Abstract
The present study was designed to fabricate a new chitosan-collagen sponge (CCS) for potential wound dressing applications. CCS was fabricated by a 3.0% chitosan mixture with a 1.0% type I collagen (7:3(w/w)) through freeze-drying. Then the dressing was prepared to evaluate its properties through a series of tests. The new-made dressing demonstrated its safety toward NIH3T3 cells. Furthermore, the CCS showed the significant surround inhibition zone than empty controls inoculated by
Introduction
Wound management is a significant clinical issue and is a growing economic burden across the globe. 1 An ideal wound dressing should not only provide adequate gaseous, but also imitate the extracellular matrix of skin (ECM). 2 Archana et al. 3 has concluded the essential properties of an effective wound dressing: (1) suitable water vapor permeability that maintains a moist physiological environment around the wound; (2) high gas permeability; (3) excellent fluid absorption capability; (4) a protective barrier against external microorganisms; (5) antibacterial property to suppress bacterial infection; and (6) have no cytotoxic components in case of secondary damage to nascent tissues. But there is no ideal wound dressing in the clinic now. Many new dressings are studied like the chitosan-calcium alginate dressing, 4 chitosan-vaseline gauze dressing, 5 chitosan-nano dressing, 3 lignin-chitosan-PVA composite hydrogel, 6 nanocellulose-collagen dressing 7 and so on. But each has own fatal flaws and are not widely used. Among these new dressings, we can find that collagen and chitosan are popular components because of their excellent properties. To make an ideal wound dressing, we tried to combine the collagen and chitosan in varying proportions through the freeze-dried way since chitosan and collagen are known to have different chemical properties but can complement each other and interact chemically through hydrogen bonding without any significant changes. 8
Collagen is known as a major structural component of the dermal extracellular matrix as a natural polymer which has many properties of hemostasis, biodegradability, hydrophilicity, biocompatibility, and the ability of promoting cell proliferation.9,10 Therefore, collagen is widely used in biomedical materials and occupies a superior place among polymers for applications of skin tissue engineering.11–13 A collagen dressing is a basic dressing without antibacterial property which is important for wound care. In our study, to reach an overall wound healing promoting effect, we select chitosan as another ingredient.
As reported, chitosan has a lot of excellent properties, such as biocompatibility, biodegradability, hemostatic property, anti-inflammatory effects and capability to accelerate wound closure.14–18 Chitosan is one of the natural polymers that represent a very promising source for the development of antimicrobial agents.
19
It was reported that the antibacterial activity of chitosan is related to its molecular weight. Younes20,21 reported that high molecular weight chitosan shows better antibacterial property against Gram-positive
In our study, to enhance the antibacterial activity, a 3.0% chitosan mixture with low and high molecular weight blended with a 1.0% type I collagen (7:3(w/w)) was made as a novel wound dressing to inhibit both Gram-negative and Gram-positive bacteria simultaneously. We evaluated the properties of the wound dressing through several in vitro assays and the healing capacity was assessed in full-thickness excisional wounds in mice.
Materials and methods
Materials
Low (MW ~ 2000) and high (MW ~ 800,000) molecular weight chitosan (degree of deacetylation is 95%) were obtained from Golden-Shell Pharmaceutical Co (Zhejiang, China). Type I collagen was obtained from Sigma.
Fabrication of wound dressings
A 3.0% mixed chitosan solution was made by dissolving low and high molecular weight chitosan powder (1:1, w/w) in 1.0% glacial acetic acid (v/v). Then it was stirred vigorously overnight, and then filtered to sterilize. The type I collagen was dissolved in 1.0% glacial acetic acid to make the solution. Then a chitosan-collagen solution was prepared by a combination of mixed chitosan solution and collagen solution (7:3, v/v).
To fabricate CCS and its control group dressings (collagen sponge, chitosan sponge), the solutions were transferred into a 24-well plate (2 mL for each well, Sigma), frozen at −60°C for 6 h, and then lyophilized for 24 h. The freeze-dried dressings were compressed, and sterilized by ethylene oxide, sealed-storage reserved in 4°C before using for experiments.
