Abstract
Postmastectomy breast reconstruction is limited by radiotherapy-induced tissue damage, as silicone implants are prone to capsular contracture, and autologous adipose grafts are limited by resorption and necrosis. In this study, two biomaterials were developed: an injectable decellularized omentum hydrogel-adipose composite (Adipose-dECM) using decellularized omentum hydrogel (dECM) bioactivity for tissue integration and an alginate-reinforced dECM hydrogel (Alg-dECM) for mechanical resilience. O-dECM, Adipose-dECM, Adipose, Alg-dECM, and Silicone were compared in a subcutaneous evaluation in female SD rats (n = 30). Half of the rats underwent radiotherapy (28 Gy) on day 16. The recorded outcomes included small-amplitude oscillatory shear rheology, scanning electron microscopy (porosity), enzymatic mass retention (collagenase), fibrous capsule thickness, inflammatory cell density, the ratio of type I to type III collagen, and angiogenesis. Adipose-dECM showed early postradiotherapy volume retention, although the results were not statistically significant. However, long-term retention decreased to 30.75% on day 50. Compared with the other implants, Adipose-dECM had the lowest inflammatory infiltration and reduced collagen I deposition, although its capsular thickness was similar. Enhanced angiogenesis was detected in Adipose-dECM, with significantly greater CD31+ areas in the peri-implant tissue (1.31% vs 0.10%, p < 0.0001) and septa (0.60% vs 0.07%, p < 0.0001). After radiotherapy, the CD31 level remained elevated in peri-implant regions (0.84% vs 0.34%, p = 0.0010) and septa (0.29% vs 0.06%, p = 0.0003). Adipose-dECM enhanced radiation tolerance through anti-inflammatory modulation and angiogenesis. Nevertheless, its long-term volumetric stability was substantially inferior to that of silicone, indicating the need for material-level strategies to slow degradation while preserving bioactivity. Adipose-dECM therefore shows promise as a radiation-compatible bioactive scaffold for breast reconstruction but requires further optimization for durable clinical translation.
Keywords
Introduction
Breast cancer remains one of the most commonly diagnosed cancers worldwide. 1 Despite improved survival with surgery and adjuvant chemotherapy and/or radiotherapy, postmastectomy sequelae and psychosocial morbidity continue to indicate an unmet need for durable, safe breast reconstruction. 2 Contemporary reconstruction relies primarily on either alloplastic implants or autologous tissue. Silicone implants allow the use of standardized procedures and provide predictable aesthetics 3 but are limited by capsular contracture (approximately 15%–30%, which increases to >40% with postmastectomy radiotherapy).4,5 In addition, conventional two-stage expander–implant approaches do not abrogate radiotherapy-associated complications. 6 Autologous fat grafting avoids a classic foreign-body response but results in variable resorption (30%–70%) and radiation-related adipose necrosis.7,8
Decellularized extracellular matrix (dECM, material) hydrogels retain their native ultrastructure and bioactive cues that modulate immune responses and support angiogenesis, thereby creating a proregenerative microenvironment.9–11 dECM systems with tunable gelation and stiffness values near soft-tissue values, particularly those involving adipose and omentum-derived materials, can sustain adipocyte viability, enhance neovascularization, and improve early volume retention.12–15 Nevertheless, omentum-derived dECM hydrogels show limited resistance to radiation-related stress and relatively rapid in vivo degradation, and current designs seldom address the specific demands of irradiated tissues.16–19 Accordingly, we sought to engineer dECM-based composites tailored for irradiated soft tissues.
Although the clinical translation of dECM remains limited, early feasibility has been reported for dECM used as an injectable filler or patch-type adjunct in implant-based breast reconstruction.20,21 Nevertheless, prospective clinical data for injectable omentum-derived dECM in irradiated settings are lacking. We therefore engineered an injectable, thermoresponsive adipose–omentum dECM composite (Adipose-dECM) and an alginate-shelled omentum dECM hydrogel implant (Alg-dECM), the latter of which provided shape fidelity via ionic crosslinking. 22 We hypothesized that Adipose-dECM would exhibit improved volume retention and favorable host responses under radiotherapy. To test this hypothesis, we compared volumetric retention and host-tissue responses—including fibrous capsule formation, inflammatory infiltration, collagen phenotype (type I/III ratio), and microvascularization—between Adipose-dECM and relevant control materials in both nonirradiated and irradiated animals.
dECM hydrogels have been explored for use in soft-tissue repair, yet most relevant reports lie outside radiotherapy settings and rarely offer multimaterial, head-to-head comparisons or a clear path toward clinical deployment. 23 To address radiotherapy-related breast reconstruction, we delineate and test a radiation-compatible scaffold concept in an animal model that provides pre- and postirradiation readouts under standardized implant volumes. At the materials and biology interface, we propose a dual-mechanism design—biological activity modulation (adipose as a reservoir of bioactive cues) plus morphological support (a permeable, remodelable omentum–dECM network)—that may enable early volume maintenance and tissue integration under radiation stress.12,13 We further incorporate regionalized analyses (peri-implant interface and adipose septa) to move beyond single-site or aggregate readouts and to capture coordinated integration across compartments. Clinically, we introduce a minimally invasive, injectable approach and benchmark outcomes before and after radiotherapy under conditions aligned with those of medical practice, providing a reference framework for the evolution of hydrogel-based breast implants.8,11
Materials and methods
Experimental animals and ethical compliance
Female Sprague–Dawley (SD) rats (n = 30), aged 8–10 weeks and weighing 200–230 g, were obtained from the Experimental Animal Center of the Second Affiliated Hospital of Harbin Medical University. Three SD rats from the same group were housed together in standard cages under controlled environmental conditions. The animals were provided ad libitum access to standard rodent chow and sterile drinking water. A 12-h light/dark cycle was maintained, with the ambient temperature and humidity regulated at 22 ± 1°C and 50 ± 10%, respectively. Prior to invasive procedures (injections or surgeries), the rats were anesthetized via the intraperitoneal administration of 2% tribromoethanol (Sigma‒Aldrich, USA; T48402) at a dosage of 0.15 mL per 10 g body weight.
Humane endpoint criteria were established for all experimental animals in this study and included ⩾20% body weight loss, irreversible motor dysfunction, and severe infection. At the experimental endpoints, euthanasia was performed under tribromoethanol-induced anesthesia, and death was confirmed following cervical dislocation. All the animals were euthanized at the study endpoints in accordance with the approved protocol. No cases necessitated the implementation of premature euthanasia at any time during the investigation. Implant-associated tissues were systematically harvested in accordance with predetermined experimental protocols.
All the experimental procedures were performed in strict compliance with the protocol approved by the Institutional Animal Care and Use Committee of the Second Affiliated Hospital of Harbin Medical University (Approval No. Refer to the Title Page) and conducted in accordance with the ARRIVE guidelines.
