Abstract
Purpose
In our hospital, cases of bone and soft tissue infections have been treated with continuous local antibiotics perfusion that allows for continuous circulation of antibiotics throughout the infected lesion. We termed this treatment “intramedullary antibiotics perfusion (iMAP)” for bone infection such as fracture-related infection (FRI) and “intrasoft tissue antibiotics perfusion” for soft tissue infection. Many cases are treated with both modalities. To introduce iMAP, this study focused on the patients with FRI treated with iMAP and reviewed their treatment outcomes.
Methods
We included 10 patients with FRI treated with iMAP between 2004 and 2017. The iMAP needles were inserted near the infected lesion, and an aminoglycoside antimicrobial was continuously administered. Patient characteristics, pathogenic bacteria, administered antibiotics, duration of administration, concentrations of antibiotics in blood and leachate fluid, fracture union rate, implant retention rate, and complications were studied.
Results
The mean age of patients was 59.9 years, and the mean follow-up period was 2.5 years. Affected bones were the tibia (n = 8), humerus (n = 1), and fibula (n = 1). Deep infections developed on average 29.9 days after osteosynthesis. Pathogenic bacteria were methicillin-susceptible Staphylococcus aureus (n = 6), methicillin-resistant S. aureus (n = 2), and unknown (n = 2). Average iMAP duration was 17.1 days. In all patients, infection was eradicated while preserving the implants, and fracture union was achieved without complications.
Conclusion
iMAP is a novel local drug delivery system allowing high concentrations of antibiotics to be administered without complications and is useful in the treatment of FRI.
Keywords
Introduction
Fracture-related infection (FRI) is one of the common and serious complications of osteosynthesis. 1 Patients who develop FRI early after osteosynthesis require concurrent eradication of infections and treatment of fractures, making it difficult to structure therapeutic strategies. It is often necessary to remove the implants to eradicate infections; however, removing them before fracture union can lead to instability at the fracture site. Therefore, it is desirable to eradicate infections while preserving the implants, at least until fracture union. The bacterial biofilm that forms around the implant is one of the factors that hinder FRI treatment.2–4 Moreover, antimicrobial agents at concentrations of 100 to 1000 times the minimum inhibitory concentrations (MIC) have been required to eradicate the biofilm, which is defined as the minimum biofilm eradication concentration (MBEC).5–7 Nevertheless, delivering the agents locally to the MBEC level by intravenous administration without systemic adverse effects is difficult. Another factor impeding the treatment of FRI is the inability of agents to reach the tissues with poor blood flow or dead cavities. In many FRI cases, infections spread to the surrounding soft tissues, and simultaneous treatment for the soft tissue infection is often required.
In our hospital, such difficult-to-treat bone and soft tissue infections were treated using continuous local antibiotic perfusion (CLAP), in which antibiotics are continuously circulated throughout the infected lesion. We address the treatment for osteomyelitis including FRI by “intramedullary antibiotics perfusion (iMAP)” and for soft tissue infection by “intrasoft tissue antibiotic perfusion (iSAP),” and many cases are treated with a combination of both. iMAP is a treatment method that delivers a sufficient local concentration of the agent by eluting a daily dose of antibiotics in a small amount of saline and injecting it continuously at low speed using a bone marrow needle placed near the infected lesion. The injected agent is infiltrated into the infected site and is then drained through a drainage tube. This system is expected to eradicate infections without needing to remove implants and generate a pathway for localized perfusion of antibiotics, continuously circulating an effective concentration of the agent throughout the infected lesion. Aminoglycoside is a bactericidal agent with a long history of clinical and basic evidence, and it has been reported that high-dose gentamicin is effective to eradicate methicillin-resistant Staphylococcus aureus (MRSA). Moreover, gentamicin has a superior bactericidal effect against biofilms formed by Staphylococci including MRSA.8,9 In addition, a previous study reported that local aqueous aminoglycoside administration in combination with systemic antibiotics is effective in reducing infection rates in open fractures. 10 Thus, in our practice, we initially used astromicin as the first choice antibiotic and arbekacin as the choice for resistant bacteria or unknown bacteria with less effective for astromicin treatment. After astromicin became unavailable in the Japanese market, we used gentamicin for local administration as the first choice, despite the susceptibility of the bacteria; the dose of gentamicin was adjusted by monitoring its serum concentration. The purpose of this study is to introduce the methods, concepts, and therapeutic efficacy of iMAP, which is one of the treatment modalities of CLAP. We selected patients with FRI treated with iMAP and retrospectively reviewed their treatment outcomes.
