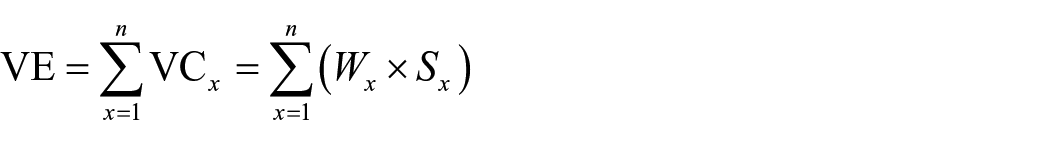Abstract
Patients with moderate to severe haemophilia A are at a higher risk of developing FVIII inhibitors that require the use of more costly and less effective treatments. The objective of this study was to determine the value of emicizumab for the prophylaxis of haemophilia A with inhibitors compared to the current therapeutic alternatives, activated prothrombin complex concentrate and recombinant factor VIIa through reflective Multi-Criteria Decision Analysis. The EVIDEM framework adapted to orphan drugs and weighted by a sample of 98 national and regional Spanish evaluators was used. Two structured evidence matrices were developed: emicizumab against activated prothrombin complex concentrate and emicizumab against recombinant factor VIIa. A multidisciplinary team of haemophilia experts rated each of the criteria. Mean and standard deviation were calculated by each criterion and discussed among all participants. Haemophilia A with inhibitors was perceived as a severe disease with high unmet needs. Emicizumab was rated with higher efficacy, therapeutic benefit and quality of life than comparators. When administered alone for the prevention of bleeding events, emicizumab had slightly better safety and tolerability profile than activated prothrombin complex concentrate and similar with recombinant factor VIIa. The inclusion of emicizumab in clinical practice guidelines was valued positively by the members of the panel. Overall, value of emicizumab was higher than activated prothrombin complex concentrate and recombinant factor VIIa, mostly because of efficacy and therapeutic benefit in reducing treated haemorrhages. Reflective Multi-Criteria Decision Analysis has proven to be a feasible method to determine the value contribution of comparative therapies in haemophilia.
Introduction
Haemophilia A is a rare congenital (X chromosome) bleeding disease caused by the deficiency of coagulation factor VIII (FVIII). It is estimated that 80%–85% of all the haemophilia patients have haemophilia A. 1 In Spain, similar to European studies,2,3 it was estimated that 84.1% of the total haemophilia patients might have haemophilia A. Approximately 51.3% of haemophilia A patients have mild haemophilia A, 16.4% have moderate haemophilia A and 32.3% have severe haemophilia A. 2
Haemophilia A has no cure, and therefore, its clinical management is focused not only on treating the bleeding events but also on preventing them. The most serious complication of the treatment of haemophilia A is the development of inhibitors, an immunologic response to the treatment with FVIII. Inhibitors are classified as low-responding (<5 BU/mL) and high-responding (>5 BU/mL at least once). Patients with low-responding inhibitors might have less clinical problems because haematosis can be ensured by saturating the inhibitor through the administration of higher doses of the deficient factor. However, high-responding inhibitors rule out the standard on demand therapy and prophylaxis and require alternative treatments which have poorer efficacy and safety profiles than factor concentrates. 3 In Spain, it was estimated that 11.6% of patients with severe haemophilia A and 4.7% of patients with moderate haemophilia A might develop inhibitors. 4 Bypassing agents are used for preventing and treating bleeding events in haemophilia A patients with inhibitors. 3
The main unmet need in haemophilia A patients is to treat patients who have developed inhibitors. 1 Recently, emicizumab has proven efficacy in reducing treated bleeding events in haemophilia A patients with FVIII inhibitors 5 and was safe when administered alone or in conjunction with recombinant factor VIIa (rFVIIa). 6
To assess the value of new drug treatments, multi-criteria decision analysis (MCDA) has been suggested as a method to support healthcare value discussions to drive the decision-making process. Evidence and Value: Impact on Decision Making (EVIDEM), a reflective MCDA framework, stimu-lates structured discussions among all stakeholders through an explicit set of quantitative and qualitative criteria. 7 This systematic, structured, objective and transparent process provides arguments for decision-making.8,9
The objective of this study was to determine the value of emicizumab for the prophylaxis of haemophilia A with inhibitors in Spain, compared to current therapeutic alternatives, through MCDA.