Preparation of decellularized omental hydrogels
Porcine omental tissues obtained from the Harbin Veterinary Research Institute (Heilongjiang Province, China) were decellularized by sequential processing. After initial rinsing with phosphate-buffered saline (PBS), the tissues were subjected to three freeze-thaw cycles (−80°C for 12 h/37 °C for 1 h) to induce cellular lysis. Lipid extraction was then performed by immersion in hexane-ethyl acetate (1:1 v/v) for 24 h with three solvent changes. Sequential ethanol washes (100% for 30 min, followed by 70% overnight at 4 °C) were performed before enzymatic digestion in 0.25% trypsin-EDTA (porcine pancreatic trypsin, EDTA 0.02% w/v; Beyotime, China; cat. C0201) for 12 h, and the resulting mixture was subsequently washed in 1.5 M NaCl with three solution changes within 24 h. The decellularized omentum was cryogenically pulverized in liquid nitrogen, filtered through 200-mesh filters, and then subjected to enzymatic digestion with magnetic stirring at 300–600 rpm. The resulting material was then digested in 0.1 M HCl, in which pepsin powder (Sigma-Aldrich, USA; 2500 U/mg) was dissolved to a final concentration of 1.5 mg/mL (w/v), corresponding to 3750 U/mL. The pH of the solution was adjusted to 7.4 using NaOH or 10× PBS. Finally, the concentration of the solution was standardized to 1.5% (w/v). The entire digestion process was carried out at room temperature for 24 h under continuous agitation. The resulting thermoresponsive hydrogel exhibited an irreversible sol–gel transition at physiological temperature (37°C).
Fabrication of the alginate-reinforced composite hydrogel
Sodium alginate (300 kDa; Engineering for Life, Jiangsu, China) was dissolved in deionized water to prepare a 5% (w/v) solution. The omentum hydrogel precursor was injected into polytetrafluoroethylene (PTFE) semicircular molds and cryomolded at −20°C. The frozen constructs were then encapsulated in alginate solution and ionically crosslinked in 3% CaCl₂ for 10 min to form crosslinked peripheral layers. The final implants were aseptically trimmed to standardized semicircular dimensions at 4°C.
Harvesting and processing of rat adipose tissue
To better approximate clinical autologous fat grafting, adipose tissue was not decellularized, thereby preserving its lipid content and bioactive components. 24 Inguinal adipose tissue was surgically excised from female Sprague–Dawley rats (n = 6). After the meticulous removal of connective tissue and vasculature, the isolated adipose pads were rinsed with PBS and manually minced by repeated orthogonal cutting until all fragments were <1 mm³, after which they the samples were loaded into sterile syringes to qualitatively confirm successful extrusion through a clinical needle and then stored at 4°C for subsequent experiments.
Experimental groups and implant design
Five experimental cohorts (n = 6/group) were established with the following configurations. In the O-dECM group (hereinafter O-dECM), 1.5% (w/v) omentum dECM hydrogel was combined with lyophilized omental matrix fragments at a 1:15 (w/w) ratio. In the Adipose-dECM group (hereinafter Adipose-dECM), omentum dECM hydrogel was homogenized with rat adipose tissue particles (1:1 v/v). The Adipose group (hereinafter Adipose) consisted of native rat adipose tissue particles, the Alg-dECM group (hereinafter Alg-dECM) consisted of alginate-encapsulated omental dECM constructs, and the Silicone group (hereinafter Silicone)consisted of a custom medical-grade silicone implant with a smooth shell (McGhan Medical). To eliminate size-related effects, the implant volume was standardized to 1.0 mL for all materials. For injectable hydrogels, the precursor volume was calculated to yield a final in situ gel volume of 1.0 mL at the time of placement. For surgically placed constructs, the implant volume was measured prior to placement, and for Alg-dECM, the alginate shell was included in the total. All the implants were placed under aseptic conditions.
The following group-specific implantation modalities were used: percutaneous injection for the O-dECM, Adipose-dECM, and Adipose groups; surgical implantation for the Alg-dECM and Silicone groups. The surgical procedure involved creating 1-cm dorsal incisions and bluntly dissecting subcutaneous pockets. On postoperative day 16, three randomly selected animals per group underwent fractionated radiotherapy (Varian Clinac IX; 28 Gy total dose, delivered in five daily fractions of 5.6 Gy; 6 MeV electron beam) using a 2 cm-diameter lead collimator for field localization under 2% tribromoethanol anesthesia (Figure 1). 25

Schematic of the implant and rat subcutaneous implantation model. (Created in BioRender, Agreement number: GW28YTR30L).
In vivo hydrogel stability assessment
Cy5 NHS ester (MedChemExpress, Shanghai, China) was reconstituted in dimethyl sulfoxide (DMSO) to 10 mg/mL and covalently conjugated to the hydrogels (0.30% w/w) during 2 h of incubation (4°C, in the dark). The unconjugated dye was removed by centrifugal filtration (Amicon Ultra, 10 kDa MWCO; Merck Millipore, Darmstadt, Germany) to yield fluorescent hydrogels (excitation wavelength: 650 nm; emission wavelength: 670 nm). In vivo fluorescence imaging and quantification were both performed on a GelView 6000Plus (Guangzhou Biolight Biotechnology Co., Ltd., China) using the instrument’s default red fluorescence channel (Cy5-compatible) with a fixed exposure time of 400 ms. The sample–camera geometry (lens, distance, alignment) and optical configuration were kept constant for all time points (0–48 h). Regions of interest (ROIs) were identical in size.
Ultrasonographic analysis
Implant morphology and vascularization were assessed using a high-resolution ultrasound system (Mindray M9; L18-4 transducer). Following dorsal depilation and acoustic coupling gel application, three-dimensional parameters were calculated using the following formula:
where H is the implant height and R is the base radius.
Hydrogel characterization
A 4°C dECM solution was loaded into a 1 mL syringe and slowly extruded through a clinical needle to qualitatively confirm dECM injectability. SEM was performed to examine microstructure and estimate areal porosity. The dECM hydrogels were examined by scanning electron microscopy (SEM; ZEISS Gemini SEM 300, Germany) to characterize their microstructural features. Areal porosity was quantified in ImageJ/Fiji (v1.53, NIH) using threshold-based binarization to compute the void-area fraction.
Viscoelastic properties were measured on a rotational rheometer (MCR 302; Anton Paar, Austria) using a strain-amplitude sweep at 25 °C with a fixed frequency of 1 Hz. Storage (G′) and loss (G″) moduli were recorded, and the loss tangent (tan δ) and complex viscosity (η*) were calculated before and after irradiation. Rheological measurements were performed at ~25 °C; the hydrogel did not undergo a sol–gel transition between 25°C and 37°C. 26
dECM hydrogel samples were pregelled at 37°C, trimmed to 50 mg (wet), and incubated at 37°C under gentle agitation in the presence of Type I collagenase (Gibco, lot 17100017) at 50 U/mL (≈0.40 mg/mL, 125 U/mg). The enzyme was prepared in PBS (pH 7.4) supplemented with 5 mM CaCl₂. The enzyme solutions were refreshed every 24 h. At 0, 12, 24, 36, 48, 60, 72, and 96 h, the samples were briefly rinsed in PBS, gently blotted, and weighed to obtain M(t); mass retention was calculated as M(t)/M₀ × 100% (n = 3 independent specimens).
