Abstract
In humans, the trachea is a conduit for ventilation connecting the throat and lungs. However, certain congenital or acquired diseases may cause long-term tracheal defects that require replacement. Tissue engineering is considered a promising method to reconstruct long-segment tracheal lesions and restore the structure and function of the trachea. Decellularization technology retains the natural structure of the trachea, has good biocompatibility and mechanical properties, and is currently a hotspot in tissue engineering studies. This article lists various recent representative protocols for the generation of decellularized tracheal scaffolds (DTSs), as well as their validity and limitations. Based on the advancements in decellularization methods, we discussed the impact and importance of mechanical properties, revascularization, recellularization, and biocompatibility in the production and implantation of DTS. This review provides a basis for future research on DTS and its application in clinical therapy.
Introduction
Tracheal defects and malformations are commonly encountered in clinical practice. Both conditions can occur congenitally or arise secondarily from tumors, trauma, and infections. As a conduit for human ventilation, the trachea connects the throat and lungs, and performs important physiological functions, including ventilation and respiratory regulation, cleaning, and immune function. Its structure includes a C-shaped cartilage ring, a mucous membrane of the respiratory tract, and muscle tissue. 1 The destruction of the tracheal anatomy impacts breathing and immune function and may be health- or life-threatening.2–4
The treatment of tracheal lesions is based on the maintenance of airtightness and continuity of the trachea, otherwise the physiological function of the trachea will be affected after treatment. Surgery is the preferred method for restoring the structure and function of the trachea. Short-length lesions can be repaired by direct anastomosis or tracheoplasty.5–8 However, if the lesion is longer than 30% of the length of an adult trachea or 50% of the length of a child trachea, 9 the diseased tissues need to be replaced. Traditional methods of replacement involve the use of autologous tissues, grafts made of artificial material, and transplantation. 8 Although these techniques have achieved good clinical survival rates and made significant progress in the treatment of long-segment tracheal defects,10,11 they still have limitations such as vascular erosion, risk of infection, limited availability of grafting materials, immune rejection, and development of malacia.11,12 However, tissue engineering – engineering tissues that are low-immunogenic, biocompatible, and durable – may solve these problems.12–14
Importantly, in tracheal tissue engineering, scaffolds provide support for cell growth, contributing markedly in tracheal regeneration. An ideal tracheal scaffold should have anatomical and morphological structures similar to those of the native trachea, and should allow cells to adhere, migrate, reproduce, and differentiate. It should also have appropriate biomechanical strength, be biocompatible and non-toxic.2,15,16 Decellularized tracheal constructs and synthetic materials are the two main methods used to prepare tissue-engineered scaffolds.12,16 Omori et al.
17
reported four cases who received biosynthetic scaffolds coated with collagen, the patients had good epithelialization of the reconstructed trachea and no significant obstruction during the follow-up of 8–34 months after surgery. Elliott et al.13,18 reconstructed a boy’s trachea using a decellularized tracheal tube seeded with bone marrow derived mononuclear cells, follow-up found neutrophil response locally in the trachea and until 18 months after surgery, the reconstructed trachea had sufficient mechanical strength, and the reconstructed trachea could meet the basic needs of life. However, in 2008, Macchiarini et al.
19
reported the first tissue-engineered airway case in
