Abstract
Aim:
To investigate the effect of different conditioning methods on remineralization potential of hypomineralized enamel and its shear bond strength (SBS) to orthodontic brackets.
Materials and Methods:
An in vitro study was conducted at Mansoura University, Egypt. Eighty premolars were collected and randomly classified into four groups (n = 20 each) as follows: Group 1. Control (sound teeth), Group 2. Demineralized, Group 3. Demineralized and treated with nanohydroxyapatite (nano-HA) varnish, Group 4. Demineralized and treated with casein phosphopeptide–amorphous calcium phosphate (CPP–ACP) paste. To detect the rate of remineralization on the surface of treated specimens, energy dispersive X-ray (EDX) analysis was used in conjunction with the scanning electron microscope (SEM). The SBS was measured with a universal testing machine. One-way ANOVA test was carried out to analyze differences between the tested groups.
Result:
SE micrographs of Group 3 and Group 4 specimens showed smoother and less porous enamel surface than that of the Group 2 specimens. EDX analysis showed highest calcium (Ca) (25.47%) and phosphorous (P) (12.76%) values for Group 4 while Group 2 showed the lowest Ca and P values (16.96%) and (10.20%), respectively (P < .001). Demineralized enamel showed lowest (3.70 MPa) SBS mean value compared to sound (10.69 MPa) and remineralized enamel (Group 3—9.90 MPa, Group 4—10.32 MPa) (P < .001).
Conclusion:
Nano-HA and CPP–ACP have equal remineralizing effect on hypomineralized enamel lesions and positive influence on SBS.
Keywords
Abbreviations
ACP: Amorphous calcium phosphate
CPP–ACP: Casein phosphopeptide–amorphous calcium phosphate
EDX: Energy dispersive X-ray
SEM: Scanning electron microscope
SBS: Shear bond strength
Introduction
The general mechanism of dental caries is attributed to bacterial adhesion to the tooth surface, with subsequent acid production due to metabolism of fermentable carbohydrates. 1 The produced acids can dissolve the mineral content of enamel, especially calcium and phosphorous, leading to “enamel demineralization or hypomineralization.”2,3 This lesion is commonly described as “white-spot lesion” (WSL) because its porosity gives a characteristic whitish appearance, and this term is used interchangeably with the terms “incipient” or “initial” enamel lesions. 4 As described by Fejerskov et al., 5 WSL is considered as the first stage of dental caries that can be detected with the naked eye. In addition to bacterial action, enamel hypomineralization may result from systemic conditions, such as molar incisor hypomineralization (MIH), or congenital conditions, such as amelogenesis imperfecta (AI).6,7 Different methods were proposed for treatment of hypomineralized enamel lesions, such as resin infiltration, sodium hypochlorite, fluoride, or calcium-phosphate containing products.2,8–11
A big challenge that may be faced by orthodontists is when they receive some patients seeking orthodontic treatment while they suffer from local or general hypomineralized enamel lesions. The challenging factor in these cases results from the fact that these lesions may decrease the adhesion between the tooth and the bonded brackets, with subsequent possibility of microleakage, and premature debonding. 12 Previous studies13–17 focused on evaluating the bond strength of orthodontic brackets to normal tooth enamel using different types of adhesives or bracket materials or even different types of curing units. However, only few studies were concerned about studying and improving the bond strength of orthodontic brackets to hypomineralized enamel.18–20 Thus, the current study aimed to assess the role of two different remineralizing agents in treatment of hypomineralized enamel lesions before application of orthodontic brackets. The agents under investigation in this study included, nanohydroxyapatite (nano-HA) varnish and casein phosphopeptide–amorphous calcium phosphate (CPP–ACP) paste. The evaluation parameters included surface topography and composition, and shear bond strength (SBS) between orthodontic brackets and the treated teeth.
Materials and Methods
Specimens’ Preparation and Grouping
A total of 80 recently extracted, sound human premolars, were collected from outpatients’ clinic, Faculty of Dentistry, Mansoura University, as they were extracted during orthodontic treatment course. The selected teeth were thoroughly cleaned, washed under tab water and stored in 0.1% thymol solution until required.21
The collected teeth were randomly classified into four groups (n = 20) as follows:
Group 1. Control (sound teeth), Group 2. Demineralized (DM), Group 3. Demineralized and treated with nano-HA, Group 4. Demineralized and treated with CPP–ACP.