FTIR analysis
FTIR spectra were obtained using a Bruker Tensor II Fourier-transform infrared spectrometer (BRUKEROPTICS, Ettlingen, Germany). The analysis was carried out on powders at room temperature and ambient humidity. Spectra were acquired in the range of 4000–400 cm−1 with a spectral resolution of 4 cm−1 and averaged over 32 scans.
Cytotoxicity test
In order to evaluate the applicability of CCS for its use in biomedical applications, its cytotoxicity was studied in vitro using the National Institutes of Health (NIH) 3T3 mouse embryonic fibroblast cells (provided by the cell bank of the Chinese Academy of Science). We set up four groups including CCS group, collagen sponge group, chitosan sponge group and the DMEM group.
Approximately 16 × 104 cells were dispensed equally into 32 wells of a 96-well plate to be incubated for 12 h, and each group owns eight wells. The four groups of different dressings were then cut into designated sizes (1.0 × 1.0 cm2), and soaked in 50 mL of cell culture medium at 37°C for 24 h to obtain sample extracts. The cells were treated with the extract of each dressing group and subcultring in the presence of 5% CO2 with 95% humidity at 37°C for 48 h. To measure the absorbance with ELIASA at 450-wavelength of each well after 2 h with a Cell Counting Kit-8 (CCK-8; Dojindo, Kumamoto, Japan). The cytotoxicity was evaluated by the relative growth rate (RGR) of NIH3T3 cells, which was calculated according to the following equation:
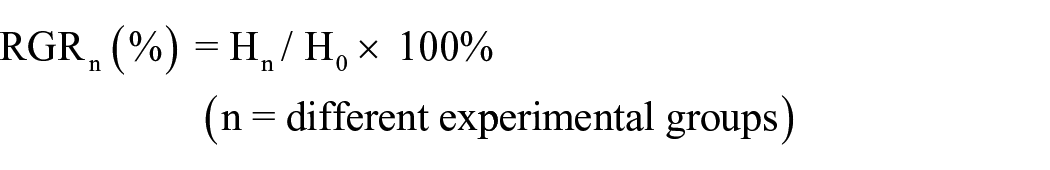
When RGRn is the cell growth of different experimental groups compared to the DMEM group, Hn is the average absorbance of different experimental groups, H0 is the average absorbance of the DMEM group. We can make judgments of cytotoxicity grades by RGR according to the GB/T 16886.5-2003 (ISO 10993-5: 1999) documents (Table 1). 22 And it can be considered to be noncytotoxic when the cytotoxicity grade is 0 or 1.
Cell relative growth rate and cytotoxicity.

Antimicrobial studies
The antimicrobial property of the dressings was evaluated by the inhibition zone diameters with filter paper. We set up four groups including CCS group, collagen sponge group, chitosan sponge group and normal saline control group (NaCl). Nutrient agar,
Animals
Adult male Sprague-Dawley rats (SD rats; weighing approximately 200–250 g, 8–14 weeks old) were provided by the Laboratory Animal Center of the Fourth Affiliated Hospital, Zhejiang University School of Medicine, Yiwu, China. The animal protocols were approved by the Ethics Committee of the Fourth Affiliated Hospital, Zhejiang University School of Medicine, and were performed according to the National Institutes of Health guide for the care and use of Laboratory animals (NIH Publications No.8023, revised 1978).