Methods
This study was approved by the Institutional Review Board of our hospital. Written informed consent was obtained from all patients whose clinical course or images are presented in this article.
Participants
Patients with FRI were included in the study that was defined and diagnosed by the following confirmatory criteria: 1. purulent drainage from the fistula, or 2. a clinical sign of deep infection confirmed by the presence of pus that excluded superficial infection in isolation. 1 Within the FRI, patients who were treated with only iMAP between 2004 and 2017 were enrolled as participants. All patients underwent surgery to construct iMAP and tried to retain implant, which did not loosen at the surgery. To exclude patients with chronic osteomyelitis, only patients with FRI within 9 weeks after the initial internal fixation were included. 11
Data collection
Patient characteristics, including age, sex, comorbidities, pathogenic bacteria, administered antibiotics (iMAP, intravenous, and oral), antibiotic concentration via iMAP, duration of administration of each agent, time required for fracture union, implant retention rate, duration of follow-up (the period during the date of the first and last medical examinations), and complications were evaluated. Pus or discharge from deep tissue and bone marrow aspiration fluid at insertion were cultured using a broad bacterial panel to identify pathogens. Fracture union was assessed by radiographs. During the follow-up period, radiographs and blood tests were performed periodically to investigate the complications of FRI treatment, such as reinfection and associated reoperation (another debridement or amputation), as well as renal damage and eighth brain neuropathy as complications of gentamicin administration. In measurable cases, the concentrations of antibiotics in the blood and leachate fluid collected from drainage bottle were measured once or twice a week during local antibiotics administration. The fluid from drainage bottle was used as a reference for local concentration.
Continuous local antibiotic perfusion by iMAP
The procedure of the iMAP pin insertion is depicted in Figure 1. Once FRI was diagnosed by the confirmatory criteria,
1
considering deep infection, surgery was performed at the earliest to construct iMAP. All procedures were performed under general anesthesia and under clean conditions, in the operating room. An unloosened implant was retained to prevent the fracture from becoming unstable. Nonviable tissues of the skin, soft tissue, and bone were debrided at the time of surgery. Sequestrum and necrotic soft tissue were easily identified for debridement; however, the extent of the debridement could not be determined due to unclear margins. In general, viable bone was assessed by “Paprika sign”; the bleeding was observed on the surface of the margin. In case of