Abstract
Introduction:
Esketamine infusion therapy is currently used to reduce the process of sensitization in patients with chronic pain syndromes. Given its anti-inflammatory, anti-depressive, and analgesic properties, this drug could be effective to treat chronic endometriosis-associated pain as well. The aim of this study was (1) to provide an overview of the available literature and (2) to present the results of our retrospective cohort study on the efficacy of esketamine infusion therapy to alleviate pain symptoms in this cohort.
Methods:
PubMed and EMBASE were searched to select publications on this topic. For our retrospective cohort study, all patients with chronic endometriosis-associated pain that underwent esketamine infusion therapy between January 2010 and 2023 in the Isala hospital in Zwolle, the Netherlands, were reviewed.
Results:
PubMed and EMBASE identified no publications on this topic, except our own case report. Retrospective analysis of 17 patients (who underwent 57 esketamine infusions in total) revealed a positive effect in 64.7% of patients. Overall the treatment was considered save and well-tolerated. Currently, eight patients continue to receive esketamine infusion therapy, others underwent endometriosis surgery (n = 2), switched pain treatment (n = 1), or were non-responders (n = 6).
Conclusions:
Esketamine infusion therapy could be a potential and a viable option to treat chronic endometriosis-associated pain in patients who are resistant to current treatment options. Our findings encourage to conduct future studies, including randomized trials. This is the first cohort study to report outcomes on the efficacy of esketamine infusion therapy for women with chronic endometriosis-associated pain.
Introduction
Endometriosis is an estrogen-dependent, chronic condition and affects approximately 10% of reproductive-aged women. 1 It is characterized by extra uterine endometrial-like glands, inducing chronic inflammation and adhesion formation which often results in subfertility and pain. The most common pain symptoms are dyspareunia, dysmenorrhea, dyschezia, dysuria, and chronic pelvic pain, of which the latter is reported by more than 60% of women with endometriosis.2,3 Diagnosis is complex as symptom presentation is non-specific and indeed, a poor relation is observed between pain symptoms and disease severity. 4 This results in misdiagnosis and a significant diagnostic delay (e.g. 7.4 years in the Netherlands). 5 Additionally, the combination of this long diagnostic interval and the misunderstood pain symptoms results in a significant reduction in quality of life, social participation, sexual intimacy, mental health, and work productivity.1,6,7 Consequently, the socio-economic burden associated with this condition is high, calculated at €9579 per woman annually. The majority of these costs are attributed to productivity loss (66%), while health care costs account for 33%. 8 In terms of mental quality of life, women with endometriosis are more prone to develop depression or anxiety disorders compared to women without endometriosis with a hazard ratio of 1.5 and 1.4, respectively. 9
Endometriosis-associated pain can be nociceptive and/or neuropathic in origin. 10 Endometriotic lesions may directly infiltrate and damage the surrounding somatosensory nervous system, resulting in neuropathic pain. 11 In addition, evidence suggests that there is continued stimulation of nociceptive pain pathways in these patients, resulting in generalized hyperexcitability of the central nervous system. 12 A pro-nociceptive environment is triggered by active bleeding at the lesion sites, activating immune responses and the release of pain-producing agents (e.g. pro-inflammatory cytokines/chemokines) in the peritoneal fluid in women with endometriosis. 13 Prolonged nociceptive stimulation induces upregulation of the N-methyl-D-aspartate (NMDA) receptor, resulting in the amplification of pain signals transmitted to the brain. 14 Consequently, women with endometriosis may develop a state of chronic sensitization, whereby pain perception becomes independent of an external stimulus. This may explain why disease classification poorly correlates with pain severity and anatomical locations of (deep) endometriosis lesions and why pain symptoms often persist despite lesion resection.4,13,15 Furthermore, the incidence of other comorbidities characterized by sensory dysfunction (e.g. irritable bowel syndrome) is higher among women with endometriosis compared to women without endometriosis. 16 As such, it is apparent that current treatment options (hormonal interventions or surgical removal of lesions), often fall short in effectively managing endometriosis-associated pain.
