Abstract
With the high global prevalence of dry eye disease and around 17% attributed to aqueous deficient dry eye (ADDE), there is an urgent need for therapies targeting lacrimal gland (LG) insufficiency. Advancements in in vitro LG analysis are essential for developing effective treatments that allow for efficient, ethical, and comprehensive evaluation of both cell-based therapies and pharmaceuticals. In light of the rapid progress in 3D bioprinting for tissue engineering, this study focuses on creating a composite hydrogel derived from decellularized porcine LG, combined with alginate. This combination leverages the bioactivity of the decellularized extracellular matrix (dECM) with the favorable biomechanical properties of alginate in a single bioink. Our results demonstrate that dECM supports the in vitro culture of LG cell types effectively, while alginate provides enhanced biomechanical stability. We analyzed the biomechanical and rheological properties of the bioink and printed 3D structures containing LG-derived cells, assessing cell viability both immediately post-printing and over a 7-day culture period. The LG-based bioink showed shear-thinning behavior, reduced cell sedimentation, and resilience to shear stresses comparable to pure alginate hydrogels. Additionally, we observed excellent viability of LG epithelial and mesenchymal stromal cells. This composite bioink, combining dECM and alginate, presents improved biomechanical properties while preserving biofunctionality, representing a promising platform for in vitro LG tissue engineering
Keywords
Introduction
Located within the superotemporal orbit is the lacrimal gland (LG), a serous gland measuring approximately 20 × 12 × 5 mm. 1 The LG’s primary function is to secrete the aqueous component of the tear film, essential for protecting the ocular surface and maintaining optical clarity of the cornea. 2 Age-related degeneration, trauma, and autoimmune diseases such as Sjögren’s syndrome or graft-versus-host disease can impair LG functionality, leading to a condition known as aqueous-deficient dry eye (ADDE). 3 ADDE can cause discomfort, sensations of foreign bodies, corneal defects, and potentially blindness. 3 Current treatments for ADDE are primarily palliative, involving eye drops or ointments. 4 Alternative treatments like salivary gland transplantation often result in quantitative (persistent hypo- or hypersecretion) or qualitative issues (hypoosmolar tear film) and have not gained wide clinical acceptance due to these challenges. 5 Regenerative approaches such as mesenchymal stromal cell therapy have demonstrated promising effects in mice, 6 and have been deemed safe after lacrimal gland injection in patients with Sjögren’s syndrome. 7 However, further clinical investigation is necessary as the observed effects in current studies remain modest.
Understanding the pathomechanisms of ADDE necessitates comprehensive in vitro analysis, analogous to the 3D models employed in heart, lung, and kidney research. 8 These models typically incorporate tissue-specific cells within organic or synthetic scaffolds. 8 dECM represents a promising organic scaffold for 3D cell culture, closely mimicking native cell microenvironments. 9 Recent works have shown that dECM hydrogels derived from porcine LG provide in vitro scaffolds with excellent cytocompatibility and high cell-matrix interaction, also promoting the cells’ physiological functions, especially secretion. 10 However, in its unmodified form, the hydrogel – similar to many dECM hydrogels – lacks certain properties essential for use as a viable bioink, such as shape fidelity and shear resistance.11,12 Further modification of the dECM LG hydrogel with genipin crosslinking has been shown to reduce biodegradation and increase scaffold stiffness while retaining biofunctionality. Still, genipin does not provide a significant advantage in the production of a dECM bioink, primarily due to its slow mechanism of action (requiring 24 h) and its lack of impact on the bioink before printing. 12 Alginate, a widely used bioink, offers excellent biocompatibility and mechanical flexibility, with the ability for adjustable crosslinking. 13 Nonetheless, alginate lacks cell-matrix interactions since cells do not have receptors capable of recognizing it. 14 Santis et al. successfully enhanced alginate’s bioactivity by incorporating dECM components. 15 The incorporation of murine lung dECM into alginate resulted in increased proliferation and metabolic activity of murine lung epithelial cells (MLE12). 15 Advancing upon these results, this study aims to incorporate the ionic crosslinking mechanism of alginate into the established dECM-based hydrogel of the LG (Figure 1). The objective is to improve shape fidelity and mechanical robustness of the hydrogel while retaining the critical advantages and biofunctionality of the native cell environment provided by the dECM.
