Abstract
Background:
Evidence of overlap between endometriosis and chronic pain conditions is emerging; however, little is known about how the pain experience differs based on the presence or absence of endometriosis.
Objectives:
In a sample of women reporting chronic pelvic–abdominal pain (CPP), the aim of this study was to characterize differences in pain symptomatology between women with and without endometriosis and to examine the influence of chronic overlapping pain conditions (COPCs) on pain among these two groups.
Design:
This was a cross-sectional study, based on an online survey.
Methods:
Participants (aged 18+ years) completed a survey collecting pain diagnoses and symptoms assessing pelvic pain severity, pain interference, and pain impact. Independent sample t-tests, chi-square, and multiple linear regression models were employed to analyze group differences in pain symptomatology and COPCs.
Results:
Of the 525 respondents with CPP, 25% (n = 133) reported having endometriosis. Women with endometriosis were younger at the onset of pelvic pain, relative to women without endometriosis (p = 0.04). There were no differences in age, race, ethnicity, or duration of pelvic pain between women with and without endometriosis. Women with endometriosis reported higher pelvic pain severity (+0.8, 95% CI = 0.4–1.1), pain interference (+5.9, 95% CI = 2.4–9.3), and pain impact (+1.9, 95% CI = 0.8–2.9). Endometriosis was associated with a higher number of COPCs (p = 0.003), with 25% (n = 33) of women reporting ⩾3 overlapping pain conditions compared with 12% (n = 45) of those without endometriosis. Women with endometriosis had a higher frequency of fibromyalgia (p < 0.001), chronic fatigue syndrome (p < 0.001), and temporomandibular disorder (p = 0.001). The number of COPCs was associated with higher pain severity, interference, and impact, independently of endometriosis.
Conclusion:
Women with endometriosis experienced higher levels of pain-related burden and COPCs compared with those without endometriosis. Pain intensity, interference, and impact increased with a higher number of pain conditions regardless of endometriosis presence.
Plain Language Summary
The presence of endometriosis was associated with a higher number of chronic overlapping pain conditions (COPCs) and greater pain symptomatology, while a greater number of COPCs corresponded to increased pain burden among women with and without endometriosis. These findings underscore the need for a more comprehensive assessment of endometriosis that addresses the full experience of the disease, including its comorbidities. A greater characterization and measurement of COPCs has the potential to facilitate the development of tailored interventions for individuals with pain comorbidities, thereby contributing to improved clinical care strategies for endometriosis-related pain.
Introduction
Endometriosis is a multifactorial gynecological disease defined by the presence of endometrial-like tissue outside the uterus. In the United States, it is estimated that endometriosis affects 4 million reproductive-aged women, 1 with prevalence highest among women aged 30–40 years. 2 Common symptoms of endometriosis include bowel and bladder problems (e.g. painful defecation and urination), dysmenorrhea, non-cyclical pelvic pain, rectal bleeding, infertility, and menstrual dysfunction, with pain being the most burdensome symptom precipitating care. Due to diagnostic delays and poor treatment efficacy, endometriosis demonstrates widespread negative impacts on quality of life, social relationships, and education/career,3 –9 thereby placing women at greater risk for depression, 10 activity impairment,8,11 loss of work productivity,3,5,8,11 work absenteeism, 5 and poorer social well-being.11,12
Although endometriosis has predominantly been conceptualized as a peripherally mediated condition, mounting evidence suggests that factors other than peripheral tissue damage contribute to the generation of endometriosis-related pain. 13 Indeed, it is increasingly being recognized that endometriosis-associated pain is heterogeneous in nature with numerous overlapping causes and a high degree of co-prevalence with other pain conditions localized in multiple bodily areas—collectively referred to as chronic overlapping pain conditions (COPCs).14,15 COPCs constitute a group of pain disorders with shared underlying pathology and include the following conditions: endometriosis, vulvodynia, irritable bowel syndrome, temporomandibular disorder, chronic fatigue syndrome, interstitial cystitis/painful bladder syndrome, fibromyalgia, tension-type and/or migraine headache, and chronic low back pain. 16 Although the etiology of COPCs is not well understood, it is believed that alterations in central nervous system mechanisms likely contribute to pain in a subset of these disorders. 