Esketamine is a non-selective NMDA receptor inhibitor and has non-opioid analgesic, anti-depressive, and anti-inflammatory effects. 17 Ketamine is a racemic mixture consisting of esketamine and arketamine. Esketamine is the left-handed optical isomer of ketamine and exhibits higher anesthetic and analgesic potency. This is attributed to its three to fourfold higher binding affinity for the NMDA receptor than arketamine (Ki = 0.3 µM and Ki = 1.4 µM). 18 Initially introduced in the 1970s as an anesthetic, (es)ketamine’s applications have since rapidly expanded to other clinical domains, including treatment of patients with treatment resistant depression and patients suffering from chronic pain. Previous studies demonstrate long-term pain reduction after (es)ketamine infusion therapy in patients with chronic regional pain syndrome (CRPS). 19 While esketamine treatment simultaneously targets multiple aspects of endometriosis (pain, inflammation, depression), its application in this group of patients is relatively unexplored and this topic has been prioritized as knowledge gap by the Dutch Society of Obstetrics and Gynecology. 20
We conducted a systematic literature search in order to provide an overview of the available literature on this topic. The primary aim of the retrospective cohort study was to investigate the efficacy of esketamine infusion therapy to reduce pain symptoms. Secondary aims were to assess treatment characteristics.
Material and methods
Literature search
We systematically searched two electronic literature databases (PubMed and EMBASE) to identify (pre)clinical studies on esketamine infusion treatment in endometriosis patients to alleviate (chronic) pain symptoms. The search strategy was developed in collaboration with an information specialists of the Walaeus Library of the Leiden University Medical Center. The literature search was performed on March 8, 2023. For our complete search strategy, see Supplemental Table 1. Case reports, case series, cohort studies, and randomized controlled trials were considered for inclusion. We excluded animal studies, in vitro studies, conference abstracts, commentaries, non-English articles, reviews, and all publications prior to 2010. In addition, a snowballing approach was used to explore publications on this topic prior to 2010. Subsequently, articles were initially screened based on title and abstract level, and subsequently at the full-text level. Inclusion criteria were (1) human studies including women diagnosed with endometriosis, (2) available full-text articles, (3) systematic administration (intravenous, subcutaneous, intraperitoneal, oral, intrathecal) of ketamine (racemic ketamine of the S-enantiomer) to alleviate pain symptoms. Publications that combined administration of (es)ketamine with another drug were also included. Two reviewers independently conducted the selection process (RdK and FvD). Discrepancies in opinion were resolved by consensus and when necessary a third reviewer (AT) was consulted.
Study design and patient selection
This single center retrospective cohort study was approved by the Medical Research Ethics Committee of the Isala clinics Zwolle (the Netherlands, protocol number 20230217). To identify eligible participants, an automatic search was conducted using the following diagnostic codes to filter the electronic patient record (EPR) system: (1) patients registered under a reimbursement code within the gynecology and obstetrics department (DBC 0307) and (2) patients that underwent intravenous esketamine infusion therapy in the Isala hospital (Zwolle, the Netherlands) between January 2010 and January 2023 (i.e. with EPRs that contained: “esketamine,” “esketamine infusion,” “parental administration”; ketamine or synonyms for esketamine (S-ketamine, S ketamine, es-ketamine, ketanest-S, esketiv, ketanest) were also included). Subsequently, each patient was individually screened for inclusion. Inclusion criteria were women diagnosed with endometriosis by means of an MRI, ultrasound, or previous laparoscopy that suffered from chronic pelvic pain for which they underwent esketamine infusion therapy.21,22 Chronic pelvic pain is defined as non-menstrual pain in the pelvic region of 6 months duration or longer, causing functional disability and requiring medical or surgical treatment.23,24 Women that received esketamine infusion therapy for other indications than endometriosis-associated pain were excluded.
Data collection
Two authors (RdK and FvD) performed retrospective data extraction from medical records according to a predefined case report form (CRF) which was created in the web-based data capture system Castor EDC. Variables of interest included (1) patients characteristics: endometriosis classification, comorbidities, surgical history, previous pain treatments and (2) esketamine infusion characteristics: dosage, treatment duration, side effects, treatment effects, treatment location.