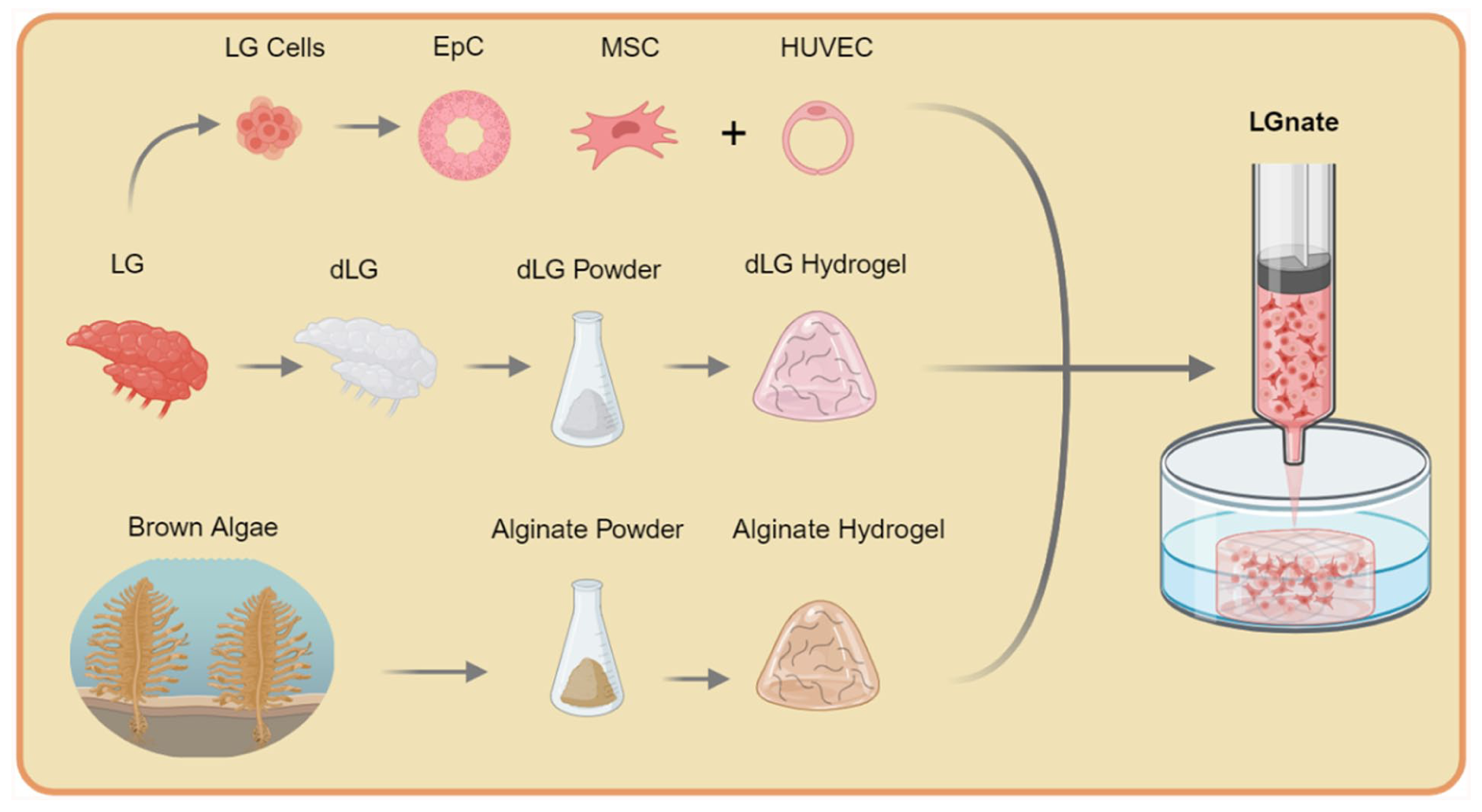
Study design. Porcine lacrimal glands (LG) were used for isolation of LG epithelial (EpC) and mesenchymal stromal cells (MSC). Further, the decellularized extracellular matrix of the LG (dLG) was processed into a hydrogel. A composite of alginate hydrogel, isolated from brown algae, dLG hydrogel and LG cells with the addition of human umbilical vein endothelial cells (HUVEC), is used to produce the LGnate bioink. Created in BioRender. Jansen, S. (2025) https://BioRender.com/qli7ahb
Materials and methods
Lacrimal gland extraction
All porcine lacrimal glands used in the experiments were freshly obtained from 8-month-old domestic pigs raised on a local farm. All experiments were conducted in accordance with the Association for Research and Vision in Ophthalmology statement for the use of animals in ophthalmic research. Excision was performed on the dead animal before any further processing steps of the meat production were conducted. Retrieved samples were used within 3 h post harvesting.
Decellularization of porcine lacrimal gland
Decellularization of the porcine lacrimal gland was performed as described before and is presented briefly. 16 The LG was prepared for decellularization under continuous agitation and sterile conditions. First, tissue was cut into approximately 3 mm pieces which were then incubated overnight at 4°C in phosphate buffered saline (PBS) containing 5% penicillin/streptomycin (P/S). The specimens then underwent a 36-h incubation at 4°C in a 1% (w/v) solution of sodium deoxycholate monohydrate (Sigma-Aldrich, Schnelldorf, Germany), with the solution exchanged thrice. Subsequently, the pieces were incubated in a DNase solution (200 U/mL in PBS, Roche, Penzberg, Germany) for 24 h. Following these treatments, the decellularized LG pieces were washed with cold PBS and then dab-dried before being stored at −80°C until further use.
Preparation of a hydrogel from decellularized lacrimal gland
