Abstract
Purpose:
Although the neutrophil–lymphocyte ratio (NLR) is a simple biomarker for inflammation, its diagnostic value for predicting surgical site infection (SSI) after spinal decompression surgery has not been extensively investigated. We aimed to determine the predictive value of NLR for SSI in patients undergoing spinal decompression surgery.
Methods:
We performed a retrospective observational study of patients who underwent spinal decompression surgery. Consecutive 254 patients were divided into an SSI group and a non-SSI group based on the presence of SSI. We evaluated which markers, including NLR, differed significantly between groups. We then determined the diagnostic cutoff values of these markers for the prediction of SSI based on the significance in the univariate analysis.
Results:
The incidence of SSI was 7 of 254 patients (2.8%). Univariate analysis showed that there were significant differences in the C-reactive protein (CRP) level at 1 day postoperatively; neutrophil and lymphocyte percentage and NLR at 3–4 days postoperatively; and CRP level, white blood cell count, neutrophil count and percentage, lymphocyte percentage, and NLR at 6–7 days postoperatively between SSI and non-SSI groups. Among these markers, the cutoff values of lymphocyte percentage and NLR at 3–4 days postoperatively for the prediction of SSI were ≤15.1% and ≥4.91, respectively. The cutoff values of lymphocyte percentage and NLR at 6–7 days postoperatively were ≤19.8% and ≥3.21, respectively.
Conclusions:
Lymphocyte percentage and NLR at 3–4 and 6–7 days postoperatively were useful markers for the early prediction of SSI in patients who had undergone spinal decompression surgery. These parameters may aid in identifying patients at higher risk of SSI after spinal decompression surgery.
Keywords
Background
With the advancement of an aging society, the number of spinal surgeries performed has been increasing. 1 –3 Accordingly, it is necessary to reduce complications, especially surgical site infection (SSI). The reported incidence of SSIs following spine surgery ranges from 0.5% to 18.8%. 4 SSI following spinal surgery often leads to reoperation, longer hospital stays, and deterioration in clinical outcomes; thus, it is considered a serious complication associated with considerable morbidity and economic burden. 5,6 Moreover, unlike general surgery, SSI after spinal surgery has a certain peculiarity; it can deepen into an epidural abscess, a pyogenic spondylitis, or meningitis. Therefore, the detection of SSI following spinal surgery during the early postoperative stage is important to allow prompt treatment in the mild phase of infection and shorten the entire treatment period.
While neutrophils are an important factor in biophylaxis against bacterial infection, lymphocytes are an important factor in nonspecific biophylaxis. 7,8 The characteristics of the initial stage of infection are increase in the number of neutrophils and decrease in the number of lymphocytes. 8,9 Recently, neutrophil–lymphocyte ratio (NLR) has been proposed as a useful biomarker for detecting bacterial infections. 10 –12 NLR is obtained by dividing the number of neutrophils (count/µL) by the number of lymphocytes (count/µL). 10 Thus, increased NLR is considered a possible sign of infection. Indeed, after posterior lumbar spinal surgery, a retrospective study showed that NLR at postoperative 7 days was a useful marker for detecting postoperative infections. 13,14 However, among patients who underwent posterior lumbar spinal surgery, the incidence and characteristics of SSI were reportedly different between those who underwent instrumentation surgery and those who underwent non-instrumentation surgery. 4,15 Moreover, laboratory markers such as C-reactive protein (CRP) and white blood cell (WBC) count are affected by addition of instrumentation. 16 Thus, in the case that laboratory markers are useful for the prediction of SSI after spinal decompression surgery, the cutoff value in spinal decompression surgery should be calculated separately from spinal instrumentation surgery. While the usefulness of NLR was established in the detection of bacteremia in emergency department, 10 diagnosis of bacterial infection among patients hospitalized for fever, 11 prediction of hospital-acquired bacterial infections in decompensated cirrhotic patients, 12 and prediction of SSI among patients following spinal instrumentation surgery, 14 there have been no reports which investigated the diagnostic value of NLR for the early prediction of SSI and its cutoff value in spinal decompression surgery. Therefore, the diagnostic value of NLR for predicting SSI in spinal decompression surgery cohort should be investigated.