Objectives
Primary objective
Given the retrospective nature of the study, our data was contingent on information documented in the EPR. We developed a three level classification system to be able to provide information on the efficacy of esketamine to reduce pain symptoms. We categorized our primary outcomes into the following groups:
Positive effect: the patient expressed pain relief during the treatment evaluation.
Questionable effect: the patient expressed uncertainty about the treatment effect of esketamine infusion.
No positive effect: the patient either did not experience any effect of the esketamine infusion treatment (non-responder), reported an increase in pain symptoms after the esketamine infusion treatment compared to the situation before, or expressed that the experienced side effects outweighed any potential positive effects.
Secondary objectives
The secondary objectives were to determine esketamine treatment characteristics (infusion time, esketamine dosage, treatment location), side effects and the impact of esketamine infusion on liver function (aspartate aminotransferase (ASAT), alanine aminotransferase (ALAT), bilirubin total, gamma-glutamyl transferase (γGT), alkaline phosphatase (AP)).
Esketamine infusion protocol
All patients were under treatment by an anesthesiologist specialized in pain management, who prescribed one or multiple treatment(s) (with a minimum interval of 12 weeks) to alleviate their chronic pain symptoms associated with endometriosis. Each patient received esketamine (Eurocept International, the Netherlands). The drug was intravenously administrated using an infusion pump. Initially, all patients received an 8-h infusion in the hospital. Both the starting and maximum infusion doses were adjusted according to the patients’ weight. The starting dose was 0.05 mg/kg/h. Subsequently the dose was gradually increased based on heart rate, oxygen saturation, blood pressure, and side effects (e.g. nausea or dissociative effects) to the maximum dosage of 0.1 mg/kg/h. Oxygen saturation and heart rate were continuously monitored (via a pulse oximeter). Blood pressure was measured every half hour while the dose was increased. Once the maximum dose was reached and the vital parameters remained stable, monitoring procedures were discontinued. In the event of unpleasant side effects (e.g. hallucinations, dizziness) the treatment dose could be reduced, or the infusion could be (temporarily) stopped. In case of nausea or vomiting an intravenous 5HT3 antagonist was administrated. Directly after the esketamine infusion, blood was drawn to determine liver function. After 30 min of observation, the patient was permitted to leave the hospital. They were not allowed to drive themselves.
In selected cases, infusion protocol was extended to 12, 24, 48, or 96 h to maximize the duration of pain symptom suppression. Another indication was to assess whether a longer infusion protocol would provide some relief in cases where minimal to no effect on pain relief was initially observed. Furthermore, if in-hospital administration of the primary esketamine infusion proceeded without complications and if the anesthesiologist deemed an outpatient setting appropriate, the patient was offered the option to undergo the next esketamine infusion at home under the supervision of a caregiver. An assessment by telephone was scheduled after the esketamine infusion with the anesthesiologist or nurse practitioner for treatment evaluation.
Statistics
The distribution of data was analyzed with the Shapiro-Wilk test. Continuous variables are presented as mean (standard deviation) or median (interquartile range), depending on their distribution. Categorical variables are presented as numbers (percentages). Descriptive analysis was performed using SPSS version 29.0.
Results
Literature search
No data on the efficacy of esketamine infusion therapy in the treatment of chronic endometriosis-associated pain has been published to date, except our own case report. 25 For further details on the results obtained from our search, please refer to the Supplemental Table 1.
Retrospective cohort study
The automatic search of EPR yielded 270 patients in total. Subsequently, the EPRs of these patients were screened for eligibility, resulting in 17 patients who met the inclusion criteria. An overview of the inclusion process is given in Supplemental Figure 1.