Moisture retention measurement
The moisture content of the tested areas was measured using a corneometer (Technology of golden light, Shenzhen, China). Six male SD rats were used in this experiment. Prior to the test, rats were anesthetized with 2% sodium pentobarbital (50 mg/kg), and then the hair was removed from the dorsal skin. Four 2 × 2 cm2 in the depilated dorsal skin were marked with permanent ink marker and covered with equal sized pieces of CCS, collagen sponge, chitosan sponge and blank sponge dressings. Before applying dressings, the humidity of the rat skin was measured as the primary moisture content (T0). After application of different dressings, moisture content of the skin was measured with a digital corneometer at different time points (25, 50, 75, 100 and 125 min). We repeated the experiment three times and took the average Tn. The skin humidity rising rate (Pn) was calculated using the formula below:
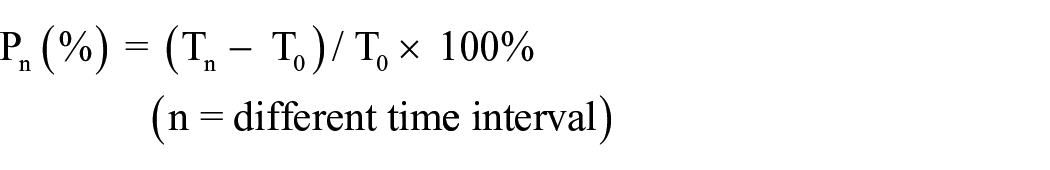
In vivo wound healing experiment
Wound model
A total of 16 male SD rats were used in vivo experiment, and they were randomly divided into two groups (
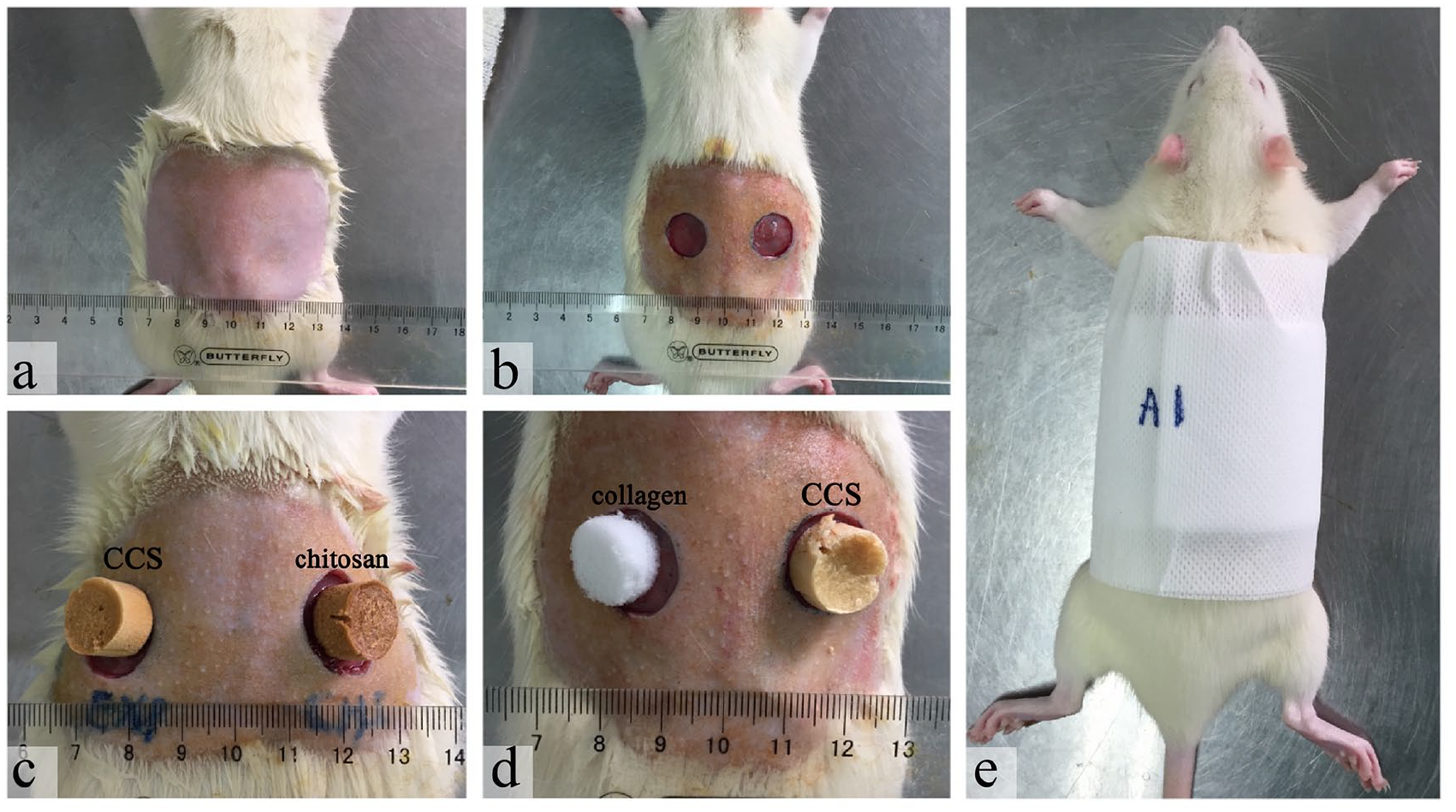
Wound model of rats: (a) the skin was prepared by shaving the desired area from the back of each animal using electric clipper after anesthesia, (b) two full-thickness round wounds of 1.5 cm in diameter down to the panniculus carnosus were created on the skin, (c) the left wounds of the rats in Group A were covered with CCS, and the right wounds were covered with chitosan sponge, (d) the left wounds of the rats in Group B were covered with collagen sponge, and the right wounds were covered with CCS, and (e) the rats were fixed with sterile gauze and elastic tape.