The dECM-based hydrogel of the LG (dLG) was prepared as previously published. 10 In brief, decellularized porcine LG were lyophilized for 48 h using a Scanvac Coolsafe freeze dryer (Labogene, Lillerød, Denmark). Tissue samples were milled (EGK 200, Rommelsbacher, Dinkelsbühl, Germany) into particles smaller than 500 µm. For sterilization, the powdered tissue underwent tyndallization involving three cycles of heating up to 95°C for 30 min in a water bath, followed by cooling down to room temperature. For enzymatic digestion, LG powder at a concentration of 10 mg/mL was treated with 4000 U/mL of pepsin (P6887, Sigma) in 0.01 M hydrochloric acid at room temperature. Following digestion, suspensions were centrifuged at 200g for 5 min (Heraeus Megafuge16, Thermo Scientific, Waltham, MA, USA). The supernatants were transferred to clean 1.5 mL tubes and stored at −80°C until use. Samples intended for gel formation were thawed on ice and mixed at a 1:10 ratio with 10× Minimal Essential Medium (Gibco, Thermo Scientific). The mixture was titrated with 1 M sodium hydroxide (Sigma) until neutralized, yielding the final dLG solution with a concentration of 8 mg/ml. Gelation was achieved by incubating for 30 min at 37°C.
Gelation of 2% alginate
Low molecular weight alginate (alginic acid sodium salt, A1112 Sigma) was dissolved in distilled water at concentrations of 2% (w/v) or 4% (w/v). The alginate solutions were stirred for at least 1 h at room temperature in 50 mL centrifugation tubes to achieve homogeneity. To ensure sterility and prevent printhead blockage, the alginate solutions were filtered through a mesh with 0.2 µm pore size (Sarstedt, Nümbrecht, Germany). For the gelation of alginate pre-gels, calcium chloride (CaCl2) (C1016, Sigma) was used as a crosslinking agent. Various concentrations of CaCl2 ranging from 20 mM to 100 mM were tested. Alginate pre-gels were stored at 7°C until used or for a maximum of 7 days.
