Abstract
Chitosan-derived hydrogels offer a highly tunable scaffold for orchestrating the multifaceted process of wound repair, yet their full potential hinges on an integrated understanding of biochemical and biophysical cues. Here, we provide a concise yet comprehensive analysis of design parameters that govern cell-matrix interactions, focusing on polymer backbone modification, crosslinking density, and incorporation of bioactive ligands. We demonstrate how precise control of viscoelastic properties viscoelastic properties, including stiffness and stress relaxation, achieved through reversible covalent bonds and supramolecular assemblies regulates YAP/TAZ nuclear localization and downstream PI3K/Akt and MAPK/ERK signaling to differentially promote coordinated keratinocyte migration, fibroblast proliferation, and endothelial angiogenesis. Functionalization with RGD peptides and heparin-binding motifs further amplifies receptor-mediated adhesion and growth factor sequestration, creating a pro-regenerative microenvironment. We critically appraise the kinetic profiles of growth factor release and immunomodulatory payloads, underscoring the necessity of spatiotemporal precision in smart hydrogel systems and summarize preclinical and clinical evidence demonstrating accelerated wound closure in diabetic and burn models. Finally, we identify pivotal challenges including the quantitative decoupling of mechanical versus molecular influences in vivo, long-term biocompatibility of modified chitosan derivatives, and translation of high-throughput screening data into clinical contexts and propose a roadmap leveraging single-cell transcriptomics and advanced imaging, and AI-driven modeling to overcome translational challenges. This synthesis delivers actionable guidelines for the development of next-generation, personalized wound therapies.
Highlights
○ Precise modulation of hydrogel stiffness and stress relaxation directs YAP/TAZ localization and downstream PI3K/Akt and MAPK/ERK pathways to optimize keratinocyte migration and fibroblast proliferation.
○ Conjugation of RGD peptides and heparin-binding domains amplifies integrin-mediated adhesion and growth factor sequestration, establishing a pro-angiogenic microenvironment.
○ Stimuli-responsive linkages and nanoparticle incorporation enable on-demand delivery of cytokines and exosomes, balancing matrix support with targeted biochemical cues.
○ Integration of high-throughput material screening with single-cell transcriptomics and advanced imaging is proposed to decouple mechanical versus molecular effects and expedite personalized wound therapy development.
Introduction
Effective wound healing requires a precisely orchestrated sequence of cellular events, among which directed cell migration is indispensable. Keratinocytes, fibroblasts and endothelial cells must collectively navigate through a provisional matrix of fibrin and fibronectin to re-epithelialize the wound bed and rebuild dermal and vascular structures. Disruptions in migratory signaling that mediated by growth factors such as EGF, PDGF and matrix metalloproteinases (MMPs) can delay closure and prolong inflammation, underscoring migration as a rate-limiting step in tissue repair.1,2
Chronic wounds including diabetic foot ulcers, venous leg ulcers and pressure injuries illustrate the clinical consequences of impaired migration. Up to 25%–50% of hospitalized patients develop pressure ulcers, and diabetic wounds often display 50%–75% reduced fibroblast and keratinocyte motility relative to acute wounds.3,4 These deficits perpetuate a non-healing state characterized by low-grade inflammation, poor angiogenesis and extracellular matrix (ECM) dysregulation. Therapeutic strategies that restore or enhance cell motility, such as chemokine delivery or engineered dressings, have therefore garnered intense interest as means to re-ignite stalled healing.
Hydrogel dressings emulate key aspects of native ECM by providing a hydrated, porous network that absorbs exudate, permits gas and nutrient exchange, and maintains a moist environment conducive to cell ingrowth.2,5 Among polymeric hydrogels, those derived from chitosan that is a deacetylated polysaccharide obtained from chitin are especially promising. Chitosan hydrogels combine biodegradability and biocompatibility with intrinsic hemostatic and antimicrobial properties, and their primary amino groups facilitate facile chemical crosslinking into three-dimensional networks.6,7 By binding endogenous growth factors and modulating immune cell behavior, chitosan gels not only serve as passive scaffolds but also actively influence cellular responses. In vitro studies show that chitosan substrates enhance fibroblast proliferation and chemotaxis, while in vivo chitosan dressings accelerate re-epithelialization and neovascularization in skin wound models. 8
Recent advances have yielded smart chitosan hydrogels capable of dynamic, stimuli-responsive behavior. Incorporation of pH-sensitive Schiff-base linkages or thermo-responsive additives enables self-healing and on-demand release of encapsulated therapeutics in response to wound-specific cues. For example, chitosan hydrogels embedded with thermo-sensitive polymers undergo sol-gel transitions at body temperature, ensuring minimally invasive delivery and in situ gelation. 9 Similarly, pH-responsive systems exploit the acidic microenvironment of chronic wounds to trigger drug release and modulate local inflammation. These smart materials thus combine mechanical adaptability with controlled biochemical signaling to better support cell migration and tissue regeneration. 10
Despite the proliferation of chitosan hydrogel formulations, critical gaps remain in our mechanistic understanding of how these materials modulate cell motility at the molecular level. While many studies report improved healing outcomes, few dissect the interplay between chitosan’s physicochemical properties, such as stiffness, viscoelasticity and charge density and key signaling pathways (e.g. integrin-FAK, PI3K/Akt, MAPK/ERK, Wnt/β-catenin) that govern cytoskeletal dynamics and directional migration. Moreover, the relative contributions of matrix mechanics versus growth factor presentation in directing endothelial sprouting and keratinocyte sheet movement have not been systematically compared.
While numerous reviews have summarized the general applications of chitosan-based hydrogels in wound healing, focusing on their antimicrobial properties, biocompatibility, and drug delivery capabilities, 7 this review distinguishes itself by emphasizing the molecular modulation of cell migration through specific signaling pathways, such as YAP/TAZ-mediated mechano transduction, PI3K/Akt, and MAPK/ERK cascades. Unlike prior works that primarily address bulk material properties or clinical outcomes, we integrate recent advances in smart, stimuli-responsive systems with emerging tools like single-cell transcriptomics and advanced imaging to quantitatively decouple mechanical and biochemical cues. This approach offers distinct advantages, including actionable guidelines for designing personalized, pro-regenerative hydrogels that optimize spatiotemporal control of growth factor release and immunomodulation, potentially reducing chronic wound healing times by enhancing targeted cell motility and angiogenesis in vivo.11,12
In this review, we critically examine recent literature on chitosan-based hydrogels through the lens of cell migration biology. We integrate findings on chitosan’s dual roles as a biochemical reservoir and biophysical cue, explore how engineered modifications influence receptor-mediated signaling, and highlight emerging smart platforms that actively orchestrate the healing cascade. By linking material design parameters to molecular- and cellular-level outcomes, this work aims to guide the rational development of next-generation chitosan hydrogels for accelerated wound regeneration.
