Abstract
Current treatment options for chronic endometriosis-associated pain are often inadequate, resulting in a rise in opioid consumption in this population. Given its analgesic, anti-inflammatory, and anti-depressive properties, esketamine may be a promising alternative treatment option. While esketamine is globally used in the treatment of chronic pain conditions, no clinical trials have been conducted to investigate the efficacy of esketamine infusion to alleviate chronic endometriosis-associated (pelvic) pain. Therefore, the objective of this double-blinded randomized controlled trial (RCT) is to assess the efficacy of 8-h infusion with esketamine versus placebo (saline) to reduce chronic endometriosis-associated pain symptoms. Secondary study objectives include pain scores, quality of life, depressive symptoms, health care utilization, productivity loss, cost-effectiveness, side effects, treatment experience, and pain coping and cognition. Premenopausal women (aged ⩾18 years) diagnosed with endometriosis (peritoneal, ovarian, deep, or adenomyosis), with an indication for laparoscopic endometriosis resection surgery and who suffer from chronic pelvic pain (NRS score ⩾6) will be eligible for inclusion. In total, we aim to include 56 participants, with a follow-up of 12 weeks. Digital questionnaires will be sent at baseline and 4, 8, and 12 weeks after the day of infusion.
Trial registration number
The EASYlight study is registered as “EASYlight-NEK” in the Clinical Trials Register (NCT06161805)
Introduction
The burden of endometriosis is high, affecting 1 in 10 women of reproductive age, of which 60% report to suffer from chronic pelvic pain.1,2 Current treatment options consist of two main strategies: conservative treatment (analgesics and/or hormonal therapy) and laparoscopic resection surgery. 3 However, the efficacy of these interventions are highly individual and side effects may require patients to discontinue hormonal treatment. 3 In addition, chronic pain symptoms frequently persists after resection of endometriosis lesions, even in the absence of lesion regeneration, suggesting a complex pain pathophysiology beyond the presence of endometriosis lesions alone.4,5 Consequently, women continue to suffer from chronic pain, resulting in a significant reduction in quality of life, social participation, sexual intimacy, and mental health, with increased rates of anxiety and depression.5–7 As a result, an increased risk for chronic opioid use, dependence, and overdose in women with endometriosis is observed. 8 In addition, these pain symptoms impair the ability to work, resulting in an high annual economic burden of €9,579 per woman, of which 66% is attributed to productivity loss. 9 Hence, there is an urgent need for safe, alternative, long-term pain treatments for endometriosis patients resistant to current treatment options. Esketamine, a non- selective N-methyl-D-aspartate receptor (NMDAR), has analgesic, anti-inflammatory, and anti-depressive properties. 10 This makes esketamine a promising treatment option for patients with chronic endometriosis-induced pain.11,12
Esketamine is the left-handed optical isomer of ketamine and exhibits higher anesthetic and analgesic potency due to its high affinity to the NMDAR. 13 Initially introduced in the 1970s as an anesthetic, (es)ketamine’s applications have since rapidly expanded to other clinical domains, including patients with therapy-resistant depression and patients suffering from chronic pain.14,15 Previous studies demonstrate long-term pain reduction after (es)ketamine infusion therapy in patients with chronic regional pain syndrome (CRPS). 16 A retrospective cohort study among 17 patients with chronic-endometriosis-associated pain who are resistant to current treatment, showed that esketamine infusion therapy had a positive effect on pain symptoms in 65% of patients. Only mild side-effects were observed during infusion. 12 However, high quality evidence supporting the efficacy of esketamine to alleviate chronic pelvic pain in endometriosis patients is lacking, and therefore this topic has been prioritized as a knowledge gap by the Dutch Society of Obstetrics and Gynecology. 17
This randomized controlled trial aims to investigate the efficacy of 8-h esketamine infusion treatment to alleviate chronic pain symptoms in patients with chronic endometriosis-associated pain.