FRI, most of the free bone fragment is not vascularized at the surgery but may be re-vascularized in the future during the fracture healing period if the infection is controlled adequately. However, as intravenous antibiotics do not reach the free fragments, they would form a biofilm; therefore, these fragments are difficult to preserve. In case of iMAP, high-concentration antibiotics solution reaches the fragments and preserves them as biofilm formation is prevented. Intramedullary antibiotics perfusion (iMAP). Two bone marrow needles (yellow arrows) are inserted proximal and distal to the fracture site in the bone, and a small amount of highly concentrated antibacterial agent is continuously infused using a syringe pump. The leachate is drained from a drainage tube placed subcutaneously. The construction of a local perfusion pathway for antibiotics allows for the sustained circulation of high concentrations of antibiotics throughout the infected lesion (yellow-dotted circle).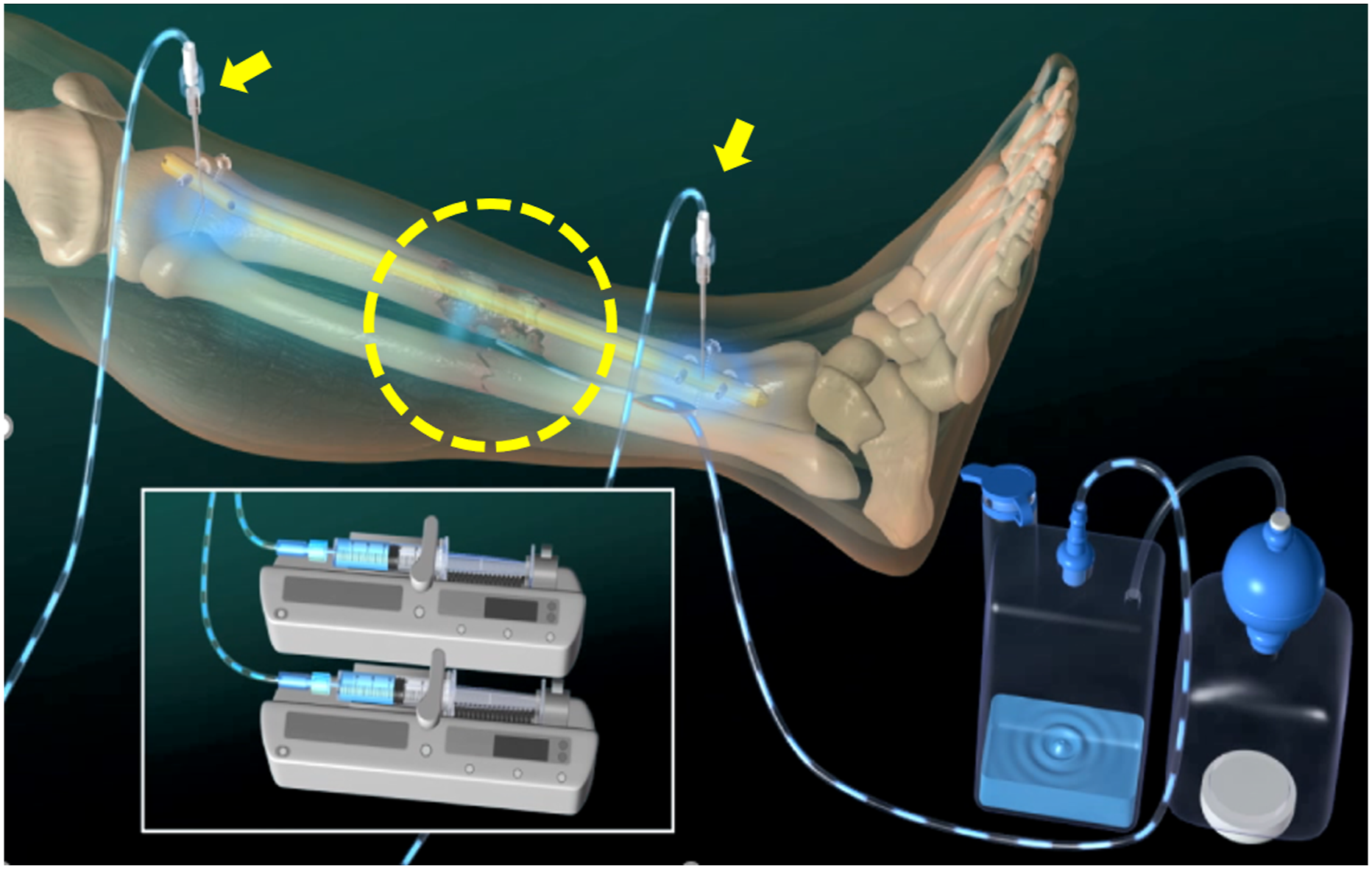
To construct antibiotics stream, the bone marrow puncture needle with a 3-mm outer diameter (Senko medical, Tokyo Japan) was used for iMAP. A 3.0-mm Kirschner wire was used to predrill 3-mm-diameter holes and insert the iMAP pin by hammering. In case of plate fixation, the iMAP pin was inserted 2–3 cm apart from the edge of the plate. In case of intramedullary nail fixation, the iMAP pin was placed at the diaphysis along the nail. Next, we visually confirmed that the saline solution that was flushed from the iMAP pin and flowed out of the fracture site (flow test). If the solution was not observed at the fracture site, we performed the flow test again after changing the insertion position or inserting a Kirschner wire through the fracture site into the medullary cavity to create a flow channel for the solution. The procedure in the operation room is completed by placing a drainage tube for the bleeding and leachate around the fracture site subcutaneously.
The perfusion system had to be managed during local antibiotics administration. After surgery, the infusion root should be filled with saline to prevent backflow of the medullary blood and initiate infusion as soon as possible at the bedside. The infusion of antibiotic solution should be controlled using a syringe pump system to maintain an accurate infusion speed. The infusion pin occasionally loosened during infusion; therefore, the fluid leak should be checked daily. Drainage tubes were often stacked by clots or debris, which were pumped out by flushing the saline from the sub-tube or pushed out by the tube sealer. If the drainage tube was stacked completely, the tube should be exchanged by another operation. Postoperative compartment syndrome should frequently be a concern, and if strongly suspected, we consider that the compartment pressure should be monitored.