Gross observation of healing wounds
On 1st, 3rd, 7th, and 14th postoperative days, gross observation was performed in two rats of each group separately. After removing the dressing, the wound sites were calculated by a transparent grid-counting method.
Histological observation
Skin regions including the margin and the entire wound bed were harvested. Tissue specimens at different time points were sectioned into 5-µm-thick sections, and stained by hematoxylin-eosin (H&E) and Masson’s trichrome to visualize the inflammatory cells, neovessels, fibroblasts, and collagen.
Statistical analysis
The data analysis was performed using the
Results and discussion
Structural features
Morphology of dressings: As shown in Figure 2(a), collagen has a crystalline appearance and filiform texture, which is very delicate. Chitosan has porous surface and loose structure. CCS was prepared by a combination of chitosan mixture with type I collagen (7:3) in a freeze-dried way. It is round and compressed, harder than both collagen and chitosan.
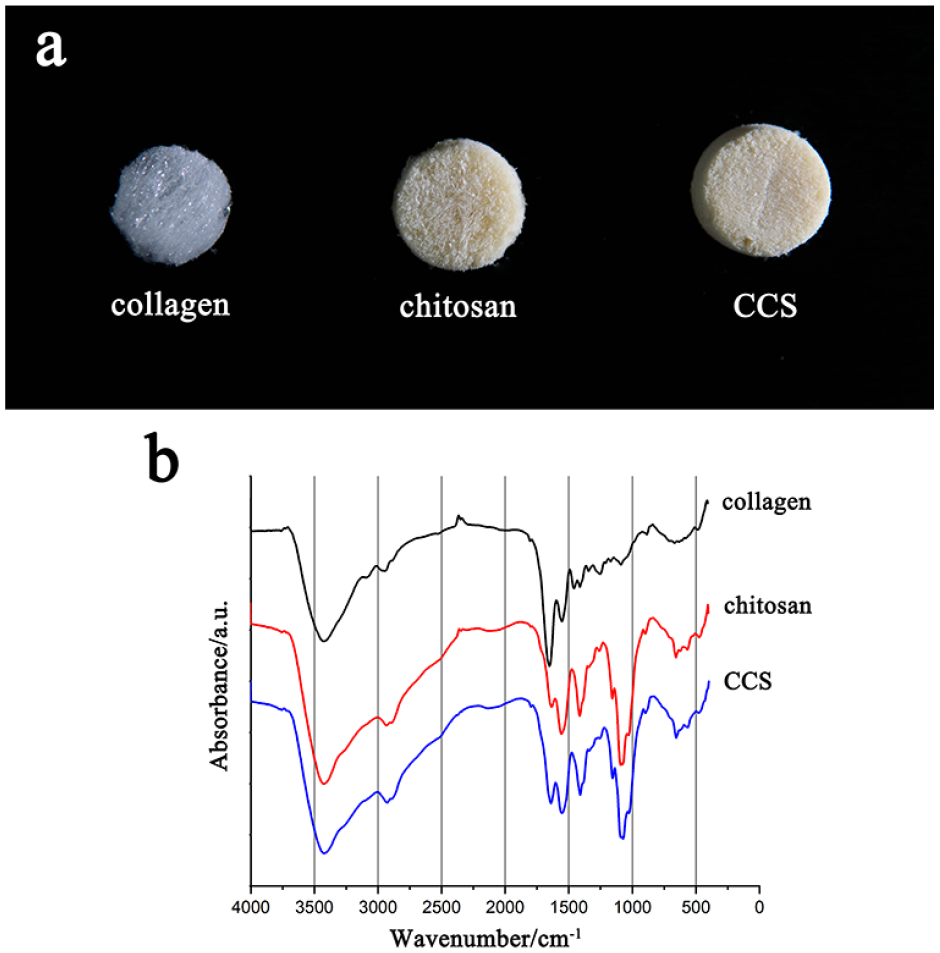
Photograph and FTIR of dressings: (a) collagen has a crystalline appearance and filiform texture, which is very delicate. Chitosan has porous surface and loose structure. CCS was prepared by a combination of chitosan mixture with type I collagen (7:3) in a freeze-dried way. It is round and compressed, harder than both collagen and chitosan, and (b) the FTIR spectra of collagen, chitosan and CCS.
FTIR spectroscopic characterization: The FTIR spectra of all the dressings were in the range 400–4000 cm−1 wave numbers. As shown in Figure 2(b), in the spectra of collage, there was a peak at 3320 cm−1, attributed to the N-H stretching, while a peak at 3400 cm−1 originating from the vibration of O-H in chitosan was detected. The speak in CCS was stronger due to the co-contribution of the collage and chitosan. Split peaks between 2880 and 2920 cm−1 attributed to C-H in chitosan were observed in CCS, indicating the successful incorporation of chitosan into CCS. Another two peaks 1585 and 1645 cm−1 were present in the spectrum of collage, which meant the C=O stretching (Figure 2(b)).
Cytotoxicity
The cytotoxicity becomes one of the most crucial indicators for evaluation of wound healing applications.
24