Preparation of bioink composite
Combining 4% (w/v) alginate pre-gel with dLG solution in a ratio of 1:1 yielded the hybrid bioink (LGnate) with a final concentration of 2% (w/v) alginate and 4 mg/mL dLG. Components of LGnate were mixed together shortly before use and from then stored on ice.
Bioprinting
Bioprinting was conducted using a pneumatic extrusion printer (Bio X, Cell Ink, Boston MA, USA) equipped with a blunt 27 Gauge nozzle of 12.7 mm length (Cell Ink) utilizing an approach based on freeform reversible embedding of suspended hydrogels (FRESH) 17 (Figure 2(a)). The preparation of the gelatin support bath was performed similar to Hinton et al. and involved the following steps performed at 4°C under sterile conditions: gelatin (G9391, Sigma) was dissolved in 150 mL of CaCl2 solution to a concentration of 6% and allowed to gel for 12 h at 4°C. The gelatin gel was then transferred to a household grade blender (Blendforce+ KB439D, Krups, Frankfurt am Main, Germany) along with additional 350 mL of CaCl2 solution. After blending for 2 min in pulse mode, the gelatin suspension was centrifuged in 50 mL centrifuge tubes at 3400g for 2 min (Heraeus Megafuge16). This process was repeated until no bubbles were visible at the top of the supernatant, indicating removal of all soluble gelatin particles. The final gelatin support bath was stored at 4°C until use for printing.

Bioprinting. (a) Setup of the freeform reversible embedding of suspended hydrogels (FRESH) bioprinting in a 12 well plate; (b) 2% alginate hydrogel specimens printed with increasing concentrations of calcium chloride in the support bath; (c) 3D model of the 3 × 3 grid used for calibration of the bioprinting process, bars of 1 mm thickness and 5 mm length, generated with slic3r; (d) 3D model of the cylindrical samples used in the bioprinting process, 8 mm in diameter, generated with slic3r; (e
Different concentrations of CaCl2 within the support bath were evaluated (Figure 2(b)). The geometries of printed specimens were defined by 3D models generated with the Slic3r software (http://Slic3r.org/) (Figure 2(c) and (d)). The inner diameter of the nozzle was determined using bright field microscopy (Leica DM 4000B, Leica Microsystems, Wetzlar, Germany) and the Fiji software. 18 A 3 mL cartridge (Cell Ink) was loaded with either LGnate or 2% alginate pre-gels. A 0.5 mm filament diameter was chosen for printing. Extruded volume rates were measured under different pressures to determine actual output. Printing accuracy and speed were confirmed by printing a 3 × 3 grid with connecting bars of 0.5 mm or 1 mm width (Figure 2(e)). Fiji software was used to measure grid dimensions. Printed structures were supported during crosslinking with a gelatin bath containing 50 mM CaCl2 in a 12-well plate at 7°C. After 30 min, the bath was dissolved at 37°C, and samples were transferred to a 48-well plate with LG medium (see 3.9) containing additional 50 mM CaCl2 for 1 h.
Biomechanics
For the measurement of Young’s modulus, unconfined compression tests of printed hydrogel specimens were performed utilizing a tensile testing machine (Zwickiline Z0.5 TN, load cell Xforce P, nominal force 10 N, Zwick Roell, Ulm, Germany). Specimens of 2 mm in height and 8 mm in diameter were compressed at a rate of 1 mm/min. For the comparison of 2% (w/v) alginate hydrogels, crosslinking was performed by incubation for 1 h in a gelatin support bath with CaCl2 concentrations of 20 mM, 50 mM, 75 mM or 100 mM. Comparison of LGnate and 2 % alginate was conducted after incubating for 30 min in a gelatin support bath with 50 mM CaCl2 at 7°C, and further 30 min at 37°C, followed by transferring samples into medium containing additional 50 mM CaCl2 for 1h at 37°C. Samples of fresh porcine LG (n = 3) or decellularized porcine LG (n = 6) were obtained with a round 8 mm tissue punch from the whole LG and measured equally.