16 Several studies have found that a higher prevalence of multiple pain conditions is associated with poorer pain-related physical function, psychological distress, increased healthcare utilization, greater pain sensitivity, diminished health-related quality of life, and lower treatment effectiveness.17,18 While the prevalence of COPCs in endometriosis is unknown, recent estimates have found that COPCs are associated with a greater frequency and duration of pain episodes, higher intensity of pelvic pain, more frequent dysmenorrhea, and greater impairments in activities of daily living.14,19
Despite evidence supporting the impact of COPCs on disease trajectory and pain outcomes, the study of multiple pain conditions has received little empirical attention in endometriosis. Likewise, previous investigations in chronic pain have examined specific COPCs in isolation, thereby ignoring their potential additive effects on pain-related functioning. Given the added burden associated with co-occurring pain, examining the association between COPCs and endometriosis may be an important step toward improving clinical care as patients with multiple COPCs may require a wider spectrum of therapeutic targets for enhanced treatment efficacy.
To address this gap, this study leveraged a community-based sample of individuals reporting chronic pelvic–abdominal pain to characterize differences in pelvic pain severity, pain interference, and pain impact between women with and without endometriosis and to examine the incidence of COPCs among these two groups. We hypothesized that a large subset of individuals with endometriosis would exhibit greater pain-related burden (i.e. pelvic pain severity, interference, pain impact) and incidence of COPCs, relative to those without a diagnosis. In addition, we hypothesized that a higher number of overlapping pain conditions would be associated with greater adverse pain outcomes.
Materials and methods
Participants
Participants were recruited via a community-based sample using SurveyMonkey in June 2022. The survey was anonymous, free, and accessible through a direct clickable link, to minimize barriers to access. The web-based dissemination strategy included invitations advertised via the SurveyMonkey email listserv, and social media posts shared across provider- and patient-led organizations including the International Pelvic Pain Society (IPPS) and the Pelvic Pain Support Network. Participants were eligible to complete the survey if they were US citizens who were 18 years of age and older and reported experiencing pelvic and/or lower abdominal pain. For the purposes of this study, only individuals reporting pelvic and/or lower abdominal pain with a self-reported pain level of ⩾2 on a 0–10 numerical rating scale (NRS) were included in analyses. No additional exclusion criteria were present. A cutoff of 2 was selected to ensure the inclusion of participants with discernible pelvic pain.
Based on a power analysis, the necessary sample size to adequately represent the population of interest (i.e. individuals with pelvic–abdominal pain) was calculated to be 385 completed surveys. This sample size was calculated based on the published US prevalence of chronic pelvic pain in the general population, which is approximately 20% or 20,000,000. 20 Sample size was calculated using a 20% expected response rate, 5% margin of error, and 95% confidence intervals (CI). To ensure sufficient responses, it was determined that a minimum of 1925 surveys would need to be distributed to yield a required sample size of 385.
Procedures
Ethics approval was provided by the University of Central Florida Institutional Review Board (IRB#00012110) and all participants provided written informed consent prior to study procedures. This online survey (Supplementary File 1) was developed based on a previous survey designed to capture women’s experiences with endometriosis including symptoms, diagnosis, and the impact of endometriosis-related pain on functioning and quality of life. 7 Items in this survey collected information on various conditions recognized for their potential contribution to pelvic pain, extending beyond the scope of endometriosis.
