Abstract
Implant-associated infections pose a significant challenge in orthopedic surgery but may be prevented using biomaterials containing antimicrobial agents such as Ag ions. This study examines the effects of Ag doping on bone metabolism following the implantation of β-tricalcium phosphate (β-TCP) doped with 0, 1, 3, and 5 at% Ag with 75% porosity. Additionally, the antimicrobial activity of Ag-doped β-TCP was evaluated against Staphylococcus aureus and Escherichia coli using shake flask tests, revealing increased antimicrobial activity with higher Ag concentrations. Cylindrical bone defects (diameter 4 mm; depth 10 mm) were introduced in the lateral femoral condyles of rabbits and treated with Ag-doped β-TCP. The rabbits were euthanized at 2-, 4-, 8-, and 12-weeks post-operation (n = 6/time point). Specimens were decalcified for histological examination using optical and scanning electron microscopy (SEM). Bone formation, residual material, and tartrate-resistant acid phosphatase (TRAP)-positive cell counts were quantified, with statistical significance assessed using one-way ANOVA (p < 0.05). Bone formation increased over time up to 12 weeks but was lower with higher Ag concentrations. Residual material decreased, while TRAP-positive cells peaked at 2 weeks and gradually declined thereafter. SEM revealed Ag accumulation in the bone marrow outside the newly formed bone. Ag doping inhibited material resorption more than osteogenesis. Bone metabolism in the defect area was delayed as Ag concentration increased, likely due to reduced osteoclast activity. This study highlights the dual effect of Ag-doped β-TCP on bone metabolism and implant-associated infections. While Ag incorporation enhanced antimicrobial potential, higher concentrations delayed bone metabolism. Optimizing Ag content is crucial to balancing infection control with effective bone regeneration, guiding the development of advanced orthopedic implants.
Keywords
Introduction
Implant-associated infections are a serious complication that occur after orthopedic surgeries. 1 The rising incidence of infections caused by antimicrobial-resistant bacteria, is a growing concern. 2 Foreign materials, such as biomaterials, can easily lead to infections because of microbial presence on their surfaces. 3 The formation of biofilm matrices comprising polysaccharides and proteins protects bacteria and reduces the effectiveness of antimicrobials. 4 Topical administration of antimicrobials effectively controls infections while preventing systemic toxicity. 5 However, no antimicrobials have been developed that are effective against all types of bacteria. 6
Numerous inorganic ions with antimicrobial properties have been identified to reduce the risk of implant-associated infections.7,8 In particular, the antimicrobial activity of Ag ions has been known for centuries. Ag ions exhibit a higher antimicrobial activity compared to other inorganic ions, including Cu and Zn. 9 They bind to the bacterial plasma membrane causing swelling, which increases membrane permeability, leading to cell death. 10 Moreover, these ions reportedly reduce the risk of bacteria-acquired resistance. 11
However, Ag ions may negatively impact surrounding tissues and cells. Hence, striking the right balance between the antimicrobial effectiveness and biocompatibility of Ag ions is necessary. 8 Therefore, when implanting Ag-containing biomaterials, determining the exact Ag content and metabolism to maximize antibacterial effect while minimizing toxicity to the host is crucial.12,13
In this study, we examined the effects of Ag on bone metabolism after implanting Ag-doped β-tricalcium phosphate (β-TCP). Implant-associated infections are usually prevented by incorporating antimicrobial substances into biomaterials through two methods, namely, coating the biomaterial surfaces with antimicrobial substances or grafting antimicrobial substances directly onto the biomaterials.7,14,15 Ag-doped β-TCP is typically categorized as a graft material. Biomaterials grafted with Ag continuously supply Ag ions until they are completely absorbed, reducing the risk of long-term infection.16–20
Ag-doped hydroxyapatite has been shown to effectively function as a bone filler for inflamed bone defects, highlighting its potential for clinical use in infected bone lesions. 21 Similarly, Ag-doped calcium phosphate bone grafts have been reported to eliminate chronic osteomyelitis in animal models, further supporting the antibacterial efficacy of Ag-containing biomaterials in vivo. 22 Additionally, osseointegration of hydroxyapatite coatings doped with silver nanoparticles has been studied in a rabbit model, revealing how Ag incorporation influences bone healing and implant integration. 23
Yoshida et al. 17 demonstrated that Ag ions can be doped into the Ca (4) sites and vacancies within the structure of β-TCP, with a maximum substitution level of 9.09 at%. However, to the best of our knowledge, the optimal Ag concentration of Ag-doped β-TCP in vivo for bone repair and material resorption has not yet been determined. 24 While previous studies have explored the incorporation of Ag into β-TCP for its antimicrobial properties, the optimal Ag concentration for maintaining a balance between antimicrobial efficacy and bone metabolism has not been well established. This study uniquely investigates the in vivo effects of different Ag concentrations (1, 3, and 5 at%) on both bone formation and material resorption, providing new insights into the role of Ag doping in orthopedic applications.17–19 To this end, we prepared 1, 3, and 5 at% Ag-doped β-TCP and examined their antimicrobial activities.