We hypothesized that NLR might have a predictive role for SSI in patients who underwent spinal decompression surgery. To test this hypothesis, the purpose of this study was to investigate whether NLR could be a useful predictor of SSI after spinal decompression surgery.
Patients and methods
Study design and population
A retrospective observational study was conducted to investigate the incidence of SSI and predictors for SSIs in patients who underwent spinal decompression surgery in the Department of Orthopaedic Surgery at Tokyo Medical and Dental University Hospital in Japan. Between April 2014 and April 2018, consecutive hospitalized patients who underwent spinal decompression surgery were retrospectively enrolled. We excluded the patients with spinal infectious diseases such as pyogenic spondylitis at the time of spinal decompression surgery and with other sources of infection, such as urinary tract infection, in the early postoperative period. The final sample included 254 patients; these patients were divided into either the SSI group or the non-SSI group based on the diagnosis.
Our institutional research ethics committee approved this study. Informed consent was waived because it was a retrospective study and patient data were anonymized.
Perioperative management
Prophylactic antibiotics were administered during and after surgery for a total of 48 h, according to the Japanese Orthopaedic Association Clinical Practice Guideline on the Prevention of Surgical Site Infections in Bone and Joint. 17 The patients received cefazolin (1.0 g, intravenous) 30 min before skin incision. The antibiotic was administered in additional doses of 1.0 g cefazolin intravenous every 3 h during surgery and every 12 h after surgery. Clindamycin was used for patients with β-lactam allergies.
Definition of SSI
Surgeons checked for SSIs every day during the hospital stay and at every outpatient clinic visit until 30 days post-surgery. SSI was defined based on the definition of the Guideline for Prevention of Surgical Site Infection, 1999, as superficial, deep, or organ/space infections occurring within 30 days of the surgery. 18 The definition of SSI in this study included a superficial and deep incisional SSI and organ/space SSI.
Briefly, superficial incisional SSI was diagnosed if at least one of the following criteria was met: Purulent drainage from the superficial incision. Organisms isolated from an aseptically obtained culture of fluid or tissue from the superficial incision. At least one of the following signs or symptoms of infection: pain or tenderness, localized swelling, redness, or heat, and superficial incision is deliberately opened by a surgeon, unless incision is culture-negative. Diagnosis of superficial incisional SSI by the surgeon or attending physician.
Deep incisional SSI was diagnosed if at least one of the following criteria was met: Purulent drainage from the deep incision but not from the organ/space component of the surgical site. A deep incision spontaneously dehisces or is deliberately opened by a surgeon when the patient has at least one of the following signs or symptoms: fever (>38°C), localized pain, or tenderness unless the site is culture-negative. An abscess or other evidence of infection involving the deep incision is found on direct examination, during reoperation, or by histopathologic or radiologic examination. Diagnosis of a deep incisional SSI by a surgeon or attending physician.
Organ/space SSI was diagnosed if at least one of the following criteria was met: Purulent drainage from a drain that is placed through a stab wound into the organ/space. Organisms isolated from an aseptically obtained culture of fluid or tissue in the organ/space. An abscess or other evidence of infection involving the organ/space that is found on direct examination, during reoperation, or by histopathologic or radiologic examination. Diagnosis of an organ/space SSI by a surgeon or attending physician.
Clinical data collection
We collected data on the CRP level, WBC count, percentages of neutrophils and lymphocytes, and NLR preoperatively and 1, 3–4, and 6–7 days postoperatively. The number of decompressed segments, comorbidities, such as diabetes mellitus and usage of steroids, operative time, intraoperative blood loss, types of surgery, and hospital stays after surgery were also recorded. NLR was obtained by dividing the number of neutrophils (count/µL) by the number of lymphocytes (count/µL). 10
Statistical analysis
Differences between the SSI and non-SSI groups were analyzed using the Mann–Whitney U-tests for continuous variables and Fisher’s exact test for nominal variables. Receiver operating characteristic (ROC) curve analysis was conducted to assess the cutoff point for the prediction of SSI. Accordingly, the dependent variable was defined as the presence of SSI, and the independent variables were determined based on the significance of univariate analysis. For all statistical analyses, JMP version 12 (SAS Institute, Cary, North Carolina, USA) was used, and a p value of <0.05 was considered to indicate statistical significance.
