Abstract
Large segmental bone defects remain difficult to treat: autografts are limited by supply and morbidity, while calcium-phosphate substitutes often deliver inconsistent outcomes. This inconsistency largely originates at the early post-implantation interface, where protein adsorption and Ca2+/PO43− flux are unpredictable and lack a scalable solution. We therefore engineered mineral-coated microparticles (MCM) by growing a conformal, low-crystallinity apatite nanolayer on porous β-TCP under aqueous, protein-compatible conditions, preserving interconnected porosity. The coating establishes a uniform, stable protein corona and attenuates burst while smoothing ion release; in a rabbit mandibular critical-size model, MCM achieved more complete bridging, denser μCT infill, and higher bone mineral density than β-TCP and β-TCP/HA. Collectively, these data support a growth-factor-free, interface-first design that delivers more consistent regeneration and is readily translatable as an off-the-shelf particulate filler.
Introduction
Large segmental bone defects arising from high-energy trauma, tumour resection, osteomyelitis, revision arthroplasty or congenital dysplasia remain difficult to manage once they exceed the critical size.1,2 Autologous bone is still the benchmark because it contributes osteogenic cells, inductive cues and a native mineral scaffold; yet finite supply, donor-site morbidity and unpredictable resorption constrain its use.2,3 Allograft increases availability but lacks viable cells and often incorporates slowly, particularly in poorly vascularised beds. 4 These limitations have accelerated the adoption of calcium-phosphate (CaP) ceramics—hydroxyapatite (HA), β-tricalcium phosphate (β-TCP) and biphasic composites—which are osteoconductive and in principle tunable in resorption. Nevertheless, clinical and preclinical outcomes with nominally similar CaPs remain inconsistent across indications and anatomical sites.5–9 The immediate post-implantation window—when proteins adsorb,10,11 ions exchange and the host immune response is first engaged12,13—varies markedly with phase ratio, crystallinity, pore architecture and surface chemistry,14,15 making early behaviour at the graft–tissue interface difficult to predict. This persistent variability defines a practical unmet need: off-the-shelf particulate fillers whose early interfacial behaviour is reproducible enough to deliver reliable regeneration in demanding defects.
While most materials strategies emphasise bulk composition or porosity,16,17 converging mechanistic work points to a different control knob: the first hours to days after implantation.18,19 In this narrow interval, blood-borne proteins rapidly assemble a protein corona that becomes the biological identity cells actually sense20–24; in parallel, initial dissolution perturbs local Ca2+/PO43− activities and pH.25,26 Corona composition and architecture are exquisitely sensitive to surface mineralogy and nanoscale morphology; on CaP substrates, lower crystallinity and defect-rich surfaces increase binding-site density and specific surface area, favouring matrix-protein adsorption and altering exchange kinetics.7–9,27 Equally important, the chronokinetics of ion release—the amplitude and smoothness of Ca2+/PO43− flux—has immunological consequences: sustained, moderate release can engage calcium-sensing pathways and bias macrophages towards a pro-repair trajectory, supporting osteoblast adhesion, matrix deposition and timely coupling of resorption and formation, whereas burst dissolution prolongs inflammation and delays interface maturation.28–31 A practical way to steer these early events—without sacrificing scaffold geometry or pore connectivity—is biomimetic mineralisation in aqueous, protein-compatible media, which can grow conformal, low-crystallinity apatite (HA-like) nanolayers that afford fine control over surface charge, wettability and dissolution/ion-release behaviour. This interface-first perspective reframes bone repair as an osteo-immunomodulatory problem: by programming protein adsorption and ion kinetics, one can set the trajectory for faster, more mature bone formation.28–33
Guided by this clinical need and mechanistic rationale, we developed a particulate bone filler in which porous β-TCP granules are clad in situ with a conformal HA-like nanolayer—mineral-coated microparticles (MCM; β-TCP@HA). Rather than serving as a passive carrier, the coating is used as an active interfacial regulator to achieve two coupled goals: (i) establish a more uniform, stable protein corona on the graft surface and (ii) dampen burst dissolution while maintaining sustained Ca2+/PO43− availability—thereby potentially supporting a pro-regenerative milieu conducive to osteogenesis without exogenous growth factors. We evaluate this strategy in a stringent rabbit mandibular critical-size defect as a translational read-out, quantifying micro-CT morphometrics and histological maturation and relating pre-implantation surface indices (adsorption plateau, burst-factor/smoothness of ion release) to in vivo regeneration. By explicitly linking programmable surface mineralogy to predictable healing kinetics, we reposition mineral coatings from adjuncts to primary interface controllers, offering a materials-and-medicine pathway towards reliable repair of critical bone defects.
Materials and methods
Materials
β-Tricalcium phosphate (β-TCP; purity 98%; initial particle size 20–200 µm) and hydroxyapatite powder (HA; purity 96%; primary particle size ~200 nm) were obtained from Shanghai Ipure (Shanghai, China). Simulated body fluid (SBF) was prepared according to the Kokubo recipe; the ionic composition is provided in Supplemental Table S1. HEPES buffer (pH 7.4), FITC–BSA (for fluorescence imaging), BSA, a micro-BCA protein assay kit, and acids for digestion (HNO3/HCl) were purchased from commercial suppliers and used as received. Anticoagulated rabbit whole blood for the haemolysis assay was obtained under institutional ethics approval SYXK2023-0017. Unless otherwise stated, all reagents were of analytical grade, and ultrapure water (18.2 MΩ·cm) was used throughout.