Materials and methods
Porous β-TCP block (control)
β-TCP (control) was prepared using CaHPO4·2H2O (Tomita Pharmaceutical Co., Ltd., Tokushima, Japan) and CaCO3 (Ube Material Industries, Ltd., Yamaguchi, Japan). CaHPO4·2H2O, and CaCO3 were weighed and mixed such that the Ca/P molar ratio was 1.50. The mixture was ground in a pot mill with water using zirconia balls; the process was continued for 24 h. Subsequently, the mix was dried at 80°C to synthesize calcium-deficient hydroxyapatite.25,26 The obtained crystals were calcined at 750°C for 1 h to convert them into β-TCP. Thereafter, an aqueous slurry of β-TCP (50 wt.%) was prepared using ammonium acrylate and alkyl phenyl ether as surfactants, which was then bubbled for 5 min and dried at 20°C for 1 week. Finally, the dried β-TCP was sintered at 1050°C for 1 h to obtain a porous β-TCP block with 75% porosity. The porosity of the products was calculated using the following formula, considering the theoretical density of β-TCP (3.07 g/cm3):
Porous 1, 3, and 5 at% Ag-doped β-TCP blocks
CaHPO4·2H2O, CaCO3, and Ag2O (FUJIFILM Wako Pure Chemical Co., Osaka, Japan) were prepared at a (Ca + 2Ag)/P molar ratio of 1.50.18,27 1, 3, and 5 at% Ag-doped β-TCP were obtained using Ca2+/Ag+ molar ratios of 99/1, 97/3, and 95/5, respectively. The drying and firing conditions were the same as those used for the β-TCP blocks.
X-ray diffraction
Phase analyses of the sintered scaffolds were performed using a diffractometer (Ultima IV, Rigaku, Tokyo, Japan) with CuKα radiation and a Ni filter. The patterns were recorded in the 2θ range of 20–45° at a scanning rate of 0.033°/s.
Antimicrobial tests
The antimicrobial activity was evaluated via the shake flask method using cuboid (10 × 10 × 20 mm) samples of the control and 1, 3, and 5 at% Ag-doped β-TCP. 28 Staphylococcus aureus (ATCC6538P) and Escherichia coli (ATCC8739) were cultured, and test samples at 1.0 × 104 colony-forming units (CFU)/mL were prepared. Two representative bacterial species were selected: S. aureus (a gram-positive pathogen commonly associated with implant-associated infections) and E. coli (a gram-negative model organism). These species were chosen to assess the antimicrobial spectrum of Ag-doped β-TCP against both types of bacterial cell walls, which differ in structure and permeability. While other bacterial species could also be evaluated, these two serve as standard models for initial antimicrobial assessment of biomaterials. Each specimen was immersed in the test sample (5 mL) in a sterilized container and subjected to shaking culture at 35°C for 24 h. A ten-fold dilution was prepared from each culture, and a 0.1 mL aliquot was spread on a standard agar medium and cultured at 35°C for 48 h. The colonies were counted, and the means of replicates were calculated (n = 3).