Demineralization
Enamel lesions were artificially generated on specimens of Groups 2 through 4 using an acidic solution as mentioned by Subramaniam et al. 22 Each tooth was immersed in 10 mL of demineralizing solution consisted of 50 mM acetic acid solution, 3 mM CaCl2_H2O, 3 mM KH2PO4, 6 mM methyl hydroxyl diphosphonate, pH 4.95, 37°C, and to create an incipient caries lesions on the enamel surface, they were kept in the solution for 160 h.2 Throughout the period of experiment, each specimen was immersed in 10 mL of artificial saliva that was freshly prepared by mixing the following chemical agents in 1 L of distilled water: sodium azide: 0.75 g, potassium monohydrogen phosphate: 0.804 g, calcium chloride: 0.166 g, magnesium chloride: 0.059 g, and sodium chloride: 1.02 g. 23
Application of Remineralizing Agents
The selected mineralizing agents were applied, depending on the manufacturer’s instructions, to the buccal surface of specimens of Groups 3 and 4 for a period of 14 days. Specimens of Group 3 were treated with synthetic 20% nano-HA varnish (ApaCare™ Cumdente GmbH, Hamburg, Germany). While specimens of Group 4 were treated with CPP–ACP paste (MI Paste Plus™ Recaldent, GC America, Alsip, Ill, 10% CPP–ACP; 900 ppm fluoride). The nano-HA varnish was applied, using an adhesive brush, for 20 seconds where it became dry and settled on tooth surface. Varnish application was repeated every other day, between which, specimens were stored in artificial saliva. 24 The CPP–ACP paste was applied following in-office nontray technique. Each specimen was dried before a generous layer of the paste was applied using gloved fingertip and left undisturbed for 5 minutes. Specimens were then stored in artificial saliva, with the paste still on tooth, to allow for dissipation and more prolonged effect of the paste. The same procedure was repeated every day during the treatment period. 25
Evaluation of Surface Topography by Scanning Electron Microscope (SEM)
Randomly selected specimens from each group were gold sputtered and then analyzed using scanning electron microscope (SEM) (JEOL JSM-5200, Japan) machine at a magnification of ×2,000.
Surface Composition Analysis by Energy Dispersive X-Ray (EDX)
To detect the rate of remineralization on the surface of treated specimens, calcium (Ca) and phosphorous (P) ions concentration was analyzed and compared to that of the untreated ones. For this purpose, energy dispersive X-ray (EDX) analysis machine (INCA X-sight, Oxford, England) was used in conjunction with the SEM.
Application of Brackets
To facilitate handling, the teeth were embedded into acryl blocks with the buccal surface facing toward the outside. For all tested metal brackets (3M Unitek™ Gemini MBT, USA) the same adhesive (TransBond Plus, 3M Unitek, Landsberg, Germany) and the same luting composite (Transbond XT, 3M Unitek, Landsberg, Germany) were applied. Teeth were etched using 35% phosphoric acid gel for 30 seconds, washed and dried. Then the primer was applied to the etched enamel surface. The bracket base was then loaded with the light-cure luting composite, which was fitted to the enamel surface with a pressure load of 500 g. Excess composite was removed carefully. Light curing was done for 60 seconds using LED curing unit (Epilar Freelight II, 1,000 mW/cm2, 3M ESPE, Seefeld, Germany).
Measurement of Shear Bond Strength (SBS)
The SBS was measured with a universal testing machine (Lloyd Instruments, Hampshire, United Kingdom). A custom, knife-edge shearing rod was important to be used in order to minimize the misalignment of the testing device. Specimens were firmly fixed and aligned precisely toward the shearing blade in the needed position using the movable platform of the vise to achieve the nearest load location to enamel surface. The long axis of the bonded bracket was placed parallel to the plunger of the testing apparatus. To guard against any deformation of the bracket during loading, 0.018-inch metal O-rings were soldered into the bracket slots in a previous step. The crosshead speed was established at 1 mm per minute, and the plunger direction was gingivo-occlusal. The load at the failure point was recorded and divided by the area of the bracket base to get the SBS expressed in megapascals (MPa). 21
Statistical Analysis
The collected data of ion concentration and SBS were analyzed by statistical analysis software (SPSS 12.0, SPSS, Chicago, Illinois). One-way ANOVA and least significant difference (LSD) test were carried out to analyze differences between the tested groups, at a significance level of P < .05.
Results
Surface Topography (SEM)
Scanning electron micrographs of the tested groups are shown in Figure 1. In Figure 1.1, enamel of the control specimen looks smooth, with some areas showing the representative fish-scales appearance of enamel rods. While in Figure 1.2, the specimen with demineralized enamel showed a highly porous sponge-like appearance. In Figures 1.3 and 1.4, SE micrographs of both treated specimens, whether with nano-HA or CPP–ACP showed smoother and less porous enamel surface than that of the demineralized one. Their surfaces showed very close similarity to that of the sound enamel of Group 1.