Methods
Study design
The current study analysed the value contribution of emicizumab (VE) compared to two drugs used in clinical practice in Spain through MCDA. Two evidence matrices were elaborated following the EVIDEM framework. Each criterion was rated by a panel of Spanish experts in haemophilia in an MCDA session in Madrid.
In Spain, comparators were selected according to the clinical practice for haemophilia A patients with inhibitors. According to the Spanish therapeutic guidance, prophylaxis might be of interest for patients with inhibitors, although it is not clear which agents should be used. 10 Activated prothrombin complex concentrate (aPCC) and rFVIIa were selected as comparators for the study. aPCC is indicated for the prophylaxis and treatment of haemorrhages in haemophilia A patients with FVIII inhibitors. 11 rFVIIa is indicated for episodic treatment and prevention of haemorrhages, 12 although it is used in the Spanish clinical practice for the prophylaxis of haemophilia A patients with inhibitors.13–15
EVIDEM framework
The EVIDEM framework adapted to orphan drugs was used. 16 The EVIDEM reflective framework stimulates structured reflection from stakeholders through a set of quantitative and qualitative criteria that integrate ethical underpinning of decision-making. 7 The used framework is composed of five quantitative domains with 10 criteria and of two contextual domains with 4 criteria. The quantitative domains are disease impact (disease severity and unmet needs), comparative outcomes of intervention (comparative effectiveness, comparative safety/tolerability and comparative patient-perceived health/patient-reported outcomes), type of benefit of intervention (type of therapeutic benefit), economic consequences from the intervention (comparative cost consequences–cost of intervention and comparative cost consequences–other medical costs) and knowledge of intervention (quality of evidence and expert consensus/clinical practice guidelines). The contextual domains are normative contextual criteria (population priorities and access, and common goal and specific interests) and feasibility contextual criteria (system capacity and appro-priate use of intervention, and opportunity costs and affordability). 17
Evidence on emicizumab and the comparators: systematic literature review
A systematic literature review was conducted to retrieve information for each of the criteria assessed in EVIDEM evidence matrix. Clinical data were obtained from Summary of Product Characteristics (SMPC), European Public Assessment Reports (EPAR) and Therapeutic Positioning Report (TPR) from the Spanish Agency of Medicinal Products and Medical Devices (AEMPS). In addition, evidence was also obtained from a biomedical literature database (PubMed), clinical trial registries, published hospital evaluation reports, national clinical practice guidelines, and national and regional health plans and strategies. Additional sources were conference web-sites, scientific societies’ websites (Spanish Society of Hospital Pharmacy and Spanish Society of Haematology and Hemotherapy) and international and national haemo-philia organisations (World Federation of Haemophilia and Spanish Federation of Haemophilia).
Evidence was synthesised and reported in two indepen-dent MCDA Evidence Matrices for each of the comparators: emicizumab versus aPCC and emicizumab versus rFVIIa. MCDA Evidence Matrices were developed following the methodology defined by the EVIDEM Collabora-tion Group, and evidence was reported in a descriptive, summarised and structured way for its subsequent revision.
Panel design and conduct
To collect insights from a broad range of perspectives, a multi-disciplinary panel of seven experts was invited to participate in a face-to-face MCDA session in Madrid. Experts were chosen according to their professional profile and experience with haemophilia patients and drug evaluation: one decision maker (J.L.T), one hospital manager (R.P.-S), two pharmacists (J.L.P., J.A.R.), two haematologists (M.T.Á.-R, A.S.) and one patient representative (anonymous) specialists in haemophilia. The session was chaired by an expert on MCDA (X.B.) who was responsible for training the experts on MCDA, providing a detailed explanation of the exercise, coordinating the session, and analysing and presenting the results.