Material and methods
This protocol outlines a double-blinded RCT that will be conducted in one (candidate) level-2 endometriosis expertise center (NEK, RdGG) in the Netherlands. 18 In addition, all members of the Dutch Endometriosis Special Interest Group (including gynecologists who are experts in endometriosis care in the Netherlands) and the Dutch patient federation “Endometriose Stichting” will be informed about the EASYlight study. Women who are eligible can be referred to participate in the trial. This study is registered as “EASYlight-NEK” in the Clinical Trials Register (NCT06161805). The Medical Ethics Committee Leiden The Hague Delft has approved this study (EU CT-number: 2023-508417-16-00).
Study population
Inclusion criteria
• Pre-menopausal women ⩾18 years
• Diagnosed with endometriosis (ultrasound, MRI, or previous laparoscopic, and/or diagnostic surgery) according to the #Enzian classification 19 . This means that endometriosis is present in the following compartments:
- Rectovaginal space (minimal A1) and/or
- Sacro uterine ligaments, cardinal ligaments, pelvic sidewall (minimal B1) and/or
- Rectum (minimal C1) and/or
- Endometriosis of the intestines, diaphragm and/or
- Adenomyosis (according to the MUSA criteria or evident adenomyosis on the MRI)20,21 and/or
- Peritoneal/superficial endometriosis (diagnosed laparoscopically and not treated during surgery)
• Resistant to current recommended lines of analgesics (paracetamol and NSAIDs) 3
• No usage of strong opioids (discontinued usage for more than 1 week prior to inclusion)
• Mild to severe chronic pelvic pain (NRS scale ⩾6)
• Eligible for endometriosis resection surgery or on the waiting list for surgical treatment
• Stable usage of hormonal therapy (no alteration within 1 month prior to inclusion)
• Able to read and understand Dutch
Exclusion criteria
• Endometriosis affecting the bladder and/or ureter
• Increased intracranial pressure
• Poorly regulated hypertension, defined as >180/100 mmHg at rest
• Patients with hyperthyroidism, malignancy, glaucoma, liver disease, epilepsy
• History of psychiatric illness (schizophrenia, psychosis, delirium, manic depression)
• Usage of xanthine derivatives or ergometrine
• Unstable angina, heart failure, history of cerebral vascular accident
• Patients suffering from an active infection
• Patients trying to achieve pregnancy and or patients who are breastfeeding
• Alcohol or drug abuse
• Patient with a known (es)ketamine allergy
• Abnormal liver enzyme levels at baseline (ASAT, ALAT, уGT, AP, Bilirubin total)
Outcomes
Primary outcome
The primary outcome is the efficacy of an 8-h esketamine infusion on chronic pelvic pain symptoms, measured by the change in the Numeric Rating Scale (NRS) score on a scale from 0 (no pain) to 10 (worst pain imaginable), pre- and 4 weeks post-treatment. Chronic pelvic pain is defined as non-menstrual pain in the pelvic region that is present for at least 3 months, more than 50% of the time, causing functional disability and requiring medical or surgical treatment.22,23
Secondary outcomes
The secondary outcomes are listed in Table 1. All patient reported outcome measures (PROMs) will be digitally sent via Castor EDC® at baseline and post-infusion. One subdomain of the EHP-30 questionnaire on sexual functioning (five questions) will also be included.
Secondary outcomes.

EHP-30: endometriosis health profile-30; EPF: electronic patient file; EQ-5D-5L: EuroQql five-dimensional five levels; HADS: Hospital Anxiety and Depression Scale; iPCQ: Productivity Costs Questionnaire; iMCQ: Medical Cost Questionnaire; NRS: numeric rating scale; PCCL: pain coping and cognition list.
NRS scores for dysmenorrhea, dyschezia, dysuria, dyspareunia, chronic pelvic pain.
Aspartate transaminase (ASAT), Alanine transaminase (ALAT), Gamma-glutamyl transferase (yGT), alkaline phosphatase (ALP), bilirubin-total.