A decellularized tracheal scaffold (DTS) is obtained by removing the cellular and nuclear components from the donor trachea through chemical, physical, and enzymatic methods (Figure 1). It presents the advantage of good biocompatibility because it lacks immunogenic components. 22 Compared to a synthetic tracheal scaffold (STS), DTS preserves the original extracellular matrix (ECM) and natural tracheal 3-D structure, facilitating cell adhesion and regeneration, and inhibiting bacteria proliferation. 23 Although STS has better mechanical strength, it lacks the bioactivities of DTS. Therefore, some studies have attempted to combine natural and synthetic materials to achieve better biomechanical strength and biocompatibility.24,25
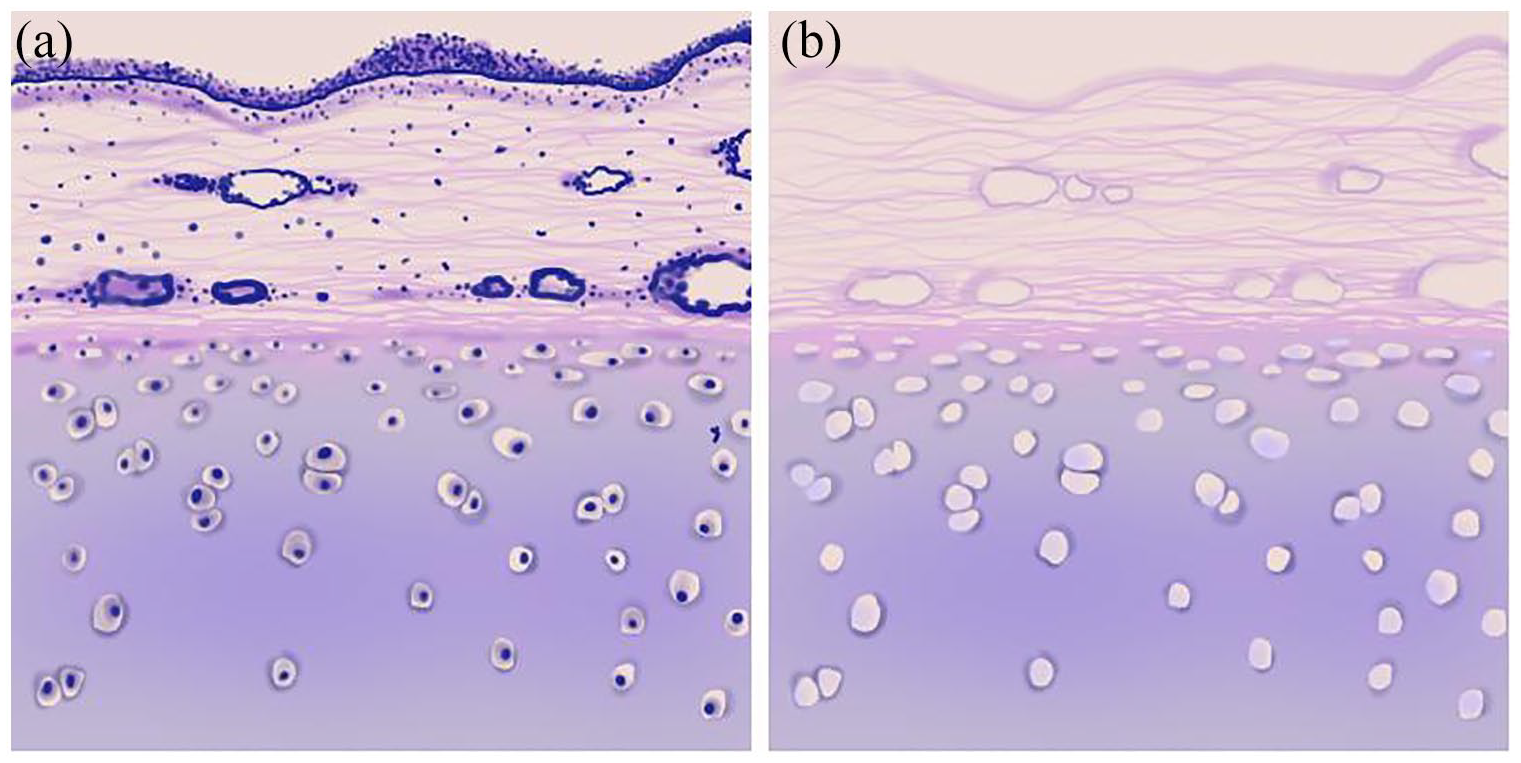
Overview of: (a) native trachea and (b) decellularized trachea.
The sources of tracheal scaffolds for decellularization are mainly divided into allograft and xenograft. Allotransplantation may minimize immune rejection and reduce the requirement for immunosuppressive therapy but has limitations such as shortage of donors. 26 Xenogeneic tissues may provide abundant right-sized trachea for tissue engineering. However, this technique may present ethical concerns, risk of rejections, and infections that cannot be ignored. 27
To date, DTS seems to be a promising alternative for long-segment tracheal reconstruction and has been clinically tried. 13 This article reviewed the advancements in DTS research and aimed to analyze the pros and cons of DTS. We also discussed future research prospects from the perspectives of DTS protocols, the mechanical properties and revascularization of DTS, the reproductive capability of cells on DTS, and the immune biocompatibility of DTS.
Production of a DTS
To obtain an ideal DTS, cellular and other immunogenic components should be completely removed from the donor trachea while maximizing the structural and mechanical components of the ECM. Compared to acellular scaffolds, cells and cellular components in incompletely decellularized scaffolds tend to elicit a proinflammatory response, 28 which negatively affects the reconstruction of tracheal tissues. 29
The three main indicators of a DTS are its biomechanical properties, recellularization, and vascularization. These largely depend on the composition and architecture of the ECM, greatly affected by the decellularization method.16,30,31 The main components of tracheal ECM include type II collagen, glycosaminoglycans (GAGs), fibronectin, and laminin. Type II collagen and GAGs are the two primary components that constitute the basic framework and structure of the DTS and determine its mechanical strength and elasticity.31,32 Loss of GAG during decellularization is significantly related to tracheal stenosis after transplantation. 33 Type II collagen and GAGs are also used as biomarkers to evaluate the chondrogenic capacity of cells. 34 Fibronectin and laminin contribute markedly in cell adhesion, migration, and revascularization.35–37 Laminins present in the basement membrane may provide a structural basis for cell adhesion and activate the signaling pathways underlying angiogenesis. 38 The laminin family is associated with re-epithelialization and neovascularization in normal tissue homeostasis. 39 Fibronectin, present throughout the lamina propria, is an adhesive glycoprotein that mediates cell adhesion. 40
The commonly used methods for decellularization include physical (such as freeze-thaw and lyophilization), chemical (such as sodium dodecyl sulfate (SDS) and Triton x-100), and enzymatic (such as DNase) methods.41–43 Repeated decellularization cycles may remove the immunogenic components of the donor tissue as much as possible. However, excessive durations of these inevitably cause the loss of GAGs in the ECM, potentially weakening the biomechanical integrity of DTS. 31 To maintain a balance between the removal of cellular components and preservation of cartilage ECM, the duration and method of decellularization are essential. Here, we list some recently developed representative DTS methods (Table 1).