Preparation of samples
Mineral-coated microparticles (MCM; β-TCP@HA)
β-TCP granules were first sieved to 0.25–1.0 mm, washed sequentially with deionised water and ethanol, and dried overnight at 60°C. For biomimetic mineralisation, the granules were immersed in SBF at 37°C at a solid–liquid ratio of 1 g:100 mL, placed on an end-over-end mixer (80 rpm), and the medium was renewed every 1 h for a total duration of 24 days. After mineralisation, particles were rinsed with deionised water and dried (60°C, 1 h). Prior to in vitro and animal experiments, samples were sterilised by γ-irradiation (25 kGy).
Physical mixture (β-TCP/HA)
For the physical blend, β-TCP was mixed with HA to a 5 wt% HA fraction using a tumble mixer for 30 min; no mineralisation step was applied. Sterilisation was performed as described for MCM.
Particle size control
Sieve windows and retained fractions were recorded. The median particle size D50 was determined by laser diffraction (Bettersize 2600, Bettersize Instruments, China).
Physicochemical characterisation
Scanning electron microscopy (SEM)
Pristine particles and post-immersion particles were examined using a Thermo Scientific Scios 2 HiVac field-emission SEM (U.S.A) operated at 10 kV. Samples were sputter-coated with Pd for 90 s prior to imaging. For each condition, images were acquired at three magnifications with scale bars of 500, 50, and 5 µm, respectively.
Fourier transform infrared spectroscopy (FT-IR)
Spectra were collected by ATR-FTIR (Agilent Cary 630, Agilent Technologies, U.S.A) over 4000–500 cm−1, with a spectral resolution of 4 cm−1 and 32 scans per spectrum.
X-ray diffraction (XRD)
Powder XRD (D8 Advance, Bruker, Germany) patterns were recorded using Cu Kα radiation (λ = 1.5406 Å) at 40 kV/30 mA, scanning 2θ = 10°–80° with a step size of 0.02° and a scan rate of 5°·min−1. Reference patterns used for phase identification were JCPDS 09-0169 (β-TCP) and JCPDS 09-0432 (HA). Rietveld refinement was optionally performed to estimate phase fractions.
Elemental analysis (Ca, P)
Calcium and phosphorus contents were determined by ICP-MS (Agilent 7900 ICP-MS, Agilent Technologies, U.S.A) following acid digestion in HNO3. Multi-element standards were used for external calibration, and results are reported as wt%.
In vitro protein adsorption
FITC–BSA was used as a tractable model protein to visualise the spatial uniformity of adsorption on the highly porous, light-scattering granules. Because fluorescence intensity on irregular particles is not directly quantitative and can be affected by imaging settings and photobleaching, adsorption kinetics were quantified by a mass-balance approach using the micro-BCA assay on the supernatant (C0–Ct). This provides a robust quantitative readout in the low-µg·mL−1 range under PBS (physiological pH/ionic strength) conditions, enabling fair comparison across materials.
Adsorption imaging
Particles were incubated with FITC–BSA (50 µg/mL in 1 × PBS, pH 7.4, ionic strength ~0.15 M) at 37°C for 1.5 h, protected from light. Samples were then gently rinsed once with PBS to remove loosely bound protein and imaged by confocal microscopy (Nikon AX R, Nikon Corporation, Japan; excitation 488 nm/emission 520 nm). Representative fields were recorded under identical settings (laser power/exposure, gain) for all groups.
Adsorption kinetics (quantitative)
Batch adsorption was performed at a liquid–solid ratio of 1 mL : 2 mg in 1× PBS (pH 7.4, ionic strength ~0.15 M) with end-over-end mixing (30 rpm) at 37°C. Supernatants were collected at 0, 0.5, 1, 2, 5, 10, 20, 40, 60, 120, 240 and 360 min and analysed by the micro-BCA assay. The adsorbed amount was calculated as:
where (C0) and (Ct) are the initial and time-t protein concentrations, (V) is the solution volume, and (m) is the mass of particles. The adsorbed fraction (%) was computed as 100×(C0−Ct)/C0 and plotted versus time. Nitrogen adsorption–desorption isotherms were used to determine the BET (ASAP2460, micromeritics, U.S.A) specific surface area (SSA),and the adsorbed mass at 360 min (last time point)was additionally normalised by SSA to yield an area-normalised metric (mg·m−2), calculated as M/(m·SSA). Independent replicates were performed as indicated. The adsorbed amount was calculated by depletion of protein from the supernatant; this assay does not distinguish reversible versus irreversible binding, and no post-adsorption desorption step was performed.