Figure 1 outlines the study timeline. Eligible participants will be informed about the EASYlight-study by their treating gynecologist. Those expressing interest to participate will receive written information about the EASYlight-study, an informed consent form, a return envelope and a pregnancy test. If recent blood pressure and/or liver enzymes (within the last 12 months at the time of inclusion) are unavailable, participants will undergo blood pressure assessment and will be requested to undergo blood sampling for liver enzyme profile evaluation. Following a minimum 3-day interval from receiving the study information, the researcher contacts potential participants to address questions and confirm their willingness to participate. If the potential participant meets the inclusion criteria, expresses willingness to participate, and exhibits no abnormalities in liver enzyme profile and blood pressure measurement, they will be invited to sign the informed consent form. Randomization between esketamine or placebo (saline) will be performed using Castor EDC® in a 1:1 ratio with non-stratified permuted blocks (4-6). Treatment allocation remains undisclosed for participants and the research team until the trial is finished and the database is locked.
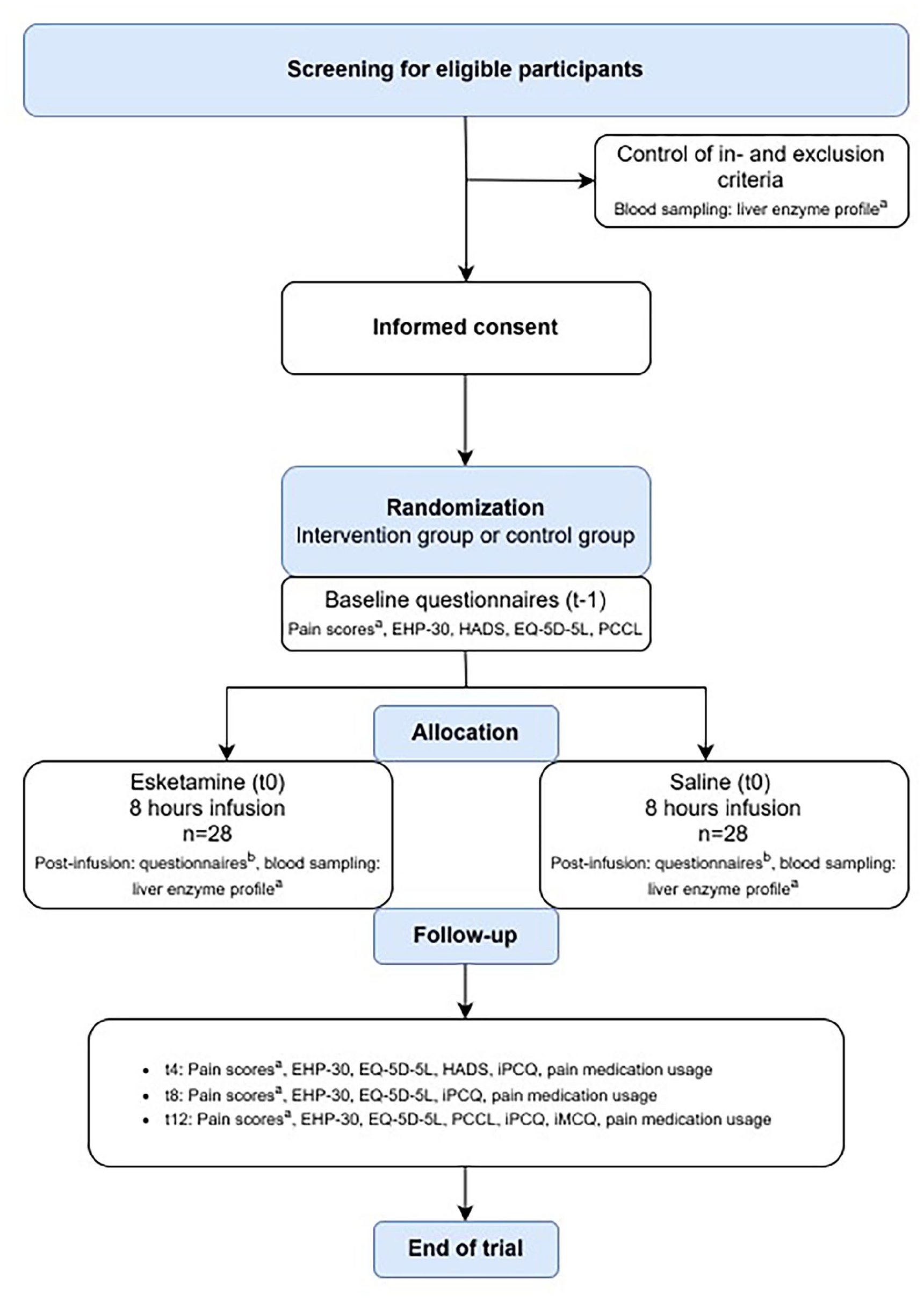
Flow diagram and study schedule. Time in weeks.