Wound healing and the role of cell migration
Wound healing is a complex process that restores skin function by coordinating cell differentiation, proliferation, migration, and Extracellular Matrix (ECM) remodeling. 13 It progresses through four overlapping stages: hemostasis, inflammation, proliferation, and remodeling. 14 Cell migration, including polarization, protrusion, attachment to neighboring cells and the ECM, as well as retraction, is required for appropriate tissue repair and homeostasis. 15 According to Brückner et al. study, cell migration plays a crucial role in various physiological processes ranging from embryonic development and immune response to wound healing and cancer metastasis. 16 During this procedure, keratinocytes collectively migrate to seal wounds. 17 Improper migration might result in problems, such as chronic, non-healing wounds. 15
Phases of wound healing
Skin healing involves successive and overlapping phases hemostasis, inflammation, proliferation, and remodeling mediated by inflammatory cells, growth factors, and cellular and extracellular components to ensure effective repair and regeneration. Following tissue injury, the coagulation pathway forms a fibrin matrix, which promotes cell migration. Hemostasis involves blood vessel constriction, platelet aggregation, and formation of a fibrin network. During inflammation, white blood cells release mediators, cytokines, platelet-derived growth factor (PDGF), and transforming growth factor-beta (TGF-β) to stimulate fibroblast activity for migration, proliferation, and collagen synthesis, while neutrophils and macrophages remove tissue debris and pathogens. The proliferation phase includes the migration of keratinocytes and fibroblasts, ECM production to form granulation tissue, MMP-mediated re-epithelialization via polarity changes, and angiogenesis. 13 In the remodeling phase, type III collagen is replaced by type I collagen, myofibroblasts produce matrix metalloproteinases (MMPs), and macrophages clear remaining tissue debris. 18 Growth factors such as epidermal growth factor (EGF), fibroblast growth factor (FGF), PDGF, and vascular endothelial growth factor (VEGF) promote healing through receptor phosphorylation and angiogenesis. 19 The fibrin clot is replaced by proteoglycans which assist in collagen maturation as well as fibronectin and hyaluronan. 20
Cell migration in wound repair
Various types of cells contribute to effective wound healing, especially keratinocytes, endothelial cells, and fibroblasts, which migrate to the wound site. Their movement is regulated by molecular signals, cytokines, and growth factors efficient repair. 21
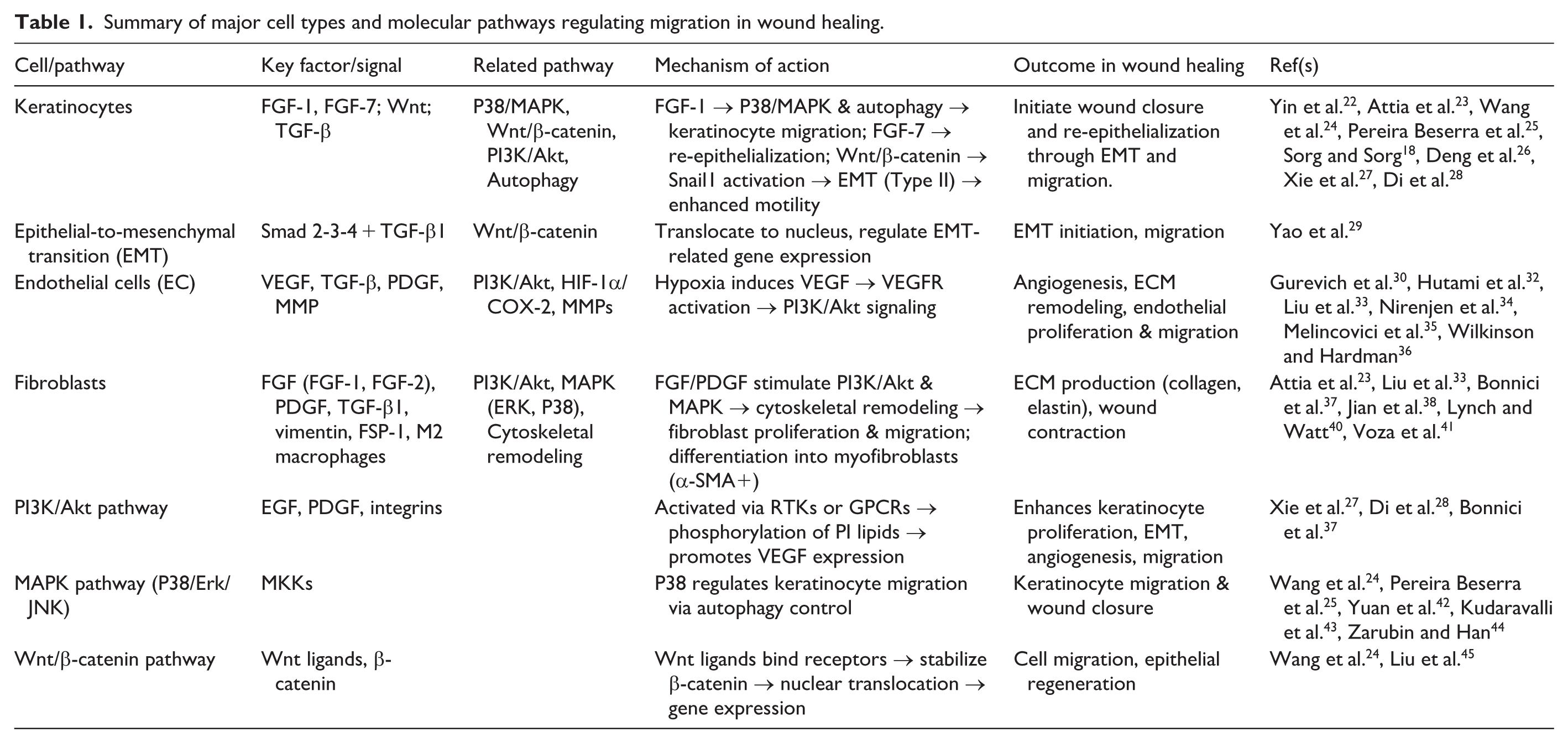
Keratinocyte migration. The initial cellular migration during wound healing is performed by keratinocytes. 22 FGF, through its various isoforms, plays a crucial role in cell motility. Specifically, FGF-1 initiates keratinocyte movement, 23 a process regulated by the P38/MAPK signaling pathway and also influenced by autophagy.24,25 Keratinocytes undergo a phenotypic change known as epithelial-to-mesenchymal transition (EMT), particularly type II EMT, which occurs during tissue repair. 18 FGF-7 selectively targets keratinocytes to drive re-epithelialization. 23 The Wnt/β-catenin pathway also regulates keratinocyte migration and initiates EMT by activating Snail1(Figure 1).26 –28

Molecular pathways modulated by chitosan to regulate cell migration. CH enhances multiple signaling cascades involved in cell migration.