Representative protocols used for tracheal decellularization and recellularization.
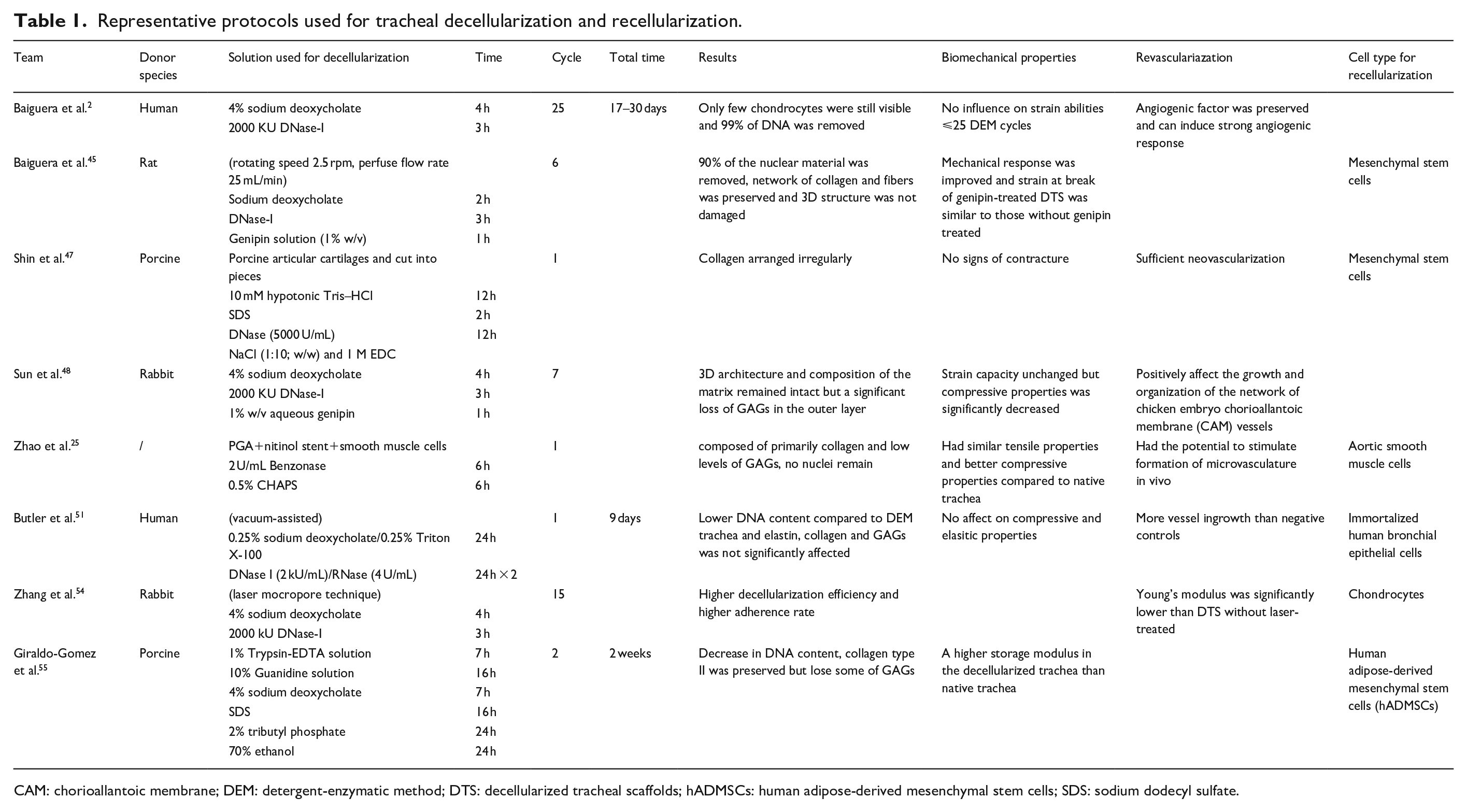
CAM: chorioallantoic membrane; DEM: detergent-enzymatic method; DTS: decellularized tracheal scaffolds; hADMSCs: human adipose-derived mesenchymal stem cells; SDS: sodium dodecyl sulfate.