The study population included women between 17 and 50 years with a mean age of 35 (Table 1). At the time of the first esketamine infusion, the majority of patients (70.6%) were found to have comorbidities, including sleep apnea, migraine, hip enthesopathy, pelvic instability, whiplash, chronic headache, depression, psychoses, PTSS, and/or burnout. The psychological diagnoses were predominantly historical, with most patients no longer experiencing active symptoms. Furthermore, the majority of patients were diagnosed with deep infiltrating endometriosis (DE) and had previously attempted multiple hormonal treatments, undergone endometriosis surgery, and frequently utilized opioids to manage pain symptoms. All baseline characteristics can be visualized in Table 1.
Baseline characteristics.
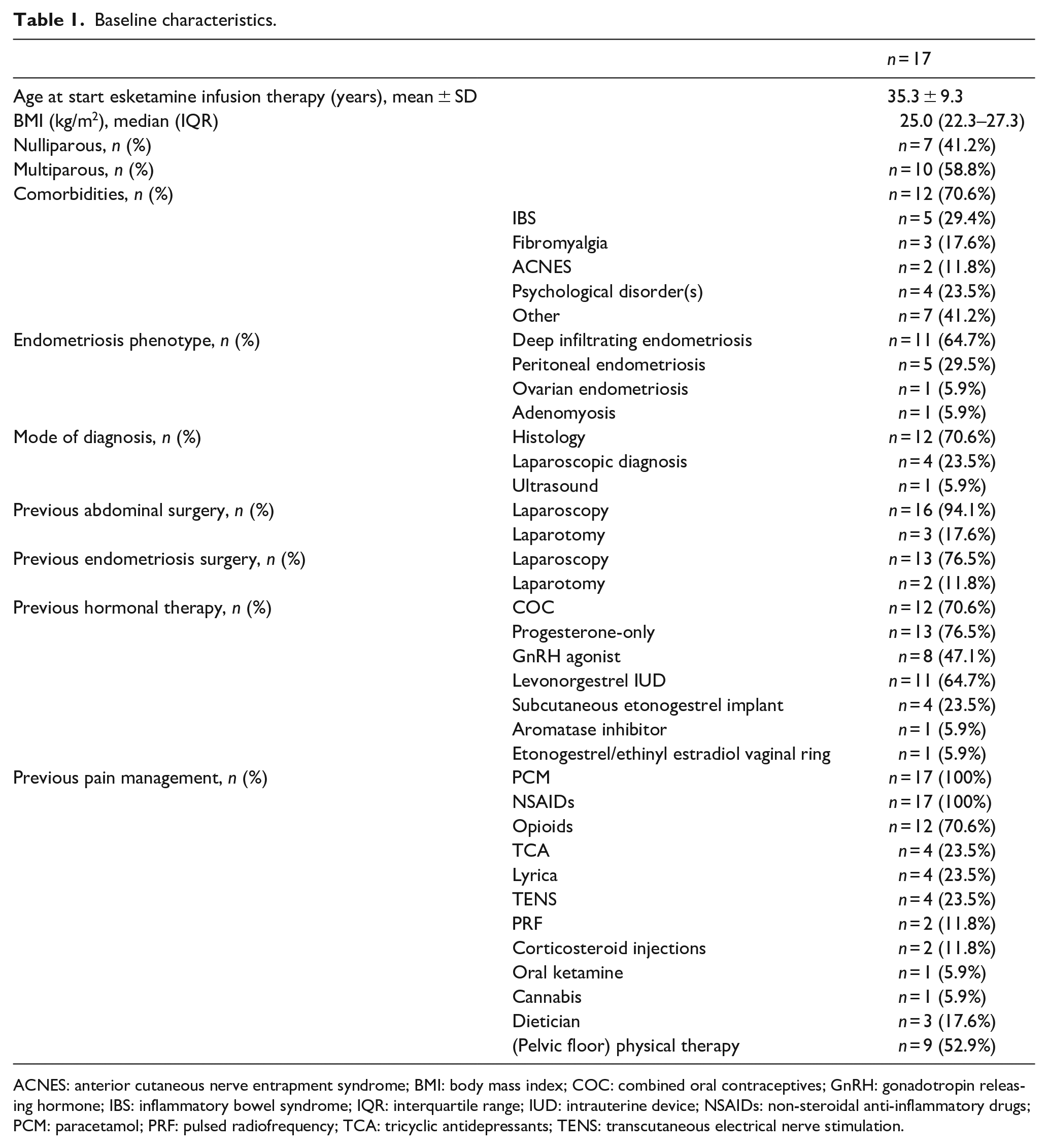
ACNES: anterior cutaneous nerve entrapment syndrome; BMI: body mass index; COC: combined oral contraceptives; GnRH: gonadotropin releasing hormone; IBS: inflammatory bowel syndrome; IQR: interquartile range; IUD: intrauterine device; NSAIDs: non-steroidal anti-inflammatory drugs; PCM: paracetamol; PRF: pulsed radiofrequency; TCA: tricyclic antidepressants; TENS: transcutaneous electrical nerve stimulation.