We used gentamicin as the first-line antibiotic for administration via iMAP. Ceri et al. 5 reported that the MBEC of gentamicin for S. aureus was 2 μg/ml, that for Pseudomonas aeruginosa was 128 μg/ml, and that for Escherichia coli was 16 μg/ml. We expected that the administered antibiotics solution would be diluted by blood or leachate and assumed that the initial concentration to be administered must be 10 times higher than MBEC. Therefore, the standard dose was 1.2 mg/mL (= 1200 μg/ml) to dissolve gentamicin 1A (60 mg) in 50 mL of saline. Depending on the severity of the infection, administration was started at a dose of 2.4 mg/mL in some cases. Administration dose was lowered, if the serum concentration of gentamicin was more than 2 μg/ml to avoid any side effects. Theoretically serum antibiotics concentration is not exceeded 2 μg/ml as a trough value, and continuous antibiotics were safely delivered during local administration. The antibiotics solution was continuously injected at 2 mL/h by using syringe pumps against the bone marrow pressure. The intramedullary antibiotics perfusion needle was withdrawn when the local and hematological findings indicated resolution of the infection. As in the case of treating osteomyelitis, intravenous antibiotics agents regarding the results of susceptibility were continued for at least 4 weeks, followed by oral antibiotics for several more months.
Results
Summary of the patient characteristics of the 10 cases.
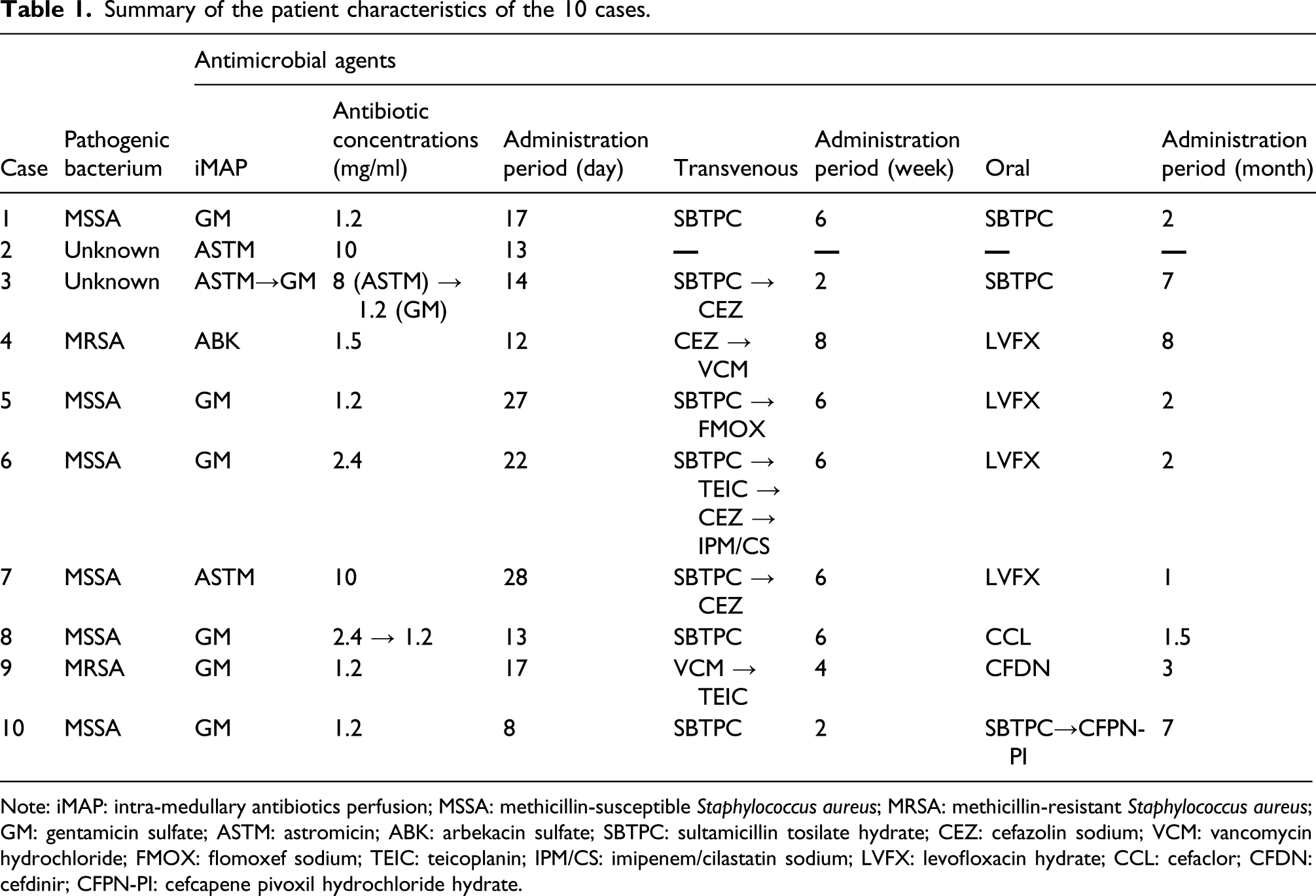
Note: iMAP: intra-medullary antibiotics perfusion; MSSA: methicillin-susceptible Staphylococcus aureus; MRSA: methicillin-resistant Staphylococcus aureus; GM: gentamicin sulfate; ASTM: astromicin; ABK: arbekacin sulfate; SBTPC: sultamicillin tosilate hydrate; CEZ: cefazolin sodium; VCM: vancomycin hydrochloride; FMOX: flomoxef sodium; TEIC: teicoplanin; IPM/CS: imipenem/cilastatin sodium; LVFX: levofloxacin hydrate; CCL: cefaclor; CFDN: cefdinir; CFPN-PI: cefcapene pivoxil hydrochloride hydrate.
