Abstract
Poly(ε-caprolactone) (PCL) and β-tricalcium phosphate (β-TCP) are promising materials for bone tissue engineering due to their complementary biodegradability and osteoconductivity. However, their individual limitations hinder clinical application. In this study, PCL@β-TCP composite scaffolds containing 5%, 10%, and 20% β-TCP were fabricated via fused deposition modeling (FDM). The effects of β-TCP content on the physicochemical, mechanical, and antibacterial properties of the scaffolds were systematically investigated. Results showed that increasing β-TCP content enhanced hydrophilicity (contact angle reduced from 89.1° to 66.9°), accelerated biodegradation (higher weight loss after 103 days in PBS), and improved antibacterial activity, as evidenced by larger inhibition zones against E. coli and S. aureus. These findings demonstrate that β-TCP incorporation effectively enhances the bioactivity and antibacterial performance of PCL scaffolds, highlighting their potential for bone tissue engineering applications, particularly in non-load-bearing contexts.
Introduction
Bone defects are a prevalent orthopedic condition that often leads to nonunion, delayed union, or nonhealing, resulting in local dysfunction and reduced quality of life for patients.1,2 Following blood transfusion, clinical data indicate that bone grafting is the second most frequently performed tissue transplantation worldwide. 3 Therefore, repairing bone defects remains a significant challenge in orthopedics.
Bone tissue engineering (BTE) focuses on creating scaffolds that simulates the body’s natural extracellular matrix (ECM) and are highly compatible with living tissue. These scaffolds are designed to gradually decompose and be absorbed by the body, helping to heal bone injuries. They support bone repair by encouraging cell growth, reshaping bone tissue, and restoring the bone’s structure and function. 4 Biological scaffolds can be produced through diverse techniques such as electrospinning, 5 phase separation, 6 freeze-drying, 7 gas foaming, 8 and three-dimensional (3D) printing.9,10 Notably, 3D printing, or so-called additive manufacturing, has become a prominent method in bone tissue engineering.11,12
The precise construction of biological scaffold architecture is essential. 13 3D printing technologies integrate advanced biomaterials with biomimetic designs to accurately create scaffolds based on digital data from targeted bone defects. 14 The inherent advantages of 3D printing for BTE stem from several key factors: Initially, 3D printing enables the creation of personalized scaffolds tailored to individual needs. 15 Simultaneously, 3D printing allows for the fabrication of intricate geometric structures, particularly complex hierarchical porous designs that are challenging to produce using traditional manufacturing methods. 16
Over the past decade, significant research efforts in bone tissue engineering have been dedicated to optimizing 3D printing technology for the fabrication of biological scaffolds. 17 For instances, Temple et al. successfully developed a specialized 3D-printed poly(ε-caprolactone) (PCL) scaffold capable of precisely constructing complex anatomical shapes and modulating porosity, emphasizing the remarkable biological properties of these scaffolds, which promote angiogenesis and osteogenesis in human adipose stem cells, thereby providing effective support for bone tissue engineering. 18 Additionally, Dang et al. made metallic copper-TCP scaffolds using 3D printing which effectively killed the cancer cells by generating heat when exposed to near-infrared light. 19 These studies aim to develop scaffolds with optimal mechanical and biological properties to support cell growth and tissue regeneration, demonstrating extensive potential for clinical applications.
Currently, the primary material used for bone repair in clinical practice is autogenous bone grafting, but due to the disadvantages of limited quantity and prolonged operation time,20,21 with the development of integration of medicine and material science, the use of artificial materials to replace autogenous bone grafting has been favored by a lot of treatment solutions. 22 However, traditional metallic bone implants face several challenges, including stress shielding, 23 potential immunogenicity, 24 and a high incidence of donor site complications, 25 limiting their effectiveness in meeting clinical needs.
Currently, degradable polymers like PCL, 26 poly(lactic acid) (PLA), 27 and poly(lactic-co-glycolic acid) (PLGA) 28 are widely utilized in bone tissue engineering. These materials are preferred for their biocompatibility, predictable degradation profiles, and non-toxicity of their degradation byproducts, ensuring no adverse effects on adjacent tissues. 29 PCL is biodegradable, biocompatible, and holds great promise for bone tissue engineering.30,31 Compared to PLA, which has been more extensively studied, PCL features a simpler synthesis process and is cost-effective. 32 However, its slow degradation rate and hydrophobicity can impede cell adhesion and proliferation, which are critical for effective bone defect repair. 30 Thus, blending bioceramics like hydroxyapatite (HAp) and with PCL to create composites is a promising approach to improve the efficacy of PCL in bone regeneration. 33
TCP is a commonly utilized bioactive ceramic, that is, absorbable and exists in three crystallographic forms: α-TCP, β-TCP, and α′-TCP. 34 Among these, β-TCP stands out for its superior hydrophilicity and biodegradability. Its chemical composition is closely akin to the crystalline structure of bone minerals, which has made it a material of extensive research in bone tissue engineering. 34 While HAp is more commonly found in bone tissue, previous studies have shown that β-TCP is more effective than HAp in promoting alkaline phosphatase (ALP) activity and the expression of osteogenesis-related genes in mesenchymal stem cells (MSCs), indicating a stronger capacity to induce osteogenesis. 35
However, the degradation rate of β-TCP is faster than the rate of bone regeneration, which can be detrimental to osteogenesis. 36 Therefore, the PCL@β-TCP composite material emerges as a promising bioscaffold material in bone tissue engineering. It effectively mitigates the individual shortcomings of PCL and β-TCP, while providing a balance of favorable physicochemical and biological properties.37–39
