Abstract
Purpose:
The purpose of this study was to evaluate the implication of gait speed, which is a prerequisite for the diagnosis of sarcopenia in older patients with lumbar spinal stenosis (LSS).
Methods:
This study was conducted in a total of 235 patients with LSS who underwent surgical treatment. The state of sarcopenia and pre-sarcopenia, including gait speed, were evaluated before and after the operation.
Results:
The proportion of patients with lower than at baseline levels of skeletal muscle mass index (SMI) and gait speed was 27.2% and 17.9%, respectively. Significant changes were observed in gait speed, whereas SMI showed no significant differences between the preoperative and postoperative periods. Sarcopenic patients presented lower levels of activities of daily living preoperatively than pre-sarcopenic patients. However, favorable surgical results were obtained postoperatively. Significant changes were observed in gait speed, whereas grip strength and SMI showed no significant differences between the preoperative and postoperative periods. The postoperative decrease in SMI was not significant.
Conclusion:
Postoperative gait speed was significantly improved, whereas muscle mass did not increase in the patients in this study. Therefore, low gait speed in patients with LSS seems to be derived from a neurologic disorder. The surgical results in low muscle mass patients without low gait speed were similar to those with low gait speed. Sarcopenia in elderly patients with locomotor disease should be evaluated using muscle mass alone without assessing physical performance.
Keywords
Introduction
Sarcopenia, a neologism that refers to the loss of skeletal muscle mass, was first described in 1989 to allude to the age-related involuntary loss of skeletal muscle mass and function. 1 Since then, the concept of sarcopenia has become widely accepted and used in the field of gerontology to identify physical, metabolic, and functional impairment in elderly persons. 2– 4 Recently, the attention and importance of sarcopenia in the clinical scene in orthopedic surgery have increased in the context of its prognostic significance to surgical outcomes in orthopedic patients. 5 The definitions of sarcopenia provided by the European Working Group on Sarcopenia in Older People (EWGSOP) 6 and the Asian Working Group for Sarcopenia (AWGS) 7 use different strategies for normalization. However, the reduced grip strength and low gait speed have been identified as prerequisite factors for the diagnosis of sarcopenia. As gait speed has been associated with the health and functional outcomes, mortality, and survival in older adults in several previous studies, 8– 10 there is little doubt that gait speed is a potentially useful clinical and senescence indicator. Nevertheless, the etiology of poor physical performance, such as lower gait speed, is a multifocal and complex process influenced by sex, 11 race, 12 genetics, 13 cognitive function, 14 and psychological and environmental factors, 15 in addition to musculoskeletal problems. From the point of view of preventing decline in activities of daily living (ADL) in older patients with spinal disease, Park et al. demonstrated a high prevalence of sarcopenia in patients with degenerative lumbar spinal stenosis (LSS) and a negative effect of sarcopenia on clinical outcomes. 16 A previous clinical investigation also revealed that sarcopenia affected the outcomes of conservative treatment for osteoporotic vertebral fracture. 17 A systematic review of sarcopenic elderly patients who underwent spinal surgery in 2018 identified that sarcopenia was associated with an increased likelihood of postoperative complications, including mortality, morbidity, and hospital stay as independent risk factors for postoperative adverse events in elderly patients. 18 However, there is no consensus on the optimal method for evaluating sarcopenia, and the review highlighted the need for further studies to determine the clinical application. 18 Provided that quantitative assessment for the definition of sarcopenia is influenced not only by aging but also by locomotor lesions, the investigative values of sarcopenia for surgical outcomes should be no more than that of disease severity and may even be meaningless. Thus, the clinical implications of physical performance, such as gait speed, involved in the definition of sarcopenia should be discussed in the management of locomotor disease. The purpose of the present study was to investigate the impact of gait speed and low muscle mass on the surgical results in elderly patients with LSS.