On the day of infusion, four participants will be treated simultaneously. The infusion may proceed only if a negative pregnancy test result was obtained on the day of (or preceding) the infusion. Study medication dosing regimen is set at 0.1 mg/kg/h as starting dosage. Dosage will be gradually increased based on heart rate, oxygen saturation, blood pressure, and potential side effects during a period of 8 h to a maximum of 0.5 mg/kg/h. Initially, the dosage will be increased in steps of 0.1 mg/kg/h every 30 min until 0.3 mg/kg/h. If side-effects are absent and the analgesic effect remains inadequate, the dosage will be further increased to a maximum of 0.5 mg/kg/h. This esketamine dosage regimen is similar to that used earlier by Sigtermans et al. 24 The following vital parameters will be measured during treatment: oxygen saturation, heart rate, and blood pressure. Oxygen saturation and heart rate will be continuously monitored (via a pulse oximeter). Blood pressure will be measured every half h while the dose is increased. Once the maximum dosage has been reached and the vital parameters have demonstrated stability, the monitoring of blood pressure will be performed every 2 h. In case side effects occur (e.g. psychedelic symptoms, nausea, and tachycardia), esketamine infusion will be (temporarily) stopped and/or the infusion dosage will be lowered. Nausea will be prevented or treated by administrating a 5-HT3 antagonist. Following the infusion treatment, participants will be observed for 30 min. Due to the potential (reversible) hepatotoxic effect of esketamine, blood will be drawn directly after the infusion to determine the liver enzyme profile. 14 The follow-up period will be 12 weeks, which is based on a previous study that showed significantly lower pain scores over an 11-week period in patients receiving ketamine versus those patients receiving placebo [1]. Digital questionnaires will be sent prior infusion (at baseline) and 4, 8, and 12 weeks post-infusion using Castor EDC® (Table 1, Figure 1). On the day of infusion, the following questionnaires will be administrated on paper: NRS scores for pain (after infusion), Bowdle questionnaire (prior and 7 h post-treatment initiation), treatment experience and allocation questionnaires.
Sample size
This study will be powered to detect a 2-point reduction in NRS score for chronic pelvic pain in the esketamine group versus the placebo group at week 4 post infusion treatment. This difference in NRS score is based on a previous trial, which reported a 2-point decrease in NRS score for non-menstrual pelvic pain as clinically meaningful. 25 Based on a power of 0.90, alpha of 0.05 (two-sided) and a standard deviation of 2.0, we calculated that 22 patients will be needed per group. Assuming that some patients will be lost during follow-up (potentially due to a scheduled surgery within the 12-week follow-up period), we plan to enroll 28 patients per group. Statistical analysis will include all patients, according to the intention-to-treat principle. In total, 56 patients will be included in the EASYlight-study.
Statistical analysis
Once all patient information and questionnaires are completed, the data will be locked and the treatment allocation will be unblinded. No interim analysis will be conducted. Descriptive statistics will be used to determine the frequency distributions of variables at baseline. We will describe data as means with standard deviations (SDs) or medians and inter-quartile ranges (IRQs), dependent on observed distributions. For dichotomous data, we will provide proportions (or percentages). Linear mixed models will be used to assess the effect of the treatment group on the NRS score for chronic pelvic pain. This model will include outcome data collected post-infusion treatment as dependent variables. All patients with at least one measurement of pain scores post-infusion will be included. Fixed effects will include randomization treatment allocation, follow-up time points (4, 8, or 12 weeks, categorical), the interaction between treatment and time point and the baseline chronic pelvic pain score (NRS score). Correlations between repeated measurements of the same patient will be modelled using a repeated effect with a correlation structure chosen based on fit of the acquired data (using Akaike Information Criterion). The primary outcome will be estimated as the difference in mean chronic pelvic pain (adjusted for the baseline value of chronic pelvic pain) at week four post-infusion between the esketamine group and the placebo group together with its 95%-confidence interval. All secondary outcomes will be described using a longitudinal mean plot and analyzed using repeated-measure linear mixed models, similar to the approach used for the analysis of the primary outcome. Treatment experience will be compared between treatment groups using T-test or Mann-Whitney U test, dependent on distribution. All reported side effects per study participant will be described. The latest version of SPSS will be used to analyze the results. A two-tailed p-value of <0.05 will be considered as statistically significant. Missing data (questionnaires) will be minimized through active monitoring and automatic reminders.
