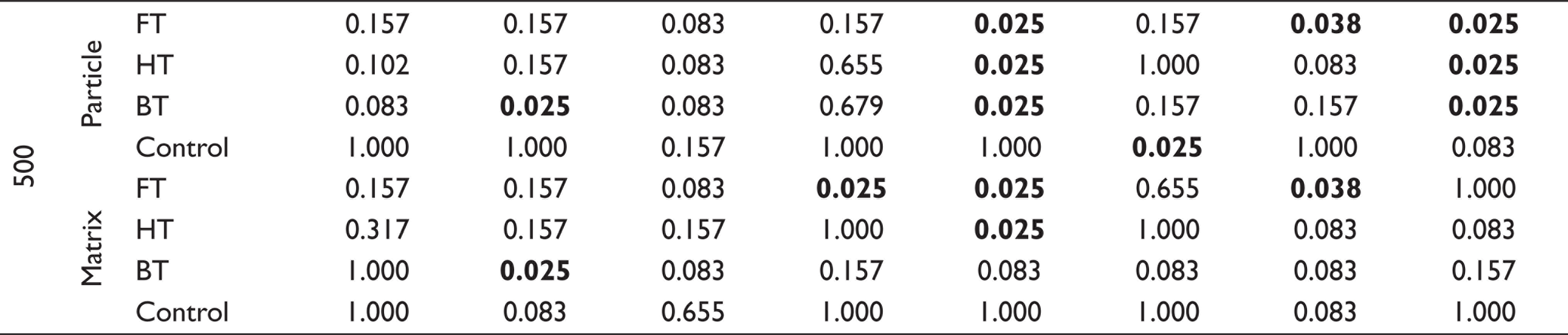
The comparison of the particle changes between the NFC and control groups revealed statistically significant differences in particles at 3,500 magnifications in all subgroups. However, the matrix changes were statistically significantly different only in the HT and BT groups. The examination at 1,000× magnification revealed differences only in the HT and BT groups, while no differences were detected in any of their subgroups at 500 magnifications. Compared to the control group, the particle and matrix changes of NHC were statistically significantly different only in the BT subgroup at all magnifications.
Compared to the control group, the particle changes in CG were statistically significantly different in all subgroups at 3,500 and 1,000 magnifications, while no significant differences were found only in the control subgroup at 500 magnifications. When the matrix changes in the CG groups were compared with those of the control group, significant differences were found in FT subgroup at 3,500 and 1,000 magnifications.
Discussion
Improvements in the surface properties of restorative materials are necessarily required for the longevity of the restoration. Surface roughness is an important physical feature of the restorative material, directly affecting the surface properties such as brightness, color, and hardness.24,25 Restorative materials in the oral cavity exhibit changes in their surface properties due to various factors. Among these factors, oral hygiene procedures play an important role. Tooth brushing as a component of daily oral care can cause roughness on the restoration surface. Bacteria and plaque retention on rough surfaces lead to many problems including dental caries and inflammatory diseases in the gingiva and periodontal tissues. 26 Therefore, it was aimed to carry out profilometer and SEM examinations in this study to investigate the surface roughness in resin-based materials including nanofilled, nanohybrid, flowable, bulk-fill composites, and CMs, and glass ionomers of conventional, light-curing, and low-viscosity types after brushing with different toothpaste types containing fluoride, hydroxyapatite, and bromelain.
The properties of resin matrices, binding agents, and fillers directly affect the surface roughness of the composite resin material. 27 The type, filler size, and filler ratio of inorganic fillers are considered to be the most effective factors.28–30 Properly polishable composite surfaces with highly aesthetic properties can be achieved by minimizing the filler size. 31 Studies in the literature reported low surface roughness values in association with reduced filler size.31,32 The purpose of using fillers of different size in the same resin material is to increase the filler ratio and ensure that rather than large particles, small particles would be removed from the surface during wear processes. 33 At baseline, CMs had the lowest surface roughness followed by the FWC, while higher values were obtained from the conventional glass ionomer group. This difference in surface roughness can be attributed to differences in interparticle spacing and filler particle size. The average size of filler particles is 0.8 μm in the CM and 0.7 μm in the FWC. This may partially explain the lower roughness obtained from these restorative materials. In our study, after brushing, no differences in surface roughness were found across the nanofilled, nanohybrid, and FWCs, and CMs with different filler size and filler quantities. Similarly, the study by Costa et al. 34 could not demonstrate any differences of roughness between nanofilled and nanohybrid composites that underwent brushing with different abrasive toothpaste types. However, Oliveria et al. 35 reported that nanocomposites exhibited lower surface roughness after brushing compared to microhybrid composites.