Histological and immunohistochemical evaluation
Hematoxylin and eosin (H&E): Explanted constructs were fixed in 4% paraformaldehyde (24 h, 4°C), dehydrated through a graded ethanol series, paraffin embedded, and sectioned at 4 µm. For each animal (n = 3 per group), three nonoverlapping fields adjacent to the implant interface at 200× magnification were sampled using systematic uniform random sampling (SURS). Briefly, the peri-implant region of interest (ROI) was delineated in ImageJ (v1.53), a 200× field template (matched to microscope calibration) was overlaid, and a random starting point was selected; three fixed-area fields were then sampled without overlap. Fields containing folds, tears, necrosis, or edge contact were excluded a priori. Capsule thickness was measured as the perpendicular distance from the implant surface to the outer capsule boundary at three evenly spaced positions per field and averaged. Inflammatory cell density was quantified by counting hematoxylin-positive nuclei within the capsule using color deconvolution and particle analysis (ImageJ). Analyses were performed in a blinded manner to group allocation.
Sirius Red staining: Collagen architecture was analyzed under polarized light (Eclipse Ci, Nikon, Tokyo, Japan) with quantitative discrimination of collagen I (orange-red birefringence) and III (green). Collagen area fractions were determined using threshold-based image analysis (ImageJ v1.53; NIH).
CD31 immunohistochemistry and determination of microvessel density: Sections were subjected to heat-induced epitope retrieval, incubated with anti-CD31 (Abcam, ab182981), developed with DAB, and counterstained with hematoxylin. For each animal (n = 3 per group), three nonoverlapping 400× fields within the peri-implant ROI were selected by SURS. Two readouts were obtained. For the CD31-positive area fraction (%), the DAB channel was isolated by color deconvolution; a fixed threshold (held constant within an experiment) was applied, and the CD31-positive area was divided by the field area. Microvessel density (MVD, vessels·mm⁻²) pixel size was calibrated using the embedded scale bar to obtain the field area (mm²). Following the Weidner method, any discrete CD31-positive endothelial cell or cluster (with or without a visible lumen) was counted as one microvessel, and large muscularized vessels were excluded. Edge objects were counted if >50% of their area lay within the ROI. Counting was performed with the ImageJ Cell Counter. For statistical analysis, the mean of three fields per animal was used. All assessments were blinded.
Statistical analysis
The animal was the experimental unit. For histology and IHC, three nonoverlapping fields per animal were averaged to obtain a single per-animal value. Endpoints included day 50 volume retention, capsule thickness, inflammatory cell density, collagen I/III fractions, CD31-positive area, microvessel density, and rheological parameters. Quantitative image analysis was performed using Fiji/ImageJ software (v1.53). Statistical comparisons were conducted in GraphPad Prism 10 (La Jolla, CA, USA), with parametric data presented as the mean ± standard error of the mean (SEM) and nonparametric data presented as the median (IQR). Intergroup differences were assessed via Student’s t test (two-group parametric), one-way ANOVA with Tukey’s post hoc test (multigroup parametric), or the Mann-Whitney U test (nonparametric). Statistical significance was defined as α = 0.05 (two-tailed).
Results
Hydrogel characterization
After lyophilization and milling, the decellularized omentum took the form of a powdered material (Figure 2(a)). At 4°C, the dECM solution was a translucent liquid suitable for injection; at 37°C, it gelled into a solid hydrogel via an irreversible sol–gel transition (Figure 2(b)). dECM solution injectability was qualitatively confirmed by slow manual extrusion through a clinical needle, which produced continuous droplets without clogging (Figure 2(c)). SEM revealed an intact three-dimensional hydrogel network (Figure 2(d)). ImageJ-based areal porosity (void-area fraction) indicated a porous microarchitecture; interconnectivity was assessed by SEM (Figure 2(e)).

Physicochemical characterization of the omentum-derived dECM hydrogel. (a) Lyophilized omentum dECM after milling. (b) Temperature response: liquid at 4°C and solid hydrogel at 37°C (irreversible sol–gel transition). (c) Injection through a fine-gauge clinical needle, forming a continuous droplet at the tip. (d) Scanning electron micrograph showing an interconnected porous network (scale bar, 40 μm). (e) Box-and-whisker plot of areal porosity (n = 6 fields): median 19.5% (IQR 14.6%–22.6%); range 10.3%–26.9%; points are individual fields. (f) Strain-amplitude sweep of storage and loss moduli (G′, G″) versus γ₀ (0.1%–100%) at ~25 °C, comparing nonirradiated and postirradiated hydrogels. (g) Complex viscosity (η*) versus γ₀ (0.1–100%) at ~25 °C under the same conditions. (h) Enzymatic mass retention at 37°C in Type I collagenase (50 U/mL in PBS supplemented with CaCl2) over 0–96 h, showing slight early swelling followed by progressive loss.
Within the small-strain plateau (γ₀ ⩽ 5%), we observed tan δ < 1 for both conditions, indicating an elastic-dominant O-dECM state. Relative to the nonirradiated O-dECM, irradiated O-dECM exhibited an overall higher tan δ, indicating a greater viscous contribution. When tan δ = 1 was used as an operational crossover criterion (G′ = G″), the irradiated O-dECM reached this point at a lower strain. At higher strains (⩾ 25%), tan δ > 1 in both groups and continued to increase, consistent with a transition to a viscous-dominant response and network rearrangement (Figure 2(f)). The complex viscosity η* decreased monotonically with γ₀; in the small-strain plateau, η* decreased from 18.83 to 17.08 Pa·s, indicating reduced flow resistance under small deformations after irradiation. In the mid-to-high strain range, the two η* curves progressively converged, suggesting similar flow behavior under large deformations (Figure 2(g)). Under 37°C Type I collagenase, the wet mass of the O-dECM hydrogel monotonically decreased, with a faster decrease during the first 24–48 h, after which it gradually stabilized. By 96 h, ~50% of the initial mass remained, indicating substantial enzyme-mediated degradation.
In vivo stability assessment of the hydrogel implants
To investigate the biological stability of the hydrogel implants, we systematically evaluated their physical characteristics and volume retention in a rat model. The hydrogel maintained liquid-phase behavior at 4°C, allowing minimally invasive administration via syringe injection, and rapidly transitioned to a translucent elastic hydrogel upon temperature elevation to 37°C. Crucially, this phase transition was irreversible after solidification. Postimplantation analysis confirmed the preservation of structural integrity with retained flexibility in the Adipose-dECM (Figure 3(a)), in contrast to the persistent rigid alginate-based outer shell observed in the Alg-dECM throughout the implantation period (Figure 3(b) and (c)).