Animals and surgical procedure
Forty-eight New Zealand white rabbits, aged 4–5 months and weighing 3.0–3.2 kg, were used in this study. Thiopental (20 mg/kg) was intravenously administered to anesthetize the animals; isoflurane (2%) was administered to maintain them in the anesthetized state. The sample size for in vivo experiments was determined based on previous studies evaluating bone formation and biomaterial resorption in rabbit femoral defect models.29–37 A minimum of six rabbits per time point was chosen to ensure statistical power while adhering to ethical guidelines for animal research. A power analysis was performed to confirm that this sample size would detect statistically significant differences in bone formation and material resorption (α = 0.05, power = 80%). The lateral femoral condyles of the rabbits were exposed, and cylindrical defects measuring 4 and 10 mm in diameter and depth, respectively, were drilled at 1 mm anterior to the insertion of the lateral collateral ligaments. The introduced defects were thoroughly rinsed with saline and then filled with gauze for hemostasis. Cylindrical blocks of β-TCP and Ag-doped β-TCP with 75% porosity were pressed to fit into the defects, and the fascia and skin were sutured in layers. Three animals were euthanized 2-, 4-, 8-, and 12-weeks post-surgery (n = 6/time point).
Histological analysis
First, the femoral condyle specimens were fixed in a solution containing 2% paraformaldehyde, 0.75 M lysine, and 0.01 M sodium periodate. Next, the specimens were immersed in a 0.5 M ethylenediaminetetraacetic acid solution for 1 week for decalcification. After they were embedded in glycol methacrylate, 5 μm thick serial sections parallel to the sagittal plane of the femoral condyle center were prepared and subjected to hematoxylin–eosin (HE) and tartrate-resistant acid phosphatase (TRAP) staining.
To evaluate osteoclast activity, TRAP staining was performed. TRAP is a widely recognized marker enzyme for osteoclasts, which are responsible for bone resorption. The number of TRAP-positive cells was quantified to assess the effect of Ag-doped β-TCP on osteoclast differentiation and function. Higher TRAP-positive cell counts indicate increased osteoclast activity, which correlates with greater bone resorption.
Quantification of bone formation and residual materials based on histological analysis was conducted in accordance with methods described in previous studies.29–37 The tissue specimens were observed using an optical microscope (VS120-S5-J, Olympus Co., Tokyo, Japan); newly formed bone and residual materials were quantified via histological analyses of the obtained images (Figure 1). The area in the drilled bone defect (4 mm in diameter) occupied by the newly formed bone and residual materials was measured using image analysis software (WinROOF, Mitani Co., Tokyo, Japan). The newly formed bone and residual materials in the defined circular area (4 mm in diameter) were quantified via color-based thresholding using this software, following a previously reported method. 29 The woven bones and lamellar bones surrounding β-TCP were considered newly formed. 30 The amount of residual Ag-doped β-TCP and the number of TRAP-positive cells in the bone defect were measured to elucidate the mechanism of Ag-doped β-TCP resorption. The amount of bone formation was measured to analyze the amount of Ag-doped β-TCP replaced by new bone.

The areas of newly formed bone and residual β-TCP were measured. To facilitate evaluation, different colors were used to distinguish residual β-TCP (blue) from newly formed bone (green) within the defect. The scale bar is 1 mm.
Scanning electron microscopy (SEM)
The decalcified and glycol methacrylate-embedded polished surfaces of the specimens treated with 1, 3, and 5 at% Ag-doped β-TCP and obtained 2-, 4-, 8-, and 12-weeks post-operation were observed using SEM (SU1510, Hitachi, Ltd., Tokyo, Japan)) at 30,000 eV and an energy-dispersive X-ray spectrometer (Apollo, EDAX Corp., Pleasanton, CA, USA).
Statistical analysis
Continuous variables were expressed as mean ± standard deviation and one-way analysis of variance (ANOVA) was performed with post-hoc multiple comparison using Dunnett’s test. All tests were two-sided and p < 0.05 was considered statistically significant. SPSS version 23.0 (IBM Japan, Ltd., Tokyo, Japan) was used for statistical analysis.