EMT process. TGF-β, released from activated platelets and macrophages, regulates both cell migration and ECM production. Among its isoforms, TGF-β1 is a well-known inducer of EMT. In this pathway, TGF-β binds to type I and II receptors, forming a complex that phosphorylates Smad2 and Smad3. These then associate with Smad4, migrate to the nucleus, and, together with other transcription factors, regulate the expression of EMT-related genes (Figure 1). 29
Endothelial Cell (EC) migration. As part of the healing response, endothelial cells begin to proliferate and migrate to the injury site, initiating angiogenesis that is a hallmark of the proliferative phase. 30 Macrophages support this process by releasing VEGF and TGF-β and enhancing endothelial migration. Hypoxia, the main trigger for angiogenesis, induces VEGF, 31 a key chemotactic factor that acts through VEGFR-1/2 and is regulated by HIF-1α and COX-2. 32 These events activate PI3K/Akt and VEGF signaling pathways, further promoting endothelial migration. 33 PI3K enhances VEGF and PDGF release from platelets under hypoxic conditions, promoting angiogenesis, tissue regeneration, and ECM remodeling. 34 This process is driven by the secretion of MMP-2, MMP-3, and MMP-9, which degrade the ECM and enhance cell migration, 35 ultimately leading to increased endothelial proliferation and migration. This rapid endothelial response is critical for transitioning from the inflammatory to the proliferative phase and thereby contributes to effective wound healing. 36
Fibroblast migration. Fibroblast migration is vital for tissue repair. After injury, fibroblasts are attracted by growth factors such as FGF, PDGF, and TGF-β. FGF isoforms enhance migration through the PI3K/Akt and VEGF pathways. 33 FGF-1 increases fibroblast migration, and PI3K/AKT signaling further supports their proliferation and movement. 37 FGF-2 promotes fibroblast migration by remodeling the cytoskeleton and upregulating vimentin and fibroblast-specific protein-1 (FSP-1). PDGF also activates PDGFR, leading to MAPK-mediated fibroblast migration.38,39 These fibroblasts produce key ECM components such as collagen and elastin. 40 Under stimulation from TGF-β1, PDGF, and M2 macrophages, fibroblasts differentiate into myofibroblasts. As noted by Reinke et al. (2012), by the end of the proliferative phase, fibroblasts transform into contractile myofibroblasts that play a role in wound contraction. 41 These cells express α-SMA and actively pull the wound edges together.22,41
PI3K/Akt pathway. PI3K, a lipid kinase, phosphorylates the inositol ring of phosphatidylinositol lipids in the plasma membrane and is activated by extracellular signals through two main mechanisms: interaction with phosphorylated tyrosine residues or G-protein-coupled receptors. Growth factors such as EGF and PDGF facilitate activation via receptor tyrosine kinases, improving integrin activation and endothelial migration. This pathway also enhances keratinocyte proliferation, EMT, and VEGF expression for angiogenesis and motility (Figure 1).28,37
MAPK pathway. The MAPK signaling cascade regulates migration through its subfamilies: Erk1/2, P38, and JNK/SAPK, with P38 playing a prominent role. 42 P38 MAPKs are serine/threonine-specific protein kinases 43 that are activated by MAP kinase kinases (MKKs). 44 P38/MAPK activation regulates keratinocyte migration by controlling autophagy, highlighting its importance in wound healing.24,25
Wnt/β-catenin Pathway. Meanwhile, the Wnt/β-catenin pathway is crucial for cell migration and epithelial regeneration. 24 This pathway is initiated by the binding of extracellular Wnt ligands to membrane receptors via autocrine/paracrine mechanisms. This process stabilizes β-catenin, allowing its nuclear translocation and the expression of migration-related genes (Table 1). 45
Summary of major cell types and molecular pathways regulating migration in wound healing.
Challenges in chronic wounds
Chronic wounds, such as diabetic foot, venous, and pressure ulcers, stem from impaired keratinocyte migration, leading to cellular senescence, prolonged inflammation, disrupted angiogenesis, and inadequate blood flow and nutrient delivery. 46 This hampers healing, accompanied by infections and inflammation. 47 Extended treatment and complicated procedure, causing physical discomfort and socio-economic issues, necessitate advancing alternative approaches to successfully handle these challenges. 48
Impaired keratinocyte migration is central to chronic wounds. 49 Zhou et al. revealed functional deficits in migration of Schwann Cells (SCs) of diabetic mice, causing reduced re-epithelialization, granulation tissue, vascularization, innervation, SC migration, and fibroblast migration compared to control SCs. 50 Overproduction of proinflammatory cytokines, MMPs (e.g. MMP-9), and Reactive Oxygen Species (ROS) can disrupt angiogenesis, ECM remodeling, and tissue repair. Additionally, reduced levels of VEGF, TGF-β1, and EGF contribute to these disruptions. Impaired fibroblast migration affects type III collagen formation and angiogenesis via VEGF and angiopoietin-1. 51 Blood vessel alterations exacerbate infection risks and epithelialization impairments through keratinocyte migration disruption. 52
Chitosan hydrogels: Properties and mechanisms
The semi-synthetic biopolymer chitosan, originating from crustacean chitin, shows varied chemical properties, having its broad applications in environmental, industrial, and medical fields. In general, chitosan is prepared by treating chitin with an alkaline solution, which converts the acetamido groups into free amino groups and thus increases possible chemical reactivity and functional adaptability. Chitosan is a copolymer with β-(1→4) linkages between the units of D-glucosamine and N-acetyl-D-glucosamine. Depending on the degree of conversion, it will display different levels of solubility and chemical reactivity. The presence of amine groups facilitates numerous chemical modifications that are pertinent to its applications in biomedicine and the environment, thereby making chitosan an essential material for biomedical uses. It has been demonstrated in vitro that chitosan-based materials show little cytotoxicity. Smith et al. found that the cytotoxicity of chitosan hydrogels was very low, being comparable to that exhibited by the usual buffers (e.g. Dulbecco’s Modified Eagle Medium (DMEM) or Phosphate Buffered Saline (PBS)) across different cell types. This property, combined with structural adaptability, makes chitosan suitable for tissue regeneration and wound healing.
Hydrogel formation
The creation of chitosan-based hydrogels employs a combination of three crosslinking methods, which produce specific material properties that scientists can adjust for biomedical use, including tissue engineering, drug delivery, and wound healing applications. Under covalent crosslinking conditions, chitosan chains bond together through stable chemical connections in the presence of glutaraldehyde as the crosslinking agent. The method shows specific promise in situations that demand structures to endure long periods of time as required for load-bearing implants. 53 The method of photo-crosslinking depends on using light-activated initiators, which include UV or visible light, to establish specific network patterns. The approach enables researchers to design different stiffness levels and geometric shapes, which helps produce intricate 3D scaffolds with enhanced mechanical properties, as demonstrated in injectable chitosan hydrogels for tissue regeneration.54,55 The method of ionic crosslinking utilizes multivalent anions, including tripolyphosphate (TPP), to establish electrostatic bonds that can be reversed. These hydrogels develop rapidly at low temperatures while showing sensitivity to pH variations and ionic changes, which provides benefits for 3D bioprinting systems and pH-controlled drug delivery systems.54,56 The method of dual-crosslinking is the combination of covalent and photochemical reactions with physical ionic bonds to create hydrogels which demonstrate higher mechanical strength, improved elastic properties, and adjustable biodegradation. Scientists use a blend of these approaches to create products that deliver customizable attributes for optimized biomedical applications (Table 2). 57
comparison of crosslinking methods and biomedical applications of chitosan hydrogels.