The cell activity was measured by CCK-8. The absorbance and RGR of different groups were displayed in Table 2. It showed that there was no significant difference between NIH3T3 cells exposed to the new-made dressing extracts and the negative control groups (
Cytotoxity detection in different groups.

RGR: relative growth rate.
Antibacterial activity (inhibition zone test)
The antibacterial activity of the wound dressing should be assessed before using the dressing candidates on wound surface because wound infection is the first hurdle. 25 The antibacterial dressing would form a sterile aspect of the wound to prevent infection and promote wound healing. 26
To evaluate the antibacterial nature of the dressing, we used the method of inhibition zone where a clear zone of inhibition was displayed after 24 h around the effective dressing in the plate inoculated by

Antibacterial activity (inhibition zone test). Zone of inhibition studies of dressings against
The new dressing owns an excellent antibacterial property most due to the element of chitosan. Chitosan was found to promote the migration of inflammatory cells, which produce enough growth factors and pro-inflammatory products at the early phase of wound healing process. It can also penetrate the nuclei of microorganism where it inhibits the protein and mRNA synthesis by binding with DNA of the microorganism.27,28 The antimicrobial activity of chitosan contributes to a lower risk of infection and reduce the incidence of complications that hamper the wound healing. 29
Moisture retention measurement
Moist wound healing was invented by George Winter in 1962. Based on this theory, wound surface dehydration becomes an essential problem of serious skin defects. 30 To enhance the wound healing process, making a moist atmosphere over the wounded site is one of the most important conditions. 31 Desirable water uptake ability of a qualified material is beneficial for cell proliferation which affects the distribution of cell suspension and the transfer efficiency of nutriment and oxygen. 32 Hence good moisture retention is important for ideal wound dressing candidates. Dehydration of wounds caused by poor water retention of the dressing, results in a critical problem in impairing wound healing and influencing normal wound recovery.