Our study found that the surface roughness values increased at similar rates in bulk-fill, nanofilled, and nanohybrid composites with all toothpaste types tested. Consistent with our study findings, Shimokawa et al. 36 found that nanohybrid, nanofilled, and bulk-fill composites exhibited increased surface roughness after brushing. However, they reported that changes in surface roughness varied by different trademarks of resin-based materials. Heintze et al. 16 studied surface roughness and reported that microfilled composites of different trademarks might exhibit different surface roughness values depending on the magnitude of the force applied during tooth brushing. Those studies explained the different outcomes obtained in the presence of different factors, including the type of the abrasion tester, the force applied during tooth brushing, the number of rotations made by the toothbrush, the hardness and the number of bristles in the toothbrush, and the type and size of the abrasive in the toothpaste.
In our study, the conventional glass ionomer had the highest surface roughness. A study investigating surface roughness before and after polishing conventional and modified glass ionomers demonstrated that the higher values of surface roughness were obtained from conventional glass ionomers. 37 Comparing the surface roughness of glass ionomers with dental amalgams and hybrid composites as references after tooth brushing processes, Momoi et al. 38 reported that surface roughness of glass ionomers was higher compared to the reference materials. Those results are similar to our study findings as we obtained the highest Ra value from conventional glass ionomers after brushing. The investigators explained this finding by the poor mechanical properties of conventional glass ionomers such as low fracture strength and inadequate hardness and wear resistance. 6 Furthermore, conventional glass ionomers are heterogeneous, setting occurs in two phases, and they contain large particles (median size of about 0.73 and 6.02 μm). 39
In the literature, fluoride-containing toothpaste types were reported to cause surface roughness in restorative materials; 40 however, no studies evaluated the effects of toothpaste types containing bromelain or hydroxyapatite. Considering the results of the present study, the tested hypothesis could be partially accepted. There were no differences in the Ra values across the groups after brushing with each toothpaste type for four weeks. However, compared to the control group, fluoride toothpaste caused a significantly rougher surface on dental materials. This result can be explained by the different relative dentin abrasion values and ingredients of the toothpaste types used in the study. Fluoride toothpaste contained hydrated silica as an abrasive particle. Similarly, previous studies demonstrated that the presence of a larger number of abrasive particles in toothpaste produced higher roughness values of composite resins.12,13 The lowest RDA value was reported for hydroxyapatite (40), whereas the highest RDA value was reported for bromelain (99). Although bromelain toothpaste had a high RDA value, similar surface roughness values of dental materials were obtained with fluoride and hydroxyapatite toothpastes in our study. This finding can be explained by the absence of abrasives such as silica in the content of bromelain toothpaste.
The data obtained in studies evaluating surface roughness values were confirmed visually via SEM images.16,36 In our study, the particle and matrix images of materials were examined and scored at 3,500, 1,000, and 500 magnifications according to the findings described by El-Badrawy et al. 23 We found different values of matrix and particle size for the different material types examined in our study at different magnifications. Similar results with profilometer findings were obtained at 1000 magnification. This finding can be explained by differences in the clarity of the digital SEM images depending on the degree of magnification, resolution, and the size of the electron beam.
The present study had some limitations. One limitation was the use of a Mylar strip on the surface of specimens. The use of a Mylar strip is a standard method employed by most studies to produce the smoothest surface. However, this technique is not usually used in clinical practice because the correct anatomical contour of the restoration is rarely achieved using only a strip. 41 Testing only the commonly used restorative materials could be considered another limitation of our study. Further in vitro and in vivo studies are needed to investigate the effect of whitening toothpastes on the surface roughness of tooth enamel and new dental materials such as nano-glass-ionomer cements and composites with antimicrobial and ester-free monomers.
Conclusions
In conclusion, brushing with toothpastes containing fluoride, bromelain, and hydroxyapatite may cause similar surface roughness of restorative materials. In our study, the highest surface roughness value was obtained from the conventional glass ionomer after brushing. Although FWCs and CMs showed lower roughness compared to the bulk-fill composite, there were no differences across the nanofilled, nanohybrid, and FWCs, and CMs. It is suggested that future studies aiming to examine the surface roughness of restorative materials should consider performing SEM examinations at different magnifications to evaluate findings obtained from each of these magnifications separately.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The work was supported by the Recep Tayyip Erdogan University Scientific Research Projects Foundation (THD-2018-890).