Data management
All data will be collected in Castor EDC®, a web-based clinical data management system. Data processing will be done according to the EU General Data Protection Regulation and the Dutch Act on implementation of the General Data Protection Regulation (in Dutch: Uitvoeringswet AVG). Analysis of the data will be performed with the coded data. The key that links the code to personal patient information will only be available to the local study team. Data will be stored for 25 years.
Cost-effectiveness
The economic evaluation will be performed as a trial-based cost-effectiveness analysis (CEA) and cost-utility analysis (CUA) from a societal perspective according to the Dutch guidelines. 26 For the CEA, the primary outcome parameter will be the cost per successful esketamine infusion treatment. Success is defined as a 2-point reduction in the NRS score for chronic pelvic pain compared to baseline (pre-infusion), assessed 4 weeks post-infusion. For the CUA, the primary outcome will be the cost per quality adjusted life-years based on the EQ-5D-5L scores, to provide data on the cost per QALY gained. The time horizon will include the period between the day of infusion and study endpoint (i.e. 12 weeks). Multiple imputation will be used to deal with missing data. Differences in mean costs and effects (QALYs) between strategies (esketamine or placebo) will be compared with bootstrapping using 1000 replications. In a net-benefit analysis, costs will be related to QALYs and presented in a cost-effectiveness acceptability curve. In a cost-effectiveness acceptability curve the probability of cost-effectiveness for esketamine infusion treatment compared to placebo will be shown for different values of the willingness to pay, including the Dutch threshold values ranging between 20,000 and 80,000 euro per QALY. 27 Sensitivity analyses will be performed to evaluate the robustness of our results. These will include analyses from a healthcare sector perspective and using EQ-VAS utilities instead of EQ-5D-5L utilities. Analyses will be performed using Stata and Excel. Both health care costs (secondary and tertiary care) and cost of loss of productivity for paid and unpaid work will be included. The iPCQ and the iMCQ questionnaires will be used to monitor absenteeism and presenteeism from paid and unpaid work and to monitor healthcare resource use (i.e. gynecology visits, emergency room visits, hospitalization, ambulance care). Healthcare utilization and productivity losses will be evaluated using cost prices from the Dutch costing manual. 28 Medication use will be valued with prices derived from www.medicijnkosten.nl.
Discussion
The EASYlight-study is the first randomized controlled trial to assess the efficacy of esketamine infusion therapy as treatment for chronic endometriosis-associated pain. In contrast to current treatment options, esketamine targets multiple key aspects of endometriosis (pain, inflammation, depression). However, its application in this population is relatively unexplored and this topic has been prioritized as a knowledge gap by the Dutch Society of Obstetrics and Gynecology. 17
The diagnostic interval of endometriosis is long, approximately 7 years. 29 During this interval, before adequate treatment is initiated, remodeling of pain pathways may occur. Moreover, chronic changes of neural processing regions in women with endometriosis-associated chronic pelvic pain have been demonstrated. 30 Endometriosis-associated pain can be nociceptive and/or neuropathic in nature. 31 Lesions may directly infiltrate and damage the surrounding somatosensory nervous system, resulting in neuropathic pain. 32 In addition, evidence suggests ongoing stimulation of nociceptive pain pathways in women with endometriosis, resulting in generalized hyperexcitability of the central nervous system. 33 A pro-nociceptive environment is triggered by active bleeding at the lesion sites, activating immune responses and the release of pain-producing agents (e.g. pro-inflammatory cytokines/chemokines) in the peritoneal fluid in women with endometriosis. 4 Prolonged nociceptive stimulation induces upregulation of the NMDAR, resulting in the amplification of pain signals transmitted to the brain. 14 Consequently, women with endometriosis may develop a state of chronic sensitization, whereby (the intensity of) pain perception becomes independent of an external stimulus. Furthermore, there is also a high risk for cross-organ sensitization, where pain is perceived from adjacent structures due to the convergence of neural pathways. 4 Together, this may also explain why disease classification poorly correlates with pain severity and anatomical locations of (deep) endometriosis lesions and the poor postsurgical pain relief in some women (11.8%) affected.4,34–36