Summary of antimicrobial agents used in the 10 cases.
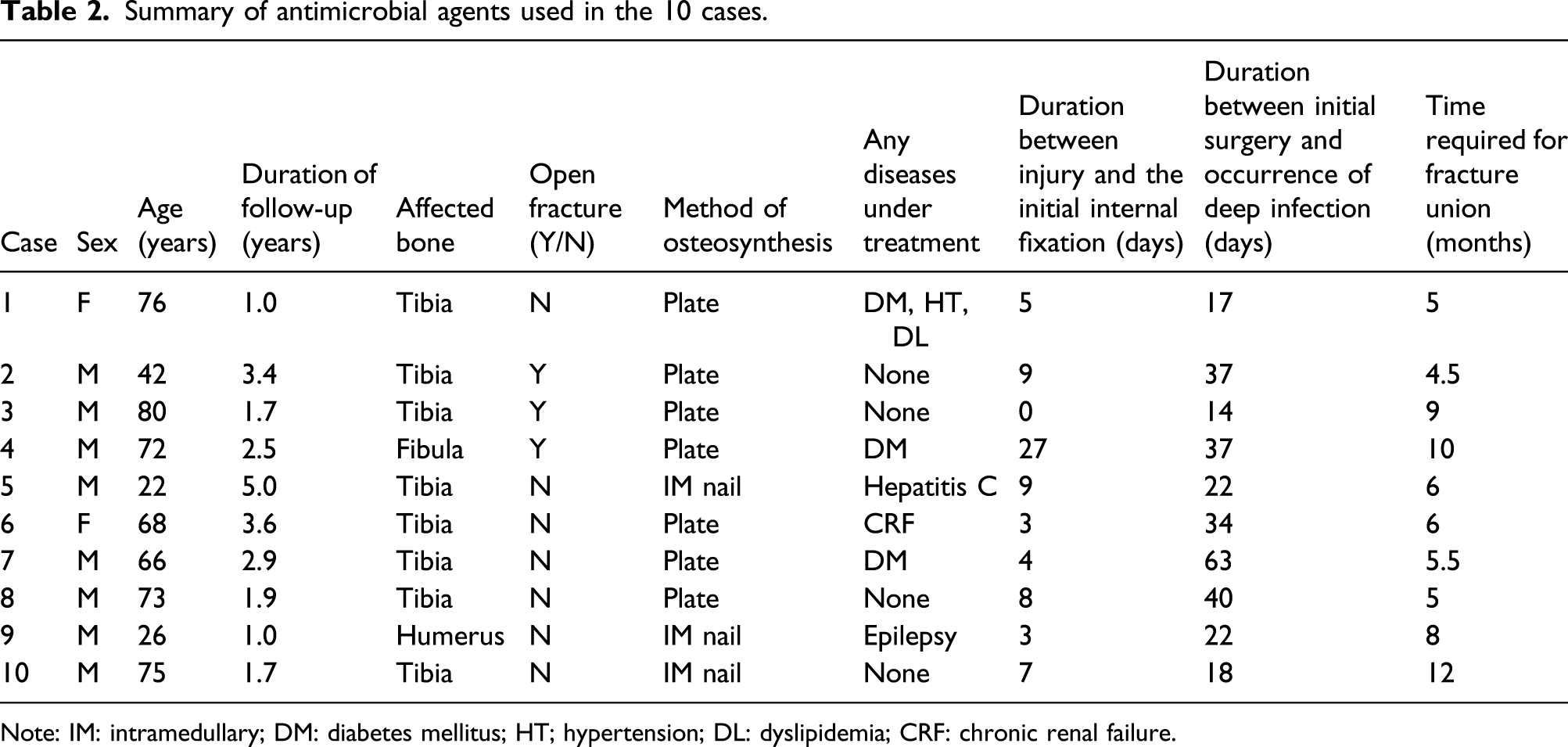
Note: IM: intramedullary; DM: diabetes mellitus; HT; hypertension; DL: dyslipidemia; CRF: chronic renal failure.