In 2010, Baiguera et al. 2 used a chemical-enzymatic method to decellularize the human trachea. After the washing and sterilization steps, the tissue was successively treated with 4% sodium deoxycholate and 2000 KU DNase-I to completely remove the cells and nuclear contents. This protocol was repeated for 25 cycles to obtain a completely decellularized scaffold. The results showed that although 99% of the DNA was removed, a few chondrocytes were still detected in the central part of the cartilaginous area. This protocol is now the most widely-used method for obtaining an ideal DTS. However, it is time-consuming, putting some structural components of the ECM at risk of loss during the process. This method was later termed the detergent-enzymatic method (DEM), first reported in 1975. 44
In 2014, Baiguera et al. 45 modified the DEM protocol to decellularize the trachea. After being fixed to tubular steel adapters, the scaffold was put into a perfusion bioreactor called “InBreath” allowing for the decellularization solvent to circulate. After six DEM cycles, the scaffold was treated with a genipin solution (1% w/v). This method reduced the decellularization time by effectively increasing the diffusion of solutions into the trachea and removing 90% of the nuclear components. However, dynamic decellularization may affect the microstructure of the scaffold, thus decreasing mechanical performance. 46 Therefore, in this study, genipin was used as a cross-linking agent to improve the mechanical response, compensating for the loss of GAGs that undermined it.
In 2015, Shin et al. 47 generated a DTS through a combination of physical, chemical, and enzymatic methods. They acquired porcine articular cartilage, cut them into fragments, and treated them with 10 mM hypotonic Tris–HCl and 1% SDS. The cartilage pieces were then incubated with DNase (5000 U/mL). After these treatments, those fragments were freeze-dried and pulverized using an auto cryogenic sample crusher to produce porcine cartilage powder (PCP). Finally, the PCP was mixed with NaCl (1:10; w/w) and 1 M N-(3-dimethylaminopropyl)-N’-ethyl carbodiimide hydrochloride (EDC), and shaped into a disk using a mold. After seeding mesenchymal stem cells (MSCs) onto the PCP scaffold, the survival rate reached 91.6%. Neo-cartilage started to form 6 weeks after implantation into rabbits and the reconstructed area showed no contracture compared to the normal adjacent area. Additionally, sufficient vascularization and epithelialization were observed after the implantation. However, collagen irregularly arranged in the neo-cartilage revealed a disturbed microstructure of the ECM.
In 2016, Sun et al. 48 optimized the DEM to decellularize donor trachea. After seven cycles of DEM, the scaffold was treated with a 1% w/v aqueous genipin solution. Although hematoxylin and eosin (H&E) staining showed that nuclei were almost completely removed in the non-cartilaginous tissue, a small number of nuclei and cell debris remained in the lacunae. Moreover, there was a considerable loss of GAGs in the outer layer of the cartilage after seven DEM cycles. Genipin helped to make the ECM structure denser. Therefore, compared to the native trachea, the genipin-treated DTS had better tensile and compressive properties. Other studies have revealed that genipin may reduce inflammation and the rate of degradation during storage, and has a proangiogenic effect.49,50
In 2016, Zhao et al. 25 developed a novel method for obtaining a DTS. They first prepared a polymer-stent composite (outer layer: polyglycolic acid mesh; inner layer: nitinol stents) and then seeded it with smooth muscle cells. After the ECM had formed and the PGA had been largely degraded, the researchers used high ionic strength salts, 2 U/mL Benzonase solution, and 0.5% CHAPS solution to remove the cellular components, leaving only extracellular structural proteins. They combined polymer and decellularization techniques, taking advantage of both the synthetic tracheal scaffold and DTS. Histology of the scaffold revealed that no nuclei remained after decellularization, and that it was primarily composed of collagen and low levels of GAGs. The scaffold showed better mechanical strength than the native trachea and had good angiogenesis properties and biocompatibility.
In 2017, Butler et al. 51 applied a new technique that shortened the entire process to 9 days. The trachea was lyophilized prior to decellularization. The tissue was treated with 0.25% sodium deoxycholate/0.25% Triton X-100, followed by DNase-I (2 kU/mL)/RNase (4 U/mL) to remove cellular and DNA components. In contrast to previous methods, the entire process was performed in an autoclaved chamber generating a vacuum environment (1 Torr). Compared to the DEM, vacuum-assisted-decellularized trachea had a lower DNA content and similar ECM components (elastin, collagen, and GAGs), cytocompatibility, and biomechanical properties. Furthermore, using a vacuum may accelerate the process and minimize the loss of structural components caused by long-term storage.49,52
In 2017, Xu et al. 53 used a laser micropore technique to harvest a porous scaffold with appropriate mechanical strength. They first treated the trachea with a CO2 laser (output power 10 W, pulse width 17 ms, spot diameter 250 μm; repeated four times). Then, they used DEM for decellularization. Compared to the native trachea, the laser-treated DTS had similar strength and elasticity. In 2019, Zhang et al. 54 reported that laser-treated DTS had a higher decellularization efficiency and adherence rate. However, increasing numbers of decellularization cycles may cause further loss of GAG, affecting the mechanical strength (decreased Young’s modulus).
