Abstract
Strontium has been widely used in bone repair materials due to its roles in promoting osteoclast apoptosis and enhancing osteoblast proliferation. In this work, synthesis and the effects of Sr2+ doping α-tricalcium phosphate at low-temperature was studied. The setting time and the mechanical properties of α-tricalcium phosphate were controlled by varying the content of Sr2+. The synthesized compounds were evaluated by XRD, SEM, XPS, setting time, compressive strength, SBF immersion, and colorimetric CCK-8 assay. The results showed that Sr2+ can improve the compressive strength and cell activity of calcium phosphate bone cement.
Introduction
Calcium phosphate cement (CPC) has been widely used in the treatment of bone defects1,2 due to its rapid coagulation, excellent bioactivity, biocompatibility, resorbability, and non-toxicity. 3 One of the CPC’s most important solid components is based on α-tricalcium phosphate (α-TCP).
α-TCP is a metastable state of tricalcium phosphate (TCP) at room temperature, its stability range is strongly influenced by ion substitution. At present, α-TCP has become an important component of biodegradable bioceramics and bone repair composite materials, because it is more soluble and quickly convert to calcium-deficient hydroxyapatite during hydration, which is similar to human hard tissue in calcium content, crystallinity, and structure.4–6 It should be noted that the inherent low strength, high brittleness, and inappropriate setting time of CPC limited its applications in load-bearing bone repairment. Lots of methods have been proposed to improve the properties of CPC.7–9 Partial replacement of Ca2+ ions in the calcium phosphate with bioactive inorganic ions is an effective way.10–15
Strontium is a microelement in human bones, 16 it has a strong affinity to bones due to its role in promoting osteoclast apoptosis and enhancing osteoblast proliferation. 17 Many studies have shown that Sr plays an important role in bone metabolism and enhance the mechanical properties. 18 Especially in areas with high metabolic conversion rates, low-dosen Sr has a significant effect in the treatment of osteoporosis. Therefore, Sr has achieved increasing positive results in long-term clinical treatment research and people become interested in introduce it in CPC.19,20 Studies have shown that even the introduction of very little Sr into α-TCP can promote the proliferation and differentiation of osteoblasts and inhibit the formation of osteoclasts. In vitro studies of hydroxyapatite (HA) powder have shown that Sr-doped HA has a greater amount of apatite deposition, while in vivo HA cement studies have shown that the speed of the bone layer formed at the bone cement interface increases.21–23 Therefore, Sr-loaded CPC have been developed to promote bone repair. 24 The most commonly used method to introduce Sr into α-TCP is adding strontium inorganic salts in the preparation process of α-TCP or mixing strontium inorganic salts with α-TCP simply. However, these methods are either expensive or complicated. Besides, the simple mixing process cannot guarantee the uniform distribution of strontium, which will reduce the performance of CPC.25,26
In this work, Sr2+ was introduced into α-TCP by a method of low temperature synthesis. Compared with the traditional methods, the products with smaller particle size and higher reactivity was obtained. The effects of Sr2+ doping on calcium phosphate were evaluated by measuring its setting time, compressive strength, mineralization in vitro, and cytotoxicity.
Experimental procedure
To prepare α-TCP cement, all used chemicals were analytical reagents (Sinopharm, China). Calcium nitrate tetrahydrate (Ca(NO3)2·4H2O), strontium nitrate tetrahydrate (Sr(NO3)2·4H2O), diammonium hydrogen phosphate ((NH4)2HPO4), and β-cyclodextrin (β-CD) are used as raw materials.
The sample according to the molar percentage of Sr2+ to total metal ion content were labeled as α-TCP (0%), 1Sr-α-TCP (1%), 2Sr-α-TCP (2%), 3Sr-α-TCP (3%), 4Sr-α-TCP (4%), 5Sr-α-TCP (5%). First, Ca(NO3)2·4H2O and Sr(NO3)2·4H2O were dissolved in β-cyclodextrin saturated solution (total metal ion concentration of 0.4 M), and then the solution was kept at room temperature for 24 h. The same volume of 0.267 M diammonium hydrogen phosphate solution was slowly added to the above solution, ammonium hydroxide was added with constant stirring to make the pH between 10 and 11. The obtained product was stirred for 30 min and filtered, washed with deionized water and ethanol several times, then dried at room temperature. Finally, the dried powder was calcined at a temperature of 550°C–700°C for 3 h.