Participants completed general demographic questions (i.e. age, sex, race/ethnicity), followed by a series of items querying on the presence or absence of pelvic pain, duration of pelvic pain, age of pelvic pain onset, diagnosis of overlapping pain conditions (i.e. endometriosis, vulvodynia, irritable bowel syndrome, temporomandibular disorder, chronic fatigue syndrome, interstitial cystitis/painful bladder syndrome, fibromyalgia, migraine or chronic headaches, and chronic low back pain), and pain characteristics (i.e. pain severity and interference, pain impact). Survey questions were closed-ended with multiple-choice options. Participants were allowed to omit any questionnaire items that they felt uncomfortable responding to. Prior to dissemination, the survey was tested with four anonymous participants to ensure that the questions were clear and relevant (i.e. the questions gathered the intended information about pelvic pain).
Pain outcome measures
Pain severity
Pain severity was measured by having participants rate their average pelvic pain using a 0–10 NRS (0 = no pain, 10 = worst imaginable pain). The NRS is considered the gold standard for pain evaluation, has high reliability and validity, 21 and provides sufficient discriminative power for patients to describe their pain intensity. 22
Pain interference
Pain interference was measured using the short version of the Brief Pain Inventory (BPI). 23 The BPI is a validated instrument and consists of seven items and measures the influence of pain on daily functioning in the past 7 days (i.e. general activity, mood, walking activity, normal activity outside the home or with housework, relations with other people, sleep, and enjoyment of life). Response options include 0 (does not interfere) to 10 (completely interferes). The pain interference scale was summed for a total score ranging from 0 to 70, with higher scores indicating a greater degree of interference from pain. The BPI has acceptable internal consistency and reliability 23 and demonstrated high reliability in this sample (α = 0.92).
Pain impact
Drawing from our prior research on endometriosis, respondents completed a 5-item questionnaire developed for this study. Questionnaire items were crafted based on qualitative data derived from women experiencing endometriosis-associated pelvic pain. 7 Items were modified in this study to query on the impact of “pelvic pain” on social life, education/career, ability to start or raise a family, sexual intercourse, and interactions with healthcare professionals. The following statements comprise the questionnaire, “I live my life as usual, as if I did not have pain,” “My pelvic pain disrupts my education/career,” “My pelvic pain interferes with my ability to start or raise a family,” “I avoid or have to stop sexual intercourse due to my pelvic pain,” and “I received conflicting information from different healthcare professionals about my pelvic pain.” Responses were provided using a 5-point scale ranging from 1 (strongly disagree) to 5 (strongly agree). Total scores range from 5 to 25, with higher scores indicating greater pelvic pain burden. In this sample, Cronbach’s alpha was 0.72.
Statistical analysis
Continuous measures were summarized as means and standard deviations and categorical measures were summarized as counts and percentages. Independent sample t-tests (reporting t-test statistics and degrees of freedom) were used to compare pain severity, pain interference, and pain impact between women with and without endometriosis. Chi-squared tests were used to assess differences in the frequencies of COPCs between women with and without endometriosis, as well as differences in patient characteristics. Multiple linear regression analysis (reporting F-test statistics and degrees of freedom) was used to examine the association between number of COPCs, with models also including endometriosis as a fixed effect and an interaction term between endometriosis and COPCs. R-squared terms were calculated to assess the proportion of variation in the study outcomes predicted by the statistical models. Normality assumptions were evaluated with graphical analyses, utilizing visual inspection of Q-Q plots. All analyses were conducted in SAS JMP Pro 17 (SAS Institute Inc, Cary, NC) and significance level was set at p < 0.05 (2-tailed).