The 17 patients collectively received a total of 57 esketamine infusions, with infusion durations ranging from 8 to 96 h (Table 2). Among the infusions that endured 24 h, 69.2% were administrated in a home setting. On average, an interval of 3.1 months was scheduled between each infusion session. An overview of esketamine dose (mg) per hour per patient can be visualized in Supplemental Figure 2. Mild side effects were reported during the majority of infusions (Table 2). There were two instances where the infusion had to be temporarily halted in two separate patients. In one case, the infusion was briefly interrupted due to constriction in the infusion line. After reconfiguring the infusion line, the patient experienced dizziness, which was possibly attributed to a relatively higher rate of infusion upon re-infusion (Figure 1, patient 1). Consequently, the infusion was paused for 30 min and then resumed. Another patient experienced hallucinations and exhibited blurred speech (Figure 1, patient 16). Consequently, her infusion was temporarily halted and subsequently resumed at a lower dosage.
Esketamine infusion therapy characteristics.

IQR: interquartile range; PCM: paracetamol; PRF: pulsed radiofrequency.
One missing date, one patient excluded due to two non sequential treatments (76 months between both treatments). bASAT, ALAT, γGT, AP, Bilirubin total. cFive missing values.
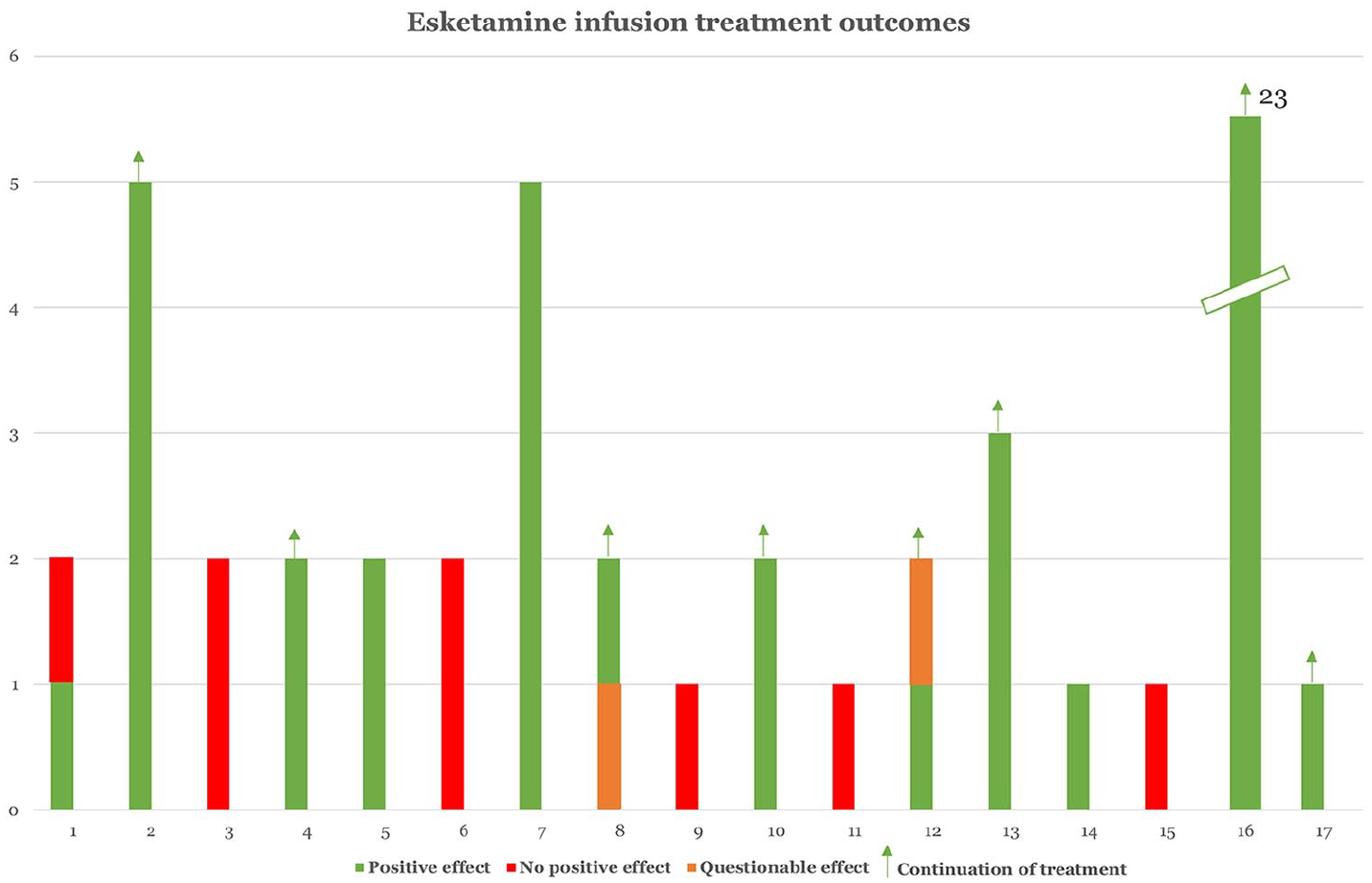
Esketamine infusion therapy outcomes.
Following her second esketamine infusion, one patient (Figure 1, patient 1) reported an increase in abdominal pain symptoms that was treated with oxycodone. The anesthesiologist could not link these symptoms to the administration of esketamine. Likewise, the gynecologist did not identify any connection between endometriosis and this sudden increase in abdominal symptoms since the patient was under treatment of a