Results
X-ray diffraction
The major X-ray diffraction (XRD) peaks of 1, 3, and 5 at% Ag-doped β-TCP correspond to the characteristic β-TCP peak (JCPDS#09-169) (Figure 2). In the XRD patterns, no peaks corresponding to silver oxide or pure silver were observed, suggesting that the added silver ions were incorporated into the crystal structure of β-TCP. This result is consistent with previous reports 18 in which β-TCP containing silver was synthesized using silver nitrate as a precursor. It also indicates that similar materials were successfully synthesized in this study using silver oxide as the starting material.

XRD spectra of β-TCP (control) and 1, 3, and 5 at% Ag-doped β-TCP. The patterns of Ag-doped β-TCP agreed with those of β-TCP.
Antimicrobial tests
Table 1 presents the mean viable bacteria counts (CFU/sterilized container) ± standard deviations of S. aureus and E. coli. The S. aureus count with 1 at% Ag-doped β-TCP was approximately 1/2600 of the control counts, but their E. coli counts did not differ. Both S. aureus and E. coli counts with 3 and 5 at% Ag-doped β-TCP were fewer than 1/107 of the respective control counts.
Results of antimicrobial tests. The 1 at% Ag-doped β-TCP exhibited antimicrobial activity against S. aureus, while no significant effect was observed against E. coli. The antimicrobial activity of Ag-doped β-TCP increased with higher Ag concentrations. Marked antimicrobial activities of the 3 and 5 at% Ag-doped β-TCP were observed against both S. aureus and E. coli, as evidenced by a substantial reduction in CFU counts.

CFU: colony forming units.
Histological evaluation
Bone formation was observed in all materials at all time points; however, bone formation, material resorption, and TRAP-positive cell count differed among specimen groups.
HE staining
New bone formation in the defect was observed 2 weeks after implantation of the control and 1, 3, and 5 at% Ag-doped β-TCP groups (Figure 3). The amounts of bone formation increased over time up to 8 weeks post-operation for the control and up to 12 weeks post-operation for the Ag-doped β-TCP groups (Figure 4). The amount of bone formation was lower for the Ag-doped β-TCP groups with a higher Ag concentration. Compared with that of the control, the amount of bone formation at 2-, 4-, 8-, and 12-weeks post-operation were not significantly different in the 1 at% Ag-doped β-TCP group. Conversely, the amounts of residual materials decreased over time for the control and 1, 3, and 5 at% Ag-doped β-TCP groups. The amounts of residual materials were not significantly different at 2- and 4-weeks post-operation but were significantly higher at 8- and 12-weeks post-operation for the Ag-doped β-TCP groups compared to the control group (Figure 5).

Photomicrograph of hematoxylin-eosin staining for each experimental group (from left to right: durations 2, 4, 8, and 12 weeks). The scale bar is 1 mm. (a), (b), (c), and (d) show the results for β-TCP at 2, 4, 8, and 12 weeks post-operation, respectively. (e), (f), (g), and (h) he results for 1 at% Ag-doped β- TCP at 2, 4, 8, and 12 weeks post-operation, respectively. (i), (j), (k), and (l) show the results for 3 at% Ag-doped β-TCP at 2, 4, 8, and 12 weeks post-operation, respectively. (m), (n), (o), and (p) show the results for 5 at% Ag-doped β-TCP at 2, 4, 8, and 12 weeks post-operation, respectively.

Bone formation versus time. The amounts of bone formation increased with time up to 12 weeks after 1, 3, and 5 at% Ag-doped β-TCP implantation, and the amounts of bone formation were lower when the concentration of Ag was higher. The amounts of bone formation at 2, 4, 8, and 12 weeks post-operation were lower, but not significantly, for the 1 at% Ag-doped β-TCP group compared with those for the control group.