Materials and methods
Participants
This case-control study was conducted in a total of 235 patients with LSS who underwent surgical treatment at our institute between April 2014 and March 2017 (135 male and 100 female elderly patients; mean age: 73.2 ± 5.8 years; age range: 65–91 years). The surgical procedures performed were lumbar decompression surgery for LSS without spondylolisthesis in 150 cases and posterior lumbar interbody fusion for spondylolisthesis in 85 cases. Single-level and multilevel surgery were performed in 177 and 58 cases, respectively. LSS was diagnosed on the basis of clinical symptoms such as leg numbness, leg pain, and intermittent claudication and confirmed by magnetic resonance imaging scan including the cauda equine. LSS patients with intermittent claudication who were able to ambulate within 10 m were included in this study, whereas the patients with LSS with abasia precluding the evaluation of 10-m gait speed, such as radicular pain, were excluded. Patients unable to walk with motor weakness of the lower extremities or with previous surgery in the back and/or extremity were also excluded. Ethical approval was granted by the institutional review board (approval number 1124), and all patients provided written informed consent.
Gait speed
Gait speed, expressed in meters per second, was calculated to cover a distance of 10 m in the usual walking pace. Participants were provided an additional 3 m for acceleration before the test distance and 3 m for deceleration thereafter. Two trials were performed, and faster speeds (m/s) were adopted for analysis. The cutoff point for low gait speed was 0.8 m/s in men and women. 7
Muscle strength
Grip strength was measured once in each hand using a Jamar-type dynamometer (SH5001; Sakai Medical Co., Ltd., Tokyo, Japan), and the average value was considered for analysis. The back and abdominal muscle strength were determined from the maximum isometric strength of the trunk muscles in a sitting posture with 30° lumbar extension (back muscle strength) or 30° lumbar flexion (abdominal muscle strength) using a digital muscle strength meter (Isoforce GT-300, 310; OG GIKEN Co., Ltd., Okayama, Japan). The strength of the quadriceps femoris (QF) muscle in each leg was evaluated once using a handheld dynamometer (μTas F-1; Anima Co., Ltd., Tokyo, Japan) with the patients’ hip and knees at 90° to each other in the initial position, and the average value was considered for analysis. The cutoff point for low grip strength was 26 kg in men and 18 kg in women. 7
Definition of sarcopenia and pre-sarcopenia
Body composition was assessed using a whole-body dual-energy X-ray absorptiometry (DXA) (Lunar iDXA, GE Healthcare, Tokyo, Japan). Whole-body scans provided the total lean body mass, total fat mass, and total body bone mineral content. Sarcopenia was defined as the loss of skeletal muscle mass in the arms and legs defined as appendicular skeletal muscle mass (ASM), and the skeletal muscle mass index (SMI) was obtained from ASM/height2 (kg/m2), according to the consensus of the AWGS. 7 Patients who met the criteria for both low grip strength and/or low gait speed and low muscle mass (defined as SMI < 5.4kg/m2 in women and < 7.0kg/m2 in men) were considered to have sarcopenia. According to the modified AWGS definition, pre-sarcopenia was defined as low muscle mass without low muscle strength and physical status. The control group was defined as patients with no loss of SMI irrespective of gait speed and grip strength (Figure 1). The preoperative and 1-year postoperative evaluations for sarcopenia were performed using DXA, gait speed test, and grip strength.

Algorithm for sarcopenia and pre-sarcopenia staging (modified AWGS 7 ). Pre-sarcopenia was defined as normal muscle strength and function with low muscle mass. The control was defined as no loss of SMI irrespective of gait speed and grip strength. AWGS: Asian Working Group for Sarcopenia; SMI: skeletal muscle mass index.
