Abstract
Introduction
Traditional wound dressings (e.g. gauzes, foams, rubbers, and membranes) can impede wound healing by causing infection, irritation, and secondary injuries during dressing changeover. In recent decades, investigations on wound dressings have suggested that hydrogel could provide a humid microenvironment, stimulate endogenous cell proliferation, maintain gaseous exchange necessary for tissue breathing, and repair epidermal or dermal injury. 1 Bioactive substances, macromolecules, and biocompatible materials, especially natural polymers derived from plants, have been evaluated as primary materials or ingredients in producing hydrogels used in wound healing. 2
Konjac glucomannan (KGM) is a safe, neutral polysaccharide derived from the tubers of
In this study, we used the one-pot approach to produce an interpenetrating hydrogel matrix by introducing KGM into the BSP solution to create a novel hydrogel based on BSP for wound treatment. The changes in microstructures and physical performance of the produced hydrogels with varying KGM concentrations, as well as the interplay between KGM and BSP, were the focus of this investigation. Impactions on the hydrogels’ cytotoxicity and wound healing performance were also examined
Methods
Materials
KGM (product number, K875397; CAS number, 37220-17-0; purity, ⩾ 95%), which had a viscosity of 1.5 × 104 mPa·s (1 g in 100 mL of H2O, 30°C), was bought from Shanghai, Macklin Biochemical Co., Ltd., (China). BSP (product number, PY191203bj; purity, ⩾ 95%) was obtained from Puyi Biotechnology Co., Ltd., (Nanjing, China) with a viscosity of 280 mPa·s (1 g in 100 mL H2O, 25°C). NaOH and HCl were of analytic grade.
Preparation of blend hydrogels
BSP was dissolved in deionized water with continuous stirring at predetermined concentrations in separate beakers and degassed for 2 h at ambient temperature. KGM of different weights was then slowly added and dissolved in the BSP solutions with continuous stirring. After 4 h of gelatinization in molds (round mold for physical and chemical examinations, diameter = 10 mm, thickness = 10 mm; square mold for cytotoxicity and animal experiments, length = 100 mm, thickness = 5 mm), NaOH (0.1 mol/L) was applied to modify the pH to 10.0 ± 0.5 to form hydrogels, followed by 24 h of equilibration at 25
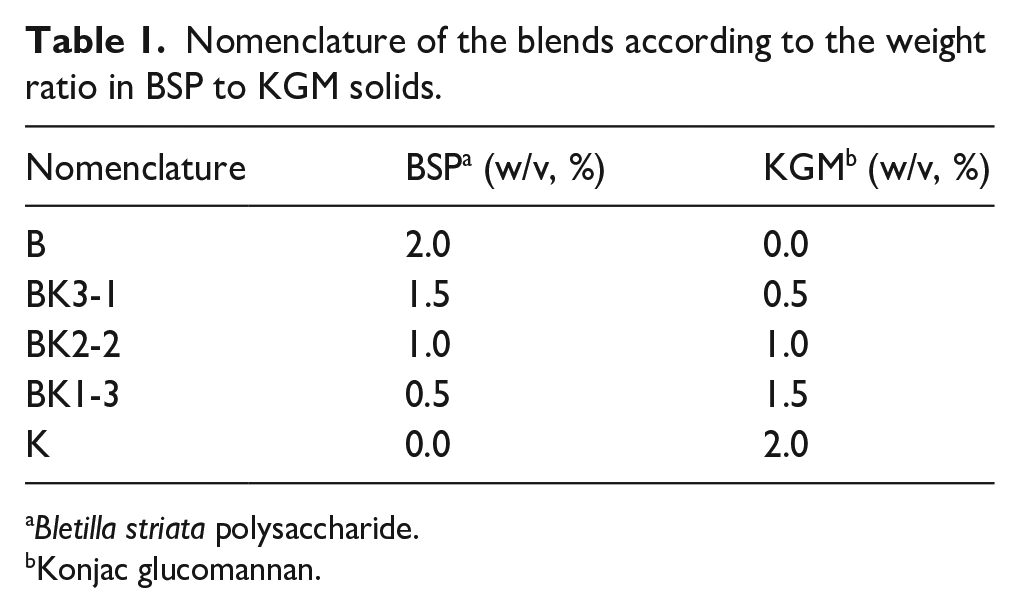
Nomenclature of the blends according to the weight ratio in BSP to KGM solids.
Konjac glucomannan.
Measurement of mechanical properties
The hydrogels’ hardness and cohesiveness were evaluated at room temperature using a CT3 texture analyzer (Brookfield, MA, USA) with a cylindrical 5 -mm-diameter probe, a compression rate of 1.0 mm/s, and a trigger force of 3.0 g. For each ratio, six samples were prepared, and each measurement was performed at least three times independently. The mean values were analyzed.
Water-holding capacity test
At room temperature, samples were cut into square pieces (10 mm × 10 mm × 5 mm), weighted (