SE Micrographs (×2,000 Magnification) Showing Enamel Surface of (1) Control Specimen, (2) Demineralized Specimen, (3) Demineralized Specimen After Treatment with Nano-HA, and (4) Demineralized Specimen After Treatment with CPP–ACP

Surface Composition Analysis (EDX)
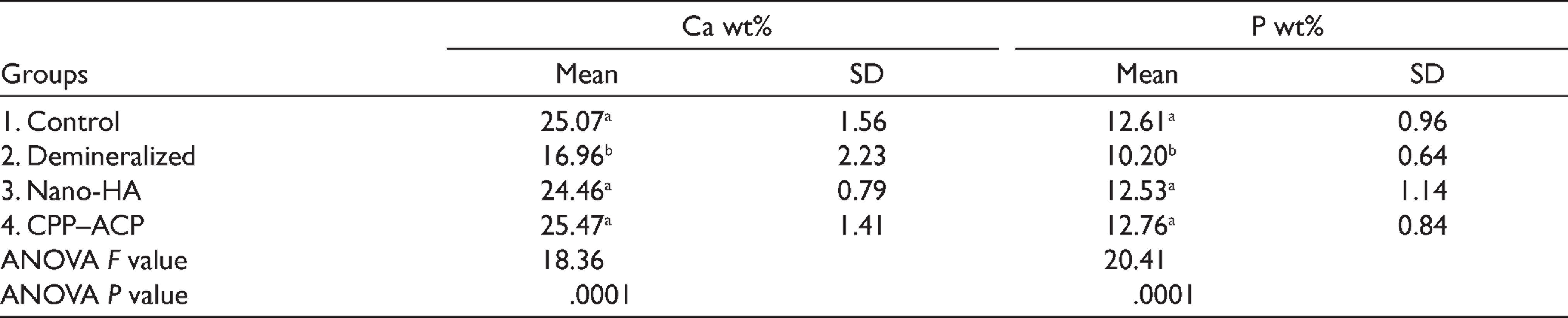
Mean weight percent (wt%) of Ca and P ions and standard deviations are presented (Table 1). It can be noted that the highest Ca (25.47%) and P (12.76%) values were recorded for Group 4 (treated with CPP–ACP), while Group 2 (demineralized) showed the lowest Ca and P values (16.96 %) and (10.20 %), respectively. ANOVA test showed significant differences (P = .0001) between the studied groups, both for Ca and P content. LSD test showed significant differences between Group 2 (demineralized) and each of the control (Group 1) and treated Groups (3 and 4). On the other hand, insignificant differences were found between Groups 1 (control), 2 (treated with nano-HA), and 3 (treated with CPP–ACP).
Mean and SD for Calcium and Phosphorous Content (wt%)
Shear Bond Strength
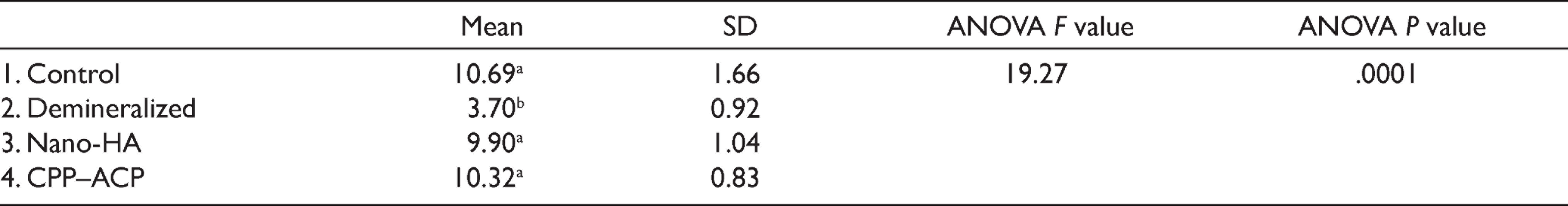
Means and standard deviations of SBS (MPa) for the studied groups are shown in Table 2. The current results showed that the highest SBS was recorded for Group 1 (10.69 MPa), while Group 2 (demineralized) showed the lowest mean value (3.70 MPa). ANOVA test revealed a significant difference (P = .0001) between the tested groups. Further analysis with LSD test showed a significant difference between Group 2 (demineralized) and each of the other three groups. While the difference between the control (Group 1) and both treated Groups (3 and 4) was insignificant. Comparing the two treated Groups (3 and 4) revealed an insignificant difference between the applied remineralizing agents.