In 2019, Giraldo-Gomez et al.55,56 developed a new protocol based on a previous study that only required two cycles (2 weeks). The connective tissue surrounding the trachea was removed, following which the trachea was successively incubated in a 1% trypsin-ethylenediaminetetraacetic acid (EDTA) solution (in phosphate buffer solution (PBS)), 10% guanidine solution (in Tris buffer), and 4% sodium deoxycholate (in an ultrasound bath) to quickly and effectively remove cellular components. Next, DNase-I enzyme was used to hydrolyze the DNA chain. After decellularization, DNA was significantly decreased and type II collagen was preserved; however, some GAG molecules were lost. The DTS obtained in this study proved to be biocompatible and suitable for cell seeding. Dynamic mechanical analysis and thermal assessment showed no significant difference between the DTS and the native trachea (Figure 2).

Overview of trachea decellularization and potential cellular sources for recellularization.
Mechanical properties of DTS
Mechanical properties are crucial for maintaining the shape and function of the engineered tissues. 57 Failing to achieve the correct mechanical strength may cause DTS collapse and rupture during a live transplantation. Therefore, the trachea requires proper mechanical strength to maintain its biological functions. Only the right mechanical strength allows the DTS to function and perform bioactivities similar to those of the native trachea.31,58 Studies have shown that the tensile modulus (Young’s modulus) of the central part of the trachea is 3.33 MPa, while the average tensile modulus of the trachea is suggested to be 4.4 MPa.58,59 Therefore, if the DTS is to function biologically, its mechanical strength should be close to these indicators. Importantly, the mechanical properties of DTSs are determined by their structure and composition. Type II collagen and GAGs are the two main components of the ECM33,60 and contribute markedly in its mechanics and cartilage regeneration. Collagen fibrils build the mesh structure, whereas GAGs occupy the space between the fibrils; this structure forms the tensile and compressive properties of cartilage.32,61,62
Longitudinal flexibility (tensile and compression) and lateral stiffness are usually measured as mechanical indicators when evaluating the mechanical properties of a DTS. Numerous current studies use uniaxial tension tests to evaluate the mechanical properties of DTS, which measure the DTS as a radially symmetrical object.2,56,63 These tests apply a force to the DTS in a uniaxial direction that increases at a certain rate until DTS collapse or rupture. In addition to these indicators, expansion compliance should be considered. 64 In 2010, Remlinger et al. 65 pumped saline through a DTS, causing the pressure to mimic the natural movement of breathing, and tested the change in pipe diameter under different pressures to evaluate the expansion compliance of DTS. In 2012, Haykal et al. 9 also used (both negative and positive) pressure to change the air flow discharge on the wall of the DTS to simulate breathing movements, and then used a micro-computed tomography 66 scanner to measure the intraluminal volume of the DTS. In 2017, Den Hondt et al. 67 developed a new non-destructive method based on Haykal’s protocol.
However, the decellularization process inevitably influences the content of type II collagen and GAGs. Therefore, numerous methods have been used to reduce the damage to the ECM or improve the DTS stability.9,63,68–71 For example, Lange et al. 52 performed the decellularization process in a vacuum environment, allowing the solutions to penetrate deep into the tissue. The results showed that the DNA content of the vacuum-assisted scaffold was <50 ng/mg of wet tissue, and no difference was recorded between the soluble collagen/GAG content of the groups after treatment with or without vacuum. Baiguera et al. 45 used a dynamic decellularization method to prepare the solutions, improve tissue contact, and increase the efficiency of reactions. Using this method, DNA content was reduced from 1.4 ± 0.1 to 0.2 ± 0.01 ng/mL (a decrease of almost 90%), whereas elastin and GAGs content did not appear to decrease significantly. Importantly, the mechanical properties of the DTS were comparable to those of the native trachea. All these measures helped shorten the decellularization time, which may reduce the influence of the decellularization process on the structure of the DTS.
Attempts have also been made to find an ideal cross-linking approach that can stabilize the biomechanical integrity of DTS. Synthetic cross-link reagents, such as glutaraldehyde and diepoxy compounds, have been used to cross-link decellularized tissues.72,73 However, their application is clinically restricted because of cytotoxicity, calcification, and mismatched mechanical properties, leading to a failed transplantation.73–75 Therefore, natural-derived cross-linking reagents such as genipin, are preferable for cross-linking. For example, Haag et al.