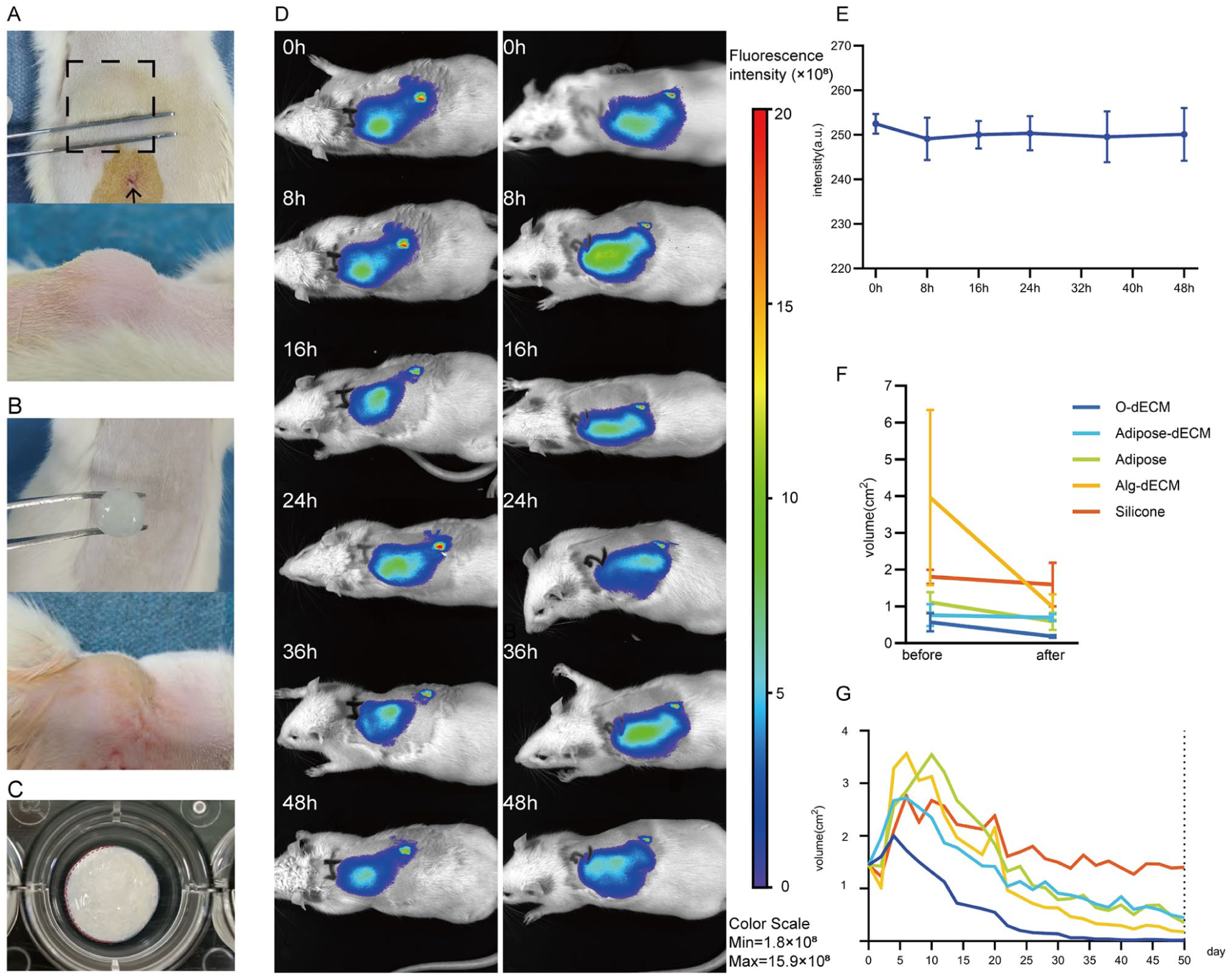
In vivo stability of breast implants based on hydrogel or control materials in a rat model. (a) Representative macroscopic view of Adipose-dECM implanted subcutaneously on postoperative day 0 (dashed box, implant contour; arrowhead, injection track). (b) Alg-dECM placed in a surgically created subcutaneous pocket. (c) Dashed outline denotes the alginate outer shell. (d) Longitudinal in vivo fluorescence imaging (0, 8, 16, 24, 36, and 48 h) demonstrating the sustained localization of the Cy5- labeled hydrogel. (e) Quantification of fluorescence retention at 48 h. (f) Postirradiation volumetric retention (28 Gy delivered on day 16): Adipose-dECM exceeded O-dECM, Adipose, Alg-dECM, and Silicone under standardized terminal volumes (1.0 mL). (g) Day-50 volume retention trends: Adipose-dECM volume remained higher than O-dECM, Adipose, and Alg-dECM volumes but below Silicone volume.
In the in vivo stability evaluation, the fluorescence localization of the subcutaneously implanted Cy5-labeled hydrogel was sustained at the administration site for more than 48 h according to longitudinal imaging. The consistent fluorescence intensity profile supported structural stability and localization (Figure 3(d) and (e)).
Radiation exposure significantly altered volume retention patterns across the experimental groups. Alg-dECM displayed minimal radiation tolerance (24.75% retention), whereas Adipose-dECM and Silicone showed increased stability (91.17% and 88.41%, respectively). The Adipose-dECM composite architecture exhibited significant efficacy in counteracting radiation-induced adipose tissue atrophy, achieving 91.17% irradiated retention, significantly surpassing those of both Adipose (53.37%) and O-dECM (32.26%) (Figure 3(f)). Longitudinal monitoring over 50 days revealed that Adipose-dECM was relatively stable (30.75% retention) compared with O-dECM (1.22%), Adipose (24.20%), and Alg-dECM (12.03%) (Figure 3(g)).
These findings suggest that the dual mechanisms of Adipose-dECM, in which mechanical stabilization is coupled with sustained bioactive signaling, may synergistically preserve early morphological integrity via hydrogel–adipose interactions. However, the day 50 retention of Adipose-dECM remained substantially lower than that of Silicone (30.75% vs 95.21%), indicating that the current formulation does not achieve durable volumization. In contrast, the <15% retention observed for O-dECM and Alg-dECM is unlikely to support clinically meaningful long-term volume retention, suggesting that these materials are unsuitable as standalone implants.
Tissue compatibility analysis of hydrogel-based implants
To assess the effects of radiation on hydrogel implant biocompatibility, we conducted a histological evaluation of fibrous capsule formation and inflammatory cell infiltration via hematoxylin-eosin (H&E) staining (Figure 4(a)), and collagen deposition was quantified using Sirius Red staining (Figure 4(b)).