Criteria weighting and scoring
The EVIDEM framework adapted to orphan drugs and weighted by 98 Spanish regional evaluators was used. 18 Scoring was performed considering the information presented in the MCDA Evidence Matrices. Quantitative criteria were scored using an ordinal scale that ranged from 1 to 5 for non-comparative and from −5 to 5 for comparative criteria. Contextual criteria were scored in a scale from −1 to 1 depending on the negative, neutral and positive impact of the intervention to each contextual criterion. 17
Data analysis
Data were collected individually and transferred to a common database to analyse and discuss the results with the expert panel. Data analysis was run in Microsoft Excel. The emicizumab value scores were analysed in the usual scale for each criterion. A descriptive analysis of the value of each criterion was conducted separately. The mean, standard deviation (SD) and range of minimum and maximum scores were calculated.
The analysis of the VE in each quantitative criterion (VC x ) was calculated as the product of its weight (Wx) and its normalised scores (Sx) provided by the panel of experts.
The overall MCDA VE was calculated as the sum of all partial value contributions of each of the quantitative criteria, with the maximum value being +1 and the minimum −1
The evaluation of qualitative criteria was performed on a qualitative scale, defined as positive, neutral or negative impact. The scoring of these options was transformed into a numerical scale corresponding to +1 point when the impact was considered positive, 0 point when the impact was considered neutral and −1 point when the impact was considered negative. The scores were adjusted to be presented as percentages that represented the percentage of experts who would consider the impact of emicizumab as positive, neutral or negative given the Spanish context.