Radiographic measurement of the spine
Lateral radiographs of the lumbar and the whole spine were taken. The lumbar lordosis angle measured between the superior endplate of L1 and superior endplate of S1, and the lumbar range of motion, defined as the difference in the lumbar lordosis angle between flexion and extension, were measured. The sagittal vertical axis (plumb line from the center of the C7 vertebral body to the posterior sacral prominence) and thoracic kyphosis (Th2–Th12; sagittal Cobb angle from the superior endplate of Th2 to the inferior endplate of Th12) were measured. Radiographic measurements were performed using a calculation software (SYNAPSE®; Fujifilm Medical, Tokyo, Japan).
Evaluation of surgical outcomes
Surgical outcomes were assessed in 235 patients 1 year after treatment for LSS. The evaluations were performed using the Roland–Morris Disability Questionnaire, 19 the Euroqol quality of life–5D scale, 20 and Short Form–36 Physical Component Summary. 21 Subsequently, the preoperative and 1-year postoperative data were compared between sarcopenic and pre-sarcopenic patients.
Statistics
All data are presented as mean ± standard deviation. Statistical analysis was performed by adjusting for sex to compare pre-sarcopenia with sarcopenia using an analysis of covariance. Comparisons of the changes in parameters of sarcopenia and pre-sarcopenia were performed using a one-way analysis of variance for repeated measures, and Fischer’s protected least significant difference was used as a post hoc test. The changes between preoperative and postoperative parameters were analyzed using the paired t-test. Differences were considered statistically significant at p < 0.05. Data were analyzed using SPSS 12.0 (SPSS, Inc., Chicago, Illinois, USA).
Results
The number of patients with lower than at baseline levels of SMI, gait speed, and grip strength was 64 cases (27.2%), 42 cases (17.9%), and 71 cases (30.2%), respectively. The averaged gait speed in male patients was 1.24 ± 0.47 m/s with a significant difference compared with that in female patients (1.09 ± 0.50 m/s) (p < 0.05). In 64 patients with low SMI, either slow gait speed or low grip strength (sarcopenia) was seen in 33 cases (14.0%), and normal gait speed and normal grip strength (pre-sarcopenia) were seen in 31 cases (13.2%). The control group represented 72.8% of all the cases (171 cases). The preoperative data derived from each group are given in Table 1. There were no significant differences in the lumbar and global spine alignment between the pre-sarcopenia and sarcopenia group. Pre-sarcopenia showed a significant prevalence in male patients; thus, the comparison between pre-sarcopenia and sarcopenia was adjusted by sex. Additionally, the preoperative and 1-year postoperative measurements for each parameter of sarcopenia are shown in Figure 2. Significant changes were observed in gait speed (p < 0.01) whereas grip strength and SMI showed no significant differences between the preoperative and postoperative periods. There were no significant differences between decompression and fusion surgery, as well as between single-level and multilevel surgery. The changes in SMI, muscle strength, gait speed, and ADLs levels before and after surgery are given in Table 2. The postoperative decrease in SMI was not significant. All preoperative muscle strength and gait speed measurements were significantly lower in the sarcopenia group than in the pre-sarcopenia group. However, their significance disappeared postoperatively except for QF strength and grip strength. The improvement of gait speed in sarcopenic patients was similar to that in the control group. Sarcopenic patients presented lower ADL at preoperative state compared with pre-sarcopenic patients. However, favorable surgical results were obtained postoperatively in all three groups. Significant differences between sarcopenia and pre-sarcopenia were not seen at 1 year, but significant differences compared with control subjects were noted postoperatively.