The NMDAR plays a vital role in the process of central sensitization. 37 It is an excitatory glutamatergic receptor present at spinal and supraspinal sites and is involved in the afferent transmission of nociceptive signals. NMDAR antagonists, such as esketamine, can halt the excessive barrage of nociceptive afferent input to the brain (i.e. reverse the process chronic sensitization) and is therefore used to treat patients with chronic pain syndromes. At the moment, 31 pain clinics in the Netherlands use esketamine in the treatment of chronic pain (maximal doses of 27.5 mg/h (in an outpatient setting) and 25.0 mg/h (in an inpatient setting)). 38 In the current study, we expect that the optimal dosage for the most participants, free from significant side effects, will be around 0.3 mg/kg/h (or 21 mg/h for a 70-kg patient), which is similar to the dosage administrated in other pain clinics in the Netherlands. An 8-h infusion protocol was chosen due to the significant burden imposed on patients by a multiday protocol, the challenges associated with implementation of such a protocol in our healthcare system and perhaps a multiday protocol is unnecessary given the fact that various pain syndromes in the Netherlands are treated with only 8 h of esketamine infusion.11,12,38
In contrast to opioid consumption, esketamine infusion treatment can be strictly monitored. This is an important advantage, given the four times higher opioid consumption among women with endometriosis compared to non-endometriosis patients, resulting in a higher opioid dependency and potential addiction. 8 In addition, it can also be administrated in a home setting, enhancing cost efficiency.12,38 While multiple side effects have been observed in clinical trials including psychedelic symptoms, nausea/vomiting, cardiovascular side effects (e.g. tachycardia) and (reversible) hepatotoxicity, esketamine infusion treatment is safe when administrated in a clinical setting.12,39
Regarding other non-hormonal treatment options, multiple studies have investigated the effect of acupuncture, physiotherapy, Transcutaneous Electrical Nerve Stimulation (TENS), Chinese medicine, nutrition, exercise, or psychological interventions on (chronic) endometriosis-associated pain. 3 However, insufficient high-quality evidence is available to recommend any non-medical intervention as mono-treatment to reduce pain symptoms. 3 Nevertheless, in selected patients these interventions could be beneficial to enhance the quality of life.40–45
We hypothesize that esketamine infusion treatment could serve as an alternative treatment option for chronic endometriosis-associated pain when regular treatment options are ineffective. Future implementation could be to use esketamine infusion as a bridging therapy until surgery in case of severe pain symptoms. Theoretically, esketamine may also be implemented as maintenance therapy in strongly selected patients with pain symptoms not responding to current strategies, but outcomes on long-term repeated administration of esketamine infusions (i.e. development of pharmacodynamic, metabolic, and tolerance, resulting in tachyphylaxis and loss of analgesic effect) are not available and are warranted to ensure-long term safety and efficiency. 46 However, it is essential to first establish proof of principle on whether a single 8-h esketamine infusion treatment is efficient in reducing chronic pain symptoms within this patient population, which will be provided by this RCT.
Footnotes
Acknowledgements
The authors would like to thank Bianca De Bie (chair of the Dutch endometriosis patient federation) for her assistance in evaluating patient information and Nan van Geloven (bio-statistician), who was consulted for the sample size calculation.
Data availability statement
The dataset generated during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is funded by STIMAG, a Dutch foundation for innovative research to improve health care. This study is also facilitated by the scientific board of the Reinier de Graaf hospital. The esketamine in this study will be reimbursed by Eurocept International with no influence on the course or outcomes of the study. They were approached by our research team.