All rheological measurements were performed with an oscillatory rheometer (Kinexus PRO; Netsch, Selb, Germany). Flow curves were obtained by loading non-gelated bioink (pre-gel) samples (n = 3) between a cone (diameter 60 mm; 1° angle) and plate (diameter 65 mm) geometry. Measurement of storage and loss modulus was done by loading hydrogel specimens (n = 4) between two parallel plates, diameter of the upper plate was 8 mm. A constant nominal force of 0.5 N was applied and gap size held constant at this point during testing. At a frequency of 1 Hz amplitude sweep tests were performed with strain ranging from 0.001 to 1.0.
Isolation and culture of cells
3T3 CCL 92 mouse fibroblasts
3T3 CCL 92 Swiss albino mouse fibroblasts (3T3 fibroblasts) (ATCC, Manassas VA, USA) were purchased and stored at −80°C until used. DMEM (D0819, Sigma) with 10% FCS (FCS superior, Biochrom, Berlin, Germany) and 1% P/S was used. 3T3 fibroblasts were routinely kept in culture at 37°C and 5% CO2. Cells were grown up to 70% to 80% confluency and passaged every other day. Applications of 3T3 fibroblasts comprised the co-culture of LG epithelial cells as well as analysis of cell sedimentation in line with established benchmark assays for bioinks. 19
Human umbilical vein endothelial cells
Human umbilical vein endothelial cells (HUVECs) (icell - h110, iCell Bioscience Inc, Shanghai, China) were procured and stored in liquid nitrogen until used. HUVECs were cultured in endothelial cell medium (EndCM) (VascuLife, Lifeline Cell Technology, Frederick MD, USA) at 37°C and 5% CO2. Cells were grown up to ~70% confluency until passaged or used for experiments.
Mesenchymal stromal cells
Freshly acquired porcine LG not destined for the decellularization process were kept in PBS during transport and storage until the beginning of cell isolation. Cell isolation occurred according to Roth et al. and will be described in short. 20 LG tissue was thoroughly washed with PBS and cut in halves. Adding 500 µL of Dulbecco’s modified eagle Medium (DMEM) (D0819, Sigma) to half a gland, the tissue was cut continuously with a scalpel into fine pieces until acquiring a pulp like consistency.
For the isolation of mesenchymal stromal cells (MSCs) the pulp-like tissue was aspirated and spread in a cell culture flask. Tissue pieces were shortly left to dry and attach to the surface. Subsequently, 25 mL of MSC Medium containing 500 mL Alpha MEM (Gennaxon Bioscience, Ulm, Germany), 75 mL FCS, 5 mL P/S, and 5 mL of 2 mM L Glutamine (Biochrom, Berlin, Germany) were added carefully to avoid lifting tissue pieces. Cells were maintained in an atmosphere of 5% O2 and 5% CO2.