Results
Participant characteristics
Demographic and clinical characteristics of the study sample are provided in Table 1. The study sample comprised 1221 individuals who responded to the survey, of which 779 self-identified as female. Of the 779 females, 254 individuals reported no pelvic or lower abdominal pain, leaving 525 participants in this study. Approximately one-quarter (n = 133) of the sample reported having endometriosis, while 75% did not have endometriosis (n = 392). Of the total sample, a majority (n = 357, 68.2%) of women were younger than 40 years and 59.2% (n = 311) self-reported White race. Over one-third (n = 192, 36.9%) reported having their pain for more than 4 years, with pain starting at age 29 years or younger for 68.3% (n = 358) of women in the sample. For age when pelvic pain started (χ2 = 13.4, df = 6, p = 0.04), women with endometriosis tended to be younger at the onset of pelvic pain; 78.2% (n = 104/133) of women with endometriosis had pelvic pain onset before 30 years of age compared with 65% (n = 254/391) of women without endometriosis. There were no statistically significant differences in age (χ2 = 7.2, df = 5, p = 0.26), race/ethnicity (χ2 = 8.0, df = 7, p = 0.33), or duration of pelvic pain (χ2 = 10.3, df = 5, p = 0.06) between women with and without endometriosis. Missing sociodemographic data include the following: current age (n = 1), age of pelvic pain onset (n = 1), and duration of pelvic pain (n = 5).
Sample characteristics.
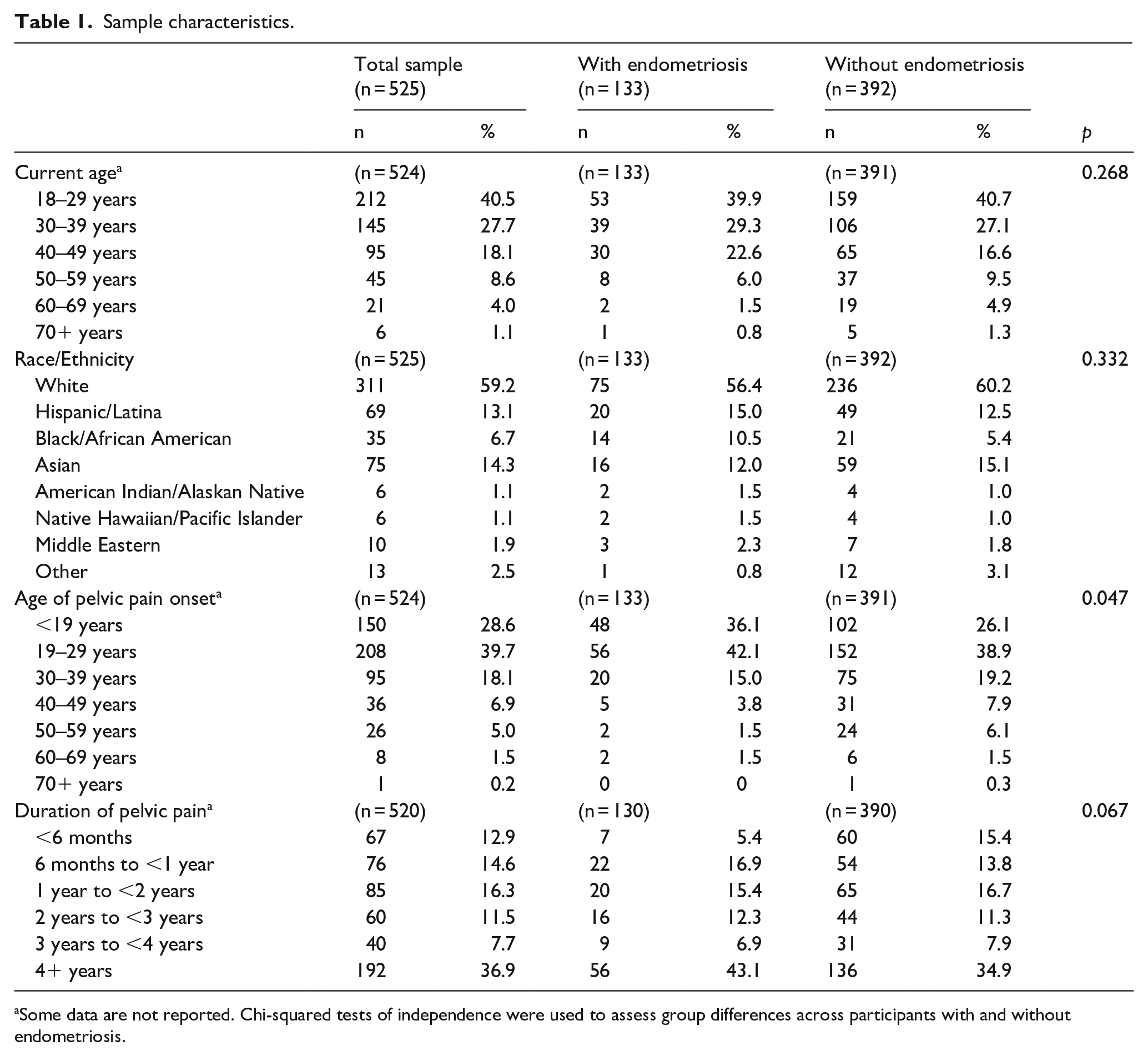
Some data are not reported. Chi-squared tests of independence were used to assess group differences across participants with and without endometriosis.