where
Swelling degree
Weighted (

where
Thermogravimetric analysis
3–5 mg of the freeze-dried BSP solution, KGM hydrogel, and blend hydrogels were thermogravimetrically analyzed (TGA) in a nitrogen stream at 30°C–350°C with a heating rate of 5°C/min on a TGA Q500 thermogravimetric analyzer (TA Instruments, DE, USA). X-ray diffraction characterization
X-ray diffraction characterization
The freeze-dried samples were crushed, sieved (200 mesh) and then subjected to X-ray diffraction (XRD) analysis using a Bruker D8 Advance X-ray diffractometer (Bruker, Karlsruhe, Germany) and Ni-filtered Cu Kα (λ = 0.154 nm) radiation at a target voltage of 40 kV and a current of 40 mA within a scanning-angle range of 2θ = 10°–60° (interval of 0.02°).
Fourier transform infrared spectroscopy
After vacuum-drying for 72 h, the KBr pellet method was used to perform Fourier transform infrared spectroscopy (FTIR) 400–4000 cm−1 with a Nicolet 6700 spectrometer (ThermoFisher Scientific, MA, USA). The data was gathered at a spectral resolution of 2 cm−1.
Microstructure characterization
The samples were frozen at −80°C, followed by vacuum-drying (Biobase, BK-FD10P, Jinan, China). The resulting sponges were cut into square pieces (5 mm × 5 mm × 5 mm). A scanning electron microscope (SEM, JSM-6360LV, Japan) with a 10 kV accelerating voltage was used to view both parallel and perpendicular sections.
Cytotoxicity
Single-ingredient BSP could not form a gel. Therefore, the BSP solution was dried, and the resulting sponge was used in this experiment as well as the animal experiments. The cytotoxicity of the BSP sponge extract, BK2-2 hydrogel extract, and KGM hydrogel extract was assessed using the cell counting kit-8 test (CCK-8, Solarbio, Beijing, China) by treating L-929 fibroblasts (provided by Wuhan GeneCreate Biological Engineering Co., Ltd.) in 96-well plates with these entities as described previously. 13 Cell L-929 was cultured in Dulbecco’s modified eagle medium (DMEM, Sigma, MO, USA) supplemented with 10% FBS (Gibco, NY, USA). The freeze-dried BSP solution and hydrogel were cut into small pieces. Samples (50 mg) were immersed in a medium (5 mL) containing DMEM supplemented with 10% FBS for 24 h at 37°C. The extracts were subsequently diluted in stages (10.00, 5.00, 2.50, and 1.25 mg/mL) with DMEM medium containing 10% FBS, yielding a series of dilutions. After seeding L-929 cells (5 × 103 cells/well) in 96-well plates for 24 h at 37°C with 5% CO2, the medium was replaced with 100 μL extract dilutions of various concentrations. Following 24, 48, and 72 h of culture, 10 μL CCK-8 reagent added to each well and incubated for 2 h in the dark. A microplate reader was used to measure the optical density (OD) at 450 nm (Multiskan FC, ThermoFisher Scientific, USA). The negative control consisted of untreated cells and the medium, whereas the background only consisted of the medium. Equation (3) was used to calculate cell viability (CV):

where
Animal experiments
Animal ethics approval
The animal experiments were carried out in compliance with the Animal Ethics Committee on the Ethics of Animal Experiments’ recommendations for animal care. Every attempt was made to minimize animal suffering.
Full-thickness wound test
After 14 days of acclimatization, male KM mice (

where
Histological analysis
The wound sections were subjected to histological examination. On the third, seventh, and fourteenth days, six mice from each group were sacrificed. All animals were sedated with an intraperitoneal dose of 1% sodium pentobarbital (12 mL/kg). After being preserved in 10% formaldehyde, the target tissues were embedded in paraffin. Hematoxylin & eosin-stained slices were examined (×200) under a microscope (Leica, Wetzlar, Germany).
Statistical investigation
All experiments have been repeated a minimum of three times. Using the statistical program Origin 2019b, a one-way analysis of variance was done, followed by Fisher’s least significant difference test between groups. The data were presented in the form of the mean ± standard deviation. The difference was statistically significant if the
Results
Mechanical properties
The maximal force achieved during the probe’s downward fall is represented by hardness. It gradually increased as the KGM content raised from 0.5% to 2.0%, as shown in Figure 1(a). Cohesiveness is defined as the compression degree between the gel and the probe before the break, which displayed a downward trend as KGM concentrations increased from 0.5% to 2.0% (Figure 1(b)).