In vitro ion release
Samples were immersed in simulated body fluid (SBF) at a solid–liquid ratio of 1 g:20 mL under gentle orbital agitation. The tests were conducted at 37°C; the pH of the immersion medium was nominally 7.4 according to the standard SBF formulation (unless otherwise stated). Aliquots were collected at 10 min, 20 min, 30 min, 1 h, 2 h, 3 h, 4 h, 5 h, 6 h, 7 h, 14 h, 21 h, 28 h, 42 h, 56 h, 70 h and 84 h. After each sampling, an equal volume of pre-warmed SBF was added back to maintain a constant total volume. Calcium and phosphate concentrations in the supernatants were quantified spectrophotometrically using the o-cresolphthalein complexone (o-CPC) method for Ca2+ and the ammonium molybdate method for PO43−, respectively; matrix-matched calibration curves and reagent blanks were included for each run. Cumulative release (%) at time (tk) was calculated with replacement correction as
where (Ci) is the measured concentration at sampling point (i), (V) the total medium volume, (Vs) the sampled volume, and (Mtotal) the total mass of Ca or P in an identical mass of particles determined by acid digestion (see Elemental analysis). After the release test, SEM imaging of the immersed particles was performed at the same magnifications as for the pristine samples (scale bars: 500, 50 and 5 µm). In this work, coating stability is operationally defined as morphological continuity and resistance to pore occlusion/delamination after SBF immersion, rather than crystallographic phase invariance. Accordingly, post-immersion SEM was used as a qualitative readout of morphological integrity (continuity/occlusion/delamination) of the mineral shell after exposure to the simulated physiological medium.
Cytocompatibility and haemolysis
Cell proliferation (extract method)
Mouse fibroblasts L929 were cultured in Minimum Essential Medium (MEM) supplemented with 10% foetal bovine serum (FBS) and 1% penicillin–streptomycin at 37°C in 5% CO2. Material extracts were prepared at an extraction ratio of 0.2 g·mL−1 in complete MEM at 37°C for 24 h, then cleared (centrifugation and/or 0.22 µm filtration as appropriate). Cells were seeded in 96-well plates at 1 × 103 cells·well−1 and allowed to attach for 24 h, after which the culture medium was replaced with the test extract or blank culture medium (control). Cell proliferation/viability was assayed by MTT at 24, 48 and 72 h following the kit instructions (incubation with MTT, dissolution of formazan crystals in DMSO). Absorbance was read at 570 nm (reference 630–690 nm). Viability (%) was calculated as:
Each condition was tested in n = 3 independent wells and repeated in 3 biological replicates.
Haemolysis
Fresh anticoagulated rabbit whole blood was diluted 1:8 (v/v) with 0.9% NaCl. Test materials were incubated with the diluted blood at 37°C for 4 h with gentle mixing. The following controls were included: negative control (NC), 0.9% NaCl; positive control (PC), deionised water (alternatively 0.1% Triton X-100). After incubation, samples were centrifuged and the supernatant absorbance was measured at 540 nm. The haemolysis index (%H.I.) was calculated as:
All measurements were performed in triplicate per condition, and results are reported as mean ± SD.
Animal experiments (rabbit mandibular defect model)
Ethics and animals
All procedures were approved by the institutional animal care and use committee (No. IACUC-20230714-01) and complied with relevant guidelines. Ten-month-old New Zealand white rabbits (body mass ~3 kg) were acclimatised under controlled housing conditions: ambient temperature 22 ± 2°C, relative humidity 40%–70%, light intensity 150–200 lx with a 12 h/12 h light–dark cycle, and noise level < 50 dB. Standard diet and water were provided ad libitum.
Grouping and time points
Animals were randomly assigned to four groups—Model (empty defect), β-TCP, β-TCP/HA, and MCM (mineral-coated microparticles)—using a computer-generated block randomisation sequence (generated in R using the sample() function), with allocation concealed until implantation. Primary endpoints were set at 4 and 8 weeks; an extended endpoint at 12 weeks was planned for longitudinal observations. The planned sample size was n = 6 rabbits per group per time point.
Surgical procedure
Anaesthesia was induced with pentobarbital and maintained with isoflurane; local infiltration with lidocaine–epinephrine was used for haemostasis and analgesia. Penicillin was administered prophylactically pre-operatively. Through a lateral extraoral approach, the mandibular body was exposed and a critical-size defect (10 mm in diameter × 4 mm in depth), selected based on previously reported rabbit mandibular critical-size defect models,34,35 was created using a trephine bur under continuous saline irrigation. According to allocation, defects were either left unfilled (Model), filled with β-TCP/HA (HA mass fraction 5%) or with MCM granules. Wounds were closed in layers. Postoperative care followed institutional guidelines; flunixin meglumine (0.2 mg/kg) was administered for analgesia.
Imaging analyses
At harvest, gross photographs were taken with a scale grid. Dental radiographs (IPET-400,Bio-Proton Biotech (Suzhou),China) were acquired at 60 kV with a source-to-sample distance of 30–40 cm. Radiographs were coded prior to analysis and scored using the Lane–Sandhu system by investigators blinded to group allocation (and time point where applicable). Micro-CT (μCT, SKYSCAN 1276, Bruker, U.S.A) scanning was performed at 60–100 kVp with a voxel size of 5–20 µm and an integration time of 100–2000 ms. Segmentation thresholds were selected by grey-level histogram inspection combined with visual verification. Quantitative μCT metrics were computed within predefined regions of interest (ROI) spanning the defect, including bone volume fraction (BV/TV, %), bone mineral density (BMD) and trabecular number (Tb.N, mm−1).