50
cross-linked DTS with genipin after nine cycles of the DEM and found that the nuclear contents decreased from 1000.0 ± 8.9 to 29 ± 0.2 mg/mL (i.e. almost 97% was removed), but the ECM framework remained intact and appeared more compact. Mechanical tests showed a significant increase in the secant modulus, and the deformation characteristics remained unchanged after cross-linking. Sun et al.
48
compared different indicators of mechanical properties between native, decellularized, and genipin-treated decellularized. They found that after being cross-linked with genipin, the tensile modulus of the scaffold reached 5.51 ± 0.65 MPa (native trachea 4.62 ± 0.57 MPa, decellularized trachea 3.57 ± 0.23 MPa,
Revascularization of DTS
Vascularization is critical to the survival of a DTS. It occurs within a few weeks after transplantation and helps in metabolic circulation and the distribution of nutrients after transplantation.80–83 A good blood supply helps avoid various transplant-related complications such as collapse, necrosis, stenosis, and infection. Additionally, adequate vascularization improves DTS function and promotes the growth of tissues and nerves.84,85
After the implantation of a DTS, a wound-healing inflammatory response contributes to vascularization. Vasculogenesis and angiogenesis are the two main stages of vascular formation. 86 Vasculogenesis is the process by which endothelial progenitor cells are activated and migrate to the repair site to differentiate into endothelial cells and participate in the formation of the vascular network. 87 Angiogenesis refers to the formation and maturation of new vessels in the vascular network. This process is driven by the need for nutrients and oxygen 83 and is followed by arteriogenesis, which refers to maturation of the vasculature. 88
Some growth factors are involved in the regulation of revascularization, and studies have shown that the application of growth factors accelerates and promotes revascularization. Vascular endothelial growth factor (VEGF) is related to vascular organization and the elongation, migration, and proliferation of endothelial cells, and is widely-used to promote revascularization.89–91 Tan et al. 92 developed a perfusion system to continuously perfuse VEGF through the DTS to facilitate neovascularization. Following this treatment, erythrocytes were found throughout the DTS, and new vessel formation occurred earlier than in the control group. Furthermore, Baiguera et al. 2 used basic fibroblast growth factor (bFGF) as an indicator of the angiogenic potential of DTS. Some studies have reported that bFGF is related to the migration, differentiation, and proliferation of endothelial cells. 93 Platelet-derived growth factor (PDGF) and angiopoietin 1 also have the potential to enhance vascular formation and maturation.94,95 In the revascularization of DTS, the correct proportion and proper combination of growth factors are very essential.
Some studies have investigated the factors associated with the speed and potential of revascularization of the DTS. Partington et al. 31 used 25 cycles of DEM to decellularize the trachea, which retained the basement membrane structures in their original location. Angiogenic factors, including bFGF and laminin, were retained, thus supporting neovascularization. Walles et al. 96 pre-implanted the DTS with endothelial progenitor cells, and the vascular network started to endothelialize after 3 weeks. This study showed that the pre-seeding process mayfacilitate the growth of new blood vessels and prevent graft thrombosis and failure. 97 Moreover, pre-vascularization of DTS in vitro or in vivo prior to implantation may also facilitate the rapid vascularization of DTS.86,98
Recellularization of DTS
Recellularization is another vital process that ensures survival of the DTS. In the natural trachea, the external surface is occupied by chondrocytes, whereas the luminal surface is lined with a ciliated pseudostratified columnar epithelium maintaining the normal immune barrier function of the trachea. 99 Thus, there are two main cell types that need to be seeded onto the scaffold: chondrocytes and epithelial cells. To achieve ideal recellularization, the cells should be functional, survivable, easy to harvest, and not cause an immune response. Additionally, the technique to harvest these cells should also be convenient, safe, and least traumatic.