In recent years, a multitude of studies have shown that PCL@β-TCP composites have significant potential for enhancing tissue regeneration.40,41 In a study by Ghezzi et al., PCL@β-TCP composite scaffolds were found to have improved hydrophilicity on their surfaces. 42 Moreover, research by Baykan et al. indicated that these composites can stimulate endochondral bone formation in bone defect sites in Wistar rats. 43 Additionally, another study confirmed that this composite material supports cell adhesion, proliferation, and osteogenesis in MC3T3-E1 cells. 44
Microbial infections can severely compromise cell viability and heighten the likelihood of implant failure in biological scaffolds. Consequently, there is a growing interest among researchers in developing scaffolds from materials with inherent anti-bacterial properties. 45 Many studies have demonstrated that PCL-based biological scaffolds can release biomolecules, such as growth factors or antibacterial substances, to promote tissue regeneration and resist infections. For instance, research by Felice et al. has shown that PCL-ZnO nanofiber scaffolds exhibit antibacterial properties against Staphylococcus aureus, highlighting the potential of such materials in combating infections. 46 Previous studies have demonstrated that β-TCP gradually degrades in the physiological environment, releasing calcium ions (Ca2+) and phosphate ions (PO43−). These ions can alter the chemical composition of the local microenvironment, thereby inhibiting bacterial growth and reproduction. Additionally, calcium ions can bind to the negatively charged groups on bacterial cell membranes, disrupting the normal function of the cell membrane and leading to bacterial death. 47 Moreover, phosphate ions may also help regulate the local pH, creating conditions that are unfavorable for bacterial survival. 45 However, there is a need for more research on the antibacterial properties of PCL@β-TCP composites to fully leverage their potential in clinical settings.
In this study, PCL-based bioscaffolds with 5%, 10%, and 20% mass fractions of β-TCP were fabricated via FDM 3D printing, and their physical and chemical properties were comprehensively evaluated. Drawing on previous research, we sought to two main novel questions: (i) whether incorporating β-TCP to adjust the degradation rate of PCL-based bioscaffolds can better match the bone regeneration rate and (ii) whether β-TCP incorporation enhances the bacteriostatic properties of PCL-based bioactive scaffolds, mitigating surgical graft infection risks and improving clinical applicability, while also attempting to investigate the underlying bacteriostatic mechanisms.
Materials and methods
Materials
The material PCL employed in our study was acquired from SOLVAY (Alorton, IL, USA) and β-TCP powder was procured from Kunshan Chinese Technology New Materials Co., Ltd. (Kunshan, China).
Before incorporation into the composite matrix, the morphology of the commercial β-TCP powders was examined by scanning electron microscopy (SEM) to assess particle size distribution, surface texture, and agglomeration state. These characteristics can influence the dispersion behavior and interfacial bonding between β-TCP and PCL during composite preparation.
3D-printed PCL, PCL@β-TCP scaffolds fabrication
Composite blending: Utilizing a two-roll rubber compression molding open mixing mill machine (by the Guangdong Lina Industrial Co., Ltd., Guangdong, China), we meticulously blended PCL with β-TCP in incremental mass fractions of 5%, 10%, and 20%. The resulting composites were systematically organized into three distinct experimental groups, designated as PCL@β-TCP5, PCL@β-TCP10, and PCL@β-TCP20, each reflecting the respective mass fraction of β-TCP incorporated. A control group consisting of pure PCL was also established.
Preparation of composite filaments: A granulator (by the Henan Xingda machinery Co., Ltd., Henan, China) pelletized the mixed materials. To ensure compatibility with the 3D printer, the mixed material was processed into 3D-printable filaments with a twin-screw extruder (by the Thermo Fisher Scientific, Bremen, Germany).
3D printing of scaffolds: A digital model of the composite biological scaffold with a cylindrical shape, 15 mm in bottom diameter, 3 mm in height, and 50% porosity was designed using SolidWorks 2022 software. Then, scaffolds were printed from the prepared composite materials via a FDM printer (by the Shenzhen Snapmaker Technologies Co., Ltd., Shenzhen, China).
Printing parameters were set as follows: the printing speed was 13 mm/s, the room temperature was kept at 16 °C, the nozzle temperature was 80 °C, and the heated bed temperature was 45 °C.
Morphological characterization
Macromorphology characterization: The dimensions of each scaffold, including diameter and height, were accurately measured using vernier calipers (by the Shanghai Tools Factory Co., Ltd., Shanghai, China), while their weight was determined with a precision balance (by the Kunshan Science Instruments Co., Ltd., Kunshan, China).
Micromorphology characterization: Images of PCL@β-TCP scaffolds were captured at a magnification of 70× using a scanning electron microscopy (SEM; by the HITACHI, Tokyo, Japan). Additionally, the microstructure of β-TCP powder was observed at various magnifications: 1,000×, 2,000×, 5,000×, and 10,000×.
X-ray diffraction (XRD) characterization
Test samples with 20 mm in diameter and 1 mm in thickness were prepared from PCL@β-TCP composite materials containing different mass fractions of β-TCP using a plate vulcanizer (by the Guangdong Lina Industrial Co., Ltd., Dongguan, China). Measurement conditions were set to 40 kV voltage, 15 mA current, 5°–40° 2θ range, and 4°/min scanning speed. Data were analyzed with XRD software MDI Jade 6 and plotted using Origin 2022.
Melt flow rate (MFR) test
The melt flow index of PCL@β-TCP composites with different β-TCP concentrations was assessed by heating to 125 °C under a 2.5 kg load in a melt flow rate tester. (Chengde Dajia Instrument Co., Ltd., China), following the ISO 1133-1 standard.