Powder X-ray diffraction (10°–40° (2θ), 10° min−1, 40 kV, and 150 mA) data were collected using Ni-filtered Cu-Kα radiation on Rigaku DMAX-2500PC diffractometer. Rietveld refinement of 5Sr-α-TCP sample was performed by the Rietveld method using the software GSAS. 27 The data were collected within 2θ angle range from 10° to 40° with a step size of 1° min−1. The morphology of the synthesized powder was characterized by Hitachi SU-70 field emission scanning electron microscope (SEM) at 15 kV acceleration voltage. Size distribution was determined by dynamic lighting scattering (DLS, Malvern Zetasizer Nano ZSE, England). Specific surface areas (SSA) of the powders were obtained according to the BET method using a JW-BK300D through the gas adsorption measurements. X-ray photoelectron spectroscopy (XPS) analysis was carried out on a ThermoFisher K-Alpha spectrometer using the monochromatic Al Kα excitation source (1486.6 eV).
The setting time was measured using a Gillmore Needles method according to ASTM C266-89 standard. The liquid was a 0.1 M citric acid solution. The solid power was the mixture of 5 wt% HA powder and α-TCP powder with different Sr2+ content. The α-TCP cements were prepared by mixing the powder with liquid under a liquid/powder ratio of 0.5 mL/g. Each formulation was tested three times, and the average value was used as the final result.
The compressive strength of the sample was measured by a electromechanical universal testing machine (CMT5105). The cement pastes were cast into stainless steel moulds (Ф 6 × 12 mm3) and stored in a 37°C, 100% humidity box for 24 h. The specimens were then removed from the moulds and immersed in 0.1 M K2HPO4 solution at 37°C for 6 h. After that, dried overnight. At least three samples were tested, and the compressive strength was expressed as the average of the measured values ± standard error. The porosity of the samples were tested by Archimedes method in deionized water.
The cements were placed into polystyrene flasks containing simulated body fluid (SBF) with ionic concentrations (Na+ 142.0, K+ 5.0, Ca2+ 2.5, Mg2+ 1.5, Cl− 148.8, HPO4− 1.0, HCO32− 4.2, SO42− 0.5 mmol L−1 ) nearly equivalent to those of human blood plasma for 7 and 14 days to evaluate the mineralization behavior of the cements in vitro. 28 The pH of SBF is 7.40 exactly at 36.5°C. The cement pastes were cast into stainless steel moulds to make a cylinder with a size of Ф 6 × 2 mm3 under the pressure of 5 MPa and stored in a 37°C, 100% humidity box for 24 h. Then samples were soaked in SBF at 37°C. After soaking in SBF for the above time, the samples were taken out, washed with water and dried at 37°C for 24 h. The surface of the specimen were examined by XRD and SEM and the sample weight was tested. The experiment was repeated three times to validate the results.
The cytotoxicity of the synthesized samples were evaluated by means of CCK-8 assay using L929 cells. L929 cells (100 μL of 1.0 × 105 cells/mL) were cultured in a 96-well plate at 37°C, 5% CO2 for 24 h. The samples with different content of TCP powders (0.5, 1, 1.5, and 2 mg/mL dispersed in sterile cell medium) were added to 96-well plate and incubated for 24, 48, and 72 h at 37°C. L929 cells treated only with culture media were fixed as a positive control. To each hole, 100 μL CCK-8 was added and incubated at 37°C for 2 h. The absorbance was measured at 450 nm using Elisa reader multiplate (TECAN INFINITE F50). Finally, cell viability was calculated according to the control sample.
Results and discussion
Figure 1(a) shows the XRD spectra of the synthesized 5Sr-α-TCP powder sintered at different temperature. It can be seen that when the temperature was below 650°C, the product was in an amorphous state. When the temperature was 700°C, β-TCP appeared. Therefore, 650°C was chosen as the sintering temperature to prepare Sr2+ doping α-TCP. It can be seen in Figure 1(b) that although the content of Sr2+ doping was different, all the diffraction peaks of the final product remained consistent with α-TCP (ICSD #29-0359). Notably, all the peaks shifted to the lower angles, which is maybe because Sr2+ were incorporated into the α-TCP lattice and cause structural changes. According to Bragg’s law, the XRD diffraction peaks all shifted to the left, indicating that the distance between parallel atomic planes increases. The reason for this larger spacing can only be that the lattice receives the stress of the Sr atoms. The Bragg’s law is as follows:

where

(a) XRD spectrum of 5Sr-α-TCP powder sintered at different temperature and (b) XRD spectra of Sr-α-TCP sintered at 650°C with different doping levels.
The first-principles calculation of the divalent substitution of Ca2+ in TCP polymorphism reported by Matsunaga et al. showed that certain Ca2+ in the α-TCP crystal structure tend to be preferentially substituted. 29 The ion radius of Sr2+ (118 pm) and Ca2+ (100 pm) are very close. Thus, Sr2+ are easily to enter the lattice structure of α-TCP and replace certain position of Ca2+. Since the size of Sr2+ is larger than Ca2+, the lattice parameter of α-TCP would increase when Ca2+ were substituted by Sr2+. Table 1 and Figure 2 show the results of α-TCP and 5Sr-α-TCP investigated using the Rietveld method. The increased in the lattice parameters of 5Sr-α-TCP proves that Sr2+ has been incorporated into the α-TCP crystal structure.
Lattice parameters of α-TCP and 5Sr-α-TCP.


Refined diffraction patterns of 5Sr-α-TCP.
The XPS results of 5Sr-α-TCP are shown in Figure 3. There is only one broad peak in Figure 3(a), on account of Sr3d5/2 (133 ± 0.5 eV), Sr3d3/2 (135 ± 0.5 eV), and P2p (132–133 eV) lines are located close to each other, the broad peak is the overlap of Sr3d and P2p peaks. The typical peak position at 269.5 and 280.1 eV related to Sr3p3/2 and Sr3p1/2 in Figure 3(b). The peaks corresponding to Sr 3d and 3p both proved that the presence of Sr in α-TCP. 30

Sr 3d (a) and Sr 3p (b) XPS spectra of 5Sr-α-TCP.
Figure 4 shows the setting time of calcium phosphate cement. We can see that Sr2+ doping would prolong the setting time of cement. With the increase of Sr2+ content, the setting time increased first and then decreased, reached the maximum when the content of Sr2+ was 3 mol%. The initial setting time and final setting time were 24 and 47 min respectively, which was much higher than that of undoped samples.

Setting time of the synthesized samples.
Table 2 lists the BET specific surface area of α-TCP and Sr-α-TCP. It can be seen from the table that Sr2+ had a significant impact on the specific surface area of α-TCP. The specific surface area of the doping samples first decreased and then increased, and reached a minimum of 16.325 m2/g when the Sr2+ concentration was 3 mol%. In comparison to samples synthesized by conventional methods, the SSA of α-TCP is significantly higher. 31 Higher specific surface area can ensure full contact between powder and liquid during hydration and increase reaction activity. Specific surface area decrease caused by Sr2+ doping extended the setting time.
Specific surface area (SSA) distribution.