Group differences in pain outcomes
Group differences were observed for pelvic pain severity (t[520] = 3.9, p < 0.001), pain interference (t[520] = 3.4, p < 0.001), and pain impact (t[520] = 3.4, p < 0.001). When compared with women with pelvic–abdominal pain not associated with endometriosis, women with endometriosis reported significantly greater pain severity (+0.8, 95% CI = 0.4–1.1), pain interference (+5.9, 95% CI = 2.4–9.3), and pain impact (+1.9, 95% CI = 0.8–2.9), translating to 16% higher pain severity, 25% higher pain interference, and 15% higher pain impact (Figure 1).

(a) Mean differences in pain severity, (b) pain interference, and (c) pain impact between women with and without endometriosis. Error bars indicate 95% confidence intervals. Means ± SDs are reported for each group.
Group differences in COPCs
Endometriosis was associated with a higher number of COPCs (χ2 = 15.9, df = 4, p = 0.003). One-quarter (25%, n = 33) of women with endometriosis reported three or more overlapping pain conditions relative to 12% of women with pelvic–abdominal pain not associated with endometriosis (Figure 2). When comparing across individual conditions, women with endometriosis had significantly higher rates of chronic fatigue syndrome (χ2 = 18.0, df = 1, p < 0.001), fibromyalgia (χ2 = 14.8, df = 1, p < 0.001), and temporomandibular disorders (χ2 = 10.7, df = 1, p = 0.001) (Figure 3).

Differences in the proportion of women with overlapping pain conditions (total) between women with and without endometriosis.

Differences in the proportion of women with overlapping pain conditions (across each condition) between women with and without endometriosis.
Table 2 represents associations between the number of COPCs and pain outcomes across groups. An increase in the number of COPCs was significantly associated with greater pelvic pain severity (F(4,512) = 7.4, p < 0.001), pain interference (F(4,512) = 18.0, p < 0.001), and pain impact (F(4,512) = 10.3, p < 0.001). No statistically significant (p > 0.05) interactions emerged between endometriosis and COPCs, signifying that the association between pain outcomes and the number of overlapping pain conditions was independent of endometriosis diagnosis. R-squared values for multiple linear regression models were as follows: pain severity (R2 = 0.10), pain interference (R2 = 0.19), and pain impact (R2 = 0.15).
Mean differences in pelvic pain severity, pain interference, and pain impact across the number of overlapping pain conditions, stratified between women with and without endometriosis.