Biological properties
Chitosan is an ideal material for hydrogel production due to its excellent biodegradability, non-toxicity, and natural antimicrobial activity. 58 Its strong non-toxicity stems from the stable glycosaminoglycan backbone, ensuring compatibility with cells and tissues. As a naturally derived polysaccharide, it undergoes enzymatic degradation under physiological conditions. Suba Rajinikanth B et al. reported that enzymes like lysozyme and chitinase cleave its molecular chains, enabling efficient resorption in the body.59,60 This makes chitosan especially valuable in applications where gradual integration into regenerating tissue is required. 61
Chitosan, due to its excellent biodegradability, non-toxicity, and natural antimicrobial activity, is an ideal material for hydrogel production. 58 Its strong non-toxicity is attributed to its stable glycosaminoglycan backbone, which ensures compatibility with cells and tissues. This material also affects the rate of enzymatic degradation. Enzymatic degradation assessment using lysozyme demonstrates the biodegradability of chitosan as a naturally derived polysaccharide under physiological conditions. Rajinikanth et al., reported that enzymes such as lysozyme and chitinase break down its molecular chains, enabling efficient absorption in the body. 59 This makes chitosan particularly valuable in applications where gradual integration into regenerating tissue is required. 61 Recent studies in applied biomaterials highlight the tunable degradation rates of chitosan-based composites, making them suitable for long-term wound applications62,63
Chitosan and its derivatives, due to differences in their cell wall structures, exhibit antimicrobial activity against different bacteria. This effect relates to chitosan’s positive charges, enabling strong bacterial surface interactions. According to Dazhong Yan et al., chitosan’s positive charges in acidic conditions enable strong binding to bacterial surfaces, causing membrane disruption, ion displacement, and cell death. 64 This way chitosan-based hydrogels exert potent antimicrobial effects by disrupting bacterial membranes and preventing infection. 58
Chitosan supports cellular functions by interacting with integrins, essential receptors for adhesion and signaling. Mochizuki et al. showed that chitosan combined with ECM-derived peptides enhances fibroblast adhesion and spreading via αvβ3 and β1 integrins, initiating focal adhesion (FA) complex formation. 65
Molecular modulation of cell migration via chitosan hydrogels
Chitosan enhances therapeutic effects via PI3K/Akt, Wnt, and MAPK pathways, promoting cell adhesion, proliferation, and migration. 11 Its mechanical characteristics guide migration, FA, and cytoskeletal formation through Yes-associated protein (YAP), Transcriptional coactivator with PDZ-binding motif nuclear (TAZ), and FAK. Functional modifications, bioactive agents, and biomaterial integration enable precise cellular interactions, further influencing migration. 66
Mechanotransduction and chitosan hydrogels
Mechano-transduction transforms mechanical forces into biochemical signals within cells. FAs and the cytoskeleton are essential mechanical components in directing biomaterial-based mechanical regulation through mechano-transduction pathways. In this context, chitosan scaffolds, with properties like tensile strength, viscoelasticity, and stiffness, regulate cell adhesion and cytoskeleton. 11
Mechanical signals and cell motility are regulated by chitosan’s stiffness and elasticity during wound healing. Stiffer hydrogels increase fibroblast migration, platelet spreading, and endothelial activity. Increased stiffness promotes YAP/TAZ localization, driving cell proliferation mechano sensitively, and may create a favorable environment for cell motion. Consequently, these proteins may also activate plasminogen activator urokinase (PLAU), and TGF-β receptor Ⅲ, further enhancing keratinocyte proliferation. 66 Specifically, hydrogel stiffness and stress relaxation play pivotal roles in directing YAP/TAZ-mediated signaling toward downstream pathways such as PI3K/Akt and MAPK/ERK. In matrices with higher stiffness and slower stress relaxation, cells sense sustained mechanical forces through integrins and focal adhesions, leading to YAP/TAZ nuclear translocation. This activation enhances PI3K/Akt signaling by promoting Akt phosphorylation, which supports cell survival, anti-apoptosis, and directed migration via cytoskeletal reorganization. Simultaneously, YAP/TAZ stimulates MAPK/ERK cascades through interactions with upstream regulators like Ras and RAF, resulting in transcriptional activation of genes involved in proliferation (e.g. c-Myc) and motility. These mechanosensitive responses are critical for optimizing keratinocyte and fibroblast migration in wound healing, as evidenced by studies showing that tunable hydrogel mechanics can rescue impaired migration in chronic wound models.66 –68 Viscosity impacts mechano transduction by improving adhesion, migration, proliferation, and survival via adhesion-ligand binding and myosin contraction. Elevated viscoelasticity increases integrin β1 and vinculin expression, influencing cell proliferation, reducing apoptosis, and modulating collective migration. 69
Matrix stiffness affects cardiac fibroblasts (CFs) behavior through mechano transduction, involving FAs and integrins that link the ECM to the cytoskeleton and activate FAK and Rho-associated protein kinase (ROCK). Soft hydrogels limit FA maturation, whereas stiffer hydrogels boost tyrosine phosphorylation of FAK and paxillin, fibroblast spreading, and cytoskeletal organization. Substrate stiffness induces novel cytoskeletal structures, activating adhesion molecules (e.g. integrin αvβ3, FAK, paxillin, RhoA), facilitating cytoskeletal reorganization and ECM-actomyosin interactions (Figure 2). 70

Hydrogel stiffness and stress relaxation regulate mechano transduction and cell migration.
Viscoelastic tuning of chitosan hydrogels differentially modulates the behavior of key cell types involved in wound healing through YAP/TAZ-mediated activation of PI3K/Akt and MAPK/ERK signaling. In keratinocytes, increased hydrogel stiffness (e.g. 10–50 kPa) promotes YAP/TAZ nuclear localization, enhancing PI3K/Akt signaling to drive actin polymerization and lamellipodia formation, thereby accelerating collective migration and re-epithelialization; however, excessive stiffness (>100 kPa) may inhibit migration via over-activation of MAPK/ERK, leading to hyperproliferation without directed motility.71,72 For fibroblasts, moderate viscoelasticity with stress relaxation (e.g. loss modulus ~0.5–2 kPa) activates YAP/TAZ to upregulate PI3K/Akt and MAPK/ERK pathways, fostering myofibroblast differentiation, ECM deposition, and chemotactic migration, while low stiffness (<5 kPa) reduces YAP/TAZ activity and impairs proliferation.66,73 In endothelial cells, dynamic viscoelastic cues (e.g. shear stress-relaxing matrices) induce YAP/TAZ-dependent PI3K/Akt signaling for tip cell extension and MAPK/ERK for sprout stabilization, promoting angiogenesis; stiff, non-relaxing hydrogels can disrupt this balance, leading to aberrant vessel formation.74,75 These cell-specific responses highlight the need for tailored hydrogel designs to optimize coordinated wound repair.