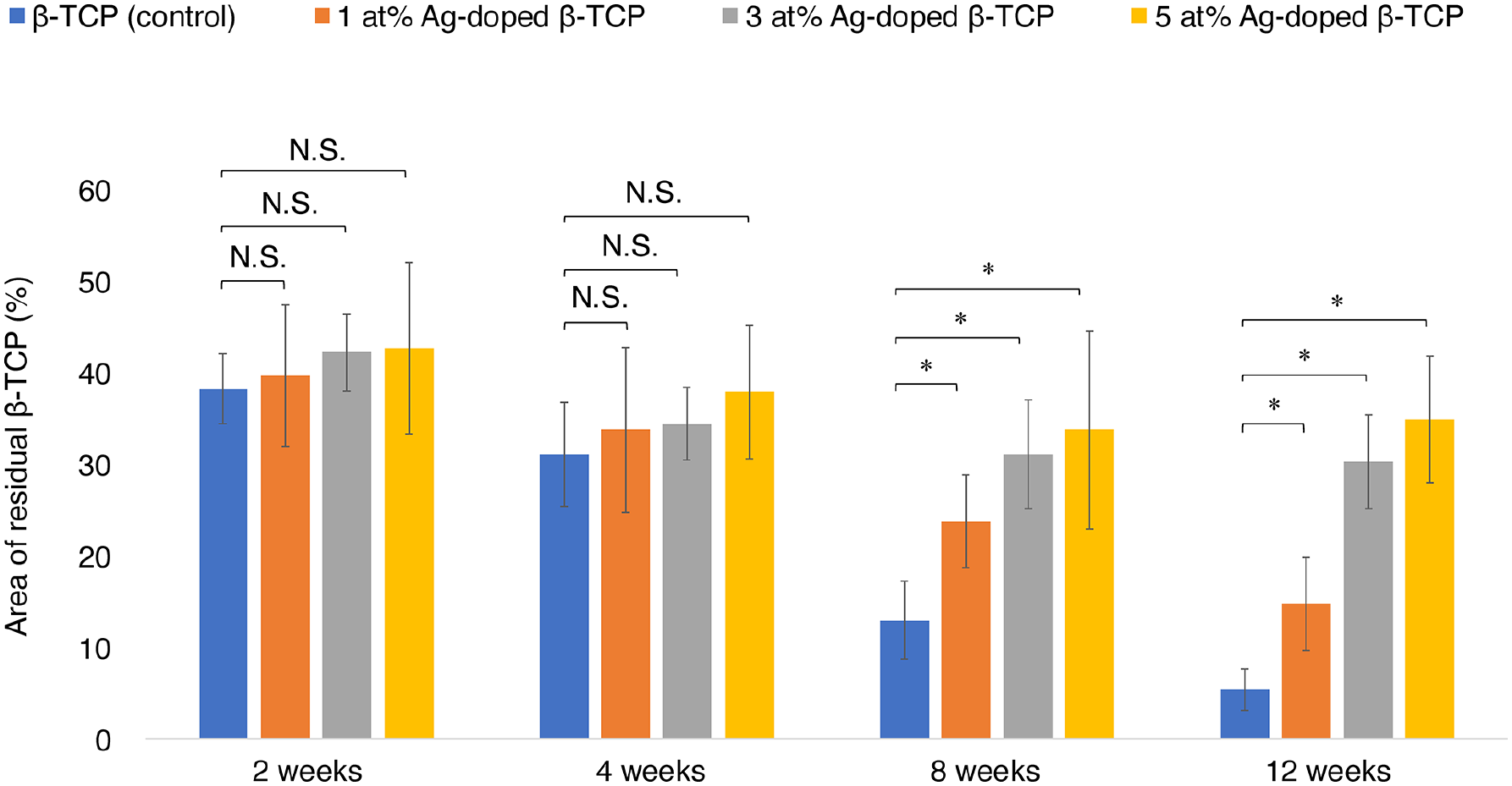
Material degradation versus time. The amounts of residual materials decreased with time after 1, 3, and 5 at% Ag-doped β-TCP implantation, and no significant difference was observed at 2 and 4 weeks post-operation. However, a significantly major difference was observed at 8 and 12 weeks post-operation for the 1, 3, and 5 at% Ag-doped β-TCP compared with that of the control group.