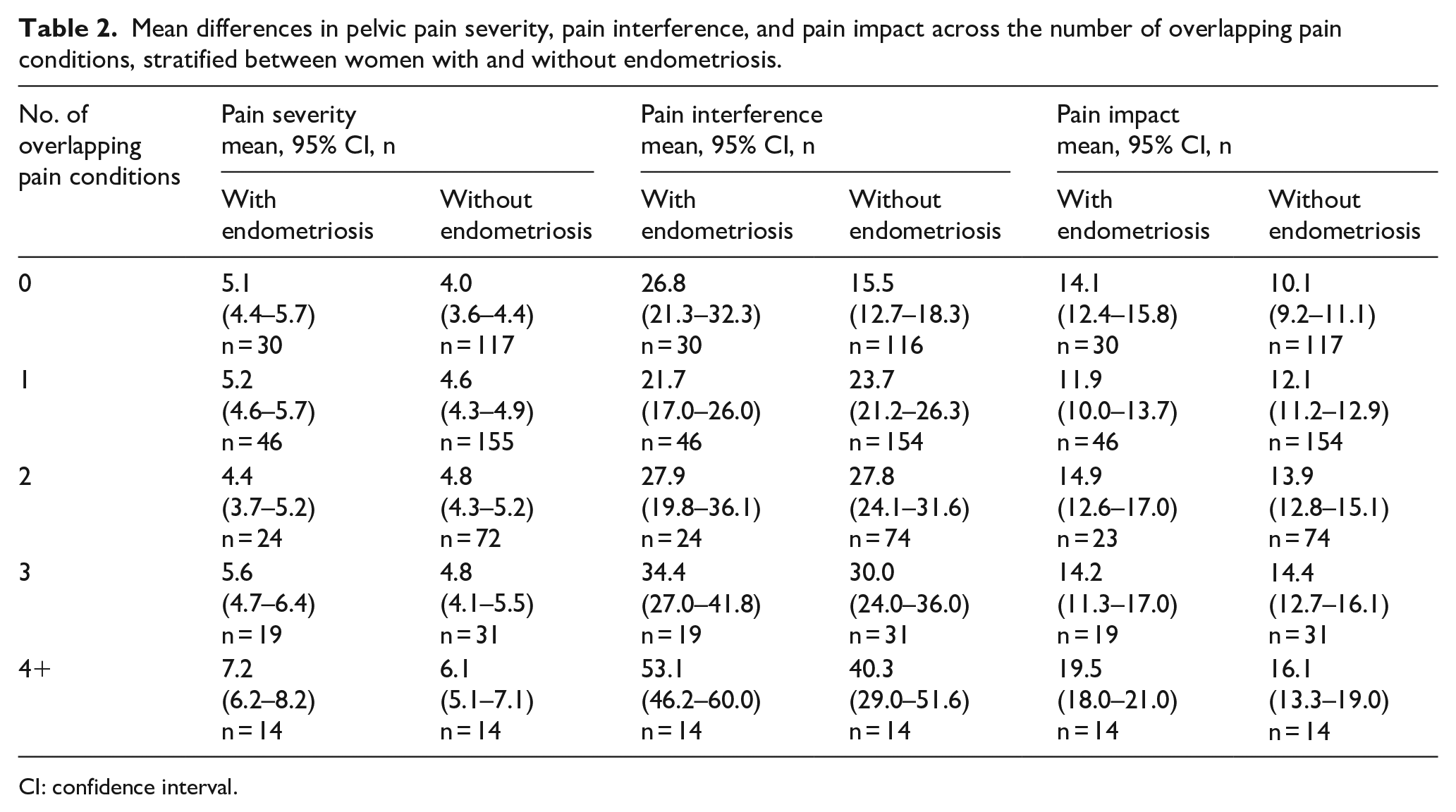
CI: confidence interval.
Discussion
Endometriosis is associated with considerable psychological and physical burden that is in part attributable to the high incidence of pain that accompanies the disease. Although evidence suggests high comorbidity between endometriosis and functional and/or pathological diseases,24,25 few studies have examined the role of co-occurring pain conditions on pain and disability-related outcomes in endometriosis.19,26 Through a large community-based sample of individuals with chronic pelvic–abdominal pain, this study investigated the relationships between pelvic pain severity, pain interference, and pain impact between women with and without endometriosis and examined the frequency of COPCs among these two groups.