Differential scanning calorimetry (DSC)
At a constant heating rate, four groups of materials with varying β-TCP mass fractions were heated above their melting points. DSC machine (by the TA Instruments, New Castle, DE, USA) determined the melting points of composites with different proportions. A second heating cycle was performed to eliminate thermal history.
Contact angle test
Test samples were made via hot pressing mixed material particles using a plate vulcanizer. A contact angle goniometer (by the Shanghai Zhongchen Digital Technology Equipment Co., Ltd., Shanghai, China) measured each sample’s contact angle. Three samples per group were analyzed, with the contact angle determined through image processing.
Mechanical test
The granular samples were melted and poured into molds to make cubic specimens. A multifunctional testing machine (SHIMADZU, Japan) was used by GB/T 1040.1-2018 to apply tensile force. Displacement-load curves were plotted by software Origin 2019b, and the modulus of elasticity was calculated from their linear parts.
The compressive modulus was calculated using the formula below:
Cm (MPa) is the compressive modulus, s (N/mm) is the slope of displacement-load curve, and r (mm) is the diameter of scaffold, h (mm) is the height of scaffold.
In vitro degradation tests
Scaffolds were weighed, disinfected with 75% ethanol for 30 min, air-dried, and immersed in 20 mL of PBS (by the Beijing Leagene Biotechnology Co., Ltd., Beijing, China) in 50 mL sterile centrifuge tubes at 37 °C. After 103 days, they were rinsed, dried at 37 °C, and re-weighed. The weight loss rate was calculated using the formula below:
Δ (%) is the weight loss rate, W0 (g) is the initial scaffold weight, and Wt (g) is the weight after 103 days in PBS.
Antibacterial properties characterization
Zone of inhibition test
Bacterial samples (Escherichia coli and Staphylococcus aureus) from glycerol stock solutions (Aoqi Ke-hua Medical Supply Chain Management Service Co., Ltd., Tianjin, China) were cultured overnight in 50 mL of LB agar medium (Aladdin Industrial Corporation, LA, USA) at 37 °C with shaking at 220 rpm.
After overnight culture, spread 10–20 μL of culture on LB agar plates. Place scaffolds from per group at the center. Incubate plates at 37 °C for 24 h, then observe antibacterial zones around scaffolds and measure their diameter and clarity.
Statistical analyses
Three independent samples from each group were analyzed, with test results reported as the mean ± standard deviation (SD). The statistical evaluation of these results was performed using one-way analysis of variance (ANOVA), followed by Tukey’s post-hoc test for multiple comparisons. Data normality and homogeneity of variances were verified using the Shapiro–Wilk and Levene’s tests, respectively. When assumptions of ANOVA were not met, a non-parametric Kruskal–Wallis test was applied. A p = 0.05 or less was set as the threshold for statistical significance. All statistical analyses and graphing were conducted using Origin.
Results
Morphological observations
Figure 1 displays the SEM micrographs of β-TCP powder at magnifications of 1,000×, 2,000×, 5,000×, and 10,000×. The powder predominantly consists of irregularly shaped spherical particles or agglomerates that are uniformly distributed. Particle sizes vary from 0.15 to 5.0 μm.

SEM micrographs of β-TCP powder at magnifications of (a) 1000×, (b) 2000×, (c) 5000×, and (d) 10,000×.
Porosity is key to bone scaffolds’ permeability and mechanical properties. It’s usually designed to mimic human cancellous bone’s 50%–90% range. In our study, scaffolds had 50% porosity. Figure 2 shows PCL@β-TCP composite bone scaffolds are cylindrical, about 15 mm in diameter and 3 mm high. Higher β-TCP concentrations changed their color from white to ivory, indicating low variability and confirming the high reproducibility of 3D printing for PCL-based bone scaffolds.

Representative images of macromorphology and SEM observations per group: (a) pure PCL scaffolds, (b) PCL@β-TCP5 scaffold, (c) PCL@β-TCP10 scaffold, and (d) PCL@β-TCP20 scaffold.
Concurrently, we examined the SEM morphology of PCL@β-TCP scaffolds with varying mass fractions of β-TCP. At 70× magnification, the PCL@β-TCP composite scaffolds displayed a smooth and flat surface around the pores, closely aligning with the intended design. Comparative analysis of the images (Figure 2) indicates that the surface roughness of the scaffolds modestly increased with higher β-TCP content.
The observed increase in surface roughness is attributed to the micro-nano porous structure of β-TCP particles on the composite surface. This leads to an inhomogeneous and slightly rough texture within the scaffolds’ pores, which benefits the interaction between the scaffolds and bone tissue, stimulating cell adhesion, growth, migration and differentiation through increased mineralization.48–50
X-ray diffraction (XRD) results
The XRD curves of the composite scaffolds are shown in Figure 3. PCL is characterized as a semi-crystalline polymer, whereas β-TCP is a crystalline material. The XRD analysis shows clear, distinct peaks for PCL and specific diffraction peaks for β-TCP, notably the (220) and (311) crystallographic planes.51–53 In the composites comprising PCL and β-TCP, the characteristic peaks of PCL are consistent across various compositions, with no additional peaks detected. This absence of new peaks indicates that no new crystalline phases are generated within the PCL@β-TCP composite material.