Bioactive modifications
Chitosan hydrogels (CS-HG) function as ideal scaffolds for sustained growth factor delivery, enhancing cell migration. 76 FGF signaling is involved in collective cell migration. Xu et al. (2017) provided a novel mechanism in which FGF and Wnt signaling were tightly coupled to maintain fibroblast cell migration through β-catenin. 77 Moreover, CS-HG loaded with FGF-2 provide a controlled-release matrix, creating a chemotaxis gradient that accelerates fibroblast motility.5,58,78 PDGF is also crucial for fibroblast migration. 38 Piran et al. (2018) used chitosan nanoparticles to control PDGF-BB release, promoting its effects. 79 By binding to the PDGFR, it triggers dimerization, autophosphorylation, and tyrosine kinase activation, and MAPK-based fibroblast migration. 38 VEGF, another growth factor, has a substantial impact on the migration of endothelial cells. This process is enabled by the secretion of MMP-2, MMP-3, and MMP-9, which decompose the ECM and boost cell migration. 35 Similarly, CS-HG loaded with VEGF generates localized signals to amplify endothelial migration and neo vessel formation.80,81 Together, these functionalized CS-HG systems focus their bioactivity on promoting directional keratinocyte, fibroblast, and endothelial migration, expediting re-epithelialization and angiogenesis, key processes in wound healing and closure. 82
The functionalization of hydrogels with bioactive materials provides favorable results in wound healing by affecting cell migration. 83 Capillary endothelial cell migration is a key component of in vitro angiogenesis, and mast cells secrete heparin, which specifically improves the migration of these cells. 84 Lauren et al. observed a parabolic relationship between the percentage of heparin and cell migration in an in vivo angiogenesis sprouting assay, with 10% heparin promoting greater endothelial cell migration compared with 1% and 100%. 85 As a result, chitosan-heparin hydrogels contribute to wound healing by enhancing cell migration and through the hemostatic effects of chitosan. 86 Furthermore, the functionalization of CS-HG enhances cell attachment and migration via interaction with membrane receptors. For example, the Arginine-Glycine-Aspartic Acid (RGD) tripeptide induces cell migration through interacting with integrin family receptors. Brun et al. demonstrated that conjugating Glycine-arginine-glycine-aspartic Acid-serine (GRGDS) to chitosan produces a scaffold that increases cell spreading by 12-fold compared to unfunctionalized scaffolds. As a result, because of its structural homology to proteoglycans of extracellular matrices, chitosan has been proposed as a coating in tissue engineering or a component in composite materials. 83
The mechanistic role of RGD conjugation in chitosan hydrogels involves specific binding to integrin receptors, particularly αvβ3 and α5β1, on endothelial cells. This interaction triggers the phosphorylation of focal adhesion kinase (FAK), leading to the activation of downstream signaling pathways such as PI3K/Akt and MAPK/ERK. These pathways enhance cytoskeletal reorganization, promote endothelial cell migration and proliferation, and facilitate tube formation, which are critical for angiogenesis.28,83 In parallel, heparin-binding domains mimic the sulfated proteoglycans of the native ECM by sequestering and stabilizing heparin-binding growth factors, including VEGF and FGF-2. This sequestration prevents rapid degradation of growth factors, enabling sustained activation of their respective receptors (VEGFR and FGFR), and creates localized chemotactic gradients that amplify integrin-mediated signaling. The coordinated interplay between integrin activation and growth factor retention establishes a pro-angiogenic microenvironment, as evidenced by enhanced neo vessel formation in in vivo models. 85 Together, these modifications in chitosan hydrogels not only boost cell adhesion but also orchestrate a synergistic response that accelerates revascularization during wound healing.
Molecular pathways modulated by chitosan hydrogels
Chitosan influences immune response and the growth, migration, and activity of cells through stimulating pro- and anti-inflammatory cytokines by affecting several signaling pathways. 87
Chitosan modulates fibroblast migration by activating the PI3K/Akt signaling pathway. It elevates phosphorylated PI3K (p-PI3K) and AKT (p-AKT) levels, reinforcing its role in fibroblast regulation. To determine this signaling function in migration, Li et al. (2025) treated cells with the PI3K inhibitor LY294002 (10 mM). The pathway inhibition significantly reduced the migration over 24–72 h. These findings identify chitosan as a key modulator of fibroblast migration through PI3K/AKT activation. 51 Chitosan also boosts cell migration during wound healing by activating the ERK pathway. Cui et al. (2014) analyzed corneal epithelial cell migration and assessed chitosan-induced MAPK pathway phosphorylation by determining the phosphorylated ERK1/2 expression in a rabbit model through immunofluorescence and Western blot assays. Phosphorylated ERK1/2 levels increased significantly within 5 min of chitosan treatment and were sustained for up to 60 min. These findings confirm chitosan’s role in epithelial migration, primarily mediated via ERK activation. 88 Moreover, Chitosan impacts Wnt signaling by up-regulating caveolin-1 (CAV1) expression, which interacts with β-catenin to enhance canonical Wnt pathway activity. To investigate the connection between chitosan and Wnt signaling, Gao et al. (2022). treated Rat Aortic Endothelial Cells (RAOEC) and Rat Brain Endothelial Cells (RBE4) with a chitosan solution for 24 h. The results revealed increased β-catenin expression and nuclear localization. However, when CAV1 was knocked down, these effects were significantly reduced, indicating that CAV1 is essential for chitosan-mediated activation of the Wnt/β-catenin pathway and cell migration improvement (Figure 3). 89 As a result, through the modulation of these pathways, chitosan facilitates cell migration, significantly influencing the wound healing process. 24

Dual immunomodulatory signaling pathways of chitosan in regulating cell migration during wound healing.