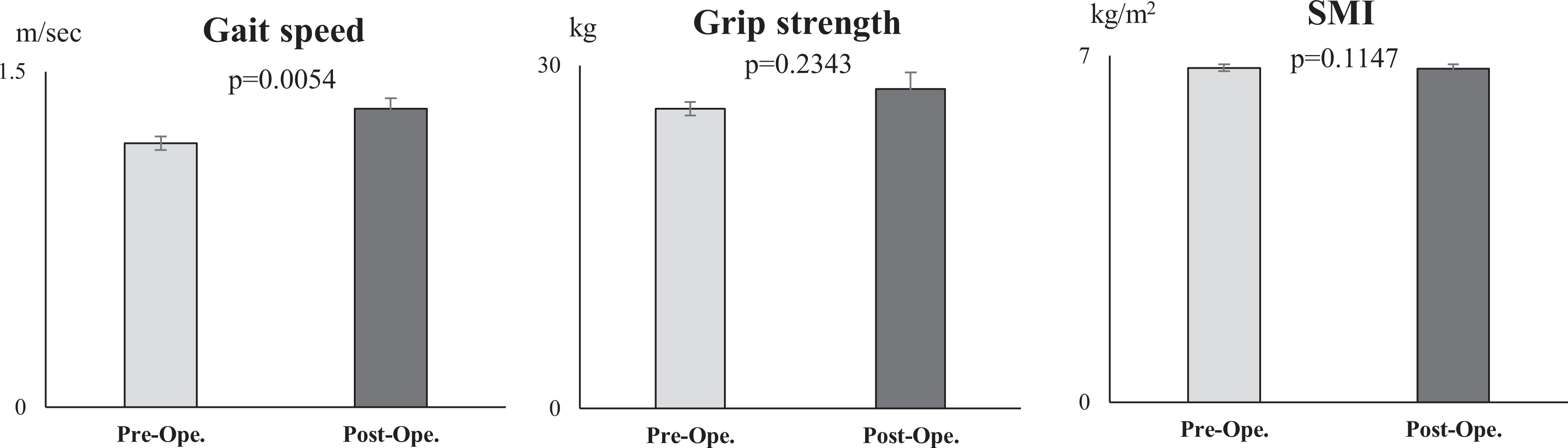
Preoperative and postoperative evaluation of sarcopenia in surgical patients with LSS. The preoperative and 1-year postoperative evaluations for each parameter of sarcopenia are shown. Significant changes were observed in gait speed, whereas grip strength and SMI showed no significant differences between the preoperative and postoperative periods. LSS: lumbar spinal stenosis; SMI: skeletal muscle mass index.
Preoperative comparisons among sarcopenia, pre-sarcopenia, and control group.
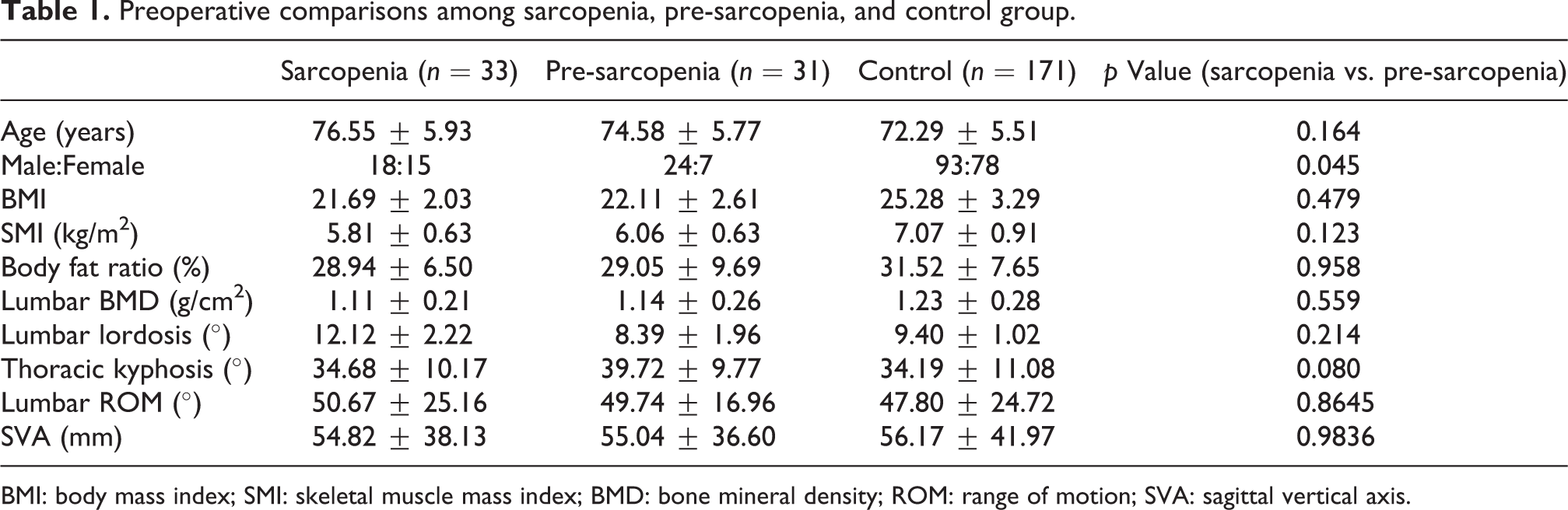
BMI: body mass index; SMI: skeletal muscle mass index; BMD: bone mineral density; ROM: range of motion; SVA: sagittal vertical axis.