Results
Scores for emicizumab
The quantitative criteria of the evidence matrices of emicizumab versus comparators were rated, and results were discussed by the panel of experts. The estimated mean score, SD, median and range (minimum and maximum) for each of the analysed quantitative criteria of emicizumab compared to aPCC and rFVIIa are shown in Figure 1.
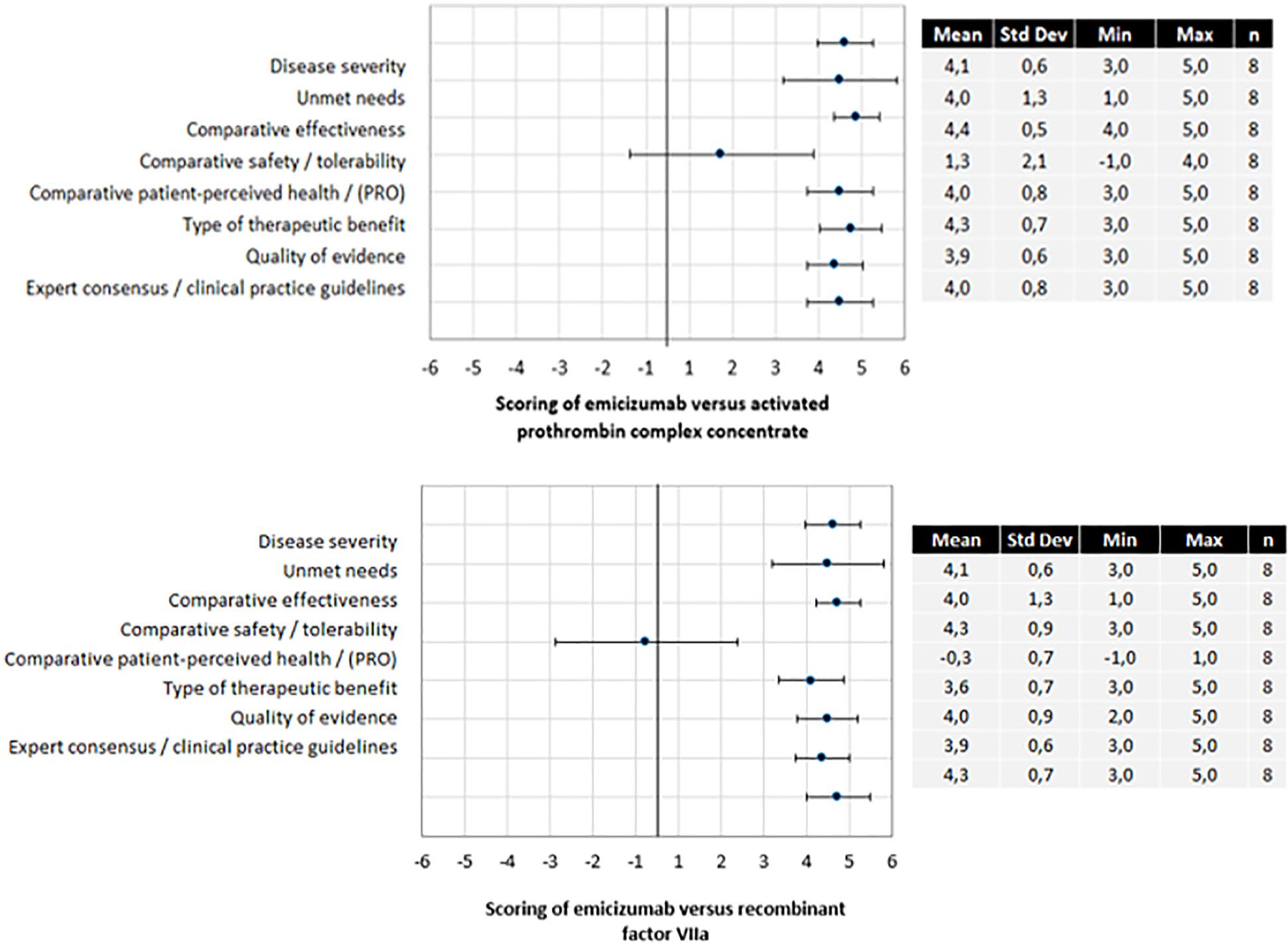
Results of the quantitative criteria scoring of emicizumab compared to aPCC and rFVIIa.
Experts perceived haemophilia A with inhibitors as a disease of high severity (mean ± SD: 4.1 ± 0.6) and with important unmet needs (4.0 ± 1.3), especially when compared with haemophilia A without inhibitors.
The highest mean score with the highest consensus (in the case of the comparison between emicizumab and aPCC) was given to higher efficacy of emicizumab versus aPCC and rFVIIa (4.4 ± 0.5 emicizumab/aPCC and 4.3 ± 0.9 emicizumab/rFVIIa). Relative patient-reported outcomes also scored high (4 ± 0.8 emicizumab/aPCC and 3.6 ± 0.7 emicizumab/rFVIIa). When emicizumab was administered alone for the prevention of bleeding episodes, comparative safety and tolerability were perceived as slightly better than aPCC (1.3 ± 2.1) and similar to rFVIIa (–0.3 ± 0.7).
According to the experts, emicizumab adds higher therapeutic value than the comparators because it might produce a change in the clinical course of the disease (4.3 ± 0.7 emicizumab/aPCC and 4.0 ± 0.9 emicizumab/rFVIIa).
Quality of evidence of emicizumab was considered positive (3.9 ± 0.6), meaning that the pivotal trials and studies from which evidence was obtained were considered adequate. Panellists considered that emicizumab should be included in the clinical practice guidelines as soon as possible (4 ± 0.8).