Epithelial cells
Epithelial cells (EpC) of the lacrimal tissue were isolated in co-culture with 3T3 fibroblasts adhering to an established protocol. 16 Cell culture flasks were coated with a feeder layer of mitomycin (M4287, Sigma) treated 3T3 fibroblasts. Analogous to MSC isolation the tissue pulp from porcine LG was aspirated and spread evenly on the feeder layer. Epithelial cell culture medium (EpCM), consisting of 500 mL of DMEM F12 with 50 mL FCS, 0.4 mg/ml hydrocortisone, 0.1 nM cholera toxin, 0.18 mM adenine, 5 mg/mL transferrin, 5 mg/ml insulin (all Sigma), 10 ng/ml epidermal growth factor, and antibiotics/antimycotics (15240096, Thermo Scientific), was cautiously layered into the flask and cells were kept at 37°C and 5% CO2.Two-fold trypsinization ensured removal of the feeder cells in the first step and harvest of the EpC in the second step. 16
3D cell culture
Porcine LG derived EpC and MSC as well as HUVEC were suspended in the bioink before printing. For culturing the different cell types together in one hydrogel sample, LG Medium (LGM), a 1:1 Mixture of EpCM and EndCM, was used. To offset the degradation of alginate, the CaCl2 concentration of LGM was elevated to 2 mM. The used ratio of 5/10 EpCs, 4/10 HUVECs and 1/10 MSCs was previously found to be adequate. 21 All bioinks were prepared as previously described with a total cell density of 106 cells/ml. 2% (w/v) alginate bioink without any cells was run as blank. Cylinders of 6.4 mm in diameter and 2mm in height were printed and crosslinked for 1 h (30 min at 7°C, 30 min at 37°C) in gelatin support bath with 50 mM CaCl2 and an additional 1 h in LGM with 50 mM CaCl2. The printed specimens were then kept in a 48 well plate with 800 µL of LGM at 37°C and 5% CO2. The medium was exchanged every other day.
Viability assessment
Specimens in 3D cell culture were analyzed on days 3 and 7. The alamarBlue Assay (Thermo Scientific) was performed according to the manufacturers protocol. In brief, medium was replaced with 200 µL of LGM containing 1:10 alamarBlue cell viability reagent and samples were then incubated for 3 h. To accommodate for increased consumption within the hydrogels, the solution was mixed thoroughly with a pipette before transferring 100 µL into a 96 well plate with black walls and clear bottoms (Greiner Bio One, Kremsmünster, Austria). Results were acquired with a photometric microplate reader (Spark, Tecan, Männedorf, Switzerland).
Cell sedimentation
Sedimentation of cells within the bioinks was assessed utilizing a previously published assay procedure. 19 Viable 3T3 fibroblasts were stained by incubating for 30 min in 4 µM Calcein (Thermo Scientific) and then suspended in bioinks with a density of 106 cells/mL. Cell laden bioinks were transferred into 70 µL cuvettes (Eppendorf, Hamburg, Germany) and incubated at 37°C for 1 h. Medium was used as control. Cuvettes were carefully rotated 90° prior to imaging (Leica DM 4000B, Leica Microsystems). Post processing of the images was performed with the Fiji application, and images were split into four vertical sections of equal size. Resulting images were utilized to calculate a sedimentation coefficient (δ) for the bioinks following an established approach 19 :
With n := number of sections evaluated in one cuvette (n = 4) and ci := cell density of each section. Normalizing the coefficient to 1, the inverse (δ−1) was used.
Thus δ−1 = 1 indicates no observed sedimentation and δ−1 = 0 represents complete sedimentation within a sample.
Statistical analysis
Data are expressed as mean ± standard deviation. Statistical analyses were performed using an unpaired Student’s t-test, one-way ANOVA followed by Tukey’s post hoc test, or two-way ANOVA followed by Fisher’s least significant difference (LSD) test, as appropriate, using GraphPad Prism 10. A p-value < 0.05 was considered statistically significant.
Results
Bioink calibration
To define the exact nozzle diameter, a total of three nozzles was imaged and measured (Figure 2(f)). The inner diameter of the nozzles was 225.6 ± 6.2 µm. At predefined pressures, the volume output of the printer was recorded (Figure 3(a)). For pressures below 7 kPa continuous extrusion of LGnate filament was not feasible. The same was true for pressures below 4 kPa and 2% alginate. At exactly 7 kPa the filament output was 2.178 ± 0.15 µLs−1 for 2% alginate or 1.746 ± 0.14 µLs−1 for LGnate. Hence for printing a filament of 0.5 mm in diameter, a printing speed of 11.1 mms−1 was needed for 2% alginate and a speed of 8.9 mms−1 for LGnate. Validating the configuration method was done with printing and measuring three by three grids. A grid with 0.5 mm thick bars was printed with 0.473 ± 0.09 mm actual thickness. Bars with 1 mm thickness resulted in an actual thickness of 0.980 ± 0.11 mm.