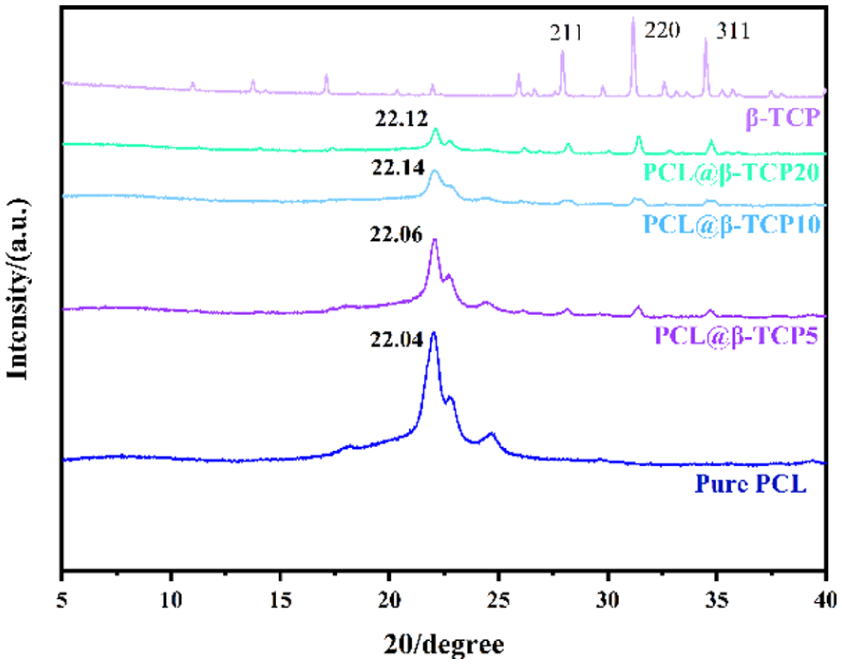
XRD curve of the different materials.
MFR of PCL@β-TCP composite material
MFR is a critical factor in the 3D printing of PCL-based scaffolds. MFR influences both the accuracy of the printed components and the adhesion between layers. Specifically, a larger MFR reduces nozzle clogging, while an excessive flow can compromise the quality of the final product. 54
As detailed in Figure 4, our study found that the incorporation of β-TCP into PCL significantly reduces the material’s fluidity, resulting in a marked decrease in MFR. Additionally, our research indicates that the influence of β-TCP on MFR is not uniform; as the concentration of β-TCP increases, the MFR progressively declines.
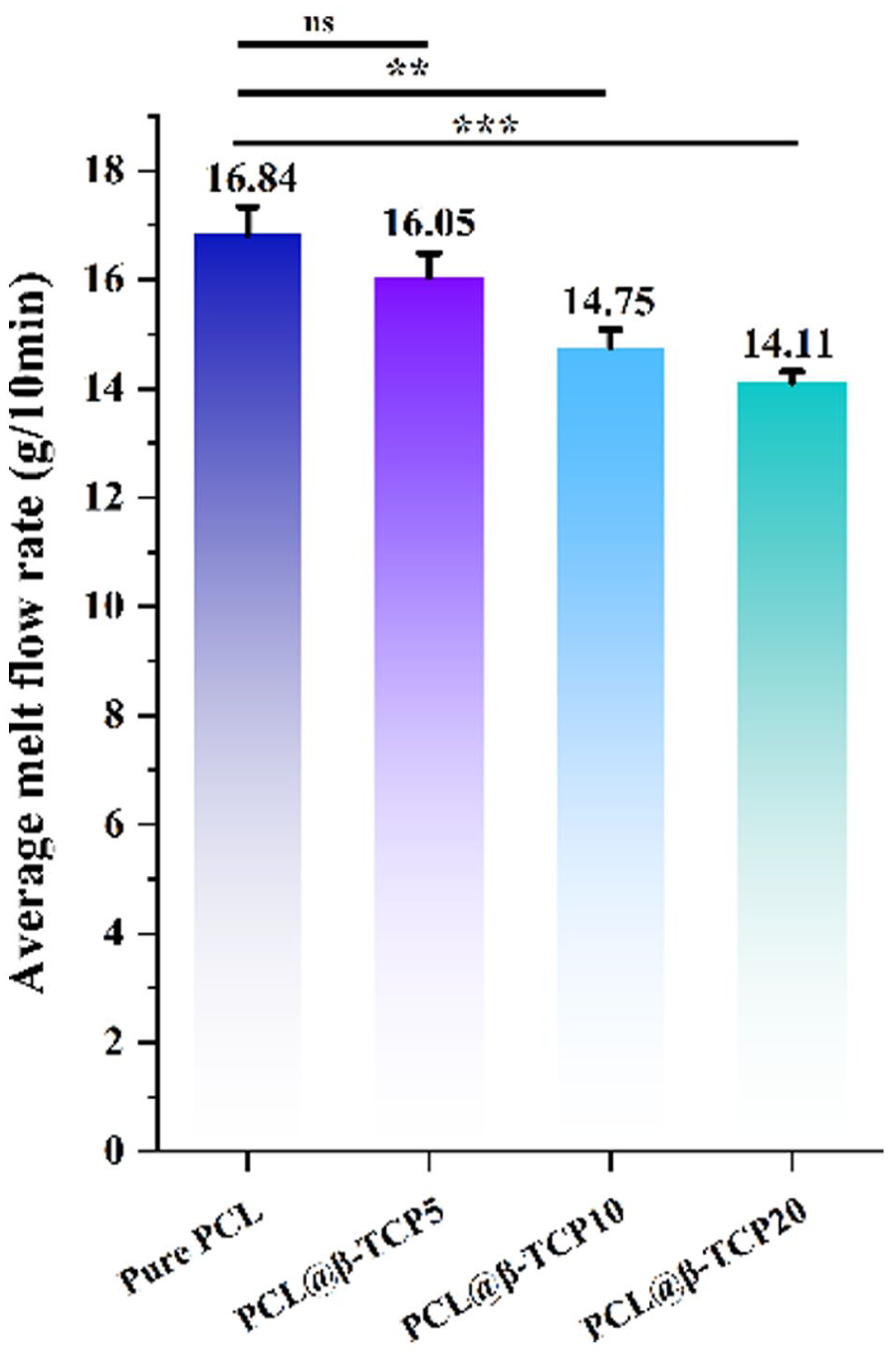
Average melt flow rate changes of PCL@β-TCP materials depend on the incorporation of β-TCP. Data are shown as mean ± SD. n = 3 samples/group. One-way ANOVA.
