Abstract
Background:
Percutaneous vertebral augmentation (PVA) under local anesthesia has been widely used to treat osteoporotic vertebral compression fractures and vertebral body tumors. However, the occurrence of spinal cord or nerve root dysfunction may result in poor prognosis for patients. The aim of this study was to analyze the causes of transient paraplegia in 12 patients undergoing PVA.
Methods:
The medical records of 12 patients with transient paraplegia during PVA in our hospital were analyzed. Data, including operation, vertebral, anesthetic dose, operation time, recovery time, and follow-up, were extracted.
Results:
Among the 12 patients, ranging in age from 62 years to 83 years, with a mean age of 74 years, 8 were females and 4 were males. The average anesthetic dose injected per vertebral body was 6.38 ml. Patients required an average of 218.75 min to recover sensation and movement completely. However, the amount of anesthetic injected into each vertebral body was not related to the time required for complete recovery. Follow-up showed that all patients had regained normal bilateral sensation and motor function. Postoperative visual analog scale and Oswestry Disability Index values of the 12 patients were significantly improved compared with preoperative values.
Conclusion:
The complication of transient paraplegia was caused by local anesthetic drugs infiltrating into the spinal canal and inhibiting nerve conduction in the spinal cord.
Introduction
Percutaneous vertebral augmentation (PVA) includes percutaneous vertebroplasty (PVP) and percutaneous kyphoplasty (PKP). Both PVP and PKP have the characteristics of restoring local biomechanical stability and relieving pain, which significantly improve the quality of patients’ life, and have fewer complications compared to conservative treatments like bed rest. While both of these treatments have been currently recognized as safe, simple, and minimally invasive surgical interventions, serious complications may occur during these procedures.
The complications of PVP and PKP mainly occur at one of the following three stages: (1) the puncture stage (these include soft tissue hematoma formation, rib and pedicle fracture, adjacent organ injury, as well as spinal cord and nerve injury) 1,2 ; (2) the bone cement injection stage (may include various symptoms caused by bone cement leakage) 3 ; (3) the postoperative phase (these include surgical vertebral refracture and adjacent vertebral collapse). 4
The occurrence of spinal cord or nerve root dysfunction may result in poor prognosis for patients. Thus, the aim of this study was to present a case series of 12 patients who had PVA with complications and analyze the causes of transient paraplegia.
Patients and methods
Twelve patients who had transient paralysis after undergoing PVA were retrospectively included. These patients were admitted into our hospital for severe back pain and limited spinal activity between January 2009 and January 2017. Data, including diagnosis, operation, vertebral location, anesthetic dose, operation time, recovery time, and follow-up, were extracted.
All patients had received conservative treatment before admission, but their symptoms did not improve at all (visual analog scale (VAS) ≥ 6, Oswestry Disability Index (ODI) ≥ 68%). Before surgery, X-ray and magnetic resonance imaging (MRI) or bone scan examinations were used to confirm the diagnosis.
Preoperative examinations were used to exclude surgical contraindications such as coagulation mechanism disorder, systemic infection, and spinal cord injury. Written informed consent was obtained from each patient before the operation. PKP or PVP was performed under local anesthesia by the same surgeon, who had 15 years of experience. Local anesthetic, consisting of 5% lidocaine and 1% ropivacaine at an equal dose, was administered. The target point was located at the body surface of pedicle projection under X-ray guidance. The local anesthetic was injected subcutaneously to the periosteum. If the patient experienced intraoperative pain, the surgeon introduced the local anesthetic by syringe needle into the vertebral body through the work channel. Generally, 5–7 ml was injected into every vertebral segment. Because of the occurrence of transient paralysis, every patient was followed-up for 6 months. Unfortunately, two patients who had cancer with vertebral metastases died of organ failure within 6 months.
General data, including gender, age, and diagnosis, are listed in Table 1. Surgical data, including operation, vertebral location, anesthetic dose, operation time, recovery time, and leg sensation and movement, are listed in Table 2.
Patient data.