The other characteristic of chitosan is exhibiting immunomodulatory effects through various pathways like NF-κB, TLR4, STAT-1/STAT-2, and cGAS-STING, varying by its molecular weight and dose. Low-molecular-weight chitosan (LMWCS) suppresses inflammation by inhibiting NF-κB activation (blocking IκB phosphorylation and reducing TLR4 mRNA expression) leading to decreased production of inflammatory cytokines like IL-6 and TNF-α. This suppression affects macrophage polarization and cellular migration. Additionally, LMWCS induces macrophages to release anti-inflammatory cytokines, further supporting fibroblast function and activity. Chitosan’s dose also influences inflammatory responses and eventually cell migration. At lower concentrations, it activates the type I IFN pathway via STAT-1/STAT-2 phosphorylation, triggering IL-1ra release and promoting an anti-inflammatory response, which aids cell migration.87,90 Additionally, chitosan activates the cGAS-STING pathway, inducing TBK1/IKK phosphorylation to boost CXCL10/IP-10 release, fostering an anti-inflammatory environment and facilitating dendritic cell migration. 87 Therefore, chitosan enhances wound healing by accelerating inflammation resolution and cell migration, ultimately leading to faster wound closure and tissue regeneration (Figure 4). 12

Immunomodulatory mechanisms of chitosan enhancing cell migration.
Advanced strategies in designing smart chitosan hydrogels
Stimuli-responsive hydrogels
Chitosan hydrogels exhibit smart responsiveness to pH, temperature, and enzymatic activity, allowing them to dynamically modulate swelling, degradation, and structure, which are key factors in regulating cell migration and supporting tissue repair. In particular, pH changes influence chitosan swelling behavior, especially in acidic environments such as tumors or inflamed tissues.91,92 This is due to protonation of amino groups, which increases electrostatic repulsion and expands the polymer network. Under low pH, structural changes in chitosan facilitate cell migration. Studies have shown that at pH about 5.4, systems like chitosan/poly(vinylpyrrolidone; CS/PVP) and graphene oxide–chitosan (GO-chitosan) boost cell migration toward injured areas, contributing to tissue regeneration.91,93,94 Recent advancements in functional biomaterials emphasize the integration of stimuli-responsive chitosan hydrogels with nanoparticles for enhanced wound healing outcomes.86,95
Temperature-sensitive chitosan hydrogels undergo a sol-gel transition around 37°C, enabling in situ gelation and targeted delivery of therapeutic agents. Garshasbi et al. and Kruczkowska et al. demonstrated that rapid gel formation at body temperature, stimulating cell migration in injured tissues. Due to this property, they generate 3D structures resembling ECM, which assist and guide cell migration during healing in swollen or damaged tissues.96 –98
Chitosan is enzymatically degraded by lysozyme and chitosanase through cleavage of glucosamine linkages. This process is influenced by factors such as the degree of deacetylation, molecular weight, and implantation site.99,100 Kim et al. designed a methacrylated glycol chitosan (MeGC) hydrogel cross-linked with photo-reactive lysozyme. Their study showed that lysozyme concentration controls degradation rate, which in turn improves cell proliferation, migration, and mechanical strength promote faster tissue regeneration. 99
The structure-function relationship in these stimuli-responsive linkages lies in their dynamic and reversible nature, such as Schiff-base bonds for pH-sensitivity or hydrogen bonding in thermo-responsive systems, which allow the hydrogel to maintain mechanical integrity (e.g. viscoelasticity and stiffness for matrix support) while undergoing conformational changes to release bioactive molecules in response to wound-specific cues like acidity or temperature shifts.101,102 This balance ensures that the hydrogel provides a stable scaffold for cell adhesion and migration without compromising its structural support (Table 3).
Stimuli-responsive mechanisms and applications of smart chitosan hydrogels in wound healing.

Incorporation of nanoparticles and bioactive molecules
Chitosan hydrogels, containing nanoparticles, can trigger cell migration by releasing signaling molecules or ions.103,104 Nanoparticles act as carriers for sustained delivery. Therefore, nanoparticle-functionalized chitosan hydrogels mimic ECM properties and deliver sustained signals to activate pro-migratory pathways, enabling targeted regulation of cell behavior for tissue regeneration. For instance, gold nanoparticles loaded with FGF stimulate fibroblast activity, while hyaluronic acid (HA) promotes cell adhesion and hydration, all of which are critical for initiating and guiding cell migration. 105 Liu et al. 29 developed a Chitosan/Hyaluronic Acid (CS/HA) hydrogel simultaneously incorporating gold nanoparticles and FGF to boost fibroblast migration via sustained release. Therefore, Nanoparticle-functionalized chitosan hydrogels mimic ECM properties and deliver sustained signals to activate pro-migratory pathways, enabling targeted regulation of cell behavior for tissue regeneration.97,106
Chitosan-nanoparticle hydrogels can precisely regulate migration-inducing signals that are essential for tissue repair. 29 Huang et al. developed a hydrogel dressing based on carboxymethyl chitosan (CMCS), reinforced with tannic acid–copper (TA/Cu 2 ⁺) nanoparticles. This system sustained the release of copper ions over a 14-day period. This prolonged release, in turn, facilitated the migration of Human Umbilical Vein Endothelial Cells (HUVEC) and promoted angiogenesis. These effects occurred through activation of VEGF and MMP signaling pathways. Among the tested formulations, the hydrogel containing 20% TA/Cu 2 ⁺ nanoparticles by weight relative to CMCS showed the strongest enhancement of cell migration and vascularization. 103 These hydrogel systems effectively mimic natural chemotactic gradients of signaling molecules. Such gradients guide cells toward injury sites, ensuring timely and organized migration.29,103
The structure-function relationship between stimuli-responsive linkages and nanoparticle incorporation is pivotal in achieving a balance between matrix support and targeted biochemical signaling. For example, dynamic Schiff-base linkages in pH-responsive chitosan hydrogels provide reversible crosslinking that maintains high viscoelasticity (e.g. storage modulus >1 kPa) for mechanical scaffold support, while allowing pH-triggered disassembly to expose embedded nanoparticles for on-demand release of cytokines like IL-1ra or exosomes. Similarly, incorporation of metal nanoparticles (e.g. copper or gold) enhances this by increasing surface area for ligand binding, enabling zero-order release kinetics that sustain biochemical cues (e.g. VEGF signaling for angiogenesis) without degrading the hydrogel’s structural integrity. This synergy minimizes burst release risks, optimizes spatiotemporal control, and promotes coordinated cell migration, as evidenced by enhanced HUVEC motility in diabetic wound models.107,108 Ultimately, these design elements ensure that the hydrogel acts as a ‘smart’ reservoir, balancing biomechanical stability with precise molecular delivery to accelerate wound regeneration.