gonadotropin-releasing hormone (GnRH) agonist. Another patient experienced temporary weakness in both legs after esketamine infusion (Figure 1, patient 3). Additionally, in another case, the patient (Figure 1, patient 9) reported the treatment as a “bad experience” due to nausea, dizziness and un unpleasant sensation of “not being oneself.” Consequently, she did not wanted to continue with esketamine infusion therapy. After six infusions in four patients, a marginal elevation in γGT, AP, ASAT, ALAT levels were noted (Supplemental Table 2). These deviations had no consequences, all patients were able to proceed with esketamine infusion therapy.
Eleven out of 17 patients (64.7%) reported pain reduction following their first esketamine infusion (Figure 1). During the second infusion, one patient (patient 1), did not experience this effect anymore. Patient 8 and 9 experienced both positive and questionable effects during their two esketamine infusions. Overall, when considering all esketamine infusions, 11 out of 17 patients (64.7%) have experienced a positive effect on pain reduction (patients 2, 4, 5, 7, 8, 10, 12, 13, 14, 16, and 17). Currently, eight patients are continuing to undergo esketamine infusion therapy. Of the remaining patients, two underwent subsequent laparoscopic endometriosis resection surgery, one switched to pulsed radiofrequency treatment (PRF), and six patients were non-responders (Table 2).
Discussion
Esketamine infusion therapy could be a potential effective treatment to alleviate chronic endometriosis-associated pain. The data from our current cohort demonstrates that esketamine infusion therapy for chronic endometriosis-associated pain appears to be safe and effective for the majority of patients to reduce pain symptoms. This study is the first to summarize data on the efficacy of esketamine infusion in this particular patient population.