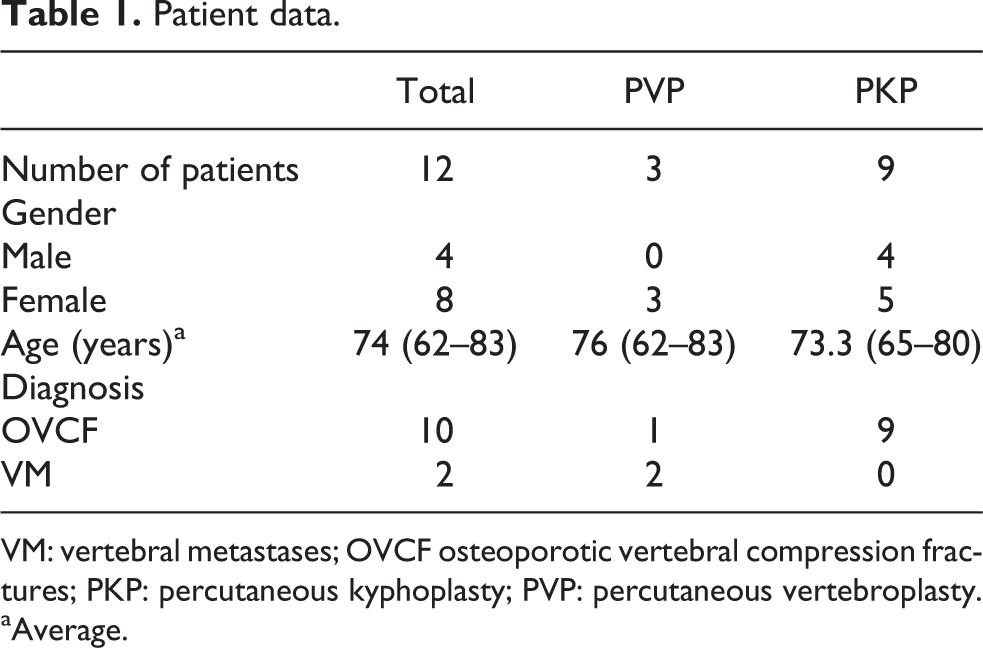
VM: vertebral metastases; OVCF osteoporotic vertebral compression fractures; PKP: percutaneous kyphoplasty; PVP: percutaneous vertebroplasty.
aAverage.
Details of patients with complications.
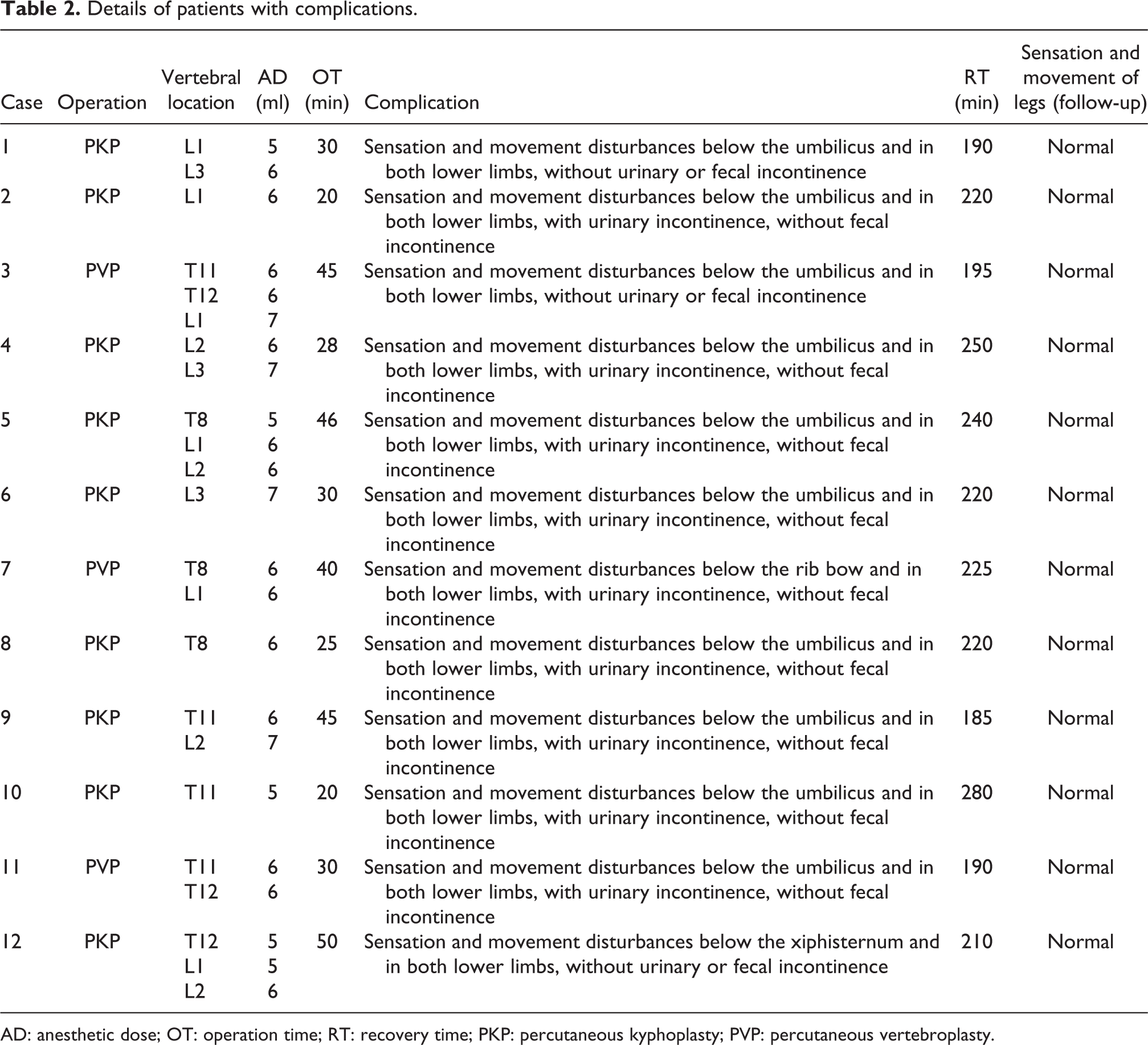
AD: anesthetic dose; OT: operation time; RT: recovery time; PKP: percutaneous kyphoplasty; PVP: percutaneous vertebroplasty.
Characteristics and treatment of transient paraplegia
Among all patients, four presented with impaired leg sensation and movement immediately after the operation, and eight had symptoms of paraplegia when they returned to the ward after surgery. All patients showed progressive numbness and decreasing muscle strength in both legs. Their tendon reflex could not be elicited and their anal sphincters were in a relaxed state. The patients had different sensory disturbances in the chest or abdomen, with or without incontinence symptoms. All patients had negative neuropathological signs. Their heart rate, blood pressure, and oxygen saturation were within the normal range.
All patients received MRI tests (see Figure 1) of the spinal cord and nerves immediately after presenting with symptoms of paraplegia. Fortunately, there was no spinal cord puncture injury or bone cement leakage. However, every patient who had minor spinal cord injury was injected with methylprednisolone 200 mg intravenously, after which the paraplegia in both legs began to improve. First, all patients regained muscle strength and movement function in both lower limbs, then the sensation in different areas of the chest and abdomen, as well as the lower extremities, began to recover. Most of the patients completely regained both movement and sensation of the lower limbs in about 4 h.

Postoperative images of all 12 patients are presented as sequential X-ray, CT, and MRI (T1, T2, T2-weighted fat saturation sequence scan). CT: computed tomography; MRI: magnetic resonance imaging.
Preoperative patients independently completed the ODI and VAS. Postoperatively, X-ray and computed tomography were performed to observe the distribution of the bone cement (Figure 1). The ODI scores and visual analog scores were followed up 1, 3, and 6 months postoperatively. Patients with vertebral metastases received further treatment in the Department of Oncology.