A reduced MFR might point to production challenges such as injection molding difficulties, thereby restricting the material’s applicability in industrial fields requiring high fluidity. Despite the lower MFR, the PCL@β-TCP composites maintained their ability to undergo key processing steps like composite pelletizing, composite filament extrusion, and 3D printing, with a satisfactory level of proficiency.
DSC results of PCL@β-TCP composite materials
Figure 5 presents the DSC curve of PCL@β-TCP composites with different β-TCP content, which records the heat flow changes with temperature. During the melting process, the DSC curve will display a distinct peak of heat absorption, and the temperature at the peak corresponds to the melting temperature. The DSC curve shows that as β-TCP content increases, the melting point of the composite material rises slightly, from 54.02 °C (pure PCL) to 54.74 °C (20 wt.% β-TCP composite). This is likely due to the unique molecular interactions formed between PCL and β-TCP upon mixing.
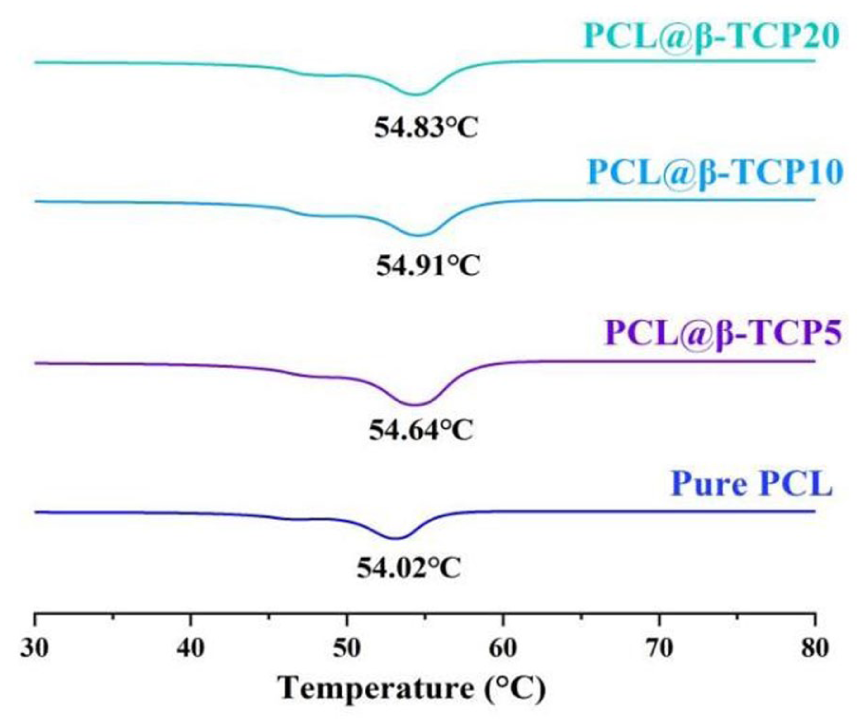
DSC curves display the melting points of PCL@β-TCP materials with varying β-TCP content.
Hydrophilicity test results
The water contact angle is a parameter that measures the hydrophilicity or hydrophobicity of a material, where its size is inversely proportional to the hydrophilicity or hydrophobicity of the material. The smaller the water contact angle, the better the hydrophilicity; whereas a larger angle indicates higher hydrophobicity. Figure 6 shows the results, which measure hydrophilicity via water contact angles of PCL scaffolds with different β-TCP mass fractions. The data demonstrate that the hydrophilicity of PCL@β-TCP composites increases with higher β-TCP content. The PCL@β-TCP group containing different β-TCP mass fractions showed significantly higher hydrophilicity compared to the pure PCL group (p < 0.05). Specifically, the water contact angle decreases from 89.10° ± 1.00° for pure PCL to 66.90° ± 1.10° for the PCL@β-TCP20 composite. This reduction in the water contact angle indicates a strong positive correlation between the hydrophilicity of PCL@β-TCP composites and the β-TCP content.

Contact angle test results. Data are shown as mean ± SD. n = 3 samples/group. One-way ANOVA.
The result of rheology
Rheological properties are essential for the application of materials suitable for 3D printing. Thus, this study used a heating rheometer to investigate the rheological behavior of materials between 75 °C and 125 °C.
In rheology, the storage modulus (G′) measures the elasticity of materials, while the loss modulus (G″) measures the viscosity. Figure 7(a) shows the storage modulus of the composite material declines with rising temperature, signifying a reduction in its elasticity. Moreover, the addition of β-TCP leads to a higher storage modulus, indicating greater elasticity.

The result of rheology: (a) storage modulus (G′), (b) loss modulus (G″), and (c) loss tangent (tan δ).
Figure 7(b) illustrates a trend in the loss modulus with temperature that mirrors the behavior of the storage modulus. The loss modulus increases with the rising β-TCP mass fraction, indicating β-TCP promotes the viscosity of composite material as well.