Current treatments to suppress endometriosis-associated pain include hormonal treatment and/or surgery. 26 Hormonal treatments have all proven to significantly reduce pain symptoms compared to placebo, but the efficacy and side effects remain highly heterogeneous in daily practice. 26 In addition, hormonal treatments such as GnRH agonists, may have severe side effects on bone density, mental wellbeing, and sexuality.27,28 With regard to surgery, 11.8% of women who undergo endometriosis lesion resection report no improvement of pain. 29 It is likely that these women have developed a state of sensitization, possibly induced by the long diagnostic delay of endometriosis. 5 In addition, it is known that pain perception is also influenced by other factors such as psychological and physical stress, hormone status, and pain coping mechanisms. 30 The NMDA receptor plays a vital role in the process of central sensitization. 31 The NMDA receptor is an excitatory glutamatergic receptor present at spinal and supraspinal sites and is involved in the afferent transmission of nociceptive signals. NMDA receptor antagonists, such as (es)ketamine, can halt the excessive barrage of nociceptive afferent input to the brain from persistent tissue damage and are therefore used to treat patients with chronic pain syndromes.
Mangnus et al. conducted a survey on the utilization of esketamine for chronic pain (mainly CRPS) in the Netherlands, revealing that esketamine is used in 31 pain clinics for chronic pain. 32 Notably, endometriosis is not reported as an indication in this study. 32 Regarding the esketamine dose regimen, the reported esketamine dose in these clinics is significantly higher compared to the dose administrated in our study. Mangnus et al. report maximal doses of 27.5 mg/h (in an outpatient setting) and 25.0 mg/h (in an inpatient setting). To compare, the maximal dose (mg/h) in our cohort was 12.5 mg/h (Supplemental Figure 2). Potentially, this difference in dose protocol is the result of the difference in patient population. Indeed, in accordance with prior studies, that reported favorable outcomes of (es)ketamine therapy in CRPS patients, the treatment dose was higher in comparison to the current study. 19
Whilst esketamine is used in the majority of pain clinics in the Netherlands to improve chronic pain symptoms, the evidence supporting (es)ketamine infusions to provide long-term pain relief is limited.33,34 Currently, esketamine is registered as an anesthetic and as an antidepressant in the treatment of patients with treatment resistant depression, and its use for the treatment of chronic pain is off-label. 35 This highlights the need for a randomized controlled trial with long-term follow up to assess whether esketamine infusion therapy significantly improves chronic endometriosis-associated pain. Recruitment for this study is ongoing (ClinicalTrials.gov NTC06161805).
In contrast to surgery or hormonal treatment, esketamine targets three key aspects of endometriosis simultaneously: pain, inflammation, and depression. Therefore, we propose that esketamine infusion therapy could serve as an alternative treatment option for chronic pain associated with endometriosis when conventional therapy fails, to bridge the time to surgery (exemplified by patient 5 and 14, Figure 1) or even as maintenance therapy (exemplified by patient 16, Figure 1). Whilst most described side-effects in this study (Table 2) are common side-effects of esketamine, outcomes on long-term repeated administration of esketamine infusions (i.e. development of pharmacodynamic, metabolic, and tolerance, resulting in tachyphylaxis and loss of analgesic effect) are not available and are warranted to ensure long term safety and efficiency.33,36
Another positive aspect of esketamine infusion treatment is that it can be strictly monitored, in contrast to opioid consumption. This is an important advantage, given the four times higher opioid consumption among women with endometriosis compared to non-endometriosis patients, resulting in a higher opioid dependency and potential addiction. 37 In addition, it can also be administrated in a home setting, enhancing cost efficiency.