Histology
For decalcified histology, specimens were fixed in 10% neutral-buffered formalin, decalcified in acid decalcifier, paraffin-embedded, sectioned, and stained with haematoxylin & eosin (H&E) and Masson’s trichrome. For undecalcified resin histology (subset), samples were fixed, dehydrated, embedded in methyl methacrylate, sectioned and stained as detailed in Methods. ROIs were predefined to cover the entire defect. Collagenation rate (%) was defined as the area fraction of Masson’s trichrome collagen-positive staining within the predefined defect ROI (collagen-positive area/total ROI area × 100). Whole-slide images (or stitched fields) were analysed in ImageJ using colour deconvolution (Masson’s trichrome vector) followed by a fixed threshold applied identically across groups. For each animal, three sections were quantified and averaged. Quantitative outcomes included new bone area fraction (%), osteoblast counts (per ROI), collagenation rate (%), and material retention rate (%). Two observers, blinded to group, performed scoring; inter-rater agreement (κ) was calculated and reported.
Data processing and statistics
Data are presented as mean ± SD (n denotes the number of rabbits per group per time point unless otherwise stated; see figure legends for exact n). Sample sizes were chosen based on prior rabbit mandibular defect studies and feasibility constraints, and no a priori power calculation was performed. For multi-group comparisons at a single time point, one-way ANOVA with Tukey’s post hoc test was used; for two-group or paired time-point comparisons, the Student’s t test (or Welch’s t test when variances were unequal) was applied. When normality or homoscedasticity was not satisfied, Kruskal–Wallis with Dunn’s multiple comparisons or Welch’s ANOVA with Games–Howell post hoc was used as appropriate. For time-course datasets (protein adsorption, ion release), inference was restricted to plateau values or pre-specified key time points; cumulative release values were corrected for sampling/volume replacement. Statistical significance was set at α = .05, with annotations: ns (p ⩾ 0.05), *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001. Analyses and plotting were performed in GraphPad Prism v10.1and R v4.4.2.
Results
Conformal apatite coating establishes a porous β-TCP core–shell architecture
We prepared mineral-coated microparticles (MCM) by growing a conformal apatite layer on porous β-TCP granules under aqueous, protein-compatible conditions. The preparation route and intended core–shell configuration are shown schematically (Figure 1(a)). Multiscale SEM verified surface mineralisation while retaining pore connectivity (Figure 1(b)): at low magnification (500 µm) MCM preserved the macroporous granular geometry of the parent β-TCP; at 50 µm the sharp facets and open pore mouths of β-TCP were covered by a thin, continuous mineral lamina that bridged micropores; at 5 µm the coating appeared as densely packed nano plate/needle-like features, with visible pore openings and no evidence of occlusion. FT-IR spectra showed phosphate bands consistent with apatite on MCM while retaining signals attributable to β-TCP (Figure 1(c)). Powder X-ray diffraction preserved the characteristic β-TCP reflections, with additional apatite features observed on the coated samples (Figure 1(d)). Elemental analysis (ICP, wt%) reported Ca2+ and PO43− contents for β-TCP, β-TCP/HA (physical blend), and MCM (Figure 1(e)). Under extract conditions, cytocompatibility and blood compatibility assays indicated no abnormal responses: cell proliferation at 24/48/72 h was comparable to the blank control as annotated (Supplemental Figure S1A, S1B) and haemolysis ratios were not different from the negative control across materials (Supplemental Figure S1C).