Interestingly, the loss modulus curve of PCL@β-TCP20 shows a trend of increasing at low temperatures (75 °C–85 °C) followed by decreasing at high temperatures (85 °C–125 °C), suggesting that the incorporation of β-TCP particles influences the rheological characteristics of the PCL matrix. β-TCP particles may serve as physical cross-linking points at lower temperatures, contributing to the material’s viscosity. As the temperature escalates, these cross-linking points may become disrupted, resulting in a viscosity reduction.
Achieving a good balance between material elasticity and viscosity is crucial for high-quality 3D printing. It was observed that G″ is higher than G′ (Figure 7(a) and (b)), indicating that the viscous component dominates over the elastic component in PCL and PCL@β-TCP composites. The loss tangent (tan δ) is the ratio of G″ to G′ (G″/G′), showing the materials’ viscoelastic transition. Figure 7(c) presents that in the entire temperature range, PCL@β-TCP20 has the highest tan δ, while PCL@β-TCP5 has the lowest, and PCL@β-TCP10 and pure PCL fall in between. This suggests that composites with a low mass fraction of β-TCP exhibit more elastic behavior than pure PCL due to interactions between β-TCP and PCL, while viscous behavior remains dominant with higher β-TCP content due to the physical cross-linking effect of the particles.
Modulus of compressibility
This study evaluated the compressive modulus of PCL and PCL@β-TCP composite scaffolds using a multifunctional material testing machine. The objective was to evaluate the effect of β-TCP on the mechanical properties of these composites and to determine their suitability as bone-mimicking materials.
Figures 8 and 9 shows the displacement-load curve of PCL@β-TCP composite scaffolds with varying mass fractions of β-TCP under static tensile load. Table S1 shows the compression modulus calculated according to equation (1) for each group of materials, which is a key indicator of the material’s mechanical properties.

(a) Compressive modulus of composite materials and (b) compressive modulus bar chart.

Scaffolds degradation rate of pure PCL, PCL@β-TCP5, PCL@β-TCP10, and PCL@β-TCP20.
The results reveal the incorporation of β-TCP increases the modulus of elasticity of PCL-based scaffolds. However, none of the composites meet the rigorous mechanical criteria required to accurately replicate cortical bone. The compressive modulus of human cortical bone ranges from 1.2 × 104 to 1.8 × 104 MPa 55 ; the highest compressive modulus was found in the PCL@β-TCP20 group samples, at 2.7130 × 103 MPa. This value is comparable to the compressive modulus of human cancellous bone, which ranges from 1 × 102 to 5 × 103 MPa.55,56
Composite scaffolds degradation in vitro
Bone typically requires 6–12 weeks for significant healing, with the rate of recovery influenced by factors such as age, gender, injury location, severity, and type. 57 The degradation rate of bone scaffold materials should ideally match the rate of bone healing. Previous studies have demonstrated that PCL degrades at a slower rate than bone heals.58,59 To evaluate the biodegradability of bioscaffolds, we measured the weight loss of a 50% porous PCL@β-TCP composite scaffold immersed in PBS for 103 days. Table S2 and Figure 9 present the weight loss data for the PCL scaffold over 103 days. The results show that an increased β-TCP content correlates with a higher degradation rate, indicating that the addition of β-TCP significantly enhances the biodegradability of PCL-based scaffolds.
Antibacterial effect
Figure 10 and Table S3 show that β-TCP addition enhances scaffold antibacterial activity. Higher β-TCP levels increase inhibition of both Gram-positive and Gram-negative bacteria, as measured by inhibition zones on LB agar plates inoculated with E. coli and S. aureus.
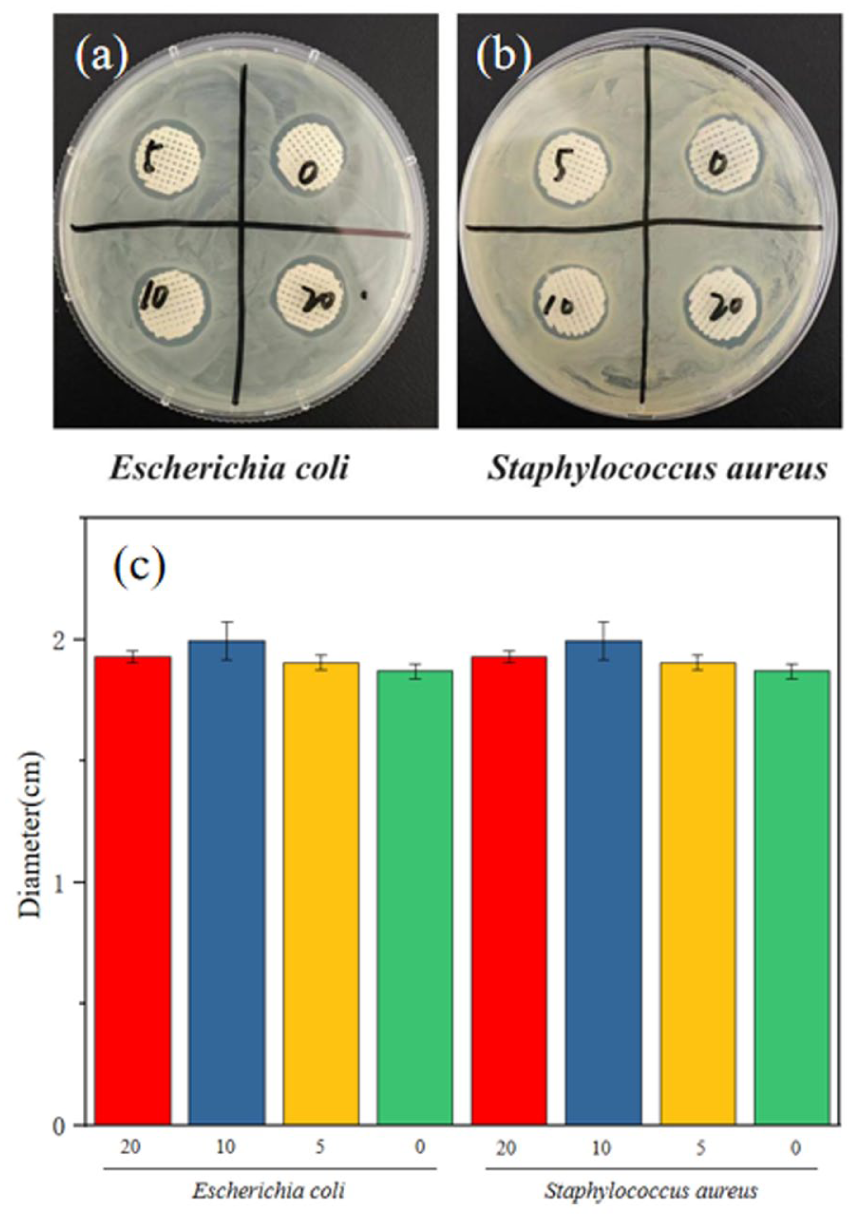
Antibacterial activity of composite scaffolds against E. coli (a) and S. aureus (b), (c) bar chart of the diameter of the antibacterial zone. The numbers indicate different groups: 0 indicates the pure PCL group and 5, 10, and 20 denote the composite scaffolds (PCL@β-TCP5, PCL@β-TCP10, PCL@β-TCP20) experimental groups.
Discussion
Bone defects, especially prevalent among the elderly, represent a significant global health challenge.60,61 While bone grafts are the traditional choice for large segmental defects, they come with notable drawbacks. Autologous grafts face limitations in availability and cause donor-site morbidity, 62 whereas allogeneic grafts risk immune rejection and disease transmission. 1 Biodegradable scaffolds have gained prominence as a promising alternative for bone repair.63,64 For weight-bearing applications, ideal bone-engineered scaffolds need to be bioactive, possess interconnecting porosity, and have sufficient strength. 65