The DLS results and SEM images of α-TCP with 0–5 mol% Sr2+ are shown in Figure 5. The undoped sample was mainly composed of elongated rod-shaped particles with a length of 300–500 nm and a width of 100–200 nm. The substitution of Sr2+ for Ca2+ did not change the morphology of α-TCP. The average particle size of α-TCP with 0–5 mol% Sr2+ are shown in Table 3. In contrast, the average size of α-TCP with 0–5 mol% Sr2+ was 400–700 nm. Compare with α-TCP, the average size of Sr-α-TCP was larger. The DLS results also suggested that the presence of Sr2+ increased the grain size and the size distribution was more uniform.
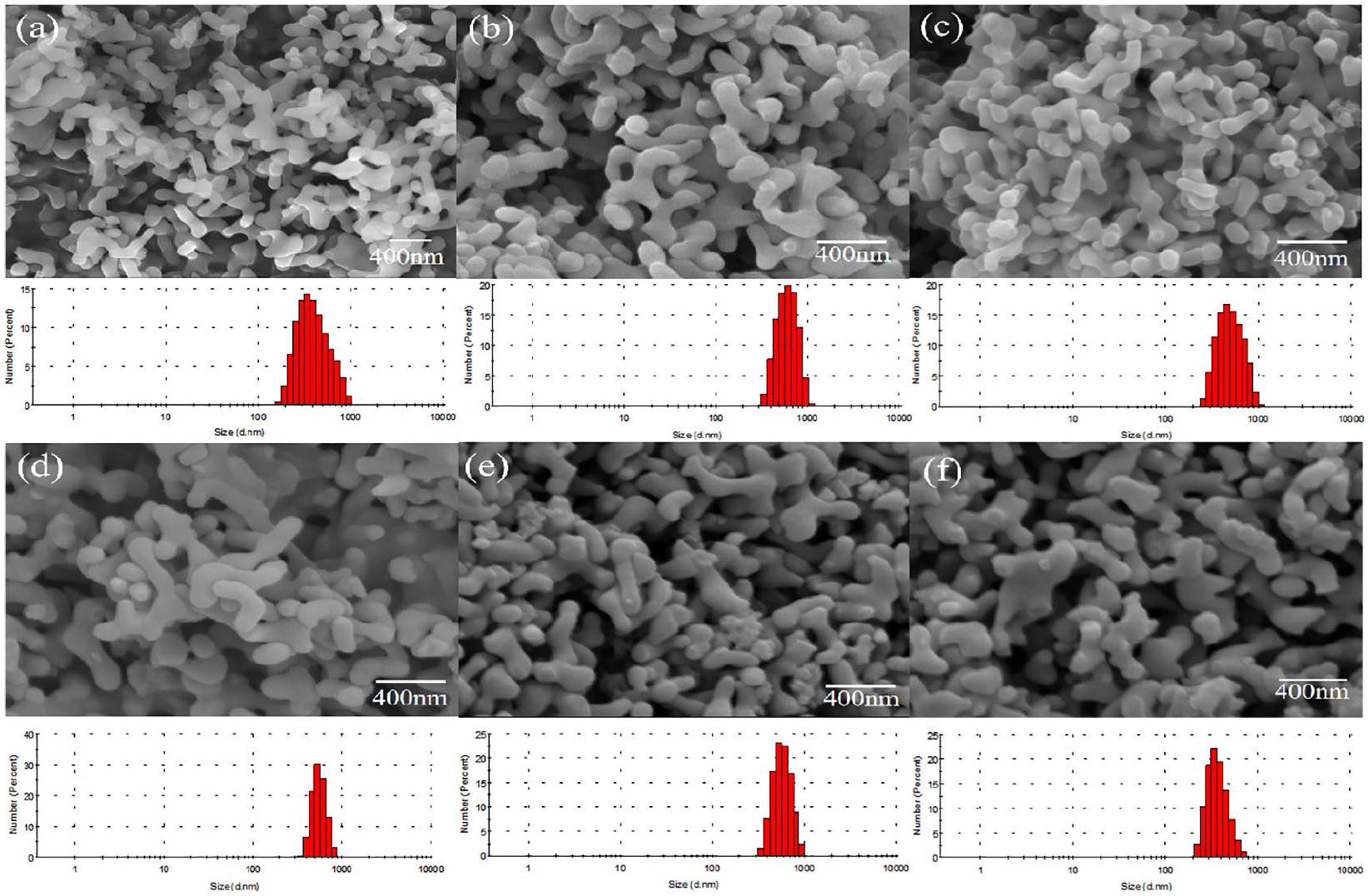
DLS results and SEM images of α-TCP powders without Sr (a), 1 mol% of Sr (b), 2 mol% of Sr (c), 3 mol% of Sr (d), 4 mol% of Sr (e), and 5 mol% of Sr (f) calcined at 650°C for 3 h.
Average particle size.