Transition in gentamicin concentration in patient 8. The straight line shows the concentration in the blood, and the dotted line shows the concentration in the leachate fluid. The latter was used as a reference for local concentration. The numbers in the white square indicate the concentration of gentamicin administered from iMAP. We started the administration of gentamicin at 2.4 mg/mL (gentamicin 2A (120 mg) + saline 50 mL) via iMAP, but because the blood concentration of the agent on the fourth day was very high (4.9 μg/mL), we changed the administration dose to 1.2 mg/mL (gentamicin 1A (60 mg) + saline 50 mL). As a result, gentamicin concentration in the blood promptly declined. In contrast, the concentration in the leachate fluid remained high, suggesting that a sufficient concentration was maintained locally. In this way, the characteristic of this treatment is that the blood concentration can be easily controlled by changing the dosage concentration via iMAP. Note: intramedullary antibiotics perfusion.
Case presentation 1 (patient 1 in Table 1)
The patient was a 75-year-old woman with diabetes mellitus. She sustained left closed tibial and fibular distal shaft fractures in a traffic accident (Figure 3(a)). Pre-injury activities of daily living were T-cane gait exercises. We performed open reduction and internal fixation (ORIF) for both fractures using locking plates on the fifth day after the injury (Figure 3(b)). On the 14th postoperative day, purulent discharge was observed from the surgical wound (Figure 3(c)). The deep infection of the FRI was easily diagnosed; thus, emergency surgery was performed on the same day. A subcutaneous pocket filled with necrotic tissue and infected fluid was curetted. A drainage tube was placed subcutaneously, and iMAP needles were inserted proximal and distal to the fracture site in the tibia (Figure 3(d)). We confirmed that the injected saline using iMAP needles was properly drained through the tibial fracture site to the drainage tube. A standard dose of gentamicin 1.2 mg/mL was continuously perfused at 2 mL/h with the implants preserved (Figure 3(e)). The pathogenic bacterium was MSSA. Local findings improved promptly after the initiation of iMAP and were removed 14 days after insertion. Intravenous and oral administrations of antibiotics were continued for 4 and 8 weeks after removal of implants, respectively. At 1 year after surgery, no signs of recurrence of infection were observed, and fracture union was achieved. The patient was able to walk with a T-cane as before the injury (Figure 3(f)). Clinical images of patient 1. (
Case presentation 2 (patient 5 in Table 1)