We performed the moisture retention assay by applying different dressings on the back of modeled rats and measuring the moisture content rate by a corneometer. The moisture rates of CCS and chitosan sponge groups were increased more rapidly than the collagen and blank sponge groups (

Moisture retention measurement. The moisture rates of CCS and chitosan sponge groups were increased more rapidly than the collagen and blank sponge groups (
We find the groups with chitosan had better moisture retention. We guess it is related to the hydrophilic nature of chitosan which affects the extent of swelling of matrices. 3 It is re-affirmed that chitosan is an active component for wound dressings as we select.
Gross observation of healing wounds
Gross observation was used to compare the healing patterns of the rat transcutaneous full-thickness dermal wounds and the easiness of removing dressings from the wounds was also evaluated. All 16 cases achieved 95% re-epithelialization by the end of the experiment. The results showed that the wound area in CCS group healed more perfectly than in the control groups on days 3, 7 and 14 (

Gross observation and quantitative analysis of healing wound area covered with different dressings.
The wound dressings remained to adhere to the excisional wound surface and absorb the wound exudate, as previously reported in the literature. 32 In addition, we found our new dressings were easy to be removed from the wounds in subjective analysis.
H&E and Masson’s trichrome staining
Wound healing is a complicated and dynamic biological process involving a serious procedure for tissue growth and regeneration, which consists of hemostasis, proliferation, migration, inflammation and tissue modeling. Histological studies are used to evaluate the wound healing process in different groups with different dressings. Tissue samples were harvested for histological analysis at various time points in the experiment.
On the third postoperative day, abundant capillary buds, inflammatory cells, and neovessels were observed in all groups, but the proliferation of fibroblasts was only seen in CCS and collagen sponge groups. On the seventh postoperative day, there were still visible inflammatory cells in CCS and chitosan sponge groups, but relatively sparse in CCS. And we could observe the number of new capillaries and fibroblasts increased more rapidly and orderly in CCS than in other groups. On the 14th postoperative day, we could observe that there were more neovessels and fibroblast proliferation, especially in CCS group. The fibroblast cells were arranged in the horizontal direction orderly in CCS and collagen sponge groups. Dermal fibroblasts act as the first line of defense against injuries and significant fibroblast proliferation plays an important role in wound healing. 33 Besides, epithelialization also could be found in the boundary area around the wounds (Figure 6).