TRAP staining
Numerous TRAP-positive multi-nucleated cells were present on the surfaces of the control and 1, 3, and 5 at% Ag-doped β-TCP groups 2-, 4-, 8-, and 12-weeks post-operation (Figure 6). The number of TRAP-positive cells peaked at 2 weeks post-operation and gradually decreased thereafter over time for all materials (Figure 7). No significant differences at 2-, 4-, 8-, and 12-weeks post-operation were observed between the 1 at% Ag-doped β-TCP and the control groups. The numbers of TRAP-positive cells were significantly lower at 2- and 4-weeks post-operation for the 3 and 5 at% Ag-doped β-TCP groups compared to the control group; however, the differences were not significant at 8- and 12-weeks post-operation.

Photomicrograph of tartrate-resistant acid phosphatase staining (from left to right: durations 2, 4, 8, and 12 weeks). The scale bar is 400 μm. (a), (b), (c), and (d) show the results for β-TCP at 2, 4, 8, and 12 weeks post-operation, respectively. (e), (f), (g), and (h) show the results for 1 at% Ag-doped β-TCP at 2, 4, 8, and 12 weeks post-operation, respectively. (i), (j), (k), and (l) show the results for 3 at% Agdoped β-TCP at 2, 4, 8, and 12 weeks post-operation, respectively. (m), (n), (o), and (p) show the results for 5 at% Ag-doped β-TCP at 2, 4, 8, and 12 weeks post-operation, respectively.

Mean number of TRAP-positive cells. The number of TRAP-positive cells peaked at 2 weeks postoperation and then gradually decreased with time for the 1, 3, and 5 at% Ag-doped β-TCP groups. No significant differences were observed at 2, 4, 8, and 12 weeks post-operation between the 1 at% Ag-doped β-TCP and control groups, but significantly lower counts at 2 and 4 weeks post-operation were observed for the 3 and 5 at% Ag-doped β-TCP groups compared with that of the control group.
Scanning electron microscopy
For the 1, 3, and 5 at% Ag-doped β-TCP groups, after 2, 4, 8, and 12 weeks, Ag accumulated in the bone marrow outside the newly formed bone. Elemental analyses revealed the presence of multi-nuclear cells with a strong Ag peak at 2.98 eV. SEM images of the area with Ag accumulation revealed HE-stained multi-nucleated cells that were TRAP-negative (Figure 8).

High-magnification images of 5 at% Ag-doped β-TCP at 12 weeks post-operation: (a) HE staining, (b) TRAP staining, and (c) SEM image. Arrows (⇧) indicate osteoclasts and arrowheads (Δ) indicate Ag accumulation. Ag accumulation was observed in multi-nucleated cells within the bone marrow outside the newly formed bone. (d) Chemical elements of bright spots (Δ) in the SEM images show a strong Ag peak at 2.98 eV.
Discussion
β-TCP has been previously reported as an excellent bone graft material for bone replacement, which is also confirmed in this study.29–37 The feasibility of using Ag-doped β-TCP as an antimicrobial biomaterial was examined. The effect of doped Ag on the metabolism of β-TCP was studied using 1, 3, and 5 at% Ag-doped β-TCP. The optimal concentration of doped Ag with respect to favorable bone formation rate, material resorption rate, and antimicrobial activity was verified.