The patient was a 22-year-old man with hepatitis C who could walk independently before the injury. He was injured in a traffic accident and was diagnosed with right closed tibial and fibular shaft fractures (Figure 4(a)). Open reduction and internal fixation with an intramedullary nail was performed for the tibial fracture on the ninth day after the injury (Figure 4(b)). High-grade fever was observed on the 14th day after internal fixation, and localized swelling with heat was noted around the fracture area and the knee. Purulent discharge was observed upon puncture of both the areas (Figure 4(c)). The infection was suspected to have spread to the knee joint via an intramedullary nail, and emergency surgery was performed on the same day. Clinical images of patient 5. (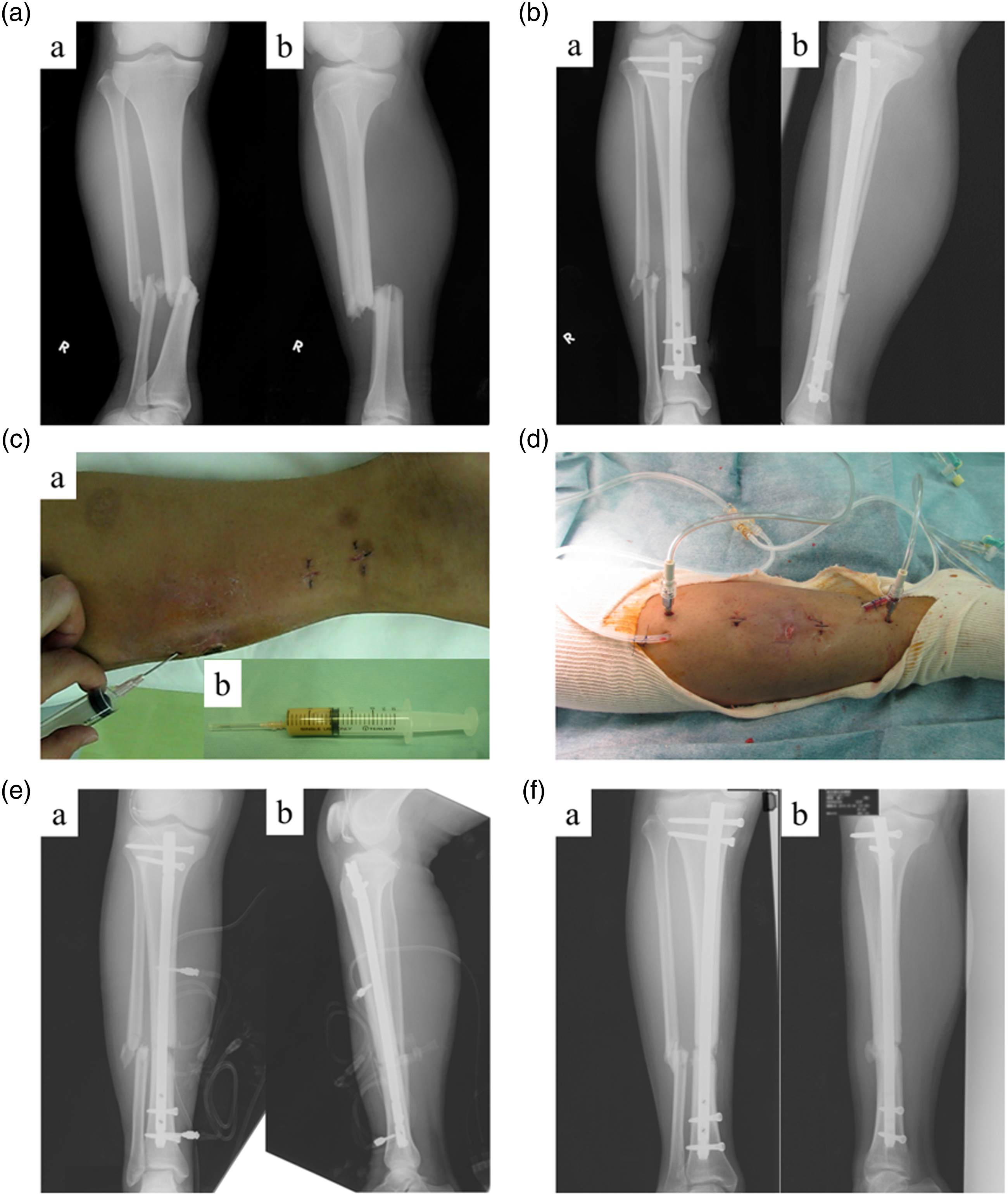
Two incisions were made around the fracture site, and curettage was performed. After placement of the subcutaneous drainage tube, the iMAP pin was inserted in the proximal and distal parts of the fracture area (Figure 4(d)). Bone marrow infusion was started at the dose of 1.2 mg/mL as in case 1, and purulent leachate was drained. Two drainage tubes were placed in the knee joint after intra-articular irrigation during arthroscopy (Figure 4(e)). The pathogenic bacterium was MSSA. Local findings improved promptly after iMAP was applied, and iMAP pins were removed 27 days after insertion. Intravenous and oral administrations of antibiotics were continued for 2 and 8 weeks after removal of iMAP, respectively. There were no signs of recurrence of infection at 5 years after the treatment. Fracture union was achieved, and the patient could walk without pain (Figure 4(f)).
Discussion
Fracture-related infection after osteosynthesis is usually difficult to treat. Especially in early postoperative cases in which implant retention is desirable, the biofilm that forms around implants and soft tissues with poor blood flow may hinder treatment. Bacteria existing within biofilms have been reported to be significantly less susceptible to antibacterial agents because their structural features prolong the penetration of agents by many orders of magnitude.12,13 Effective antimicrobial concentrations are generally assessed by MIC; however, MBEC is required to prevent the growth of biofilm, which is reported to be 100–1000 times the MIC depending on the type of bacteria.5–7 However, high blood levels of antibiotics can cause systemic side effects, and extremely high concentrations of antibiotics have been reported to inhibit the replication of osteoblasts and cause cell death. 14 In this study, no patient exhibited systemic side effects. The fracture union was achieved in all cases, suggesting that cell death due to exposure to high concentrations of antibiotics, a concerning prognostic factor, did not occur, and biological viability at the fracture site was preserved. Therefore, we believe that iMAP can safely deliver high concentrations of antibiotics to the infected lesions.