Recently, adult stem cells have been widely-used as cell sources. These include MSCs (both bone marrow- and adipose-derived), embryonic stem cells (ESCs), and induced pluripotent stem cells (iPSCs). 100 Due to their ability of self-renewal and potential for pluripotency, these cells present advantages compared to traditional chondrocytes. For example, MSCs are a widely-used cell type for the recellularization of DTS. 66 They are mainly isolated from the bone marrow and adipose tissue and are reported to be immunoprivileged cells101,102; harvesting MSCs only requires fine-needle aspiration. 100 Additionally, previous studies have reported that the application of MSCs accelerates revascularization and regeneration of the epithelium, and helps avoid complications after implantation.103,104 ESCs are another type of cell source used in tissue engineering. To obtain an ESC, researchers either implant the nucleus of a somatic cell into a nuclear-free oocyte, or use a defined chemical method to transform an oocyte without fertilization. 105 ESCs differentiate into functional chondrocytes. However, their clinical application is restricted due to ethical issues and potential tumor-inducing risks. iPSCs are derived from human fibroblasts and can differentiate into chondrocytes to form cartilage.106,107 They can also treat neurological disorders and some genetic diseases. 108 However, the use of iPSCs is complicated by the limited number of methods to induce their differentiation into chondrocytes, the risk of immune rejection, and ethical concerns.105,109,110 Chondrocytes may also be used for recellularization. There are many sources of chondrocytes, and they are usually isolated from the ribs, auricle, and nasal septal cartilage, most of which are non-weight-bearing cartilages.111,112 The auricle is the simplest site for harvesting cartilage to be used for regeneration. The nasal septum is another applicable cartilage source that has mechanical properties similar to those of the trachea, and its epithelial tissue may also be utilized. 113 However, the harvesting of chondrocytes is still relatively difficult, and their growth capacity is limited.
The re-epithelialization of DTS is an important part of recellularization. Several studies have reported that epithelial cells can be isolated, cultured, and functional when seeded onto DTS. 114 To harvest epithelial cells, many sites have been tested for regeneration, such as the trachea, nasal cavity, buccal mucosa, and skin. 115 Epithelial cells are the most widely-used cells to regenerate the epithelium of the DTS because they can differentiate into pseudostratified columnar epithelium (as in the native trachea). However, methods to induce differentiation remain inconvenient and unstable. Stem cells, which are easier to differentiate into ideal cell types, seem to solve these issues. MSCs, ESCs, iPSCs, amniotic fluid stem cells, and human umbilical cord blood-derived MSCs may also be applied in the re-epithelialization of DTS. These cells may proliferate and differentiate to maintain homeostasis and repair defects in the epithelium. 116
Sometimes, the different types of cells used for recellularization can cause an immune response in recipients after transplantation. 117 MSCs, because of their lack of MHC-II and low expression of MHC-I, seldom cause severe rejection in recipients.118,119 ESCs also present low levels of MHC-I and -II, but after transplantation with DTS, ESC can upregulate the MHC-I and -II expression and cause an immune response when faced with certain cytokines.120,121 As for iPSCs, there are reports of teratomas formation and immune response.118,122 To avoid this, fully differentiated iPSCs are recommended. 117
In the future, easier to harvest, more biocompatible and more readily available cells should be further investigated to facilitate recellularization. Additionally, techniques should be tailored to induce cell differentiation in a more stable and convenient manner.
Biocompatibility of DTS
Biocompatibility is one of the most important indicators for evaluating the DTS. Residual nuclear components, cell debris, and antigens in the ECM from incomplete decellularization may trigger an inflammatory response affecting the biocompatibility of DTS. 29 However, it is technically difficult to completely remove these antigens. Partington et al. 31 showed that even after 25 DEM cycles, DNA fragments were still detected in the DTS. DNA has high adhesiveness, thus tending to remain and adhere to the ECM. However, its side effects may be minimized as long as the fragment is smaller than 300 bp. 123
When a DTS with cellular components is implanted into the recipient, the host inflammatory response is activated by mononuclear cell infiltration, and macrophages express polarization toward an M1 phenotype.29,124 An ideal decellularized scaffold would promote the remodeling effect of tracheal tissue by inducing M2 phenotype macrophage differentiation.28,29,125 However, Ma et al. 126 used a micropump to perfuse decellularization solution through the bilateral carotid arteries to of Brown Norway rats to decellularize the larynx. They removed the mucosal epithelium, submucosal glands, and perichondrium but retained the intrachondral cells. After being implanted into a Lewis rat, the scaffold did not cause rejection, and neovascularization was observed. This may be because chondrocytes and the cartilage matrix do not express major histocompatibility complex-I and -II (MHC-I and MHC-II) antigens and do not elicit immunological rejection.126–128 Furthermore, chondrocytes are not nourished by direct blood supply, and the ECM – consisting of collagen, protein, and polysaccharides, functions as a physical isolation. Pan et al. 129 treated the trachea with 5% sodium perchlorate (NaClO4) during decellularization. They performed cytotoxicity tests for leaching solutions of NaClO4-treated DTS and found good cell activity. They also applied immunohistochemical methods to detect the remaining MHC after decellularization and found that the expression of MHC-I and MHC-II antigens in ECM was negative. Their study showed DTS treated with NaClO4, which is non-toxic and can help maximize the rate of cell removal, showed good biocompatibility.
Haykal et al. 130 used two different protocols to decellularize the trachea. They found that MHC proteins might remain in the cavity of submucosal glands during decellularization, potentially leading to the subsequent activation and proliferation of CD4+ T cells. They then used MSCs and tracheal epithelial cells to recellularize the DTS and found an inhibitory effect on CD4+ T cells and an increase in CD4+CD25+Foxp3+ regulatory T cells, suggesting an active immunomodulatory effect of recellularization.