Results
Demographics
The incidence of SSI was 7 of 254 patients (2.8%). Patients were divided into an SSI group and a non-SSI group according to the presence of an SSI. The SSI group included 7 male patients (2.8%), whereas the non-SSI group included 159 male and 88 female patients (Table 1). The mean age at surgery was 66.7 (standard deviation (SD): 9.6) years in the SSI group and 65.8 (SD: 14.3) years in the non-SSI group. The operational features were as follows: mean operative time, 190 (SD: 66) min in the SSI group and 155 (SD: 80) min in the non-SSI group; and mean intraoperative blood loss, 158 (SD: 140) mL in the SSI group and 127 (SD: 161) mL in the non-SSI group. There was no significant difference between the groups with respect to any other background parameter (Table 1). As expected, the duration of hospital stay after surgery was significantly longer in the SSI group.
Demographic data of the patients.a

SSI: surgical site infection; HbA1c: hemoglobin A1c; SD: standard deviation.
a Data are presented as mean (SD) or as counts.
b Statistically significant (p < 0.05).
SSI cases
Among the seven patients who developed SSI, five underwent debridement. The other patients were treated with antibiotics. The positive rate of wound culture in the SSI group was 100%. The results of the wound culture are shown in Table 2. All patients recovered (Table 2).
Patient characteristics (SSI group).
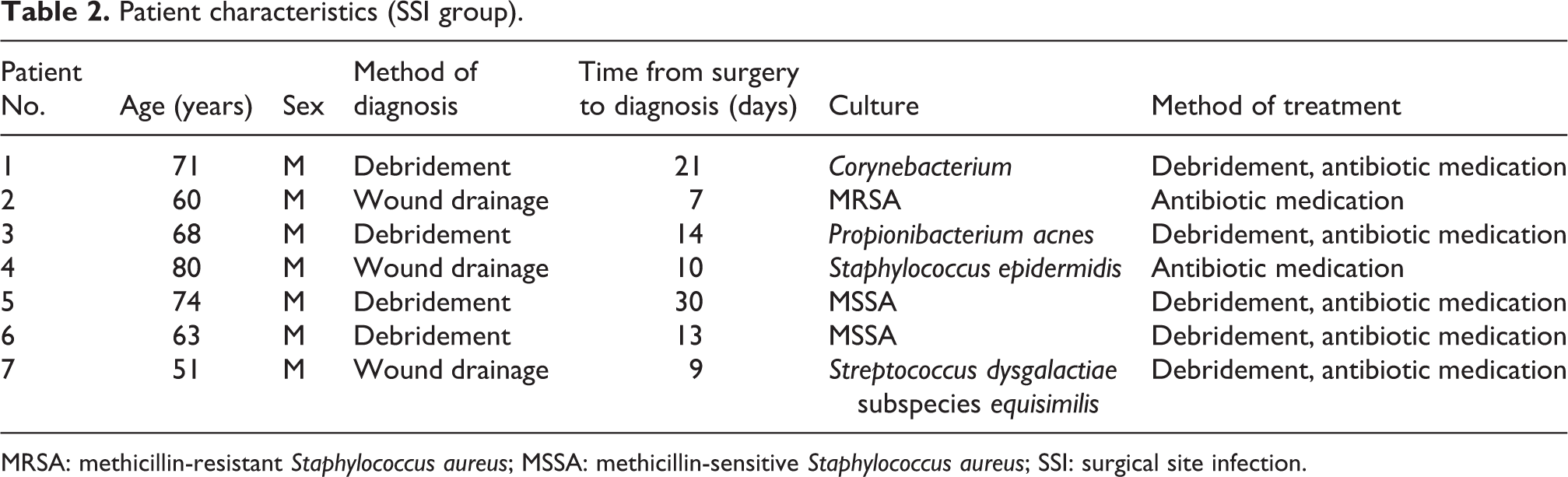
MRSA: methicillin-resistant Staphylococcus aureus; MSSA: methicillin-sensitive Staphylococcus aureus; SSI: surgical site infection.