PCL is widely used for bone tissue scaffolds due to its biocompatibility and biodegradability, but its hydrophobic properties are a disadvantage. 66 β-TCP is a bioceramic material with a mineral composition similar to bone and teeth. 34 It is biodegradable and has the potential for bone tissue engineering, but lacks sufficient mechanical strength.34,67 Therefore, forming composites with other biopolymers is advantageous. 68
Many studies have confirmed that PCL@β-TCP composite bone scaffolds have good induced osteogenic activity with certain mechanical strength: Niu et al. found that Large PCL@β-TCP artificial bone effectively promoting MC3T3-E1 cell adhesion, proliferation, and new bone formation. 44 Shi et al. conducted β-TCP scaffold coated with PCL as biodegradable materials for dental applications. 69 Another study showed that spinal fusion cages with different PCL: beta-TCP ratios all met the mechanical properties of human cancellous bone. 70 The most common manufacturing method for PCL@β-TCP bioscaffolds is 3D printing, which allows precise design and manufacture of bone scaffolds tailored to the patient’s specific anatomy and bone defects 65 (Table S4).
In this study, PCL@β-TCP bioscaffolds with different β-TCP mass fractions (5%, 10%, and 20%) and 50% porosity were fabricated using FDM 3D printing technology. Their macroscopic and microscopic morphologies, material phases and structures, rheological, mechanical, biodegradation rates, and hydrophilic properties were comprehensively evaluated. The scaffolds showed excellent size consistency, demonstrating the high precision and reproducibility. β-TCP particles were uniformly dispersed in the PCL matrix, and the surface roughness of the β-TCP bioscaffolds was slightly enhanced, which is conducive to osteoblast adhesion and new bone formation. 71 XRD results showed that any new crystals were not generated by adding β-TCP to PCL-based scaffolds, indicating a physical mixture without chemical bonding. The crystal structure of β-TCP was retained, which is beneficial for promoting osteoblast adhesion and mineralization. Rheological results showed that β-TCP incorporation affected the rheological properties of the PCL matrix, enhancing elastic recovery and reducing high-temperature flow, which is beneficial for FDM 3D printing. However, high β-TCP mass fraction (20%) materials maintained viscous behavior at elevated temperatures, affecting flow and forming. In vitro degradation experiments showed that β-TCP incorporation increased the degradation rate of PCL-based scaffolds. The 20% β-TCP scaffold degraded 7.42% in 103 days, balancing biodegradation and osteogenesis rates. Water contact angle assays showed that β-TCP significantly improved the hydrophilicity of PCL-based bioscaffolds, facilitating cell adhesion and osteogenesis, but also increasing the risk of hydrophilic bacterial infections. One of the major limitations of the current application of bone scaffolds is the risk of infection associated with grafting surgery. 45 In order to investigate whether the increase in hydrophilicity brought about by the incorporation of β-TCP would diminish the antimicrobial effect of the PCL-based scaffolds, the present study was carried out to conduct the Bacterial Circle of Inhibition assay. Antimicrobial experiments showed that the addition of β-TCP increased the resistance of PCL-based bioscaffolds against both Gram-positive and Gram-negative bacteria, and the resistance was comparable to that of the two bacteria. It has been shown that hydrophilic scaffolds are able to form a water film to prevent the adhesion of hydrophobic bacteria such as Gram-negative bacteria. As for its resistance to hydrophilic bacteria such as Gram-positive bacteria, the researchers guessed that the metal ions (e.g. Cu2+, Ag⁺) that can be released by β-TCP can bind to proteins and enzymes on the bacterial cell membrane, disrupting their structure and function and inhibiting bacterial growth and reproduction, in conjunction with the previous studies.72,73 Future research will explore this mechanism further using Inductively coupled plasma-mass spectrometry (ICP/MS) experiments. Based on the current research results, we plan to first test the cell compatibility and cytotoxicity of the scaffold, and then focus on animal model experiments to evaluate the bone regeneration efficiency, biodegradation kinetics, and biocompatibility of the scaffold in critical size bone defect models of rats or rabbits, in order to verify its clinical translational potential. The research content includes long-term implantation studies, monitoring new bone formation, angiogenesis, and immune responses. At the same time, due to the mechanical strength of the scaffold not being able to meet the needs of load-bearing bone repair, we plan to introduce nano hydroxyapatite to enhance the interfacial bonding strength of the composite material, and adjust the printing model to design a structure with better load-bearing effect, thereby expanding the application prospects of this study in bone tissue engineering.