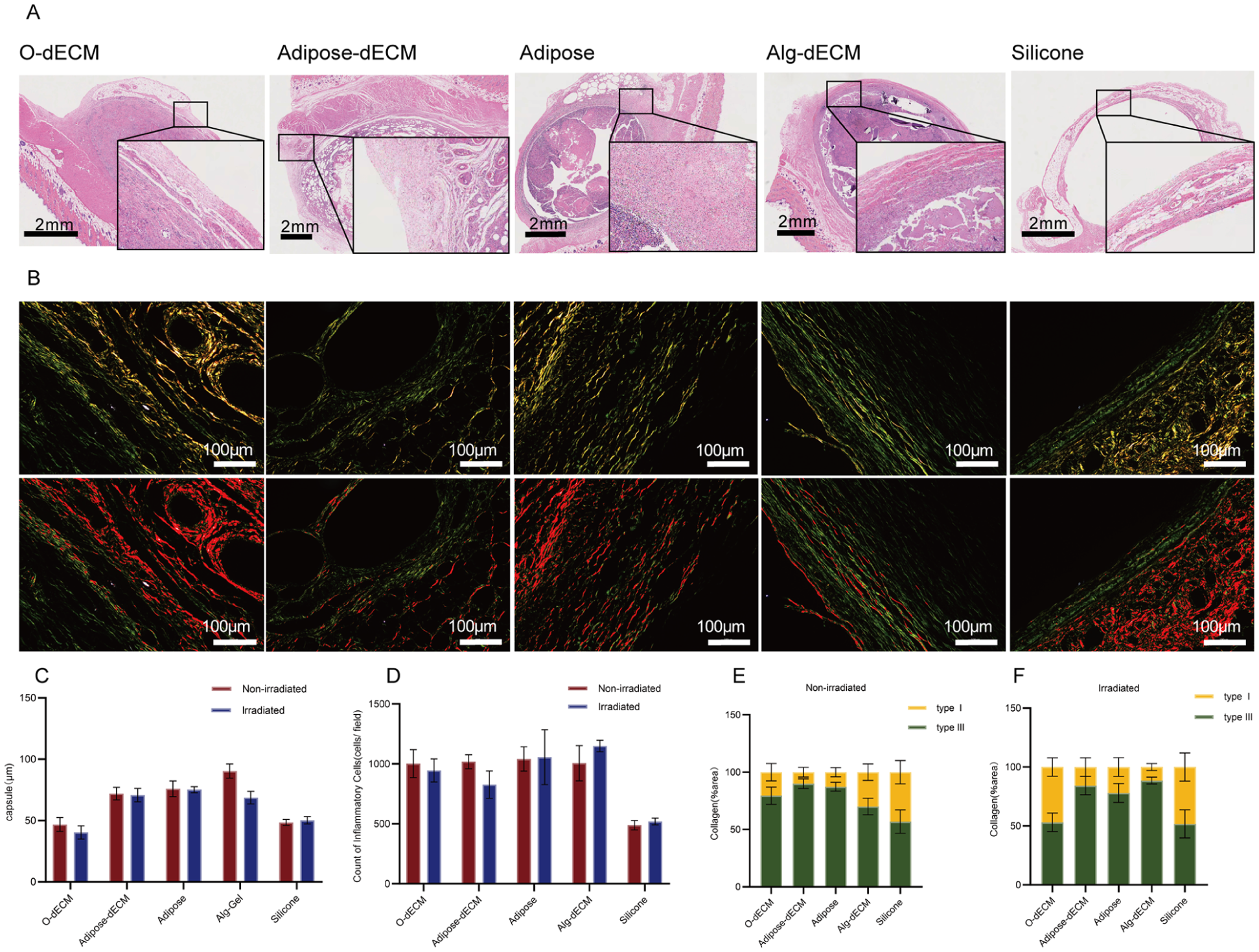
Histological and compositional analyses of peri-implant tissues across groups. (a) Representative H&E images of peri-implant tissues in O-dECM, Adipose-dECM, Adipose, Alg-dECM, and Silicone (scale bar, 2 mm). (b) Sirius red polarization results showing collagen distribution (orange–red: type I; green: type III) (scale bar, 100 μm). (c) Fibrous capsule thickness in the irradiated (28 Gy) and nonirradiated subgroups; Alg-dECM showed significant thinning after irradiation (p = 0.0132). (d) Inflammatory cell counts: in Adipose-dECM, irradiation was associated with a ~22% reduction versus Adipose. (e) Collagen composition in nonirradiated animals: the type I fraction in the Adipose-dECM group was lower than that in the Silicone group. (f) Postirradiation collagen composition: the type I fraction in Adipose-dECM remained below that in Silicone.
An analysis of fibrous encapsulation revealed comparable capsule thicknesses between the O-dECM (46.75 ± 17.11 μm) and Silicone (48.43 ± 7.59 μm) groups. Under nonirradiated conditions, Adipose-dECM resulted in greater capsule thickness (71.95 ± 15.46 μm) than Silicone did but a significantly thinner capsule than Alg-dECM (90.30 ± 17.30 μm) and Adipose (75.90 ± 19.21 μm). Enhanced measurements revealed a slight reduction in the thickness of the adipose-dECM (70.71 ± 16.29 μm), in contrast to the significant decrease in the thickness of the Alg-dECM (90.30 ± 17.30 μm to 68.65 ± 18.7 μm; p = 0.0132). The other groups exhibited marginal increases in thickness (Figure 4(c)).
Inflammatory cell quantification revealed greater counts in nonirradiated Adipose-dECM (1018 ± 176.5 cells/field) than in Silicone (487.8 ± 118.9 cells/field), with values comparable to those of O-dECM (1002 ± 350.7 cells/field), Adipose (1041 ± 304.8 cells/field) and Alg-dECM (1006 ± 442.6 cells/field). The analysis revealed reduced infiltration in Adipose-dECM (826.7 ± 342.2 cells/field), which was 22% lower than that in Adipose (1056 ± 687.7 cells/field) and lower than that in O-dECM (945.4 ± 287.7 cells/field) and Alg-dECM (1150 ± 146.0 cells/field) but higher than that in Silicone (519.8 ± 86.06 cells/field) (Figure 4(d)). Adipose-dECM uniquely reduced the radiation-induced inflammatory response.
Sirius Red staining revealed that the nonirradiated collagen composition of Adipose-dECM (type I: 10.01%; type III: 89.99%) closely resembled that of Adipose (type I: 12.69%; type III: 87.31%). Measurements of irradiated samples revealed increased type I collagen in Adipose-dECM (15.79%), although it remained below the levels observed in Adipose (22.01%), O-dECM (46.94%), and Silicone (48.29%) and exceeded that in Alg-dECM (11.54%) (Figure 4(e) and (f)).
Analysis of angiogenesis in hydrogel-based implants
Ultrasonographic examination revealed enhanced vascularization in peri-implant regions and parenchymal tissues of animals in both Adipose-dECM and Adipose (Figure 5(a)). To evaluate the effects of the implants on angiogenesis, vascular density was quantified via the immunohistochemical staining of CD31 in the surrounding connective tissues and adipose septa. Preirradiation CD31 staining was performed for Adipose-dECM and Adipose because only these groups showed ultrasound-detectable vascular changes at baseline. Following radiotherapy, O-dECM, Alg-dECM, and Silicone maintained baseline values of vascular parameters, whereas both Adipose-dECM and Adipose presented greater CD31-positive areas in the peri-implant and parenchymal regions, with particularly pronounced changes in Adipose-dECM (Figure 5(b) and (c)).