Value estimates for emicizumab
Criteria scoring was weighted by the relative importance of each criterion to estimate the overall VE versus the comparators, which is shown in Figure 2. It was observed that efficacy, disease severity and therapeutic benefit were the criteria that contributed the most to the value of emicizumab. Comparative criteria represented 46% of the VE compared to aPCC and 42% compared to rFVIIa. All the experts agreed that emicizumab showed better efficacy than the current alternatives because it demonstrated a significant reduction in the treated bleeding events. Emicizumab could also improve quality of life of the patients due to its weekly subcutaneous administration. Experts said that this would have a positive impact on patient’s life, especially for paediatric patients and their families. Adults would also benefit because their working life would be less affected due to the decrease in hospital visits. Regarding the safety and tolerability criterion, when emicizumab was administered alone for the prevention of bleeding episodes, it was perceived as slightly better than aPCC and similar to rFVIIa. Experts discussed that although the adverse events are non-serious when emicizumab is administered alone, long-term efficacy is still unknown. Some serious adverse events were reported when emicizumab was used in conjunction with aPCC. 6 Emicizumab was also perceived to have a high therapeutic benefit because it would change the clinical course of the disease by decreasing the number of treated bleeding events that would reduce morbidity.
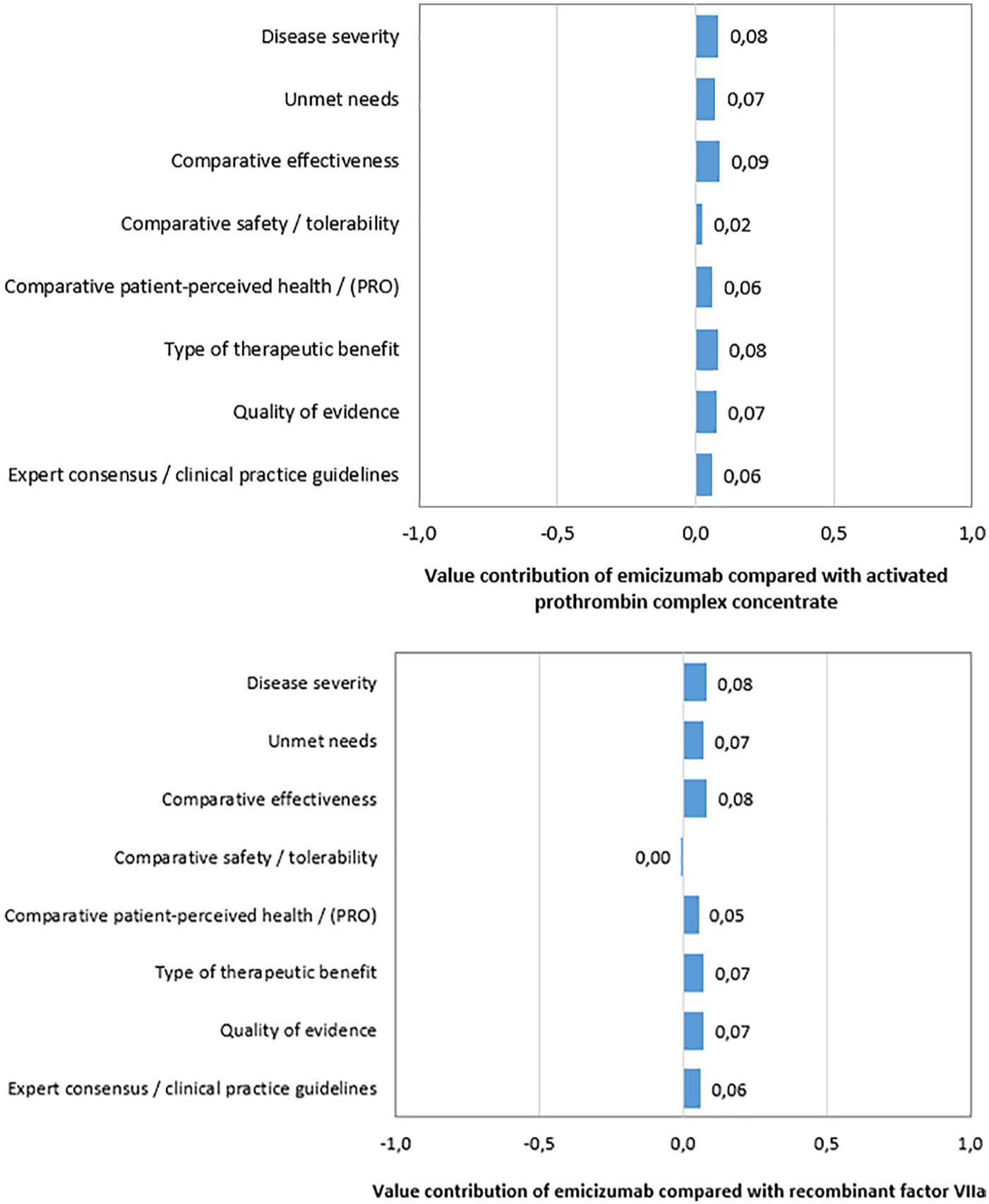
Results of the value contribution of emicizumab compared to aPCC and rFVIIa.