Mean and SD for SBS (MPa) of the Studied Groups
Discussion
Hypomineralized enamel is more porous than sound enamel due to less mineral content. 7 Thus, bonding of orthodontic brackets to this type of enamel may be weak and accompanied by extensive microleakage. 16 As calcium and phosphate ions play an essential role in remineralization, novel materials were introduced to the markets containing these ions, for prevention and treatment of enamel hypomineralization. 26 Among the most popular remineralizing products come casein CPP–ACP and hydroxyapatite HA (bioactive glass). 27 Therefore, the current study was conducted to evaluate the remineralization potential of these two agents and to assess their ability to enhance bonding of orthodontic brackets to the treated enamel lesions. For this purpose, incipient lesions were artificially created on enamel surfaces of the tested specimens, which were then treated with the selected remineralizing agents.
For evaluating the remineralization potential, calcium and phosphate ions were investigated on surface of the treated specimens using EDX analysis. Our results showed significant improvement in mineral content of the treated enamel with both types of remineralizing agents, in comparison to those of the demineralized specimens. Both nano-HA and CPP–ACP showed nearly similar remineralizing effects. Calcium and phosphate concentrations of the treated lesions were nearly equal to those of sound enamel, which proves the remineralization efficiency of the selected agents.
The effect of nano-HA particles on the present results can be attributed to their ability to supply calcium and phosphate ions to penetrate the porous surface of demineralized enamel, filling gaps between calcium crystals, and thus resulting in a uniform enamel structure with normal mineral content. 24 On the other hand, the results of CPP–ACP are explained in terms of its nature. This remineralizing agent is a milk product that is able to supply amorphous calcium phosphate (ACP) due to its content of casein phosphopeptide, which also helps ACP to bind to enamel surface. It is believed that CPP–ACP provides deeper penetration of ions into enamel lesion, resulting in remineralization of the entire lesion rather than only the surface layer. 28
The current results came in agreement with previous studies19,20,24,29 that proved the role of both agents in treatment of primary enamel lesions. However, while the present study showed equal remineralizing effect of both investigated agents, a previous study by Bajaj et al., 30 did not agree with us, as they reported a better performance of hydroxyapatite in comparison to CPP–ACP and tricalcium phosphate. This disagreement can be attributed to mismatches of the used products or different methodology.
For evaluating the ability of the tested agents, in the present study, to resist debonding of the applied orthodontic brackets, SBS test was performed. On comparing our results, specimens treated either with CPP–ACP or nano-HA showed significant improvement in SBS, as compared to the untreated demineralized specimens. CPP–ACP showed slightly better performance than nano-HA in this instance, but the difference was insignificant. It was found that the tested agents restored the bond between brackets and enamel lesions to the normal range of sound enamel of the control group. These results came in agreement with previous studies,31–33 as authors of these studies reported that pretreatment of enamel with CPP–ACP before bracket bonding significantly increased bond strength. On the contrary, Parka et al. 34 tested the effect of CPP–ACP on SBS of orthodontic brackets, and they found no significant effect of the remineralizing agent. This disagreement with our results can be related to different methodology, as Parka et al. 34 applied the mineralizing agent to nondemineralized teeth only. Although many researchers19–21,31–33 investigated the effects of different surface treatment methods on bond strength of orthodontic brackets to caries-like enamel lesions, yet, none of these studies have included a comparison between the effect of nano-HA and CPP-APC on SBS of the bonded orthodontic brackets. Thus, no previous studies were available to be compared to our current work. The present in vitro study showed both nano-HA and CPP–ACP have equal remineralizing effect and positive influence on SBS on the incipient enamel lesions. Further long-term studies are needed to evaluate the correlation between different remineralizing methods prior to orthodontic treatment and SBS of the brackets.
Conclusion
Within the limitations of the current research, it could be concluded that nano-HA and CPP–ACP have equal remineralizing effect on the incipient enamel lesions. Both agents similarly caused a positive influence on bond strength of the treated enamel to orthodontic brackets.
Authors’ Contributions
ETE: contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, critically revised manuscript, gave final approval, agrees to be accountable for all aspects of work ensuring integrity and accuracy.
MAT: contributed to conception and design, analysis, and interpretation, drafted manuscript, critically revised manuscript, agrees to be accountable for all aspects of work ensuring integrity and accuracy.
RM: contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, critically revised manuscript agrees to be accountable for all aspects of work ensuring integrity and accuracy.
SB: contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, critically revised manuscript, agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Statement
This study was scientifically and ethically approved by the Research Committee of Mansoura University (Ethical clearance number-A01100719).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