Figure 6 shows the compressive strength and porosity of α-TCP and Sr-α-TCP cement. The samples doped with Sr2+ had higher compressive strength. 26 With the increase of Sr2+ content, the compressive strength tended to increase first and then decrease. The maximum of 10.8 MPa was reached at 3 mol% Sr2+ content, which was 50% higher than that of undoped samples and reached a strength similar to cancellous bone (10–30 MPa). 32 The changes in compressive strength were related to porosity. The porosity directly reflects the compactness of the material. Generally speaking, a material with a larger porosity has a lower strength, and the relationship between strength and porosity is approximately linear. As showed in Figure 7, most undoped samples had a needle-like morphology, 33 while doped samples tended to retain their original morphology. The structure formed by the combination of the needle-like morphology and the elongated rod-shaped morphology is denser than the structure composed of a single morphology, and affects cement strength through porosity. In addition, the change in morphology also reflects excessive Sr2+ will negatively affect the α-TCP hydration process. 34

Compressive strength and porosity of samples.

SEM images of α-TCP powders without Sr (a), 1 mol% of Sr (b), 2 mol% of Sr (c), 3 mol% of Sr (d), 4 mol% of Sr (e), and 5 mol% of Sr (f) stored in a 37°C, 100% humidity box for 24 h.
The appearance of characteristic peaks associated with hydroxyapatite was noticed in Figure 8(a). The surface of the sample was fully covered by amorphous calcium phosphate on the seventh day, which indicated its excellent biomineralization ability. As the immersion time increases, an increase in the intensity of amorphous calcium was noticed in the XRD pattern. Figure 8(b) shows SEM images of CPC cement surface after immersion in SBF for 7 and 14 days. It can be observed that layered structure of apatite deposited on the sample surface. When the immersion time was 7 days, the entire cement surface was covered with a fine layer of apatite. When the immersion time extended to 14 days the apatite layer on the surface increased. Figure 9 shows the weight gain of samples after immersion in SBF solution for 7 and 14 days. As the immersion time increases, the weight of the sample increases. However, Sr-doped samples will reduce the rate of mineralization, and with the increase of Sr content, this effect becomes more obvious.

(a) XRD spectrum of the α-TCP and 3Sr-α-TCP cement after immersion in SBF solution for 7 and 14 days and (b) SEM images of the α-TCP (I, II) and 3Sr-α-TCP (III, IV) cement after immersion in SBF solution for 7 days (I, III) and 14 days (II, IV).

Weight gain of samples after immersion in SBF solution for 7 and 14 days.
Figure 10(a) shows the results of the cell viability of the sample with 48 h’s incubation time. The cell viability of all samples were determined to be above 80%, which indicated that the powder had good biocompatibility. What’s more, with the increase of Sr2+ concentration, the cell viability increased gradually. In addition, with the prolongation of culture time, the cell viability of samples increased in varying degrees as shown in Figure 10(b). Some of them even exceeded the control group cells, which reflects its good cell activity.

(a) Cell viability profile of cultured L929 cells incubated with 1–5 mol% Sr doped α-TCP for 48 h and (b) the cell viability of 3 mol% Sr doped α-TCP in different hours.
Conclusions
In this study, Sr2+ doping α-TCP were synthesized at low temperature of 650°C. Partial substitution of Ca2+ by Sr2+ leads to the changes in crystal lattice of α-TCP. Sr2+ doping will increase the setting time and compressive strength of α-TCP. Sr2+ will negatively affect the mineralization rate of α-TCP, which makes more difficult for apatite to deposite on the sample surface. The cytotoxicity test not only revealed that Sr2+ did not have any toxic effect, but instead improved cell viability. When the doped amount of Sr2+ was 3 mol%, the sample has the most excellent mechanical properties and biological properties. In addition, on the basis of Sr2+ doping, there is still much room for improvement in compressive strength. Therefore further research is needed for Sr-α-TCP cements and its in vivo application.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Major Basic Research Projects of Shandong Natural Science Foundation (ZR2018ZB0104), China Postdoctoral Science Foundation (2018M632673).