Conclusions
Our study employed FDM 3D printing technology to fabricate porous PCL@β-TCP composite scaffolds and comprehensively evaluated their physical, chemical, and mechanical properties, in vitro degradation, and antibacterial efficacy to assess their suitability as bone defect fillers. Our results demonstrated that the incorporation of β-TCP significantly enhanced PCL-based scaffolds’ bone tissue regenerative potential. This bioceramic is renowned for its osteoconductivity, biocompatibility, biodegradability, and ability to promote extracellular matrix component formation. In vitro degradation studies revealed that the scaffold’s degradation rate increased with higher β-TCP content, suggesting that β-TCP content can be modulated to tailor degradation and ion release kinetics, potentially enabling controlled drug release. Compression testing indicated that while the PCL@β-TCP composite’s mechanical properties were not sufficient for load-bearing bone repair while for cartilage repair. Their hydrophilic surface, indicated by contact angle measurements, promotes cell adhesion and proliferation. In addition, antibacterial tests show that the scaffolds’ antibacterial efficacy against both Gram-positive and Gram-negative bacteria increases with higher β-TCP content.
In summary, our study highlights the potential of PCL@β-TCP composite scaffolds for bone tissue engineering applications, particularly in non-load-bearing contexts. Future research should focus on elucidating the scaffolds’ post-degradation performance and specific cellular effects to further optimize their clinical utility. Additionally, this study investigated the impact of surface hydrophilic modification of PCL@β-TCP scaffolds on their bacteriostatic properties. We suggest that β-TCP/PCL scaffolds may have antibacterial effects via two pathways: enhanced surface hydrophilicity and metal ion release. Further experiments are needed to confirm this mechanism. However, the absence of in vivo studies in this paper limits the clinical applicability of the findings, which are anticipated to be addressed in future research.
Supplemental Material
sj-docx-1-jbf-10.1177_22808000261421820 – Supplemental material for Preparation and characterization of PCL@β-TCP composite antibacterial scaffolds by additive manufacturing
Supplemental material, sj-docx-1-jbf-10.1177_22808000261421820 for Preparation and characterization of PCL@β-TCP composite antibacterial scaffolds by additive manufacturing by Chen Zhang, Yi Qi, Binying Dai, Runyi Lin, Jinchao You, Yaomei Wang, Shunxue Xing, Yuansong Ye and De-Jing Li in Journal of Applied Biomaterials & Functional Materials
Footnotes
Abbreviations
The following abbreviations are used in this manuscript:
PCL Poly(ε-caprolactone)
β-TCP β-Tricalcium phosphate
BTE Bone tissue engineering
ECM Extracellular matrix
3D Three dimensional
PLA Poly(lactic acid)
PLGA Poly(lactic-co-glycolic acid)
HAp Hydroxyapatite
ALP Alkaline phosphatase
MSCs Mesenchymal stem cells
Ca2+ Calcium ions
PO43− Phosphate ions
FDM Fused deposition modeling
ICP/MS Inductively coupled plasma-mass spectrometry
Author contributions
Conceptualization: Chen Zhang. Data curation: Yi Qi and De-Jing Li. Formal analysis: Chen Zhang. Funding acquisition: Chen Zhang and De-Jing Li. Investigation; Yi Qi, Binying Dai, and Yaomei Wang. Methodology: Yi Qi, Binying Dai, and Runyi Lin. Project administration: Chen Zhang and De-Jing Li. Resources: Chen Zhang. Software: Jinchao You. Supervision: Chen Zhang and De-Jing Li. Validation: De-Jing Li. Visualization: Binying Dai, Runyi Lin, Yaomei Wang, and Yuansong Ye. Writing – original draft: Chen Zhang, Yi Qi, Binying Dai, Runyi Lin, Jinchao You, Shunxue Xing, and Yaomei Wang. Writing – review and editing: Chen Zhang, Yuansong Ye, and De-Jing Li.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Fujian Provincial Department of Science and technology joint innovation project (grant no. 2023Y4021), National Natural Science Foundation of China (grant no. 22205090), Natural Science Foundation of Fujian province (grant no. 2022J05232), Fuzhou Overseas Chinese Talent Project (grant no. 2025-ZD-052).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
All relevant data are within the manuscript and its additional files.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.