(a) Hardness, (b) cohesiveness, (c) water-holding capacity, and (d) swelling degree of hydrogels comprising BSP and KGM with different formulas. *Statistical difference at the 95% confidence interval (
Water-holding capacity and swelling degree
The WHC and SD of hydrogels made using different BSP/KGM ratios are shown in Figure 1(c) and (d), respectively, both of which increased with increasing KGM concentrations and obtained their maximum at a KGM concentration of 2.0%.
An appropriate swelling behavior was expected after transforming the viscous BSP solution into the hydrogel to provide a stable humid environment needed for wound healing. The SD of K increased with time and obtained the maximum (35.3 ± 4.6%) after 24 h of immersion (Figure 1(d)). The expansion curve of BK1-3 and BK2-2 followed the same pattern. After a rapid increase in the first 4 h of immersion, the increment slowed down, and then reached a plateau and maintained until 24 h. However, the SD of BK3-1 decreased slowly with time after an initial increase in the first 4 h.
Thermogravimetric analysis
All samples showed a similar thermal degradation behavior (Figure 2(a)). Following the first stage of moisture loss at low temperatures (<110°C), the primary weight loss occurred at >200°C, due to a complicated process that included the dissolution of glycosidic bonds in polysaccharides, and also sugar unit degradation. The residue mass of 28.6% was for BK1-3, 30.7% for BK2-2 and 24.4% for BK3-1, respectively, higher than K (6.3%) and B (14.3%).

(a) TG curves and (b) XRD spectra of B (lyophilized BSP solution), K (hydrogel KGM), and their blend hydrogels.
X-ray diffraction examination
The XRD spectra of B, K, and blend hydrogel were comparable, as shown in Figure 2(b), with minor differences in their diffraction peaks. All samples were amorphous materials with a broad diffraction peak at about 21.1°. The peak at 12.1° (2
Fourier transform infrared spectral analysis
Figure 3 depicted the FTIR spectra of KGM, BSP, and their blends. In comparison to BSP, no additional peaks were found in the blend hydrogel spectrum, while some differences were observed. The peaks at 2922, 2850, 1732, 1321, and 1246 cm−1 in the BSP spectrum correlating to -CH3 vibration, -CH stretching vibration, -C=O stretching vibration from the -C=O-CH3, -C-O stretching vibration, 3 and -CH asymmetric stretching vibration, respectively, shifted to lower wave numbers when KGM concentration increased. And the max blue shift was obtained in BK2-2. The peak at 1373 cm−1 (methyl’s -CH stretching vibration from -C=O-CH3) shifted to higher wave numbers. The FTIR results indicated the hydrogen bonding formed within -CH3, -CH, -C=O-CH3 and -C-O groups of KGM and BSP molecules rather than chemical reactions occurring between them.

FTIR spectra of K (hydrogel KGM), B (lyophilized BSP solution) and their blend hydrogels with different formulas between 4000 and 400 cm−1.
Microstructural characterization
The morphology of hydrogels was influenced by the ratio of different polysaccharides as shown in Figure 4. The lyophilized BSP solution had a regular laminated structure, with a loose tubular structure arranged in parallel between the adjacent lamellae. Granules and small pores were observed in each layer. Different from B, hydrogel K exhibited a typical porous structure with relatively smooth and rigid walls (~3 μm thick). The pores were loose and uneven in size. When KGM was added to BSP, the structure was strengthened without obvious cracks and phase separation on the surface. The higher the BSP content, the more similar the microstructure was to that of B. The porous structure formed by KGM was gradually regular, with increasing BSP concentrations from 0.5% to 1.5% and obtained the densest structure at the BSP concentration of 1.0% with homogeneous and smooth walls (~1 μm thick). As the microstructure of BK2-2 was much denser and more uniform than other blend hydrogels, it was used for further research.

Microstructure visualization from parallel and perpendicular sections of B (lyophilized BSP solution), K (hydrogel KGM) and their blend hydrogels at magnification 100× and 3000× for the respective images.
Cell viability assay
Figure 5(a) revealed that after 72 h of exposure to BK2-2 and B extracts at concentrations of 1.25, 5.00, and 10.00 mg/mL, CV was not significantly different. Nevertheless, cells exposed to BK2-2 extract exhibited a higher CV compared with those incubated with the B extract at 2.50 mg/mL concentration.