In line with the existing literature, our results signify that endometriosis is associated with high pain burden. 4 In particular, women with endometriosis exhibited a higher severity of pelvic pain symptomatology when compared with women reporting pelvic–abdominal pain without endometriosis. Aligning with these findings, Evans et al. 26 found that women with comorbid endometriosis and dysmenorrhea exhibited menstrual and pain-related symptoms of greater severity and higher functional pain disability, as compared with women with dysmenorrhea alone. Notably, we observed that endometriosis was associated with a greater prevalence of COPCs, with approximately 25% of women with endometriosis having three or more pain conditions, compared with only 12% of women without endometriosis. Indeed, previous reports estimate that 65% of women with endometriosis have three or more health comorbidities at the time of diagnosis, with comorbidity burden increasing to 85% 2 years later. 25 Similarly, based on a sample of 510 women with endometriosis, Leuenberger et al. 19 found that 52% experienced endometriosis-related chronic pain with approximately half also experiencing co-occurring pain conditions (i.e. migraine/headache, back pain, stomach pain, inflammatory bowel disease). Those with concomitant pain diagnoses reported higher pain frequencies, longer pain episodes, and greater pain severity. Comparable rates of comorbidity have also been observed across other pain conditions (e.g. temporomandibular disorder, fibromyalgia, migraine headache, rheumatic diseases, low back pain), with incidence of comorbidity associated with adverse outcomes including increased pain severity and interference, psychological distress, poorer social and physical functioning, heightened pain sensitivity, and greater healthcare utilization.16,18,27 –31
While the pathophysiological mechanisms underlying comorbid pain among endometriosis are not fully understood, there is a growing body of evidence signifying shared underlying mechanisms linking COPCs together, with peripheral and central sensitization playing a role in the incidence and amplification of pain. While endometriosis has largely been treated as a peripherally mediated condition, an expansive literature suggests that the severity of pain symptoms and the morphological characteristics of the disease are only modestly correlated.32 –34 Such discrepancy between symptom severity and anatomic disease burden suggests that endometriosis may have a multifactorial etiology that not only includes both peripheral modulators, but also central mechanisms that augment pain and nociceptive input. Indeed, several lines of evidence support the presence of central sensitization in endometriosis35 –37 as observed by local (i.e. pelvic sites) and widespread pain sensitivity,35,38 –40 and decreased brain volume in areas associated with pain perception, relative to individuals without pain. 41 Importantly, symptoms of centralized pain are associated with greater pelvic pain severity in endometriosis,36,37 findings which have also been observed across other pain conditions.42,43
Interestingly, we found that a higher number of COPCs was associated with greater pelvic pain severity, interference, and pain impact, findings which were irrespective of endometriosis diagnosis. This aligns with prior work demonstrating that with an increased number of pain diagnoses, symptoms become more severe. 44 As pain becomes more diffuse, it likely interferes with sexual function and physical and social activities (e.g. exercise, activities of daily living, social engagement), effects which may have negative downstream effects on psychological health. Indeed, patients with multiple pain comorbidities report higher levels of anxiety and depressive symptoms, as well as lower quality of life and higher healthcare use—all of which may reflect the overall burden that multimorbidity has on patient functioning. 45 Nonetheless, our findings suggest that the presence of COPCs may help identify patients more likely to be adversely impacted by pelvic pain, and in particular, endometriosis.