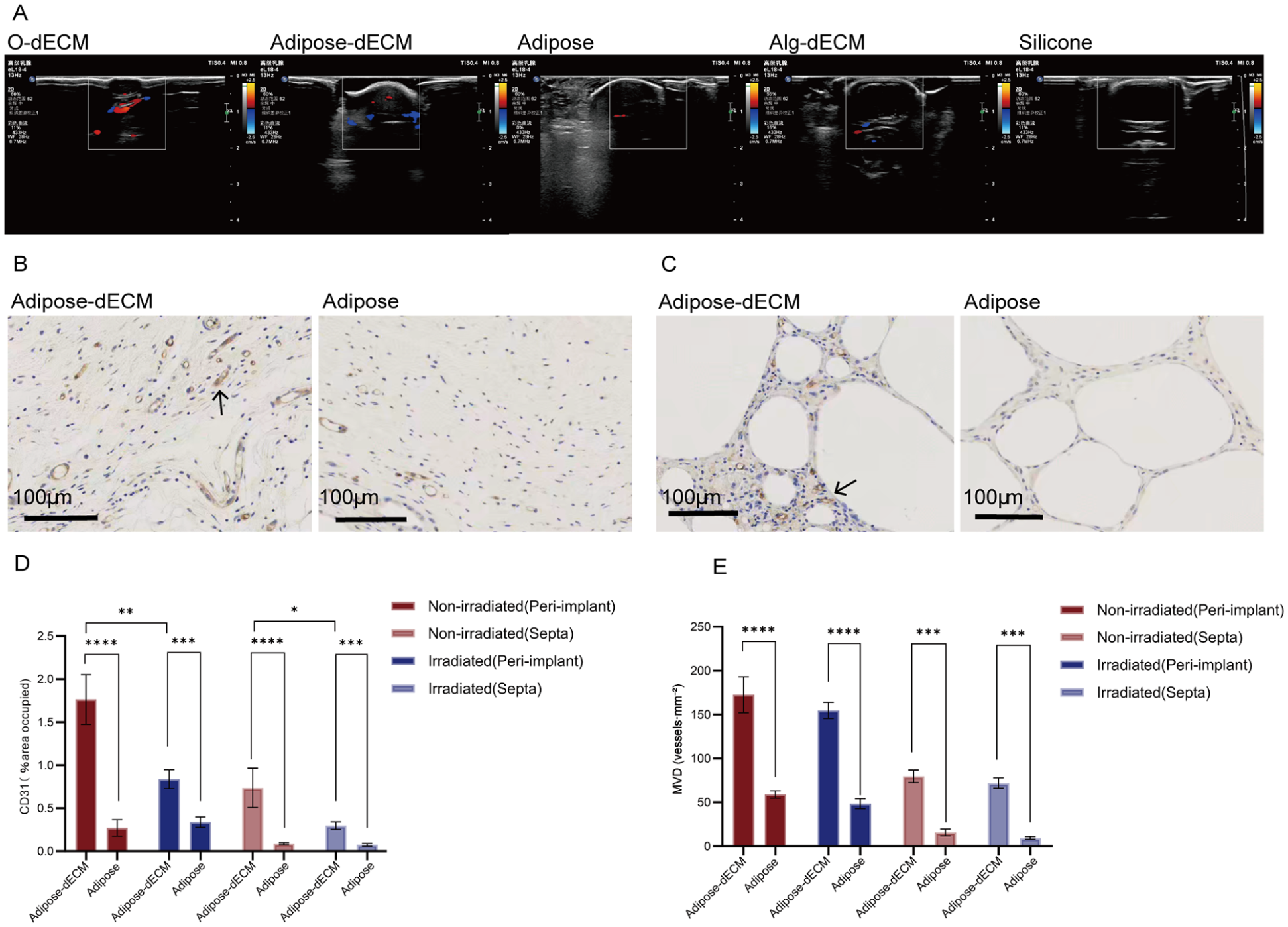
Angiogenic profiling of hydrogel-based implants. (a) Representative ultrasonographic images of O-dECM, Adipose-dECM, Adipose, Alg-dECM, and Silicone, showing color Doppler flow signals (red/blue) within the peri-implant zones. (b) CD31-positive vasculature in the peri-implant regions of Adipose-dECM and Adipose (black arrows indicate angiogenic hotspots; scale bar, 100 µm). (c) CD31-positive vasculature within adipose septa of Adipose-dECM and Adipose (black arrows; scale bar, 100 µm). (d) Quantification of the CD31-positive area. In nonirradiated animals, AdiposedECM exhibited larger CD31-positive areas than Adipose did in peri-implant tissue (****p < 0.0001) and adipose septa (****p < 0.0001). After irradiation, Adipose-dECM maintained higher CD31 positivity in periimplant regions (***p = 0.0010) and adipose septa (***p = 0.0003). Radiotherapy reduced the vascular density in Adipose-dECM peri-implant tissue (p = 0.0086) and adipose septa (p = 0.0188). (e) Microvessel density (MVD, vessels·mm⁻²) in peri-implant tissue and adipose septa for Adipose-dECM versus Adipose under non-irradiated and irradiated conditions. In non-irradiated animals, Adipose-dECM showed a higher MVD than Adipose in both peri-implant tissue (****p < 0.0001) and adipose septa (***p < 0.001); this difference persisted after irradiation in peri-implant regions (****p < 0.0001) and adipose septa (***p < 0.001). Radiation decreased MVD across regions. Bars show mean ± SD; points represent individual specimens.
Among the nonirradiated samples, Adipose-dECM samples presented significantly greater CD31-positive areas in peri-implant tissues (1.31% vs 0.10%, p < 0.0001) and adipose septa (0.60% vs 0.07%, p < 0.0001) than Adipose samples did. Measurements following irradiation showed maintenance of this differential pattern, with Adipose-dECM images demonstrating greater CD31 positivity in peri-implant regions (0.84% vs 0.34%, p = 0.0010) and adipose septa (0.29% vs 0.06%, p = 0.0003). Notably, radiotherapy significantly reduced vascular density in both the peri-implant tissues (p = 0.0086) and the adipose septa (p = 0.0188) of Adipose-dECM (Figure 5(d)).