Biomechanics. (a) Extrusion volume of 2% alginate pre-gel and LGnate bioink in relation to extrusion pressure (n = 3); (b) Viscosity measurement for varying shear rates of 2% alginate and LGnate pre-gels, (n = 3); (c) Results of unconfined compression tests of 2% alginate hydrogels printed in support baths with increasing concentrations of CaCl2 (n = 6), p20 vs 50 = 0.0023, p 50 vs 75 = 0.0028, one way ANOVA; (d) Results of unconfined compression tests of 2% alginate hydrogels (n = 6), LGnate (n = 8) and fresh porcine lacrimal gland (pLG) (n = 3), p = 0.13, one way ANOVA; (e) Stress, Storage and loss moduli of 2% alginate hydrogels acquired in amplitude sweep tests, each datapoint represents the mean value of the associated strain (n = 4); (f) Stress, Storage and loss moduli of LGnate hydrogels acquired in amplitude sweep tests, 8mm plate plate geometrie, (n = 4); (g) Maximum storage modulus of 2% alginate and LGnate hydrogels (n = 4) p = 0.001, unpaired t-test; (h) Stress at crossover (G’ = G’’) for 2% alginate and LGnate hydrogels (n = 4) p = 0.138, unpaired t-test; All error bars represent standard deviation.
Biomechanics
The shear dependent viscosity prior to gelation is a key property of viable bioinks. LGnate pre-gel showed desirable shear thinning properties in the dynamic viscosity measurement with a peak viscosity of 21.79 ± 8.4 Pa*s as opposed to 2% alginate pre-gel which behaves similar to water (Figure 3(b)). Unconfined compression tests were performed to evaluate the influence of several modalities on the stiffness of the specimens. Different concentrations of CaCl2 during 1 h of crosslinking showed significant differences in stiffness of 2% alginate hydrogels (p = 0.0013) (Figure 3(c)). Mean Young’s modulus (e) when crosslinked with 50 mM CaCl2 was 4.570 ± 0.53 kPa and therefore significantly larger than hydrogels crosslinked with 20 mM (2.90 ± 0.65 kPa) and 75 mM (2.933 ± 0.46 kPa) (Figure 3(c)). The concentration of CaCl2 during crosslinking also had a visible influence. Specimens crosslinked with 20 mM CaCl2 showed increased swelling and extended clearly beyond the initially printed geometry, while crosslinking with 75 mM CaCl2 lead to contraction and an overall less sharp geometry (Figure 2(b)). In Figure 3(d), the Young’s modulus of porcine LG as well as LGnate and 2% alginate hydrogel crosslinked with 50 mM CaCl2 is shown. No significant difference was detected between the Young’s moduli of LGnate (3.121 ± 1.51 kPa), porcine LG (6.314 ± 3.22 kPa) and 2% alginate hydrogels (4.379 ± 2.00 kPa).
Results of the amplitude sweep tests for LGnate and 2% alginate hydrogels show the strain-dependence of storage and loss moduli (Figure 3(e) and 3(f)). Specimens of LGnate hydrogel showed a maximum storage modulus (G’) of 2490 ± 145 Pa whereas 2% alginate hydrogels expressed significantly higher values with maximum G’ of 3648 ± 167 Pa (Figure 3(g)). However, comparing the stresses at crossover points, with the crossover point indicating loss of gel structure, no significant difference between LGnate and 2% alginate was observed (Figure 3(h)).
Viability
At day 3 and 7, cells cultured in LGnate hydrogels showed significantly higher cell viability (Figure 4(a)). Compared to 2% alginate viability was 4.8-fold higher on day 3 and 7.0-fold higher on day 7.