Data analysis
Baseline characteristics and disease-related information were analyzed using SPSS 22.0 (IBM, USA) statistical software.
Results
Among 2000 patients treated by PVA, 12 suffered transient paraplegia, resulting in an incidence rate of 0.6% for this complication. The 12 patients (4 males and 8 females) were between 62 years and 83 years of age, with a mean age of 74 years. The average anesthetic dose injected into each vertebral body during surgery was 6.38 ml. Patients required an average of 218.75 min to recover sensation and movement completely. However, the amount of anesthetic injected into each vertebral body was not related to the time required for complete recovery.
Follow-up examinations showed that all patients regained normal bilateral sensation and motor function. Postoperative VAS and ODI of the 12 patients were significantly improved compared with preoperative values (p < 0.0001, Table 3).
Preoperative and postoperative VAS and ODI.a

VAS: visual analog scale; ODI: Oswestry Disability Index; SD: standard deviation.
aData are presented as mean ± SD. Paired sample’s t-test.
b p < 0.0001 (compared with preoperative values).
Discussion
During PVA, spinal nerve puncture injury should be considered first when double-limb paraplegia occurs. Once spinal cord or nerve puncture injured, the patient will immediately present partial dysfunction of sensation and movement of the legs. In these cases, during the puncture stage, the sensation and movement of the lower extremities were normal in all of our patients. Immediate MRI did not show any abnormalities in the spinal cord. Therefore, the possibility of spinal cord puncture injury or bone cement leakage causing the patient’s transient paraplegia in these cases should be excluded.
Spinal cord concussion is defined as transient paralysis or sensory disturbance that subsides within 72 h. 5 –7 It features transient loss of sensation, movement, anal reflexes, and sphincter function below the plane of injury. In most cases, the pathological characteristics present a small amount of focal hemorrhage forming in the central gray matter of the spinal cord and a few degenerative changes in parts of nerve cells or axons. 8 A few weeks later, the bleeding in the spinal cord tissues will be completely absorbed and spinal cord function will completely recover. 6 In some cases, spinal cord concussion occurs without radiographic abnormalities, including on MRI. 9 , 10 As Nesnidal et al. 8 reported on the timing in a retrospective study, 24 patients with spinal cord concussions recovered from their symptoms within 76 h. The MRI scans of 21 (87.5%) of these patients showed no evidence of any traumatic spinal changes. All of the patients experienced a rapid resolution of their neurological deficits. In our case, during the puncture stage, frequently punching the puncture needle may vibrate spinal cord and inhibit nerve cells to be in a physiologically stagnant state. Therefore, spinal cord concussion may be one reason that causes transient paraplegia during PVA.
Methyl methacrylate (MMA) is a commonly used bone cement filling material for PVP. However, it is reported that MMA may cause the occurrence of bone cement implantation syndrome (BCIS). 11 , 12 BCIS is a series of clinical symptoms caused by MMA, including hypotension, arrhythmia, severe hypoxemia, myocardial infarction, pulmonary arterial pressure, coagulopathy, asthma attack, and so on. In order to achieve the purpose of fully filling the vertebral body, MMA applied in the vertebroplasty should be mixed at the stage of the low viscosity. If the viscosity is not well controlled, a large amount of unpolymerized monomer will be released into the blood circulation system, resulting in the occurrence of BCIS. 13 Razuin et al. 14 reported that MMA particles were found in lung, kidney, and other organs through autopsy of a dead patient. It is believed that a large number of monomers entering the blood circulation system are closely related to complications such as hypoxemia, hypotension, arrhythmia, and cardiac arrest. 15 , 16 The main mechanism may be that MMA interferes with calcium mobilization inside and outside the cell to affect the excitatory-contraction coupling process of smooth muscle, which leads to a decrease in blood pressure during MMA implantation. 17 , 18 In addition, a small number of patients performed a transient or long-term neurological dysfunction undergoing cement head cranioplasty without neurological complications related to surgery. The sensory nerve conduction velocity is significantly reduced if the finger contacts with MMA for a long term because of local neurotoxicity and axonal degeneration. 19 Gosavi et al. 20 observed a decrease in tracheal and bronchial cilia, peribronchial lymphoid follicular hyperplasia and pulmonary capillary congestion, increased concentrations of severe pulmonary edema, lungs emphysema, and pneumothorax if the rats were exposed to 0.45 ppm of MMA monomer. Thus, there is a possibility MMA may cause spinal cord injury and transient paraplegia.