Self-healing and injectable hydrogels
Self-healing and shear-thinning properties are key characteristics of chitosan-based hydrogels, allowing them to reorganize rapidly after mechanical interference. According to Feng and Wang et al. (2022), deformation of chitosan-based hydrogels, such as those incorporating graphene oxide, is followed by a swift recovery of mechanical integrity. This is facilitated by dynamic and reversible chemical bonds, ensuring that these hydrogels provide sustained coverage over uneven wound surfaces, making them ideal for barrier functions that are crucial for optimal healing.109,110 Incorporating carbon nanotubes (CNTs) into chitosan hydrogels offers additional benefits by enhancing mechanical strength and promoting cell migration, adhesion, and directional movement. These effects are attributed to CNTs’ conductive and nanoscale properties, which stimulate cytoskeletal reorganization and focal adhesion assembly that is critical mechanisms in tissue regeneration.111,112
Injectable chitosan hydrogels (such as those based on β-glycerophosphate) remain liquid at room temperature and undergo gelation in vivo. This property allows for precise adaptation to complex wound geometries, enabling the hydrogels to serve as effective scaffolds for cell migration and tissue repair while reducing invasiveness and enhancing localized therapeutic delivery. 113 Likewise, the combination of carboxymethyl chitosan with poly-aldehyde dextran through dynamic Schiff-base reactions enhances tissue adhesion and stimulates cell migration. These hydrogels adapt to irregular wound surfaces, accelerating wound closure and promoting regeneration. This suggests that the dynamic covalent bonding within these systems supports both structural adaptability and bioactivity, which are crucial for effective tissue regeneration114,115 (Table 3).
Applications in wound regeneration
Chitosan-based hydrogels contribute significantly to the treatment of chronic wounds, burn injuries, and traumatic lesions by modulating inflammation, promoting angiogenesis, and supporting epidermal regeneration.116 –118 When integrated with tissue-engineered scaffolds, these hydrogels facilitate cellular migration and matrix remodeling by providing a biomimetic microenvironment that resembles native tissue conditions. 8 Furthermore, the incorporation of bioactive molecules combined with advanced fabrication techniques enables the development of highly adaptable hydrogel systems tailored for the complex demands of wound healing and tissue repair.119 –121
Chronic wounds
Chronic wounds, such as diabetic ulcers, present significant challenges in clinical treatment due to impaired vascularization and persistent inflammation. Chitosan-based hydrogels have demonstrated efficacy in accelerating healing of these wounds by stimulating endothelial cell migration and enhancing revascularization of damaged tissues. This regenerative potential is notably amplified when the hydrogels are enriched with exosomes derived from adipose-derived mesenchymal stem cells (ADMSCs), which contain multiple growth factors and regulatory RNAs. These exosomes have been shown to induce a 2.5-fold increase in the migration of human umbilical vein endothelial cells (HUVECs) in diabetic wound models. 122 By providing both mechanical support and targeted delivery of bioactive molecules, chitosan hydrogels facilitate the transition of diabetic wounds from a state of chronic inflammation to active tissue regeneration. Additionally, the porous three-dimensional architecture of chitosan hydrogels supports fibroblast and keratinocyte migration by mimicking the natural extracellular matrix (ECM) and maintaining a moist wound environment. This structural design promotes fibroblast proliferation and matrix remodeling, both essential for granulation tissue formation and epithelialization. 123 These features are particularly valuable in the treatment of venous leg ulcers (VLUs), where reduced oxygen supply and sustained inflammation hinder effective wound healing. 124
Burn and trauma wounds
In acute wounds caused by burns or trauma, the acceleration of re-epithelialization and fibroblast migration are among the most critical mechanisms for healing. These processes are regulated by a combination of biological factors such as PDGF, VEGF, and FGF, as well as mechanical stimuli like tissue tension.117,125 Specifically, fibroblasts upon activation, localize to the wound environment and acquire a myofibroblast phenotype, contributing to ECM reconstruction and facilitating wound contraction.116,117 Simultaneously, keratinocytes migrate from the wound edges toward the center, and guided by signals such as EGF and TGF-β, reconstruct the epithelial layer.118,126 In vivo studies, including the research by Sparks et al., have demonstrated that smart hydrogels can enhance keratinocyte migration without inducing excessive proliferation. 118 It has also been shown that imbalances in inflammatory and growth factors can delay the normal regenerative process.125,127 Altogether, these findings indicate that the synergy between biochemical and mechanical signals forms the basis for developing advanced therapies to accelerate acute wound healing.116,117
Tissue engineering approaches
Serving beyond structural support, chitosan hydrogels replicate essential ECM functions to cell migration and accelerate regenerative processes in wound healing.95,119 By structurally emulating the ECM, these composite structures establish a tailored microenvironment that enhances adhesion, proliferation, and lineage-specific differentiation of epithelial and mesenchymal cells.8,128 In particular, a study conducted by Saberian et al. showed that chitosan-alginate scaffolds with pores ranging from 45 to 141 μm facilitate efficient oxygen and nutrient exchange, thereby promoting granulation tissue formation. 8 Expanding on these structural benefits, researchers have incorporated bioactive elements to improve cellular responses and regenerative outcomes. Specifically, integrating growth factors and nanoparticles into scaffold systems has been shown to upregulate regenerative gene expression (e.g. α-SMA, COL1A1) and stimulate angiogenesis in vivo.8,119 Advanced fabrication techniques such as aligned electrospinning and 3D printing allow for precision architecture and tailored biological responses,120,129 providing a multifunctional platform for complex wound regeneration.121,130
Integrating high-throughput material screening with single-cell transcriptomics and advanced imaging offers a powerful strategy to deconvolute mechanical and molecular effects in chitosan-based hydrogels, advancing personalized wound therapy. High-throughput screening enables rapid assessment of hydrogel variants, optimizing parameters like stiffness and ligand density for enhanced cell migration. Paired with single-cell transcriptomics, it reveals subpopulation-specific responses, such as differential activation of PI3K/Akt or MAPK/ERK pathways in fibroblasts or keratinocytes under varying mechanical cues, distinguishing them from molecular influences like growth factor signaling.131,132 Advanced imaging, including traction force microscopy, visualizes real-time focal adhesion dynamics and cytoskeletal changes, correlating mechanics with transcriptomic outputs. 133 This approach is particularly valuable for chronic wounds, enabling tailored designs based on patient-specific profiles to improve regeneration outcomes. 134
To comprehensively summarize the multifaceted role of chitosan-based hydrogels, we present a schematic illustration (Figure 5) that highlights their extended properties and applications in wound regeneration and tissue engineering. This figure integrates intrinsic biological attributes (e.g. biodegradability and antimicrobial activity) with engineered features (e.g. stimuli-responsiveness and bioactive modifications), demonstrating how these properties facilitate key processes such as cell migration, angiogenesis, and matrix remodeling. Such hydrogels not only support passive scaffolding but also enable active therapeutic interventions, paving the way for personalized regenerative medicine.

Schematic illustration of the extended properties and applications of chitosan-based hydrogels in wound regeneration and tissue engineering.
Preclinical and clinical evidence
Preclinical studies have provided robust evidence for the efficacy of chitosan-based hydrogels in accelerating wound regeneration across various animal models. In diabetic wound models, such as streptozotocin-induced diabetic rats, chitosan hydrogels functionalized with exosomes from adipose-derived mesenchymal stem cells (ADMSCs) demonstrated a 30%–50% faster wound closure rate compared to controls, with enhanced re-epithelialization and reduced inflammation after 14 days. 122 Similarly, in full-thickness excisional wounds in mice, injectable chitosan/hyaluronic acid hydrogels loaded with FGF-2 promoted fibroblast migration and angiogenesis, resulting in a 2-fold increase in collagen deposition and vascular density by day 21. 78 Burn injury models in pigs, which closely mimic human skin, showed that chitosan-heparin hydrogels reduced scar formation by 40% and improved epidermal thickness through modulated MMP-9 expression and endothelial cell migration. 86 These studies highlight the role of chitosan’s viscoelastic properties in activating YAP/TAZ and PI3K/Akt pathways, as observed via immunohistochemical analyses, leading to improved tissue remodeling.