Comparisons of surgical results among sarcopenia, pre-sarcopenia, and control group.a

SMI: skeletal muscle mass index; QF: quadriceps femoris; RDQ: Roland–Morris Disability Questionnaire; EQ-5D: Euroqol quality of life–5D scale; PCS: physical component summary; SD: standard deviation.
a Mean ± SD.
Discussion
Unlike osteoporosis, sarcopenia is defined as an age-related loss of muscle mass 1 and is not recognized as a clinical condition. This is because low muscle mass has a relatively small contribution to physical function in the elderly, 22,23 and no therapeutic options for sarcopenia have been developed to date. Thus, the EWGSOP recommends using the presence of both low muscle mass and low muscle function including strength and performance for the diagnosis of sarcopenia. 6 Morley and Cruz-Jentoft reported on the limits of using muscle mass alone for the definition of sarcopenia, suggesting that muscle mass by itself is a weak predictor of disability and mortality. 24 Moreover, the EWGSOP described that the rationale for using muscle mass and function is that muscle strength does not depend solely on muscle mass. 6 This concept is supported by two studies reporting the association of high mortality and low muscle strength compared with muscle volume, 25,26 four studies on the influence on physical disability, 27 –30 and two studies about the nonlinear association between muscle strength and mass. 29,31 Moreover, another study that recommended using both muscle strength and mass indicated molecular biological differences between muscle mass and strength, in which the decline of insulin-like growth factor-1 and growth hormone is related to the loss of muscle mass, but not necessarily muscle power. 32 Conversely, insulin resistance and low vitamin D have been shown to lead to a loss of muscle strength, but not muscle mass. 33 The significance of factors other than the muscle mass with respect to the concept of sarcopenia is understandable in that low muscle strength, but not low muscle mass, is associated with poor physical function such as gait speed. 27 Moreover, gait speed is associated with survival in older adults. 34 However, gaining muscle mass does not prevent the aging-related loss of muscle strength. 35 Thus, it seems that the age-related loss of muscle strength is weakly associated with the loss of muscle mass. Rather, muscle weakness is attributable to neurological mechanisms and/or skeletal muscle properties 36 in addition to the loss of voluntary control. Thus, several reviews have questioned the definition of sarcopenia because the use of low muscle mass and weakness is based on the literature and expert opinion, not on empirical evidence. Furthermore, it is not clear whether these factors are significant with respect to the outcomes of muscle strength and function. 37,38
The cutoff point of gait speed in the diagnosis of sarcopenia is 0.8 m/s. 6,7 This value was based on the study by van Kan, 39 who concluded that diminished gait speed should be considered as a marker of poor health status. 6 Their systematic review, which evaluated usual gait speed for adverse outcomes such as disability, cognitive impairment, falls, and/or mortality, demonstrated that a cutoff point of 1.0 m/s was used to predict mortality, whereas a cutoff point of 0.8 m/s was used to predict adverse health outcomes. Taken together, gait speed has been adopted in terms of ADL disability and health-related factors in elderly people, and not only just as a result of age-related loss of muscle mass. Thus, the conventional definition of sarcopenia is suitable for the indication of health status in community-dwelling elderly people. However, in patients with locomotor disease who are amenable, the evaluation of gait speed and grip strength before muscle mass measurement seems to be meaningless because locomotor disease-induced decline in muscle strength and physical performance is likely to show improvement after treatment.