Consistent with the CD31-positive area readout, the MVD of Adipose-dECM was greater than that of Adipose-dECM under both radiation statuses. In the peri-implant region, the MVD was greater for Adipose-dECM than for Adipose in nonirradiated animals (172.6 ± 61.3 vs 59.1 ± 12.5 vessels·mm⁻²; p < 0.0001) and remained higher in the radiotherapy cohort (154.7 ± 27.7 vs 48.4 ± 16.8 vessels·mm⁻²; p < 0.0001). In the adipose septa, the same pattern held (nonirradiated: 79.9 ± 21.3 vs 15.9 ± 11.5 vessels·mm⁻²; p < 0.001; radiotherapy: 72.1 ± 17.4 vs 9.6 ± 4.7 vessels·mm⁻²; p < 0.001). Grouping by radiation status further revealed that radiotherapy modestly attenuated the MVD in Adipose-dECM, yet the values remained well above those of Adipose; in contrast, Adipose exhibited a region-dependent response (a small peri-implant decrease vs a marked septal decrease) (Figure 5(e)). These MVD findings mirror the CD31-area results and indicate that Adipose-dECM maintains a denser microvascular network than Adipose does in both settings, with partial but incomplete radiation sensitivity.
Discussion
In breast reconstruction, optimal implant materials must achieve a balance between biocompatibility, mechanical stability, and tissue integration potential. 27 Our investigation provides the first experimental evidence that Adipose-dECM significantly maintains high short-term volume retention in radiation-treated implants, with 91.17% retention after radiation. However, its 50-day retention was ~31% (vs ~95% for Silicone), indicating that the current formulation is not yet suitable for long-term implantation. We favored an injectable, in situ–gelling approach to preserve permeability and remodelability, which are properties that are expected to facilitate early mass transport and neovascular ingress and may better align with minimally invasive delivery. Within this framework, we used minimally processed, nondecellularized adipose tissue to retain lipids and adipose-derived bioactive cues that could support proangiogenic and immunomodulatory processes. 28 In contrast, prefabricated hydrogel blocks typically require larger incisions and may offer less conformability to irregular defects; in the specific context of irradiated beds, these differences could influence early integration and morphological maintenance. Adipose-dECM is designed to provide a more permissive microenvironment and early volumetric support under radiation-related stress and host responses. Our animal data demonstrate feasibility and highlight room for optimization, particularly with respect to long-term shape stability.
In this study, small-amplitude oscillatory shear results indicated an elastic-dominant response (tan δ < 1). Irradiation only modestly increased tan δ and slightly reduced complex viscosity (η*), which are results consistent with mild network relaxation rather than structural failure. Scanning electron microscopy revealed a network with moderate porosity, favoring molecular transport and cellular infiltration. In vitro volumetry demonstrated an initial swelling phase followed by progressive degradation. Early interconnecting porosity and limited relaxation likely aided stress dissipation and mass exchange, where as the hydrogel degraded, the late-phase volume decreased. A low tan δ also implies lower energy dissipation and limited creep resistance, causing predisposition to resorption under cyclic loading, helping to explain late volume loss. In parallel, radiotherapy inflicts sustained injury to adipocytes and microvessels (e.g. oxidative stress, lipolysis), causing endothelial damage and perfusion decline, which are characteristics matching the 50-day volume attrition and MVD downshift we observed.
Experimentally, Adipose-dECM presented ~91% short-term retention during radiotherapy, possibly because of the synergy between the dECM and adipose components. The hydrogel scaffold provides mechanical support to adipose tissue, while the adipose component may attenuate implant immunogenicity. The porous microstructure facilitates angiogenesis and mass transport, improving early volume retention. This aligns with the concept that mechanical reinforcement stabilizes hemodynamics and buffers early radiation-related stress.29–31 In contrast, the day-50 retention rate decreased to ~31%, which was far below that of Silicone (~95%), indicating that durability remained insufficient for long-term repair. dECM degradation is a known challenge in the use of biomaterials. 32 Superimposed chronic radiation injury to adipocytes and the endothelium further drives late-phase resorption. Although a loose, exchange-friendly capsule can benefit transport, it may also contribute to some loss of volume. Encapsulation in an Alg shell did not prevent late resorption—likely owing to in vivo Alg degradation and a proinflammatory milieu; prior work suggests that cell-assisted dECM construction may mitigate resorption. 33 Practical strategies include repeated dosing, gentle network stabilization (e.g. mild chemical/physical crosslinking or interpenetrating networks), and sustained proangiogenic signaling (e.g. heparin-conjugated VEGF) to decelerate degradation and preserve morphology. Each approach must balance the potential loss of bioactivity or heightened inflammatory risk.34,35 Achieving superior biocompatibility and long-term shape retention will require a calibrated trade-off between viscoelasticity and degradability.
In the absence of irradiation, the capsule surrounding Adipose-dECM was thicker than that around Silicone but lower than or comparable to those surrounding Alg-dECM and Adipose. After irradiation, capsule thickness slightly decreased in the Adipose-dECM group, whereas that in the other groups slightly increased or did not significantly change. Although a thinner capsule is generally desirable, thickness alone does not affect functional integration. Several in vivo studies have indicated that appropriate pore sizes can promote cell infiltration and vascular invasion, yielding a broader, more active “integration zone”; thus, geometric thickness may increase, while tissue remains looser and more active. 36 From a mechanobiological standpoint, stiffer matrices more readily increase α-SMA and dense type-I collagen deposition, and softer matrices favor remodelable repair. The type III-lean collagen profile of adipose-dECM is consistent with that of a softer, remodelable capsule. In contrast, silicone’s inert, nonpermeable surface typically has a thin, relatively inactive isolating capsule. 37 These observations align with the amniotic-matrix-mediated immunomodulatory mechanisms reported by Wassmer and colleagues.38,39 Glycosaminoglycans in the interstitium—especially heparan sulfate—may both suppress TGF-β/Smad signaling to limit fibroblast activation and activate PI3K/Akt to polarize macrophages toward the M2 phenotype,40,41 together attenuating fibrosis and inflammation. 42
Before irradiation, Adipose-dECM resulted in higher inflammatory cell counts than Silicone did; after irradiation, the counts decreased by ~22% and coincided with the predominance of type III collagen, a lower type I fraction (~15.79%), and a slightly thinner capsule (still thicker than Silicone but lower than/close to those of the other controls). Unlike inert Silicone, the interconnecting porosity of hydrogels facilitates transport and cellular trafficking, widening an integration zone rather than simply thinning an isolating layer. dECM can undergo binding and present growth and chemotactic factors, recruiting immune cells and promoting repair, 35 while adipose tissue also attracts macrophages. 43 Numerous dECM studies have shown polarization toward an M2 phenotype, which is a remodeling phenotype that, together with angiogenesis and matrix turnover, yields a richer, more active capsule. 44 Thus, inflammatory counts higher than those associated with silicone are consistent with dECM-driven remodeling; at the 50-day endpoint, the system may still be in a recruitment phase. The ~22% reduction observed following irradiation further supports a buffering effect of dECM on radiation-related inflammatory and fibrotic pathways. Hydrogel-based radioprotective materials and dECM hydrogels have also been reported to attenuate radiation injury and fibrosis across multiple tissues. 45 In line with our data, enhanced microvascularization can facilitate tissue repair while polarizing macrophages toward prorepair phenotypes and restraining excessive leukocyte infiltration. These features are in accordance with our observation that inflammatory cell counts decreased relative to those in the nonirradiated state. 46 Accordingly, the available evidence supports radiation compatibility rather than mere anti-inflammatory action, highlighting hydrogels as bioactive scaffolds compatible with irradiation rather than as purely inert fillers.