The results obtained in this study confirmed the antimicrobial effect of Ag-doped β-TCP against S. aureus even at a concentration of 1 at%. The antimicrobial effect was stronger when the concentration of doped Ag was higher; 3 and 5 at% Ag-doped β-TCP exhibited marked antimicrobial activity against S. aureus and E. coli. Previous studies have demonstrated that Ag ions bond to bacterial plasma membranes, increasing permeability and leading to cell death. Additionally, Ag ions interact with thiol groups of enzymes, disrupting cellular functions and inducing oxidative stress.38–40 While these mechanisms are well established in the literature, our study focuses on evaluating the antimicrobial effects of Ag-doped β-TCP rather than directly investigating these molecular interactions. In a previous study, almost all S. aureus cells were killed owing to the antimicrobial effect of Ag in bacterial suspensions containing 45 ± 6 ppb Ag, whereas a small proportion of E. coli cells survived. 41 The results obtained herein also suggest that the antimicrobial activity of 1 at% Ag-doped β-TCP was more pronounced against the gram-positive S. aureus that has a weak cell wall structure. 24 Loh et al. 42 reported that no resistant strains were identified after exposing 876 S. aureus strains to Ag for 42 days. Conversely, the outer membrane of gram-negative bacteria has tightly packed lipopolysaccharide molecules, forming a stronger barrier. 43
The amount of newly formed bone increased over time in the 1, 3, and 5 at% Ag-doped β-TCP specimens. The amount of bone formation at 2-, 4-, 8-, and 12-weeks post-operation were not significantly different for the 1 at% Ag-doped β-TCP group compared to the control. The amount of residual materials in the Ag-doped β-TCP groups was also not significantly different after 4 weeks post-operation but was significantly higher at 8- and 12-weeks post-operation compared to the control. The decrease in TRAP-positive cells over time suggests a natural progression of bone remodeling. Osteoclast-mediated resorption of β-TCP is an essential process for material degradation and subsequent bone formation. 37 However, in the 3 and 5 at% Ag-doped β-TCP groups, the lower TRAP-positive cell counts at early time points indicate that silver may suppress osteoclast activity, potentially leading to slower material resorption. This is in line with previous studies suggesting that Ag ions have cytotoxic effects on osteoclasts at higher concentrations. While this effect may help maintain implant stability in the short term, it could also delay the turnover of β-TCP, impacting long-term bone remodeling. Therefore, optimizing the Ag concentration is essential to balance its antimicrobial benefits with its potential effects on bone metabolism. The numbers of TRAP-positive cells at 2-, 4-, 8-, and 12-weeks post-operation were not significantly different between the 1 at% Ag-doped β-TCP and control groups, but the numbers significantly decreased at 2- and 4-weeks post-operation for the 3 and 5 at% Ag-doped β-TCP groups. Thus, the results of this study suggest that doped Ag in β-TCP had a greater effect on material resorption inhibition than on osteogenesis inhibition. With increase in time after implantation, the difference in bone formation between the Ag-doped β-TCP and control groups decreased, whereas the difference in residual material increased. The observed results are consistent with previous findings on the concentration dependence of Ag cytotoxicity and the greater degree of effect on osteoclasts than on osteoblasts. 44
Although Ag-doped β-TCP demonstrated significant antimicrobial effects, the fate of released Ag ions in vivo remains a crucial factor in assessing its long-term safety. One potential concern is the risk of Argyria, a condition characterized by permanent skin discoloration due to silver accumulation in tissues. 45 Argyria has been primarily reported in cases of chronic systemic Ag exposure, such as prolonged ingestion of colloidal silver supplements or occupational exposure. 46 However, local Ag deposition from implants remains underexplored. Studies on Ag-containing orthopedic implants have suggested that silver may accumulate in surrounding tissues, 47 but no definitive conclusions have been drawn regarding its long-term effects.
In this study, SEM observations revealed no Ag accumulation within newly formed bone; instead, Ag deposits were detected in the bone marrow. This suggests that Ag released from Ag-doped β-TCP remains localized within the bone tissue for a certain period. The long-term fate of this Ag in the bone marrow is beyond the scope of this study and requires further investigation. However, its localization within the marrow may reduce the amount of silver ions reaching subcutaneous tissues, potentially lowering the risk of Argyria.
While the localized release of Ag from β-TCP is expected to be significantly lower than systemic exposure levels associated with Argyria, further studies should evaluate Ag biodistribution and potential systemic accumulation over prolonged implantation periods. Additionally, while the antimicrobial properties of Ag-doped β-TCP offer promising protection against implant-associated infections, optimizing Ag concentration remains critical to minimizing cytotoxic effects.