Esketamine can be administrated via multiple routes: intravenous, intramuscular, intranasal, inhalation, oral (liquid or pills), subcutaneous, topical, and rectal. 33 However, intravenous administration of esketamine facilitates precise dosing and allows for discontinuation or adjustment of the treatment dosage in response to side effects, unlike administration routes where the entire dosage is already absorbed. In addition, a low and steady (es)ketamine dose protocol prevents high peak plasma levels, which are more associated with side-effects. 38
During the majority of infusion sessions, mild side effects were reported (Table 2). However, these observations are consistent with anticipated side effects. 14 Apart from these mild effects, the treatment was safe and well-tolerated, except for patient 9, who opted to discontinue esketamine infusion therapy due to a “bad experience” (nausea, dizziness, and sensation of not being oneself). Furthermore, alleviation of pain symptoms was observed in 11 of 17 patients (64.7%), which is a favorable outcome, particularly given that all these patients were resistant to conventional therapy. To compare, a similar response rate (62%) was reported in a retrospective study including patients with CRPS. 39 It may be that non-responders received a dose that was too low or are experiencing symptoms not mediated through sensitized NMDA receptors, differing in pain pathophysiology from responders, as hypothesized in fibromyalgia patients. 40 Larger studies are necessary to be able to identify predictors for esketamine responsiveness.
Our study has, however, some limitations. First, the retrospective design with its inherent limitations. In this context, pain scores were not documented in a standardized manner (e.g. numeric rating scores for pain), prior and post-infusion treatment. This limited our ability to determine the decrease in pain in a quantitative manner. In addition, the duration of the treatment effect was not reported (only a single telephonic evaluation occurred 6 weeks after the infusion). This information is crucial when considering esketamine as a potential alternative treatment for endometriosis-associated pain. Nevertheless, this study is the first to publish promising data regarding the efficacy of esketamine infusion treatment in endometriosis patients experiencing chronic pain.
Conclusion
Esketamine is a versatile drug that possesses analgesic, anti-inflammatory, and anti-depressive properties. This makes esketamine a promising treatment option for patients with chronic endometriosis-associated pain as is reflected by the results of our study. However, future research in a randomized controlled trial is mandatory to further assess the efficacy of (es)ketamine infusion therapy in this patient population.
Supplemental Material
sj-docx-1-pev-10.1177_22840265241259880 – Supplemental material for Esketamine infusion therapy: A promising alternative in the treatment of chronic endometriosis-associated pain
Supplemental material, sj-docx-1-pev-10.1177_22840265241259880 for Esketamine infusion therapy: A promising alternative in the treatment of chronic endometriosis-associated pain by Rozemarijn de Koning, Florine van Dulken, Willem Brinkert, Albert Dahan, Frank Willem Jansen, Mathijs Blikkendaal and Andries Twijnstra in Journal of Endometriosis and Pelvic Pain Disorders
Supplemental Material
sj-jpg-2-pev-10.1177_22840265241259880 – Supplemental material for Esketamine infusion therapy: A promising alternative in the treatment of chronic endometriosis-associated pain
Supplemental material, sj-jpg-2-pev-10.1177_22840265241259880 for Esketamine infusion therapy: A promising alternative in the treatment of chronic endometriosis-associated pain by Rozemarijn de Koning, Florine van Dulken, Willem Brinkert, Albert Dahan, Frank Willem Jansen, Mathijs Blikkendaal and Andries Twijnstra in Journal of Endometriosis and Pelvic Pain Disorders
Supplemental Material
sj-jpg-3-pev-10.1177_22840265241259880 – Supplemental material for Esketamine infusion therapy: A promising alternative in the treatment of chronic endometriosis-associated pain
Supplemental material, sj-jpg-3-pev-10.1177_22840265241259880 for Esketamine infusion therapy: A promising alternative in the treatment of chronic endometriosis-associated pain by Rozemarijn de Koning, Florine van Dulken, Willem Brinkert, Albert Dahan, Frank Willem Jansen, Mathijs Blikkendaal and Andries Twijnstra in Journal of Endometriosis and Pelvic Pain Disorders
Footnotes
Abbreviations
NMDA: N-methyl-D-aspartate
EPR: electronic patient record
CRPS: chronic regional pain syndrome
CRF: case report form
ALAT: alanine aminotransferase
ASAT: aspartate aminotransferase
AP: alkaline phosphatase
γGT: glutamyl transferase
Author contributions
RdK, AT, FWJ, and MB contributed to the study design. RdK, FvD, WB, AD, and AT contributed to data analysis, data interpretation, and manuscript writing. All authors contributed to data interpretation and critical manuscript revision.
Data availability statement
The dataset generated during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