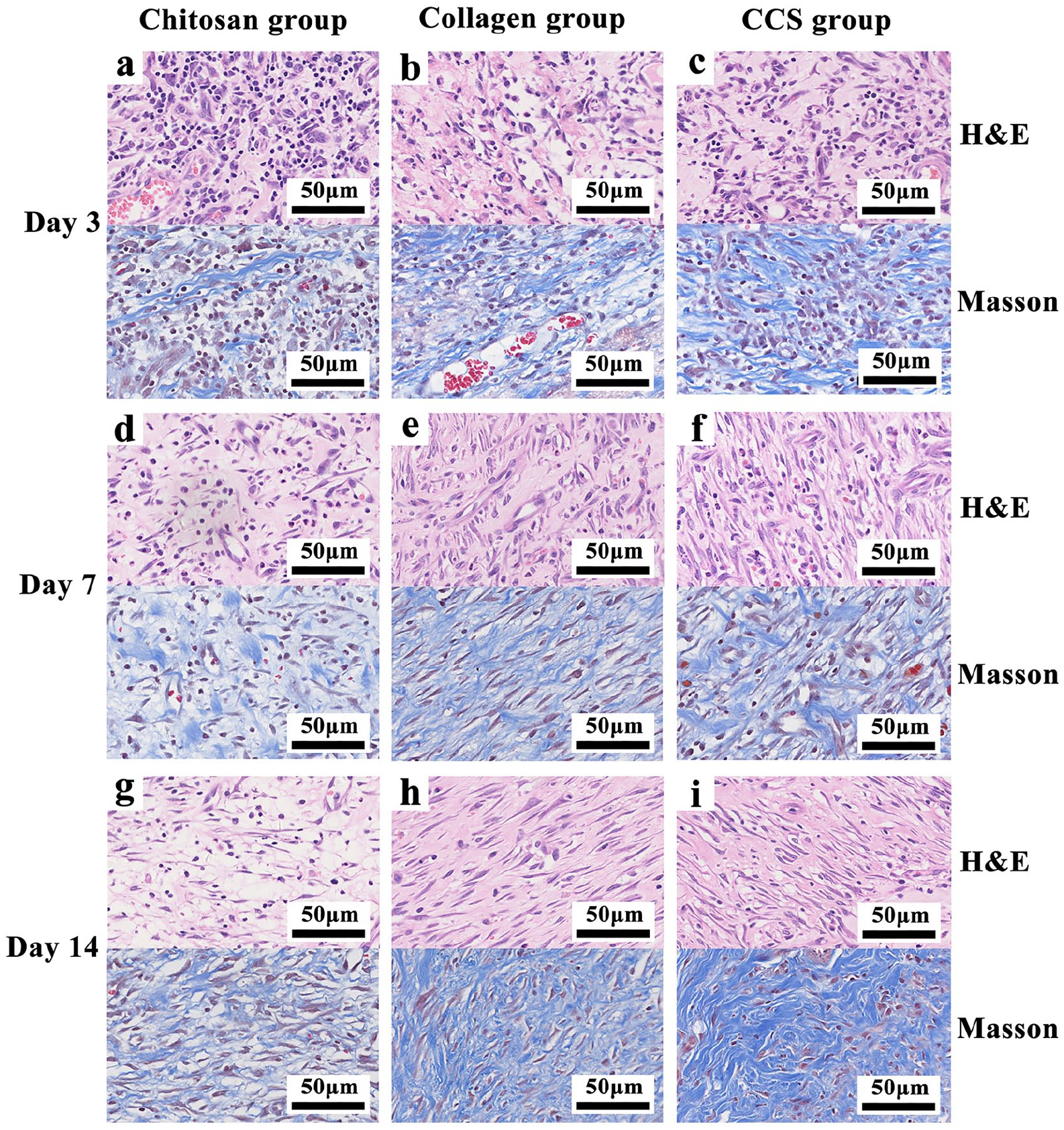
Hematein-eosin (H&E) and Masson’s trichrome staining of the wound tissues (400 X).
Histological examination provides us with crucial information on wound condition. During the wound healing process, chitosan depolymerizes to release N-acetyl glucosamine (NAG) gradually which is an important component of dermal tissue. 34 The N-acetyl glucosamine in chitosan can promote fibroblast proliferation, increase collagen deposition, and stimulate hyaluronic acid synthesis on the wound. 35 The new dressing is confirmed to be beneficial for wounds since chitosan has red blood cells binding and anti-bacterial properties, while collagen can support cell and tissue growth for skin wounds. 9
Conclusion
In conclusion, the results corroborated that the chitosan mixture with type I collagen dressing was nontoxic and biocompatible. Furthermore, apart from excellent water retention property which is crucial for reducing the risk of dehydration at wound site, our new dressings also showed significant antibacterial property against both Gram-negative and Gram-positive strains. Additionally, based on the macroscopic and histological examinations, the CCS showed excellent neovascularization and fibroblast proliferation, which were demonstrated to promoting full-thickness skin wound healing substantially. These results showed that the CCS could prove to be a promising biomaterial for wound dressing applications.
Footnotes
Contributorship
WQT and LHW researched literature and conceived the study. MXZ, WYZ, QQF, and XFW were involved in protocol development, carried out the fabrication of wound dressing and the test in vitro. CYC, BHS, and BZ carried out the animal study, as well as histopathological examinations of them. MXZ and SJW collected and analyzed the data. MXZ and WYZ wrote the first draft of the manuscript. WQT and LHW revised the manuscript and gave the final approval of the version to be submitted. All authors have approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Zhejiang Provincial Medical and Healthy Science Foundation of China (No. 2019KY757 and 2019ZD028), grants from National Natural Science Foundation of China (No. 81671918), and National Key Research Program of China (2016YFC1101004).
Guarantor
WQT