Laboratory markers
Univariate analysis showed that CRP at 1 day postoperatively; neutrophil and lymphocyte percentage and NLR at 3–4 days postoperatively; and CRP, WBC count, neutrophil count and percentage, lymphocyte percentage, and NLR at 6–7 days postoperatively were significant predictors of SSI (Table 3).
p Values for the statistical analysis of biochemical markers in the SSI and non-SSI groups.

a Statistically significant (p < 0.05).
CRP: C-reactive protein; NLR: neutrophil–lymphocyte ratio; SSI: surgical site infection; WBC: white blood cell.
Cutoff value of lymphocyte percentage and NLR for the prediction of SSI
In order to predict SSI in the acute phase, the appropriate cutoff values of these markers were determined using ROC analysis based on the p value in the univariate analysis. The cutoff value for CRP at 1 day postoperatively was ≥2.52 (sensitivity, 83.3%; specificity, 63.2%; area under the curve (AUC), 0.752); for lymphocyte percentage at 3–4 days postoperatively, ≤15.1% (sensitivity, 66.7%; specificity, 81.8%; AUC, 0.760); for NLR at 3–4 days postoperatively, ≥4.91 (sensitivity, 66.7%; specificity, 81.7%; AUC, 0.748); for lymphocyte percentage at 6–7 days postoperatively, ≤19.8% (sensitivity, 85.7%; specificity, 79.6%; AUC, 0.847); and for NLR at 6–7 days postoperatively, ≥3.21 (sensitivity, 85.7%; specificity, 77.7%; AUC, 0.800) (Table 4).
Cutoff value of biochemical markers for the prediction of SSI.
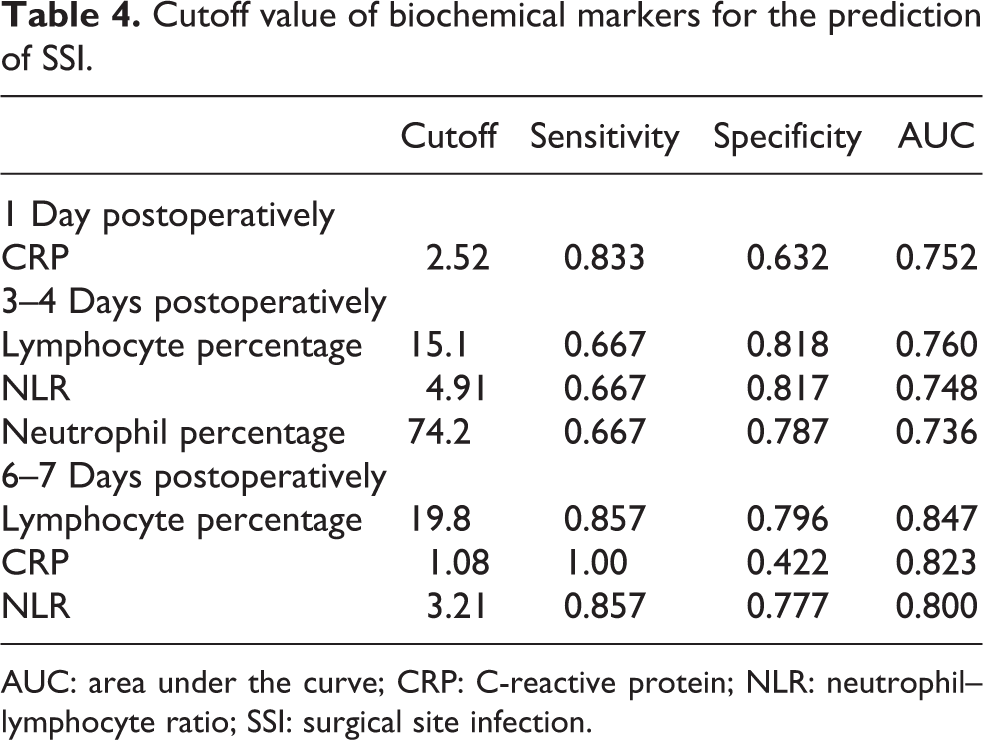
AUC: area under the curve; CRP: C-reactive protein; NLR: neutrophil–lymphocyte ratio; SSI: surgical site infection.