Non-comparative criterion represented 54% of the VE compared to aPCC and 58% compared to rFVIIa. Haemophilia A was perceived as a severe disease with important unmet needs, especially for patients who develop inhibitors. Quality of evidence was perceived positive because the clinical trial included a direct comparison between emicizumab and bypassing agents. Finally, although emicizumab is still not included in current clinical practice guidelines, expert consensus criterion was valued positively, and experts recommended its inclusion in the clinical practice guidelines.
Contextual criteria
Figure 3 shows the percentage of experts who consider the impact of emicizumab as positive, neutral or negative to the Spanish context. The population priorities and access criterion had a positive impact on emicizumab’s value, and all stakeholders perceived that the treatment was in line with the interest and objectives of the Spanish National Health System (NHS) because it targets a specific group of patients with a rare disease and high unmet clinical needs. Seventy-five percent of the panellists also agreed that the system’s capacity was adequate to implement and ensure the appropriate use of emicizumab because it does not require additional resources compared with the current alternative treatment options. Finally, the common goal and specific interests criterion was also perceived as positive by 88% of the panellists because no stakeholders’ barriers were identified.
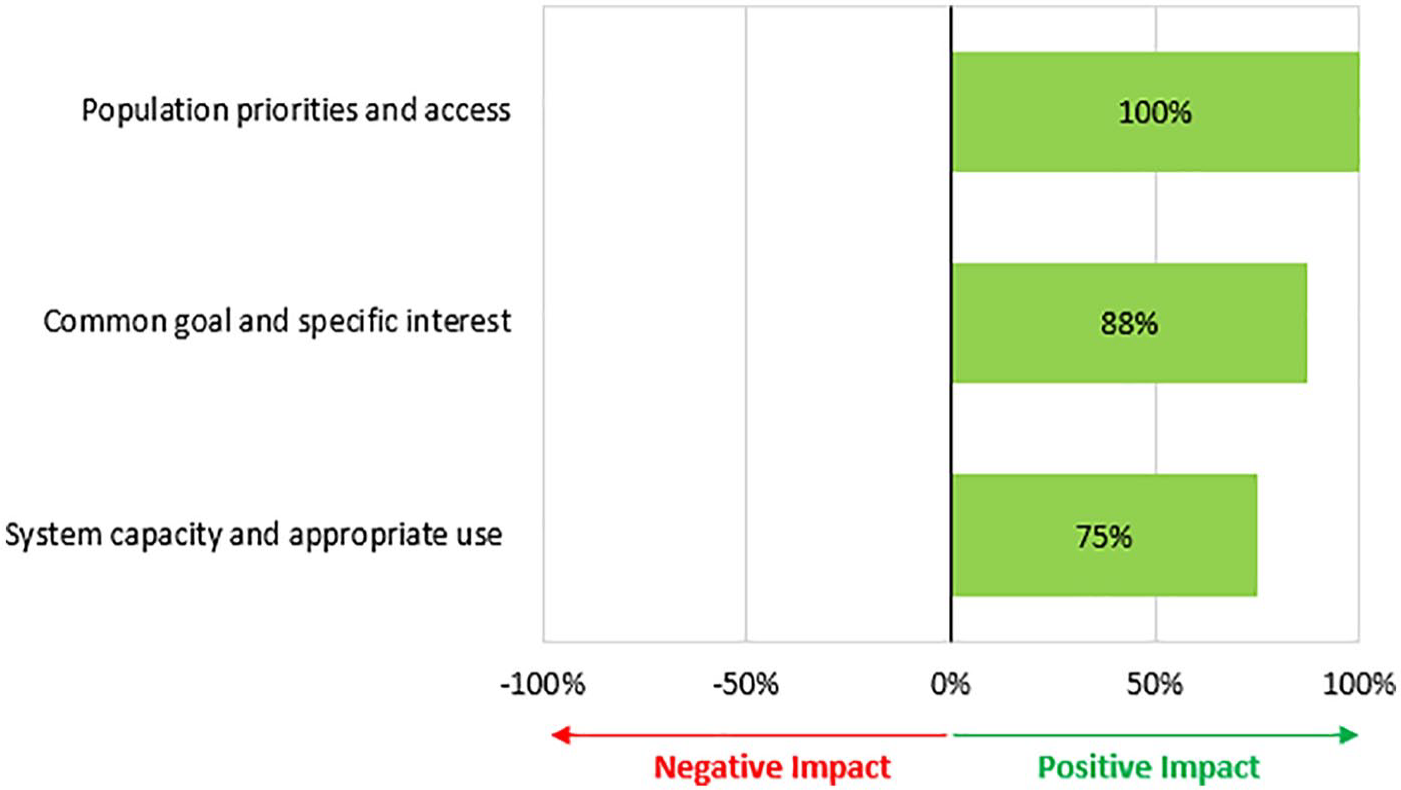
Percentage of experts who would consider the impact of emicizumab as positive, neutral or negative.
Discussion
The aim of the study was to assess the VE, a new medicine for prophylaxis of haemophilia A with inhibitors, compared to the current therapeutic alternatives in clinical practice in Spain using reflective MCDA. Haemophilia A was perceived as a severe disease with high unmet needs, especially for patients who develop inhibitors. The results obtained suggested that emicizumab might be