Ultrasonography revealed perfusion changes surrounding Adipose-dECM and Adipose. CD31 staining confirmed denser microvascular networks around the implants and within the adipose septa, particularly in Adipose-dECM. After irradiation, the number of microvessels decreased by ~18% but remained significantly above that observed for Adipose, suggesting partial resistance of the dECM-supported endothelium to radiation-induced apoptosis. 47 The porous scaffold provides conduits for angiogenic ingrowth 48 and a favorable matrix for endothelial expansion, 49 enabling vessels to grow into the implant. The immunomodulatory properties of dECM and its reservoir of proangiogenic cues (e.g. VEGF) are consistent with the combination of a type III-lean collagen milieu and increased MVD observed without irradiation. Although radiotherapy harms the endothelium and reduces perfusion, a well-perfused, cell-friendly, and elastic scaffold helps offset these effects; thus, Adipose-dECM outperforms Adipose alone overall. 50
In radiotherapy-related settings, the apparent advantage of Adipose-dECM may reflect a dual mechanism of biological activity modulation and permeable structural support. Adipose-derived components retained in the composite (viable adipocytes, lipids, and growth factors) could act as a local reservoir of bioactive cues, whereas omentum-derived dECM provides a porous, remodelable network that may sequester, present, and gradually release these signals to support angiogenesis and temper fibrotic responses under irradiation.51,52 The confinement of adipose particulates within the dECM network likely reduces early mechanical loss and results in microscale dissipation of micromotion, which could facilitate early volume maintenance and integration amid endothelial injury and reduced perfusion. Consistent with this model, Adipose-dECM showed a greater CD31-positive area and microvessel density, lower inflammatory infiltration, and a more favorable collagen I/III profile—these features are indicative of a low-fibrosis, pro-perfusion remodeling trajectory. 11
In contrast, although the Alg-dECM shell provides greater initial shape stability than the others, its mass-transfer barrier and lack of adhesive ligands may limit cell ingress and neovascularization, leading to passive encapsulation rather than active integration at the material–host interface. 8 Under irradiation, impaired microcirculation and suppressed matrix turnover further hinder reperfusion and volume preservation; in addition, inflammation-associated ionic/protein shifts may weaken Ca²⁺-mediated alginate crosslinking, thereby compromising long-term shell integrity. 53 Taken together, these considerations suggest that midterm outcomes in irradiated beds may be governed less by shell stiffness per se than by a scaffold’s permeability, remodelability, and proangiogenic integration. Mild network stabilization (e.g. gentle crosslinking or interpenetrating networks), adhesive-motif incorporation, and controlled porosity appear to be reasonable strategies for enhancing durability without sacrificing bioactivity.
Adipose-dECM was thought to provide a friendlier microenvironment than the other tested biomaterials under irradiation-related stress, with the goal of supporting early contour maintenance while accommodating tissue remodeling. In this study, the observed postirradiation readouts—including lower inflammatory infiltration, a remodeling-prone collagen profile, and microvascular indices exceeding those of adipose controls—were consistent with that design intent and with radiation-compatible behavior at early time points. Despite these favorable responses to irradiation, late volumetric durability remained limited in the current formulation, indicating that network stabilization could be further optimized. Overall, the data highlight the promise of Adipose-dECM specifically after irradiation, where tissue responses appear more permissive, while underscoring the need to translate these microenvironmental advantages into improved long-term retention.
Limitations and future directions
This study has four primary limitations. First, the 50-day observation window was insufficient to fully evaluate long-term tissue remodeling and volume retention after material degradation. Second, we did not quantitatively characterize the time-dependent release kinetics of proangiogenic factors bound to the dECM (e.g. VEGF and FGF-2). Third, the peak injection pressure and pore size distribution were not quantified. Fourth, we did not perform controlled temperature ramp/time-sweep rheology (4°C −37°C) because of the lack of an active temperature unit; future work will incorporate temperature-controlled sweeps and 37°C isothermal tests to complement the available amplitude sweep data. To address these gaps, future work will (i) implement standardized syringe extrusion force–displacement testing to quantify injectability; (ii) employ automated image analysis to derive pore metrics; and (iii) perform the cross-sectional mapping of alginate shell thickness and uniformity to enhance reproducibility and process control.
At the mechanistic and translational levels, we propose prioritizing system-wide proteomics to map the bioactive composition of mesentery-derived dECM and elucidate the functional roles of key components in tissue regeneration. In parallel, the development and application of 3D bioprinting to fabricate anatomically customized implants with improved biomechanical integration merit investigation. Finally, stem cell-augmented organoid models offer a rigorous platform for evaluating the biocompatibility and long-term performance of breast prostheses and may help accelerate clinical translation. 54
Conclusions
Adipose-dECM was designed to support tissues specifically under irradiation-related stress by fostering a permissive microenvironment and maintaining early morphology. Our results show favorable irradiation tolerance consistent with that goal but reveal that long-term volumetric retention remains an area for improvement in the current formulation. Moreover, iterative optimization aimed at reinforcing network stability without compromising bioactivity may help to better convert early, radiation-compatible responses into durable outcomes.
Footnotes
Acknowledgements
The authors thank the staff of Harbin Medical University Cancer Hospital and the Experimental Animal Center for technical support.
ORCID iDs
Ethical considerations
All experimental procedures were performed in strict compliance with the protocol approved by the Institutional Animal Care and Use Committee of the Second Affiliated Hospital of Harbin Medical University (Approval No. SYDW2024-091).
Author contribution
Lingling Tao: Software, Writing- Original draft preparation. Yajie Gong: Visualization, Formal analysis. Shuang Gao: Investigation. Abiyasi Nanding: Formal analysis. Xueqiao Yu: Data curation. Saihan Bao: Investigation. Xi Chen: Visualization. Siliang Zhang: Methodology, Validation, Writing- Reviewing and Editing. Yang Li: Supervision, Validation, Writing- Reviewing and Editing. Xianyu Zhang: Conceptualization.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Haiyan Foundation of Harbin Medical University Cancer Hospital on Key Research Program, grant number JJZD2021-09 and also by National Cancer Center Climbing Foundation for Clinical Research Program, grant number NCC201908B04; by Heilongjiang Postdoctoral Fund Financial Assistance, grant number LBH-Z17115; by National Natural Science Foundation of China, grant number 82073410, 82272623; by Key Special Projects of Heilongjiang Province Key Research and Development Program, grant number 2023ZX06C10; by Nn10 Program of Harbin Medical University Cancer Hospital, grant numbers: 2017-02; by Spring Goose Support Program of Heilongjiang Province, grant numbers: CYQN24010.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Data will be made available upon reasonable request to the corresponding author.