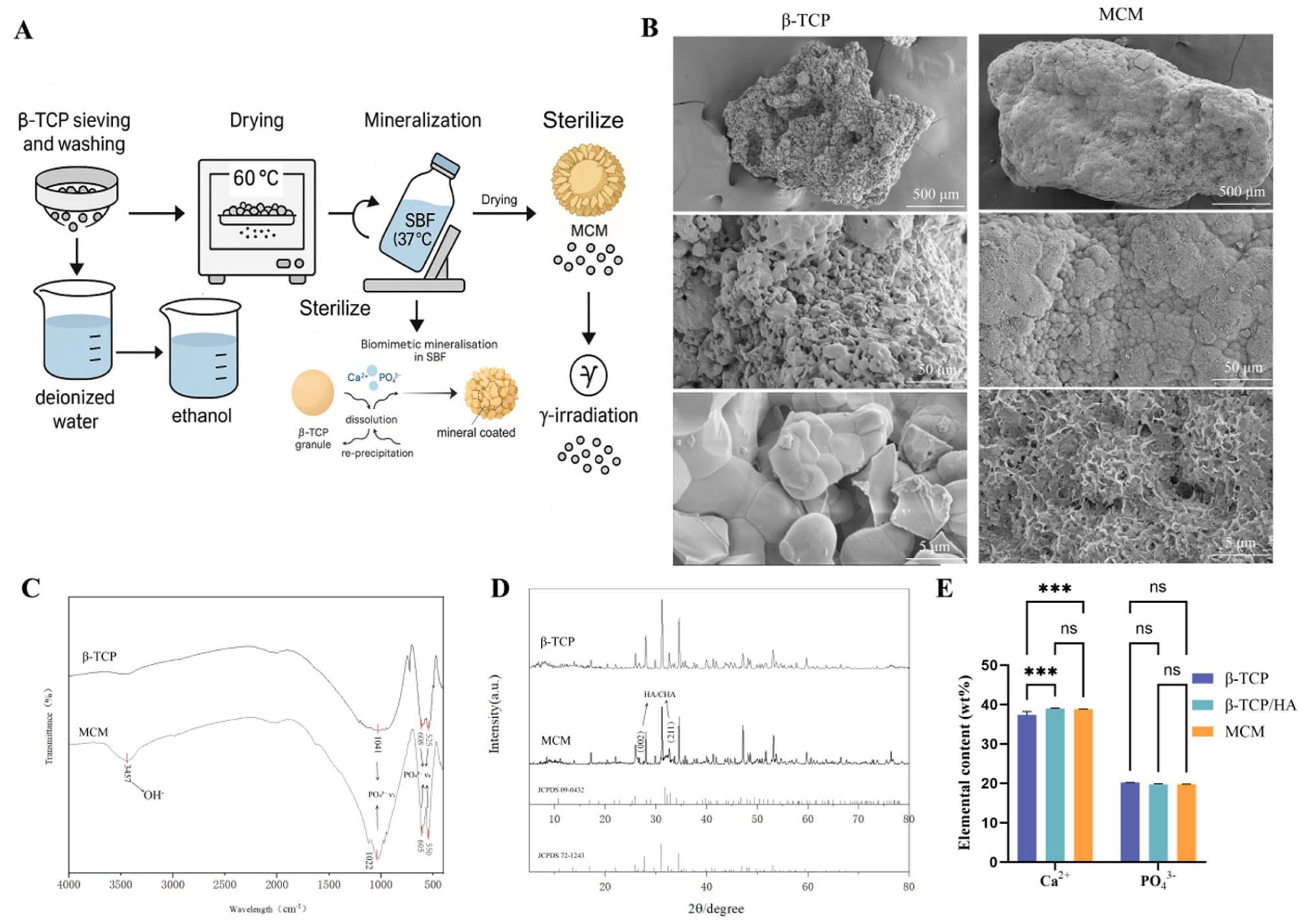
Fabrication and physicochemical characterisation of β-TCP and mineral-coated microparticles (MCM): (a) schematic sequence: β-TCP granules → immersion in simulated body fluid (SBF) → formation of mineral-coated microparticles (MCM, β-TCP@HA), (b) SEM images of β-TCP (left) and MCM (right) at low, medium and high magnification; scale bars: 500 µm, 50 µm and 5 µm (top to bottom), (c) FT-IR spectra of β-TCP and MCM (4000–500 cm−1), (d) powder XRD patterns of β-TCP and MCM with reference cards displayed below (JCPDS 72-1243and 09-0432), (e) elemental content (wt%) of Ca2+ and PO43− for β-TCP, a physical mixture β-TCP/HA, and MCM; bars show mean ± SD with significance annotations as indicated.
Coated interfaces enable uniform protein adsorption and controlled Ca/PO4 release
Representative epifluorescence of FITC–BSA showed a continuous, high-intensity layer on MCM, whereas β-TCP/HA and β-TCP exhibited patchy, lower-intensity coverage (Figure 2(a); 300 µm). Adsorption kinetics at 37°C rose more rapidly on MCM and reached a higher steady state, with an earlier plateau than the physical blend and β-TCP (Figure 2(b)). To confirm that the adsorption comparisons were not driven by specific surface area, BET SSA was determined from N2 adsorption–desorption isotherms (Supplemental Figure 2) and SSA-normalisation of the adsorbed mass at 360 min preserved the same group-wise ranking (Supplemental Table 2). Under the ion-release test conditions (SBF, 37°C), cumulative release curves indicated attenuation of the early burst on MCM and a smoother trajectory thereafter for both phosphate (PO43−) and calcium (Ca2+), while the comparators showed higher initial release (Figure 2(c) and (d)). SEM supported these measurements: before soaking, MCM displayed a conformal HA-like overlayer, β-TCP retained angular facets and the physical blend showed mixed HA/TCP domains; after soaking, β-TCP exhibited surface pitting/erosion and the physical blend exposed faceted crystalline regions, whereas MCM maintained a continuous nanoscale coating that bridged micropores without occluding connectivity (Figure 2(e) and (f); 500, 50, and 5 µm). Notably, after SBF immersion the MCM surface still exhibited a continuous nanoscale overlayer bridging micropores, with no apparent pore occlusion or gross delamination, supporting morphological stability of the mineral shell over the tested time window.

Protein adsorption and dissolution characterisation of β-TCP, a physical mixture β-TCP/HA and mineral-coated microparticles (MCM): (a) fluorescence micrographs of FITC–BSA on particle surfaces for β-TCP, β-TCP/HA and MCM; scale bars = 300 µm, (b) time course of adsorbed fraction (%) for the three materials (37°C; 1×PBS, pH 7.4 ionic strength ~0.15 M; conditions as in Methods), (c) cumulative Ca2+ release (%) versus time under the ion-release test conditions, (d) cumulative PO43− release (%) measured in the same medium and schedule, (e) SEM survey of pristine particles at low, medium and high magnification; scale bars (left to right) = 500 µm, 50 µm, 5 µm and (f) SEM survey of particles after immersion under the ion-release test conditions at the same magnifications; scale bars (left to right) = 500 µm, 50 µm, 5 µm.