an intervention of high added value because it was perceived to have greater efficacy and therapeutic benefit than aPCC and rFVIIa. Patient’s quality of life could also be improved because it has a perceived better posology than the current alternatives, and this would favour the conciliation between the everyday life and the disease. When emicizumab was administered alone, it was perceived to have slightly better safety profile than aPCC and was similar to rFVIIa. Quality of evidence and its inclusion in the clinical practice guidelines were also valued positively. Contextual criteria were valued positively by most of the stakeholders. Emicizumab was in line with the objectives and interests of the Spanish NHS because it targets a rare disease and a high unmet population, and it would not require additional organisational, training and surveillance resources for its implementation. Therefore, priority access should be given to emicizumab, especially for those patients with the highest unmet needs.
The cost of emicizumab was not formally assessed because at the time of the study, there were no real data available about emicizumab’s costs. Instead, the cost of intervention criterion was valued in an exercise using two hypothetical costs for emicizumab: the first one 20% higher than the cost of aPCC and the second one 50% higher. The other medical costs criterion assumed that emicizumab’s expenditures were lower than the comparators because it has shown greater reduction in treating bleeding events and has better posology than the current alternatives. Emicizumab’s cost of intervention criterion was perceived positively compared to rFVIIa and similar compared to aPCC. Other medical costs criterion was valued positively in both cases. Results from this exercise have not been presented because they would have over- or under-estimated the overall VE. It would be interesting to re-score the evidence matrices when real costs data become available to see how the overall VE would be affected.