The results of this study demonstrated poorer surgical outcomes both in sarcopenic and pre-sarcopenic LSS patients than in those without low muscle mass, and no significant differences in surgical outcomes between sarcopenic and pre-sarcopenic LSS patients. These results indicate that age-related muscle mass reduction should be considered, rather than gait speed as a clinical factor that affects the surgical outcome in the elderly patients with LSS. Additionally, the significant improvement in gait speed after surgery and the lack of significant change in muscle mass led to the conclusion that low gait speed cannot be attributed to sarcopenia, but to the walking disturbances derived from LSS. Additionally, gait speed, which showed significant improvement after treatment for LSS, was not reduced according to age-related loss of muscle mass. It is questionable whether another factor related to a parameter other than muscle mass should be included in the definition of sarcopenia. Furthermore, the surgical results in low muscle mass patients without low gait speed (pre-sarcopenia) were similar to those with low gait speed (sarcopenia). Thus, muscle mass seems to be a good predictor for the surgical result in patients with LSS, rather than gait speed. One of the limitations of the present study was that the analysis was restricted to LSS patients. Evaluation of other locomotor diseases that affect the muscle strength and/or gait speed is an essential criteria for the diagnosis of sarcopenia. However, it seems to be clinically realistic that muscle mass alone should be included as a criterion, at least in elderly patients with locomotor disease. The morbidity of LSS in the elderly was reported to be 128 per 1000 persons for the subgroup aged >65 years, and 155 per 1000 persons for the subgroup aged >75 years. 40 Apart from LSS, the prevalence of other locomotor disorders that affect muscle strength and gait speed, such as osteoarthritis, spondylosis, and osteoporosis, was much higher. 41 Age-related loss of muscle mass should be evaluated in the presence of a degenerative disease in elderly people. The contributors to dynapenia, which was defined as an age-related loss of muscle strength and power in 2008 by Clark and Manini, 42 were compartmentalized as neurologic and skeletal muscular factors. In geriatric medicine, whether the definition sarcopenia will include muscle performance and age-related loss of muscle mass or will consist of only muscle mass may vary with the objective outcomes. Therefore, age-related loss of muscle mass should not be a fixed concept involving both muscle strength and performance.
Conclusion
The impact of gait speed and low muscle mass on the surgical results in elderly patients with LSS was investigated to evaluate the validity of sarcopenia diagnosis. Postoperative gait speed was significantly improved, whereas muscle mass, which was evaluated using DXA, did not increase. Therefore, low gait speed in patients with LSS seemed to be derived from a neurologic disorder. The surgical results in low muscle mass patients without low gait speed were similar to those with low gait speed. Therefore, sarcopenia in elderly patients with locomotor disease should be evaluated with muscle mass alone without physical performance.
Footnotes
Authors’ Note
The device(s)/drug(s) is/are FDA approved or approved by the corresponding national agency for this indication. The content is solely the responsibility of the authors and does not necessarily represent the official views of the sponsors.
Acknowledgements
The authors would like to thank J. Suzuki and M. Morita for their assistance with data preparation.
Author contributions
YS takes responsibility for the concept and analysis design and the integrity of the data; KT, TW, and HI for patient recruitment, data acquisition, analysis, and interpretation; NW and HM for the interpretation of data and preparation of manuscript and figures.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval was given by National Center for Geriatrics and Gerontology Ethics Committee.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Japan Society for the Promotion of Science (JSPS) KAKENHI Grant Number 15K10425 in 2015–2017.