Cytotoxicity assessment against L-929 fibroblasts with the use of (a) B (lyophilized BSP solution), hydrogel BK2-2 (containing 1.0% BSP and 1.0% KGM, w/v) and K (hydrogel KGM) with different concentrations after 72 h of culture, and (b) hydrogel BK2-2 (containing 1.0% BSP and 1.0% KGM, w/v) extracts at different concentrations after 24, 48, and 72 h of culture.
For the series of BK2-2 extract dilution, a similar trend in CV was observed after treating for 24, 48, and 72 h (Figure 5(b)). The CV increased with increasing concentrations of the BK2-2 extract from 1.25 to 5.00 mg/mL, then decreased as the concentration raised to 10.00 mg/mL.
Animal experiments
The wound healing rate in KM mice was determined to explore the impact of hydrogels on wound tissue regeneration (Figure 6(a)). Hydrogel K was not tested, as its wound-healing effect has been proved. 12 Wounds treated with hydrogel BK2-2 and lyophilized BSP solution (B) healed faster than the control group, and no significant difference was observed between them. On day 14, the wounds treated with BK2-2 hydrogel and B were essentially enclosed, while a tiny wound covered with a scab was still observed in the control group (Figure 6(c)).

(a) Wound healing rate in the treatment group and control groups. (b) Hematoxylin & eosin staining of tissues in the treatment and control groups on each observation day. (c) Pictures of a wound in different groups at 0, 3, 7, and 14 days after wounding.
Wound healing is a complicated biological process with several overlapping stages, including hemostasis, inflammation, proliferation, and wound remodeling. Inflammatory cells, epidermal regeneration, and the creation of granulation tissue are important indications of wound healing. In the first few days, all of the wounds displayed bleeding, surrounding tissue edema, and exudation. All groups did not have epidermal closure 7 days post wounding (Figure 6(b)). More neovessels were identified in groups B and BK2-2 than in the control, indicating a beneficial role in the initial stages of wound healing. On Day 14, the wounds in all groups have epithelialized, while the shaped epidermis in the control group was thinner with sparse blood vessels compared with treatment groups B and BK2-2 in the micro.
Discussion
The hydrophilicity of KGM supports the moist environment required for wound healing. As the concentration of KGM raised, so did the WHC and SD of the blend hydrogel. KGM disclosed more hydroxyl groups, as well as glucose and mannose residues, 18 as a result of alkali-induced deacetylation, and numerous connections between these deacetylated molecules created a network structure, while hydrophilic groups in the molecular chain provided plenty of water-binding sites to confer exceptional water retention. In comparison to the hydrogel with added KGM only, BSP noticeably decreased the WHC of the blend hydrogels with increasing concentrations (Figure 1), owing to its defect in water absorption, which is consistent with a recent report. 5 When immersed in water, the three-dimensional network frames comprising KGM became loose as the BSP concentration increased, failing to retain the BSP molecules within it after immersion in water. The dissolving BSP decreased the weight of the residue and caused lower WHC and SD. Therefore, the higher the KGM concentration, the better the moisture retention and water uptake capacity, and the more stable the hydrogel is in a humid environment. However, as an active pharmaceutical ingredient, BSP works only when dissolved in the tissue fluid. Thus, a proper dissolution of the hydrogel was required for wound healing.
It has been proved that combing KGM improved the thermal stability of a blend
19
; the stronger the interactions amongst components, the higher the thermal stability.
20
Similarly, when heated to 350°C, TG analysis revealed a higher residue mass of blend hydrogel than K and B, with BK2-2 having the highest residue (Figure 2(a)), indicating increased thermal stability generated by the interactions between BSP and KGM, and the interactions were much stronger in BK2-2, as confirmed by FTIR with the maximum blue shift obtained in BK2-2 spectrum (Figure 3). Moreover, in XRD spectra (Figure 2(b)), the diminished peak at 12.1° (2
The interactions between BSP and KGM, on the other hand, prevented BSP from dissolving quickly in water, which was weaker than those between BSP and water. Thus, BSP molecules could slowly and steadily release from the blend hydrogel and dissolve in the tissue fluid. Does the interaction between KGM and BSP have an impact on the physiological activity of BSP? Cytotoxicity tests and animal research were carried out. BK2-2 with the densest microstructure was used as the experimental group, and B was used as a positive control.