MCDA methodology has been widely used in non-health-related areas. 19 Healthcare decision-making is characterised by its complexity due to the number of factors that need to be considered. Therefore, it is understandable that MCDA is increasingly becoming popular for supporting healthcare decision-making. 19 At the European level, the Benefit-risk methodology project developed by the European Medicines Agency (EMA) aimed to develop and test different tools and processes for balancing multiple risks and benefits, which could be used as an aid to inform science-based regulatory decisions about medicinal products. MCDA methodology was identified as the most useful tool. 20 This study showed that MCDA methodology might contribute to improve decision-making as it helps to consider different stakeholders’ perspectives and enhances reflective discussion among them. For example, in the workshop, the comparative safety and tolerability criterion had the lowest consensus among experts. MCDA contributed to understand and discuss the results obtained. In this case, some experts valued emicizumab positively because its adverse events were perceived as non-serious, while others perceived it like the comparators because long-term efficacy is still unknown.
An example of the use of MCDA to assess the value of a drug can be found in Italy where a study was performed to assess the performance of obinutuzumab for rituximab-refractory indolent non-Hodgkin lymphoma (iNHL) using MCDA. As in the present study, the EVIDEM framework was also used to estimate the overall total score which was 0.45. 21 In Spain, some studies using MCDA methodology have also been performed. An international study was performed with the aim to analyse the contribution of a range of criteria to the value of lenvatinib for radioiodine refractory differentiated thyroid cancer (RR-DTC) in country-specific contexts (France, Italy and Spain). The overall total scores estimated in Spain ranged from 0.33 to 0.38. 22 Recently, a study was performed in Spain to assess the value of dupilumab for severe atopic dermatitis compared with secukinumab for moderate-to-severe plaque psoriasis using MCDA. The EVIDEM framework was also used in this study, although an online re-test for the weights and scores was performed. The overall total scores estimated ranged from 0.39 to 0.51. 23 In the studies presented, the overall total scores were lower than the ones found in the present study, highlighting the high added VE. All studies highlighted the value of MCDA as a useful tool for improving decision-making.
The study is not exempt from some limitations. First, although weighting results might be representative at the national level (n = 98), the scoring results might be limited by the small sample of experts who formed the panel (n = 8). Nevertheless, the number of experts was in accordance with other MCDA studies,22,23 and it was very similar to the number of experts who form evaluation commissions in Spain. In addition, the clinical practice of haemophilia A is very concentrated in Spain. Therefore, experts were representative of the knowledge of haemophilia A in Spain. Second, even though an introduction on MCDA and a detailed description of the exercise were explained before the session started, misinterpretations of evidence or of a scoring scale might have occurred. Consequently, results might not reflect stakeholders’ preferences. Finally, as other MCDA studies, the information presented in the evidence matrices was limited by the information available at the time of the study. Therefore, results might change when new data on effectiveness become available.
Conclusion
Emicizumab adds higher therapeutic value compared to aPCC and rFVIIa for the treatment of haemophilia A with inhibitors, and it also satisfies important unmet needs such as higher efficacy and better quality of life. To the knowledge of the authors, this is the first MCDA that estimates the value of a treatment for haemophilia A with inhibitors. Reflective MCDA has proven to be a feasible method to determine the value contribution of comparative therapies in haemophilia, although future research would be needed to continue developing MCDA methodology to value new treatments to help healthcare decision-making.
Footnotes
Acknowledgements
We would like to thank the anonymous referees who helped us with their comments to improve the paper.
Author contributions
All authors contributed equally to this work. All authors read and approved the final manuscript.
Availability of data and material
The evidence matrices used and/or analysed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: J.L.P., R.P.-S, J.A.R. and I.C.-A. stated that they had no interests. J.L.T. and A.S. have received reimbursement for participating in an advisory board with Roche. M.T.Á.-R. has received reimbursement for speaking and participating in advisory boards with Takeda, Pfizer, Novonordisk, Roche, Sobi, Novartis, CSL-Behring, Amgen and LFB. X.B. and M.T. have acted as paid consultant to Hoffmann-La Roche.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Hoffmann-La Roche.