Defect bridging and mineral quality are enhanced and correlate with interface metrics
In a rabbit mandibular critical-size defect, gross inspection of harvested mandibles showed persistent voids in untreated defects, progressive filling with both comparators and more complete closure around MCM (Figure 3(a)). Three-dimensional μCT reconstructions revealed denser, more continuous trabecular infill with MCM relative to β-TCP/HA and β-TCP (Figure 3(b)). Notably, the untreated defects showed persistent voids with limited bridging at 8 weeks across gross observation, µCT and histology, supporting that this defect represents a critical-sized/non-self-healing challenge in this rabbit mandible setting. At the 4/8-week readout in the main figure, trabecular number (Tb.N) did not differ significantly among groups, although MCM showed a numerical increase versus both comparators (Figure 3(c)). Consistent with the reconstructions, BV/TV increased from 4 to 8 weeks in all groups and was highest in the MCM group at both time points, with the pairwise significances indicated in Figure 3(d). For bone mineral density (BMD), at 4 weeks MCM was higher than β-TCP (p < 0.05) with no significant difference versus β-TCP/HA; at 8 weeks MCM was higher than both β-TCP/HA (p < 0.05) and β-TCP (p < 0.01), and β-TCP/HA exceeded β-TCP (p < 0.05; Figure 3(g)).

Rabbit mandibular defect study design and imaging/μCT readouts: (a) gross photographs of mandibles harvested at 4 and 8 weeks for each group (scale shown on the cutting mat), (b) μCT 3D reconstructions and axial views of the defect region at 4 and 8 weeks, (c) dental X-ray radiographs with regions of interest (red boxes) at 4 and 8 weeks, (d) Bone volume fraction (BV/TV, %) at 4 and 8 weeks, (e) Lane-Sandhu X-ray score at 4 and 8 weeks, (f)linear correlation between in vitro protein adsorbed fraction (%) and in vivo new bone formation (%) across materials and (g) bone mineral density (BMD) at 4 and 8 weeks.
Radiographic scoring using the Lane–Sandhu system at 4/8 weeks aligned with these trends at the whole-mandible level: scores increased over time in all groups, with MCM exhibiting higher scores than the model cohort at both time points, and an advantage over β-TCP/HA at 8 weeks; exact pairwise significances are indicated in the bar annotations (Figure 3(e)). To relate these tissue-scale outcomes to early interface behaviour, the adsorbed-protein fraction (%) measured in vitro on the same materials correlated with new-bone formation (%) across groups (linear fit Y = 1.844X − 17.80; R2 = 0.9221; Figure 3(f)), consistent with the μCT-based bone fill (Figure 3(f)).
Organised collagen deposition with comparable material persistence across groups
Masson’s trichrome at 4 weeks showed that the model group retained voids within the defect, with dense blue-stained fibrous callus at the margins and only scant new trabeculae; around MCM, extensive, orderly blue callus was present with numerous new trabeculae; in the β-TCP and β-TCP/HA groups, abundant fibrous callus was also seen at the defect edge and around particles, but with comparatively fewer new trabeculae (Figure 4(a)). At 8 weeks, the model group still contained residual voids of varying size; marginal fibrous callus decreased and appeared looser, and bone increased with signs of maturation; around MCM, fibrous callus decreased yet remained dense and orderly, with visibly increased and more mature bone; the comparators showed decreased callus with denser alignment and modest bone maturation (Figure 4(a)). Quantitatively, the collagenisation rate was higher for MCM than Model at 4 weeks (p < 0.01), with other pairwise comparisons at 4 weeks not significant; at 8 weeks, differences among groups were not significant (Figure 4(b)). Material retention showed no significant differences among β-TCP, β-TCP/HA, and MCM at either 4 or 8 weeks (Figure 4(c)).

Histology and quantitative morphometry of rabbit mandibular defects: (a) Masson’s trichrome–stained sections at 4 and 8 weeks for the Model, β-TCP, β-TCP/HA, and MCM groups; upper panels show low-magnification overviews and lower panels show higher-magnification views of the red-boxed regions. (b) Collagenation rate (%) at 4 and 8 weeks (bars: mean ± SD; significance annotations as indicated). (c) Material retention rate (%) for β-TCP, β-TCP/HA and MCM at 4 and 8 weeks (bars: mean ± SD; significance annotations as indicated).
Greater new-bone formation and osteoblast accumulation at the graft interface
At 4 and 8 weeks, H&E sections showed that the model group retained central voids with limited marginal osteogenesis and inflammatory infiltrates that diminished over time; in defects containing MCM, the material filled the gap and was surrounded by orderly fibrous callus with numerous new trabeculae; by 8 weeks new bone visibly increased with features of lamellar maturation (Figure 5(a)). The β-TCP and β-TCP/HA groups were intermediate, with scattered material, more fibrous encapsulation/inflammation and absent or limited central osteogenesis. Quantitatively, the area ratio of new bone (%) showed that at 4 weeks MCM was higher than Model (p < 0.0001), with other pairwise comparisons not significant; at 8 weeks, MCM remained higher than Model (p < 0.0001) and was higher than β-TCP/HA (p < 0.05), with other comparisons not significant (Figure 5(b)). For osteoblast number, at 4 weeks MCM exceeded Model (p < 0.001), with other comparisons not significant; at 8 weeks, MCM exceeded Model (p < 0.0001) and β-TCP/HA (p < 0.05), while other comparisons were not significant (Figure 5(c)).