S. aureus is the most commonly encountered pathogenic bacterium in postoperative surgical site infections in the orthopedic field,15,16 with MRSA infections being especially difficult to treat. High concentrations of gentamicin are effective in eradicating biofilms formed by S. aureus, including MRSA,8,9 and the MBEC of gentamicin is lower than that of linezolid and vancomycin. 8 Although gentamicin at high blood concentrations is associated with a risk of nephrotoxicity and ototoxicity, 17 its serum concentration can be monitored during treatment and a low concentration of the antibiotic might be administered from the iMAP according to the measured blood concentration. The gentamicin concentration in the blood required some adjustment in one of our patients; it was possible to promptly reduce the gentamicin concentration in the blood to an appropriate level by decreasing the dose concentration of gentamicin administration. A study reported that exposure to high concentrations of gentamicin does not inhibit the activity and proliferation of osteoblasts or endothelial cells, which are essential for fracture union. 18
Therefore, we consider that gentamicin is suitable as an antibacterial agent for local administration and selected it as a single protocol in CLAP, regardless of antimicrobial susceptibility. Based on our experience in many cases, we currently standardized the initial dose concentration of gentamicin at the beginning of treatment to 1.2 mg/mL and the dose rate to 2 mL/h. Nevertheless, since the target of CLAP is the local infected lesion, systemic administration of antibacterial agents should be combined with intravenous and oral administration of agents depending on the results of antimicrobial susceptibility.
Antibiotic-loaded bone cement has previously been used as an effective method of treating implant-associated infection.19,20 In this method, for patients who develop infection after arthroplasty, the joint prosthesis or spacer is removed and the defect is filled with bone cement containing antibiotics, and the joint prosthesis or spacer is replaced again when the infection has settled. However, local concentrations of the antibiotics rise for the first few hours and sharply decline within the next few days in this method. Although the local concentration is several times higher than the MIC for about 1 month after bone cement placement,21,22 this is far below the MBEC required to inhibit biofilm formation. Some authors have reported that the low level of antibiotics from bone cements could cause bacterial biofilm formation on the surface of antibiotic-loaded bone cement.23,24 Conversely, CLAP can maintain the required local concentration of the antibiotics by monitoring the concentration in the leachate fluid and adjusting it accordingly. To monitor gentamicin concentrations in the leachate fluid, gentamicin levels were kept high throughout the administration period, suggesting that sufficient local concentrations were maintained during the application of CLAP. As another advantage, CLAP can approach the site of infection from a distance and does not require the placement of artifacts directly into the lesion.
The standard treatment for FRI is thorough debridement, including removal of the implant. However, removal of the implant early after osteosynthesis, when fracture union has not been achieved, contributes to the instability of fracture site and is naturally detrimental to fracture union process. In addition, even if cement containing antibiotics is placed in the defect after debridement, the concentration of antibiotics required for biofilm eradication will never be reached. CLAP allows preservation of the implant by administering a sufficient concentration of antibiotics locally, and by adjusting the local concentration appropriately, FRI can be treated without systemic side effects and without interfering with fracture union process, which we believe is a significant advantage over conventional treatment methods.
Negative-pressure wound therapy with instillation in combination with local infusion is an efficient local perfusion method for agents and has been reported to be effective in controlling infection in cases of posttraumatic osteomyelitis and infections associated with implants.25,26 We expect that the combined use of negative-pressure wound therapy and iMAP will be efficient in guiding locally administered antibiotics through the infected lesion and into the drainage tube according to the pressure gradient. Currently, we have started the treatment with iMAP in combination with NPWT for FRI in our hospital, and we plan to summarize their treatment outcomes and present it in the future.
There are some limitations to this study. First, during the initiation of this treatment, due to the absence of protocol to measure the concentrations, the data about agent concentration were not available for some patients. Second, since we basically apply iMAP to all patients with FRI early after osteosynthesis in our hospital, there are no cases treated with other methods (comparison group). We consider that it is also important to compare the treatment outcomes with those of cases treated with other methods, which is for our further study. Third, this study included only 10 patients of FRI, treated with iMAP alone in our hospital at earlier period; now, most of FRIs are treated in combination with iSAP: another perfusion system for soft tissue. Since the purpose of this study was to investigate the therapeutic effect of iMAP alone, which is the basis of this treatment method, only 10 cases treated with iMAP alone were presented. We are currently treating many patients with FRI with a combination of iMAP and iSAP, and we are preparing to summarize their treatment outcomes for a future presentation.
Conclusion
We treated FRI with CLAP by iMAP early after osteosynthesis and could eradicate infections while preserving implants in all patients. As a novel drug delivery system, CLAP has the advantage of being able to distribute high concentrations of antibiotics locally, making it a useful option to treat FRI.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This study was approved by the institutional review board of our hospital.
Informed consent
Written informed consent was obtained from all patients whose clinical course or images are presented in this article.
Consent for publication
Patients provided signed informed consent forms regarding the publication of their data and photographs.