Clinical implications
Findings from this study have important clinical relevance. Our results support the consideration of COPCs in the assessment and management of endometriosis. Traditional medical therapies often rely on treatments that suppress local or systemic estrogen levels to reduce endometrial lesions; however, significant variability exists in the degree of pain relief achieved from medical therapy; up to one-fourth of women remain symptomatic during treatment, and 59% of women report persistent pain after therapy cessation with 34% of patients reporting pain recurrence. 46 Furthermore, recurrent pain affects 30%–50% of patients undergoing surgical ablation or resection. 47
Lack of treatment response may be due, in part, to the predominant targeting of the peripheral nervous system and failure to consider the extent to which centrally mediated factors impact pain. Individual treatments that specifically target endometriosis pain and suppression of lesion proliferation overlook comorbid pain conditions which contribute to the pain experience, an effect which may explain why so many women are refractory to treatment. Overall, the results of our study suggest that COPCs may warrant systemic evaluation in endometriosis to advance clinical care. Women with overlapping pain conditions may require a comprehensive multimodal approach with a broader spectrum of therapeutic targets that extend beyond medical and surgical management. For instance, psychotherapeutic approaches (e.g. cognitive–behavioral therapy) that address the emotional burden of pain and dysfunctional cognitive beliefs (e.g. pain catastrophizing, pain uncontrollability) may be particularly beneficial for endometriosis, 48 as would treatment of pelvic floor dysfunction through physical therapy. 49 Furthermore, integrative and complementary health therapies such as yoga, acupuncture, and mindfulness-based approaches50 –52 have shown promise in mitigating endometriosis-related pain. Given that chronic pain is heavily influenced by biopsychosocial factors, treatments that are more holistic in nature, and include a combination of both pharmacological and non-pharmacological therapies, are likely to derive the most benefit for individuals with COPCs.
Limitations
There are some limitations that merit acknowledgment. First, given the nature of the survey design, responses were based on self-report; therefore, it was not possible to confirm the diagnosis of individual COPCs, including endometriosis. Future studies are warranted that include confirmation by physician diagnosis or the use of validated diagnostic medical screeners. Second, the cross-sectional nature of this study prohibits causal inferences regarding the associations between endometriosis, COPCs, and pelvic pain symptomatology. Third, we used a convenience sample recruited online which may not accurately represent the target population and limit generalizability. Fourth, the presence of central sensitization was not ascertained in this sample, thus we cannot confirm whether the prevalence of COPCs in our endometriosis sample is linked to symptoms reflective of centralized pain. Furthermore, our study population primarily consisted of adults who were White/Caucasian, thus future studies with more representation across diverse samples are needed. And finally, our study outcomes were limited to measures of pain and disability. Future research is needed to determine how COPCs impact other areas of functioning including psychological and social health. These limitations notwithstanding, our study boasts several strengths including a large sample size and we are one of the first, to our knowledge, to examine COPCs and their impact on pain outcomes in endometriosis. The present study provides a foundation for future research to explore the role of COPCs in the clinical management of endometriosis.
Conclusion
In summary, the presence of endometriosis was associated with a higher number of COPCs and greater pain symptomatology, while a greater number of COPCs corresponded to increased pain burden among women with and without endometriosis. These findings underscore the need for a more comprehensive assessment of endometriosis that addresses the full experience of the disease, including its comorbidities. A greater characterization and measurement of COPCs has the potential to facilitate the development of tailored interventions for individuals with pain comorbidities, thereby contributing to improved clinical care strategies for endometriosis-related pain.
Supplemental Material
sj-rtf-1-whe-10.1177_17455057241248017 – Supplemental material for Presence of endometriosis and chronic overlapping pain conditions negatively impacts the pain experience in women with chronic pelvic–abdominal pain: A cross-sectional survey
Supplemental material, sj-rtf-1-whe-10.1177_17455057241248017 for Presence of endometriosis and chronic overlapping pain conditions negatively impacts the pain experience in women with chronic pelvic–abdominal pain: A cross-sectional survey by Emily J Bartley, Meryl J Alappattu, Kelsey Manko, Hannah Lewis, Terrie Vasilopoulos and Georgine Lamvu in Women’s Health
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