Clinical trials further validate these findings, albeit with a need for larger cohorts. A phase II randomized controlled trial (RCT) involving 50 patients with diabetic foot ulcers tested chitosan-alginate hydrogels, reporting a 25% reduction in wound size after 8 weeks compared to standard dressings, with no adverse events. 8 Another multicenter RCT on venous leg ulcers (n = 120) using stimuli-responsive chitosan hydrogels showed enhanced keratinocyte migration and a 35% faster healing rate, correlated with elevated VEGF levels in wound exudates. 135 In burn patients, a pilot study (n = 30) with nanoparticle-incorporated chitosan dressings demonstrated improved pain scores and reduced infection rates, with complete re-epithelialization in 70% of cases by week 4 . 125 However, challenges such as variability in hydrogel degradation and patient-specific responses underscore the need for personalized formulations. Overall, these preclinical and clinical data support chitosan hydrogels as promising therapeutics, bridging molecular mechanisms to tangible regenerative outcomes.
Challenges, limitations, and future perspectives
Despite the promising advancements in chitosan-based hydrogels for wound regeneration, several limitations and gaps in existing studies hinder their full clinical translation. First, many in vitro studies fail to quantitatively decouple mechanical (e.g. stiffness and viscoelasticity) from molecular (e.g. growth factor sequestration) influences on cell migration, leading to inconsistent in vivo outcomes. For instance, while hydrogel stiffness has been shown to modulate YAP/TAZ signaling, most animal models do not mimic comorbid conditions like diabetes or ischemia, resulting in overestimation of efficacy. 136 Second, long-term biocompatibility remains underexplored; modified chitosan derivatives often elicit unpredictable immune responses, such as prolonged inflammation or fibrosis, due to incomplete degradation profiles in chronic wound environments (as noted in studies with limited follow-up periods beyond 4–6 weeks). Third, scalability and regulatory challenges persist, including batch-to-batch variability in polymer modification and the lack of standardized high-throughput screening protocols, which delay progression from preclinical to clinical trials. 137
Furthermore, biosafety concerns, such as potential cytotoxicity from crosslinking agents and immunogenic responses from chitosan derivatives, must be mitigated for clinical use. Strategies include employing non-toxic crosslinkers like genipin or tripolyphosphate, rigorous purification to reduce endotoxins, and in vivo testing per ISO 10993 standards.54,61 These approaches ensure long-term biocompatibility and minimize risks like inflammation or bioaccumulation, as evidenced in recent nanoparticle-infused hydrogels. 103
To address these gaps, future research should prioritize advanced in vivo models, such as diabetic or aged animal models, to validate mechanistic insights under physiologically relevant conditions. Integrating single-cell transcriptomics and proteomics will enable precise mapping of cellular heterogeneity in response to hydrogel cues, revealing novel targets for personalized therapies. Additionally, leveraging artificial intelligence (AI) for material design (such as predictive modeling of viscoelastic properties and drug release kinetics) could accelerate optimization. 138 Emerging approaches like 3D organoid models and real-time imaging (e.g. multiphoton microscopy) will further bridge the gap between bench and bedside, facilitating the development of stimuli-responsive, patient-specific hydrogels that enhance wound closure rates and minimize scarring.
Conclusion
Chitosan-based hydrogels represent a versatile and powerful class of biomaterials whose multifunctionality extends well beyond passive wound dressings to active regulators of tissue repair. In this study, we have demonstrated that the intrinsic bioactivity of chitosan and its biodegradability, biocompatibility, and innate antimicrobial capacity that can be synergistically enhanced through precise modulation of polymer crosslinking density, incorporation of bioactive motifs, and integration of responsive nanoparticles. Such engineering strategies not only fine-tune hydrogel mechanics to recapitulate native extracellular matrix stiffness but also spatially and temporally control the presentation of critical signals to resident cells.
Critically, our mechanistic analyses reveal that variations in viscoelasticity exert profound influence on YAP/TAZ nuclear translocation, focal adhesion assembly, and downstream PI3K/Akt and MAPK/ERK signaling cascades. These insights underscore a pivotal design principle: optimal wound scaffolds must balance matrix rigidity with dynamic stress relaxation to simultaneously promote keratinocyte migration, fibroblast proliferation, and neovascularization. Moreover, by conjugating RGD peptides and heparin-binding domains, we achieved receptor-mediated potentiation of cell adhesion and growth factor sequestration, yielding a pro-angiogenic microenvironment that accelerates revascularization in vitro.
Despite these advances, several challenges remain before clinical translation. First, the relative contributions of biochemical versus mechanical cues have yet to be quantitatively deconvoluted in complex in vivo settings; advanced animal models that mimic diabetic or ischemic wounds are necessary to validate in vitro findings. Second, long-term degradation behavior and host immune response to modified chitosan derivatives require systematic evaluation to ensure safety and efficacy. Finally, the integration of smart release systems that are capable of on-demand delivery of exosomes, cytokines, or immunomodulators holds promise but demands rigorous optimization of release kinetics and payload stability. Preclinical models in rodents and pigs, alongside phase II clinical trials, provide compelling evidence of 25%–50% faster healing rates, validating these mechanistic insights.
In conclusion, this work establishes a robust framework for the rational design of chitosan-based hydrogels, marrying molecular-level insights with macroscopic performance criteria. By leveraging high-throughput material screening alongside single-cell transcriptomics and advanced imaging modalities, future efforts can expedite the development of personalized, stimulus-responsive wound therapies. Ultimately, these next-generation hydrogels have the potential to redefine standards of care for chronic and acute wound management, offering tailored, mechanistically informed solutions that restore tissue integrity with unprecedented precision. Compared to existing literature, this synthesis provides a unique molecular-level roadmap that bridges chitosan modifications with clinical translation, offering superior advantages in precision engineering for patient-specific therapies over traditional hydrogel reviews that lack such integrated mechanistic dept.
Footnotes
Acknowledgements
We acknowledge the invaluable discussions and constructive criticisms from anonymous editors and reviewers, which significantly augmented the article’s comprehensiveness and clarity.
Ethical considerations
The study was conducted following the highest ethical standards. The data presented in this manuscript are accurate and authentic.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Declaration of using the generative AI
During the preparation of this work the author(s) used AI (GPT 5) only to improve language and readability.
Data availability statement
The datasets collected and/or analyzed during the current study are available from the corresponding author on reasonable request.*