Discussion
We investigated the early postoperative predictors of SSI after spinal decompression surgery. Univariate analysis showed CRP at 1 day postoperatively; neutrophil and lymphocyte percentage and NLR at 3–4 days postoperatively; and CRP, WBC count, neutrophil count and percentage, lymphocyte percentage, and NLR at 6–7 days postoperatively were significant predictors of SSI. The cutoff values of lymphocyte percentage and NLR at 3–4 days postoperatively for the prediction of SSI were ≤15.1% and ≥4.91, respectively. The cutoff values of lymphocyte percentage and NLR at 6–7 days postoperatively were ≤19.8% and ≥3.21, respectively. To the best of our knowledge, this is the first study to show that NLR is useful for predicting SSI after spinal decompression surgery and determine its cutoff value.
We found that lymphocyte percentage at 3–4 and 6–7 days postoperatively was significantly lower in the SSI group. A case-control study showed that lymphocyte percentage and counts and neutrophil percentage at 4 days postoperatively were reliable markers indicating SSI after posterior lumbar decompression surgery. 16 Thus, the continuous decrease in lymphocyte percentage after spinal decompression surgery is a potential indicator of SSI.
We also found that NLR at 3–4 and 6–7 days postoperatively was significantly higher in the SSI group. A retrospective study showed that the cutoff value for NLR at 6–7 days postoperatively for the prediction of SSI after spinal instrumentation surgery was 3.87. 14 Another retrospective study also showed that NLR at 4 days postoperatively could significantly discriminate the SSI and non-SSI groups after posterior lumbar spinal surgery. 13 Thus, the use of NLR would offer some diagnostic utility for the early prediction of SSI after spinal surgery. However, the results of our ROC analysis may also indicate that NLR offers no advantage over lymphocyte percentage for the early prediction of SSI after spinal decompression surgery because it has lower specificity and AUC.
Based on our research, if the lymphocyte percentage is less than 15.1% or if NLR is more than 4.91 at 3–4 days postoperatively after spinal decompression surgery, surgeons should consider the possibility of SSI occurrence. Further, if the patient has fever, redness in or around the wound, or pus coming from the wound, a more intensive diagnostic approach such as culture of the exudate (pus from the wound), magnetic resonance imaging (MRI), and blood culture is recommended. After the definitive diagnosis of SSI, surgeons should start administering antibiotics and/or perform debridement.
There are some limitations in this study. First, our data were obtained from a single institution and were based on information listed in the medical records. Second, our study was a retrospective analysis of suspected predictors. Thus, a potential bias associated with patient selection and that due to missing patient information cannot be overlooked. Third, the number of patients with SSI was relatively small. However, despite the small sample size, differences were found between the SSI and non-SSI groups. Fourth, we did not evaluate the correlation between imaging methods and blood tests. However, while MRI is traditionally thought to be the most sensitive modality for evaluation of postoperative infections, there is a high false-positive rate of MRI. 19 Moreover, it is difficult to differentiate active infection from ongoing reparative and/or fibrotic tissue using MRI. 20 Large-scale prospective studies are recommended to overcome these limitations and confirm the effectiveness of these laboratory markers on the prediction of SSI following spinal decompression surgery.
Conclusion
The current study demonstrated that lymphocyte percentage and NLR at 3–4 and 6–7 days postoperatively are useful markers for the early prediction of SSI in patients who had undergone posterior spinal decompression surgery. These markers can help identify patients at higher risk of SSI after spinal decompression surgery. More research is needed to further determine the role of lymphocyte percentage and NLR in SSI prediction and treatment.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