The primary β-TCP resorption process was biological resorption by osteoclasts, and the amount of chemically dissolved Ag was small. Therefore, the degradation of osteoclasts was presumed to release a large number of Ag ions, which resulted in a strong antimicrobial effect in vivo, although 1 at% Ag-doped β-TCP was not effective against E. coli in this study. Furthermore, 3 at% Ag-doped β-TCP exhibited a significant antibacterial effect against S. aureus and E. coli. However, bone formation and material resorption were impaired when the Ag concentration in Ag-doped β-TCP increased. In particular, in this study, after the implantation of 5 at% Ag-doped β-TCP, material resorption did not progress, and the amount of bone formation was low. Thus, Ag concentration of 1 at% in Ag-doped β-TCP was desirable from the perspective of bone formation and material resorption whereas Ag concentration of 3 at% or higher was desirable from the perspective of the antimicrobial effect.
The limitations of this study are as follows. In this study, no barrier membrane was used, as our focus was to evaluate the biological response of Ag-doped β-TCP in a bone defect model without additional external factors. Guided Bone Regeneration (GBR) membranes are commonly used to prevent soft tissue infiltration and enhance bone formation; however, they may also influence material resorption and remodeling. 48 To assess the direct effect of Ag doping, we chose to conduct this study without a GBR membrane. However, we recognize that GBR membranes could offer clinical advantages by stabilizing the graft material and promoting guided bone regeneration. Future research will explore the combined use of GBR membranes with Ag-doped β-TCP to further understand their impact on bone healing and material resorption. The number of Ag ions eluted from Ag-doped β-TCP in distilled water was below the detection limit of 10 ppm. However, this elution test is based on in vitro mechanisms, and the Ag ion release mechanism by Ag-doped β-TCP in vivo has not been clearly demonstrated. In this study, initial micro-CT data were obtained; however, quantitative analysis is still in progress, and thus, these results have not been included in the current manuscript. Micro-CT is a valuable technique for three-dimensional evaluation of bone formation and material resorption. In future research, we plan to conduct a detailed 3D analysis using micro-CT and compare the findings more explicitly with histological evaluations. This approach will allow for a clearer and more comprehensive assessment of the effects of Ag-doped β-TCP on bone regeneration and material resorption. The main process of β-TCP absorption in the body has been reported to be a cell-mediated biodegradation mechanism by osteoclasts.30,34 SEM observations of the implanted Ag-doped β-TCP confirmed the phenomenon of Ag deposition and uptake by osteoclasts. This observation may best capture the dynamics of Ag after implantation of Ag-doped β-TCP.
Conclusions
The findings of this study can be summarized as follows:
Bone formation increased over time in all groups, but higher Ag concentrations were associated with reduced bone formation.
Residual material decreased progressively, but degradation was slower in higher Ag-doped groups, suggesting that Ag influences material resorption kinetics.
TRAP-positive cell counts declined over time, with a more pronounced reduction observed in the higher Ag-doped groups, indicating a potential inhibitory effect of Ag on osteoclast-mediated resorption.
The presence of Ag-doped β-TCP influenced both bone metabolism and material resorption, demonstrating a dose-dependent effect on osteogenesis and osteoclast activity. Specifically, increased Ag concentration was associated with a gradual reduction in bone formation and a slower resorption rate of β-TCP, likely due to the inhibitory effect on osteoclast function. While the antimicrobial properties of Ag are beneficial, its impact on bone remodeling must be carefully considered to optimize its concentration for clinical applications.
Footnotes
Ethical considerations
The animal experiments performed in this study were approved by the Ethics Committee of the Jikei University School of Medicine (No. 2019-012) and were performed in compliance with the university regulations on animal experiments and in accordance with the ARRIVE 2.0 guidelines.
Author contributions
T.T. designed the study, conceptualized the main ideas. S.A., T.T., H.K., and N.I. collected the data. H.K. aided in interpreting the results and worked on the manuscript. M.S. supervised the project. S.A. wrote the manuscript with support from T.T. and H.K. All authors discussed the results and commented on the manuscript. All authors approved the final version.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The materials used in this study were provided by Olympus Terumo Biomaterials Co. (Tokyo, Japan). The authors declare that they have no competing financial interests. No benefit of any kind has been or will be received directly or indirectly by any of the authors.
Data availability statement
The data supporting the findings of this study are available within the article.