The CV increased with increasing concentrations of the BK2-2 extract from 1.25 to 5.00 mg/mL (Figure 5). The improved survival may have been due to the bioactivity of dissolved KGM and BSP, both of which promoted cell proliferation. However, the decreased CV at 10.00 mg/mL concentration was caused by the damaged nutritional balance of the medium induced by the excessive concentration of chemicals, as previously reported. 22 In our research, the CCK-8 assay demonstrated that the cells were >95% viable, which was higher than reported BSP-based hydrogels prepared via chemical cross-linking, 9 as well as strong interacted physical cross-linking. 8 The blend hydrogel might meet the essential characteristics of an ideal wound dressing, including high compatibility, as well as non-cytotoxicity to human fibroblasts. 23
In terms of healing capabilities, the blend hydrogel should be comparable to materials formed from single–ingredient BSP. The difference in wound healing rate between groups B and BK2-2 was not significant, both of which were higher than the control (Figure 6). Compared to the control group, the more neo-vessels identified in groups B and BK2-2 on Day 7, their thicker shaped epidermis and more blood vessels on Day 14 in the micro, indicating a beneficial role of BSP-based material in the wound healing procedure; additionally, the healing function of BSP did not appear to be reduced after blending with KGM. When the lyophilized BSP solution was applied to the wound, it partially dissolved and became sticky after absorbing the exudates, whereas the BK2-2 hydrogel could facilitate a proper balance between exudate and hydrogel, and result in the wet and clean wound. It was observed that BK2-2 hydrogels remained intact throughout the day, and their removal from the wound was easy. Compared with composites designed by cross-linking BSP with other polysaccharide,8,24 the BSP/KGM blend hydrogel seemed quite promising because of its outstanding wound healing rate after 14-day treatment (98.79 ± 0.73%). Moreover, the essentially healing wounds are observed in Figure 6(c) which was in accordance with the histological results.
Based on our research, the incorporation of KGM could improve the physical properties of BSP-based material without reducing the biocompatibility, fibroblast viability and healing properties of BSP. Blend hydrogel BK2-2 has potential practical applications in health care. However, more research into the mechanism of BSP release from the blend hydrogel, as well as the detailed wound healing mechanism of BSP/KGM blend hydrogel is required.
Conclusions
In summary, the hydrogel formed by BSP and KGM has the potential to be utilized as a nontoxic dressing facilitating wound healing. It was shown that the improvements in hardness, WHC, and SD were positively correlated with the increasing KGM concentration, because the frame of the hydrogel was primarily formed by KGM, with BSP dispersed uniformly to create the structure of an interpenetrating network. The densest porous structure, as shown in the SEM images, was obtained at a BSP concentration of 1.0%. The major intermolecular interaction between BSP and KGM was hydrogen bonding, as confirmed by FTIR. However, such interaction failed to keep the dissolving BSP molecules within the hydrogel after immersion in water, resulting in lower WHC and SD when BSP concentration was increased. Proper BSP dissolution was necessary for wound healing, whereas excessive dissolution decreased physical performance and broke the structure of the hydrogel BK3-1. Hence, the BK2-2 formulation (containing 1.0% BSP and 1.0% KGM, w/v) with the highest BSP content was chosen as the best. A high degree of compatibility was proved using XRD, TGA and SEM. The CCK-8 assay revealed good biocompatibility and fibroblast viability. The CV increased as the concentration of the BK2-2 extract increased from 1.25 to 5.00 mg/mL. After 72 h of exposure to BK2-2 extract at 2.50 mg/mL, a higher CV was exhibited compared to B, whereas no difference was observed at other concentrations. By accelerating epidermal and blood vessels regeneration, the full-thickness skin defect results illustrated that the treated group healed significantly faster than the control group. Although interactions between BSP and KGM may influence the fast release of BSP, no significant differences in wound healing rates were observed in groups B and BK2-2. Histologically, hematoxylin & eosin staining corroborated the above findings. As a nontoxic component, KGM can improve the physical characteristics and microstructure of the BSP-based material without decreasing its healing activity, and BK2-2 can be used as a promising wound dressing.
Footnotes
Acknowledgements
The authors would like to thank the School of Public Health, Shaanxi University of Chinese Medicine, PR China, for providing equipment.
Author contributions
Conceptualization, JS and BX; original draft writing, JS; methodology and investigation, JS and WZ; software and data curation, XL; validation, formal analysis, and visualization, LD; resources and validation, CM and DG.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Key Research and Development Program of Shaanxi Province [2018SF-290], the Natural Science Foundation of Shaanxi Province [2019JQ-563], school level scientific research project of Shaanxi University of Chinese Medicine [2021GP43], and the Innovative Team Project of Shaanxi University of Chinese Medicine [2019-py01].