Histological (H&E) assessment and morphometric quantification in the rabbit mandibular defect model. (a) Representative H&E-stained sections at 4 and 8 weeks for the Model, β-TCP, β-TCP/HA and MCM groups; for each time point, the upper row shows low-magnification overviews and the lower row shows higher-magnification views of the red-boxed regions. (b) Area fraction of new bone (%) at 4 and 8 weeks (bars: mean ± SD; significance annotations as indicated). (c) Number of osteoblasts in the predefined regions of interest at 4 and 8 weeks (bars: mean ± SD; significance annotations as indicated).
Discussion
The present study demonstrates that growing a conformal apatite overlayer on porous β-TCP granules yields a core–shell mineral-coated microparticle (MCM) architecture that modulates early interfacial events and translates into improved bone regeneration in a mandibular critical-size defect (Figure 1). At the material level, SEM, FT-IR and XRD collectively indicate that MCM is best described as a porous β-TCP core enveloped by a conformal apatite shell, rather than a simple physical mixture. The coating appears as densely packed nano-plate/needle-like features bridging micropores while leaving pore mouths open. This is distinct from the angular, dissolution-prone surface of bare β-TCP and from the heterogeneous mixed domains observed in the β-TCP/HA blend. Such a continuous, nanostructured apatite layer is expected to provide a high-surface-area template for protein adsorption and mineral nucleation,10–13 while the preserved interconnected porosity should facilitate cell infiltration and vascular ingrowth. 10 Importantly, we achieved this architecture under aqueous, protein-compatible conditions, which is relevant both for preserving bioactivity of any co-delivered biologics and for aligning with manufacturing constraints of clinical bone fillers.
The in vitro protein adsorption and ion-release data suggest that this interfacial architecture has functional consequences during the immediate post-implantation window (Figure 2). FITC–BSA formed a uniform, high-intensity layer on MCM, whereas both β-TCP and β-TCP/HA showed patchy coverage. Kinetically, MCM adsorbed protein more rapidly and reached a higher steady state than the comparators; here, the ‘higher plateau’ reflects greater protein depletion from the supernatant under the tested conditions and should not be interpreted as evidence for reduced desorption or irreversible binding, which was not assessed. This behaviour is consistent with a higher density of energetically favourable binding sites on the apatite shell and a more homogeneous surface chemistry than the physical blend, where proteins encounter a mosaic of HA and TCP microdomains. Notably, this trend persisted after normalisation by BET SSA at the 360-min endpoint (Supplemental Table 2), supporting that the observed adsorption differences reflect interfacial chemistry/architecture rather than surface-area variations alone. In parallel, MCM attenuated the early burst release of Ca2+ and PO43− and exhibited a smoother release trajectory thereafter; the comparators showed higher initial release, consistent with the more exposed and faceted crystalline surfaces observed after SBF soaking. Under SBF immersion (37°C; nominal pH 7.4), MCM retained a continuous nanoscale overlayer without obvious pore occlusion (morphological integrity), although solid-phase evolution after immersion was not quantified (XRD/FT-IR/Rietveld). Accordingly, in this study the term ‘pro-regenerative milieu’ refers primarily to physicochemical cues (protein adsorption and ion flux), rather than directly demonstrated immune-cell polarisation.
Importantly, excessive coating stability could in principle blunt dissolution-mediated cues and impede cell recruitment or remodelling; in this context, the moderated—rather than suppressed—Ca/PO4 exchange observed for MCM suggests a balance between coating integrity and ion availability. Moderating early ion fluxes may help avoid excessive local supersaturation or pH shifts while still supplying osteoconductive ions, thereby shaping the protein corona and subsequent cell responses25,26; however, osteogenic differentiation markers (e.g. ALP activity, RUNX2/OCN expression) were not quantified in pre-osteoblasts or MSCs, and any direct pro-osteogenic effect at the cellular level remains to be validated. In addition, osteoimmunology studies indicate that calcium phosphate chemistry/topography and Ca2+/PO43− flux can influence macrophage polarisation and the coupling between inflammation resolution, angiogenesis, and osteogenesis.28–33 However, we did not directly quantify immune-cell phenotype (e.g. CD68/CD206) or cytokine profiles in this study; therefore, any immunomodulatory contribution of MCM remains an evidence-supported hypothesis rather than a demonstrated mechanism, and future work will incorporate macrophage/cytokine profiling under physiologically simulated conditions.
These interfacial metrics map onto clear differences in the in vivo regeneration pattern. In a rabbit mandibular critical-size defect, 36 μCT showed that MCM generated denser and more continuous trabecular infill than β-TCP or β-TCP/HA (Figure 3). At 4 weeks, the trends in bone mineral density already favoured MCM over β-TCP, and by 8 weeks MCM surpassed both β-TCP and the physical blend, with the blend also outperforming β-TCP. Although trabecular number did not differ significantly among groups at the measured time points, the numerical increase seen with MCM is consistent with more extensive network formation that may have become statistically evident with longer follow-up or larger cohorts. Radiographic Lane–Sandhu scores at the whole-mandible level tracked these μCT findings, further supporting the conclusion that MCM promotes more complete defect bridging and earlier functional consolidation than the comparators.