Lange et al. 131 treated rabbit tracheas with a vacuum and decellularized them with Triton X-100/sodium deoxycholate and DNase/RNase. They used H&E staining to evaluate the biocompatibility of the DTS and used pan cytokeratin and CK7 to assess its ability to differentiate. The results showed that although vasculogenesis occurred after implantation into the recipient, differentiation toward respiratory epithelium was poor.
Giraldo-Gomez et al. 55 used 1% trypsin-EDTA solution, 10% guanidine solution, 4% sodium deoxycholate, and DNase-I to quickly and effectively decellularize the trachea. They evaluated the immunogenicity of DTS by implanting it subcutaneously in a mouse and evaluating the changes 1, 2, 7, and 15 days post-implantation. Compared with the native graft group, the decellularized graft group showed a similar inflammatory response on days 1 and 2 after implantation. However, the inflammation decreased on day 15, and the fibrosis was mild. Immunohistochemical analyses showed that TNF-α, a proinflammatory cytokine, declined after implantation in both the DTS and control group.
Although various new technologies and methods have been developed to improve the biocompatibility of DTS during decellularization, several challenges persist in completely eliminating the immunogenicity of DTS.
Future trends
Recently, three-dimensional (3D) bioprinting has emerged and developed rapidly. The term 3D-bioprinting refers to techniques that precisely fabricate 3D biological structures layer by layer, using living cells and biomaterials. 132 Bioink, or biomaterial ink, refers to materials used for 3D-bioprinting which is always living cells either with or without a supportive hydrogel component. 133 This technology has been explored in kidney, heart, cartilage, and other tissues. 132 In 2018, Bae et al. 134 developed a 3D-bioprinted trachea composed of epithelial cells, chondrogenic-differentiated bone marrow-derived MSC, and PCL frameworks. The trachea showed good compatibility, without stenosis or erosion, after transplantation into rabbits, and both neo-cartilage and neo-vascularization could be observed. Park et al. 135 used endothelial cells and fibroblasts encapsulated in porcine tracheal ECM as bioink to bioprint an airway within a designed PCL framework. It mimicked the microstructure and vascular network of human airway well. Overall, 3D-bioprinting is a promising technique in tracheal reconstruction, because it can not only precisely print the 3D structure of the patient’s trachea through their imaging data, but also accurately simultaneously arrange different kinds of cells in their positions layer by layer.
Conclusions
Recent years have seen marked technological progress in tracheal decellularization, and various decellularization techniques and methods of recellularization have been widely investigated. DEM – a combination of chemical and enzymatic decellularization methods, has been used as a classic decellularization method, and many efforts have been made to modify and improve it. Physical methods (vacuum, CO2 laser, etc.) have been applied to increase the contact area between the tissue and the detergent, or to accelerate and improve the efficiency of decellularization, thereby reducing the loss of structural components in the ECM. A combination of physical, chemical, and enzymatic methods for decellularization, as well as the combined use of synthetic materials and decellularization techniques, have also provided new ideas for the generation of DTS.
Maintaining the balance between the clearance rate of nuclear components and the retention rate of the ECM remains a challenge in the generation of DTS. The retention of the ECM should be maximized to retain the mechanical properties of the DTS and its ability to revascularize. To achieve this, various methods have been investigated in the decellularization process, such as the use of cross-linking agents and the application of perfusion decellularization methods, which can improve the mechanical properties of the DTS or reduce the time required for decellularization. Future studies are warranted to investigate more efficient decellularization agents and methods.
Recellularization is another essential factor for improving the survival rate of the DTS in recipients. Various types of stem cells, chondrocytes, and epithelial cells are currently used to recellularize the DTS. However, no cells are currently completely satisfactory. Therefore, efforts should be continued to find more widely-available and easier-to-grow cells for recellularization, while the technology for inducing differentiation requires optimization.
Here, we summarized the recent advancements in DTS production and listed the current challenges and limitations from various perspectives. After comparing these various studies, we believe that biocomposite decellularized scaffolds are the most promising. These scaffolds have excellent mechanical properties and good biocompatibility, because they combine synthetic materials and decellularization techniques. But how to maximize these two advantages requires more attention in the future, so that DTS can be applied in clinical settings as soon as possible.
Footnotes
Acknowledgements
We would like to thank Dawei Cui for his assistance and guidance in this research.
An authorship declaration
(i) That all authors listed meet the authorship criteria according to the latest guidelines of the International Committee of Medical Journal Editors, and (ii) that all authors are in agreement with the manuscript.
Author contributions
Lei CY and Xia C researched literature and conceived the study. Mei S and Zhou C wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Health Science and Technology Plan Project of Zhejiang Province [grant number 2020371214/2021KY506].
Guarantor
Chen Xia.