Finally, the possibility of hysterical paralysis also needs to be considered. Hysterical paralysis is mainly caused by psychological factors, and it mostly occurs in female patients. 21 The clinical characteristics of hysterical paralysis include paraplegia, hemiparesis, and single paralysis. Physical examination shows normal or enhanced tendon reflex with no pathological reflexes or other positive signs of the nervous system. The incidence of hysterical paralysis has been reported to be between 0.3% and 3.8%. 22 It is important to distinguish hysterical paraplegia from pathological paraplegia. The key symptoms of hysterical paralysis are as follows: (1) There is an absence of symptoms like anal sphincter disorder, skin dystrophy due to bedsores, or muscle atrophy. Tendon and superficial reflexes are normal. (2) The patient often suffers from mental disorders and appears intolerant and irritable. In our study, mental factors may be an important cause of morbidity for patients suffering from long-term back pain. In addition, the surgeon repeatedly asked the patients about feeling and sensation in their legs during the operation, which may be a psychological trigger to induce paraplegia. However, hysterical paralysis can be identified through careful examination. In our study, the tendon and superficial reflexes of patients were completely absent, which is not characteristic of hysterical paraplegia. Therefore, the probability of hysterical paralysis causing transient paraplegia in our patients is very low.
Based on an analysis of the occurrence and recovery process of transient paraplegia, we concluded that this complication was caused by the local anesthetic. During the placement of the vertebroplasty needle, we might have violated the dura, either medially or caudally, where there was a breach of the dura mater. While the injury to the dura was too small to be discovered on the MRI, the local anesthetic drugs were able to infiltrate the spinal canal and block the spinal cord. As a result, patients suffered progressive physical sensation disorders, muscle weakness, and dyskinesia. Due to the fact that only a small amount of local anesthetic had been used, its effect did not last for a long time. All patients regained their function of movement first, followed by the recovery of sensation. All patients returned to their preoperative state after the effect of the anesthetic drugs wore out completely. The gradual recovery process was similar to that of spinal subarachnoid anesthesia and in line with the characteristics of spinal cord nerve block.
The results of our study suggest that, during the puncture stage, adding anesthetic through the bone cement injection channel should be avoided, because it can lead to the rapid spreading of the anesthetic drugs in the cancellous bone. Furthermore, if the anesthetic drugs get into the venous circulation, they may cause general anesthesia. Thus, if the patient cannot tolerate the pain, the anesthetist should administer analgesic drugs and sedatives intravenously. As a rule, the puncturing should be done accurately, strictly following the operating specifications, in order to avoid complications. In addition, the surgery should involve no more than three vertebra segments per operation. Finally, the surgeon should pay attention to the exploration of the puncture channel during surgery to ensure the integrity of the pedicle internal wall and carefully control the amount of local anesthetic to avoid accidental anesthesia.
Conclusion
The infiltration of local anesthetic drugs into the spinal canal may temporarily inhibit nerve conduction in the spinal cord and cause transient paraplegia during PVA surgery.
Limitations
The present study had some limitations. First, based on available information, the mechanism of transient paraplegia was not elucidated clearly in this study. Therefore, it is necessary to conduct the basic experiment to explore how paraplegia occurred after PVA. In addition, although we reach the conclusion that local anesthetic drugs cause transient paraplegia by analyzing 12 cases, a large study of many patients is necessary to further validate local anesthetic drugs infiltrate into the spinal canal and temporarily inhibit spinal cord.
Footnotes
Author contribution
Guan Shi and Fei Feng contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