Histological analyses provide additional insight into the quality of the newly formed tissue. Masson’s trichrome staining revealed that MCM supported extensive and orderly fibrous callus formation with numerous new trabeculae at 4 weeks, while untreated defects retained central voids and the comparator grafts exhibited abundant fibrous tissue but fewer new trabeculae (Figure 4). By 8 weeks, MCM defects showed denser, more aligned collagen and more mature bone than β-TCP and β-TCP/HA, whereas the model group still contained residual voids and immature tissue. The quantitative collagenisation rate confirmed an early advantage for MCM versus the defect model at 4 weeks, with convergence among groups by 8 weeks as healing progressed. H&E staining similarly indicated that MCM reduced central voids, promoted lamellar-like bone maturation and supported higher osteoblast counts at both time points compared with the untreated model and, at 8 weeks, with the β-TCP/HA blend (Figure 5). Together, these data suggest that MCM promotes defect filling and yields a more organised, cellular regenerate, consistent with improved structural organisation and tissue maturation. Importantly, these histological features were mirrored by imaging-based structural endpoints, including higher BV/TV and BMD on μCT and improved Lane–Sandhu scores, supporting that the more organised regenerate is accompanied by greater μCT-based consolidation/bridging of the defect.
Interestingly, material persistence did not differ significantly among β-TCP, β-TCP/HA and MCM at either 4 or 8 weeks (Figure 4), indicating that the superior regeneration observed with MCM cannot be attributed simply to faster resorption or greater space liberation. Instead, the data are more consistent with an interface-centric mechanism, whereby a conformal apatite shell tunes early protein adsorption and ion release, leading to a more favourable microenvironment for osteoblast recruitment, matrix deposition and mineralisation. This interpretation is supported by the strong linear correlation between the in vitro adsorbed-protein fraction and the in vivo new-bone fraction across groups (R2 ≈ 0.92), as well as the alignment between μCT-based bone fill and histological outcomes. 24 However, these associations (within the 8-week window) do not establish causality and were derived from simplified in vitro assays using BSA as a model protein under static/low-agitation conditions; because in vivo perfusion and fluid shear can alter corona exchange and ion transport, longer-term follow-up and adsorption profiling in multi-protein media (serum/plasma) under dynamic flow/perfusion will be required to test remodelling outcomes and corona composition/competitive exchange. Despite these limitations, the observed cross-scale concordance supports the idea that quantifiable interface metrics may serve as practical mechanistic surrogates for predicting graft performance in vivo. While the strong linear association suggests that simple interfacial metrics may serve as practical surrogates for comparing materials in this model, these correlations do not establish causality and should be interpreted as hypothesis-generating rather than predictive in a strict statistical sense.
Future studies will (i) quantify macrophage phenotype (CD68/CD206) and key cytokines under dynamic conditions, (ii) profile competitive protein corona formation in serum/plasma and (iii) extend evaluation to 12–24 weeks and, ultimately, load-bearing models.
In summary, by engineering a conformal apatite shell on porous β-TCP granules, we created a mineral-coated microparticle that decouples bulk composition from interfacial function. MCM preserves favourable features of β-TCP—such as macroporous geometry and resorbability—while introducing a nanostructured apatite layer that promotes uniform protein adsorption and controlled Ca2+/PO43− release. These interface-level changes translate into enhanced defect bridging, higher mineral density, more organised collagen deposition and increased osteoblast accumulation in a mandibular critical-size defect, without altering overall material persistence. The close alignment between in vitro interface metrics and in vivo regenerative outcomes highlights the value of explicitly targeting the graft–tissue interface in the design and preclinical evaluation of bone-filling materials.
Conclusion
This study establishes an interface-first strategy for particulate bone fillers. By growing a conformal, low-crystallinity apatite nanolayer on β-TCP granules, we improved early protein-corona quality and smoothed Ca/PO4 release, producing faster bridging, higher bone quality and tighter bone–material contact in a rabbit mandibular critical-size defect—without exogenous growth factors. Simple pre-implantation indices (adsorption plateau; burst factor) predicted in vivo regeneration, positioning programmable surface mineralogy as a practical design handle. This growth-factor-free approach translates across indications and scaffolds, and provides a quantitative framework to couple osteoimmunomodulation with mineral kinetics towards reliable repair of critical bone defects. Future work will extend to load-bearing models and larger animals.
Supplemental Material
sj-docx-1-jbf-10.1177_22808000261431927 – Supplemental material for Interface-programmed regeneration of segmental bone defects with biomimetic mineral-coated β-TCP microparticles
Supplemental material, sj-docx-1-jbf-10.1177_22808000261431927 for Interface-programmed regeneration of segmental bone defects with biomimetic mineral-coated β-TCP microparticles by Bingyao Chen, Lufeng Yao, Keye Chen, Yanguang Zhou, Chenxin Wang, Xiaodong Xu, Jinxia Ma, Jiayu Chen, Deting Xue, Zhaoming Ye and Xiaohua Yu in Journal of Applied Biomaterials & Functional Materials
Footnotes
Acknowledgements
We thank Phil Stone Biotech (Hangzhou, China) for the offer of experimental raw materials.
Ethical considerations
All procedures were approved by the institutional animal care and use committee (No. IACUC-20230714-01) and complied with relevant guidelines.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the National Natural Science Foundation of China (82372381 and 82572778).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data supporting the findings of this study are available within the article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.