Cell based analysis of bioinks. (a) Comparison of viability in bioprinted specimens of 2% alginate or LGnate hydrogels containing porcine lacrimal gland (pLG) epithelial cells, pLG mesenchymal stromal cells and human umbilical vein endothelial cells, measured via metabolic activity (n = 6, p3 < 0.0001 p7 < 0.0001), two way ANOVA and fisher’s LSD for multiple comparisons); (b) Sedimentation coefficients of LGnate and 2% alginate (n = 4, p = 0.0063, unpaired t-test); (c) representative images of cell sedimentation of calcein stained 3T3 mouse fibroblasts in LGnate, 2% alginate and medium, scale bar 1 mm.
Cell sedimentation
Sedimentation coefficients of LGnate, 2% alginate and medium were 0.99 ± 0.0, 0.89 ± 0.0 and 0.00 ± 0.0, respectively (Figure 4(b)). Representative pictures show the cell distribution over the full fluid column height in the cuvettes after 1 h of vertical incubation (Figure 4(c)). Viable cells show bright green fluorescence due to calcein staining and differences in sedimentation correlate with the determined coefficients. While in LGnate, the dispersion remains stable, a significant part of cells dispersed in 2% sodium alginate solution has settled to the bottom of the cuvettes after 1 h of incubation. In medium all cells have settled.
Discussion
In summary, the incorporation of alginate into the dLG solution results in the formation of a bioink blend that exhibits the needed biomechanical properties essential for the fabrication of the intricate 3D structure of LGs. Furthermore, this bioink not only facilitates cell survival throughout the printing process but also sustains cell viability and proliferation for at least 7 days post-printing. This bioink blend was chosen to address some of the most frequent limitations encountered in dECM hydrogel-based research. Low mechanical strength and low shear stress resistance represent the predominant challenges of dECM scaffolds. 22 Further hurdles with regard to bioprinting comprise low shape fidelity and limited control over bioink properties such as viscosity and gelation speed. 23 The alginate component induces ionic crosslinking capability in LGnate and was shown to compensate some key limitations. Thus it was shown that LGnate represents a promising bioink, especially considering that unmodified dLG materials are not compliant with extrusion 3D bioprinting.10,12 As a result, printed LGnate constructs could not be directly compared to printed, pure dLG Hydrogels. However, this study is mainly focused on physico-chemical properties of the hybrid bioink and a more comprehensive biological characterization of the bioink in comparison to pure dLG hydrogels 10 will be subject of upcoming studies.
For one, the stiffness of a matrix is known to influence cell proliferation, function and even differentiation. 24 Hence in respect to a 3D in vitro model it is beneficial to generate a stiffness as close to the native tissue as possible. Crosslinking 2% alginate in a gelatin support bath containing 50 mM CaCl2 yielded the highest Young’s modulus of the printed hydrogels. The lower values measured for higher CaCl2 concentrations are unusual, since increasing the concentration should result in higher stiffness. 25 A likely explanation is that in higher concentrations the gelation occurred too fast and resulted in a crosslinked shell and thus heterogeneously crosslinked layers of reduced overall stiffness. The established crosslinking process following biofabrication resulted in Young’s moduli of 4.379 ± 2.00 kPa for 2% alginate hydrogels and 3.121 ± 1.51 kPa for LGnate hydrogels. These values do not significantly differ from the measured fresh porcine LG and also fit the range of healthy human LG albeit at the lower end (6.17 ± 2.91 kPa). 26 Even though all LGs examined by Cerit and Sedur were considered healthy, factoring in age of the patient yields mean Young’s moduli of 6.89 kPa for LGs from patients younger than 40 and of 3.79 kPa for LGs of patients older than 40. 26 Hence possible modification of the crosslinking process and the alginate concentration could be implemented to steer LGnate hydrogels towards characteristics of a ‘younger’ LG. Especially considering age as one major factor for insufficiency of the LG. 27 In that regard, tunable stiffness of LGnate scaffolds might represent a key property for example in mimicking aging by varying scaffold stiffness.
Another result of relevant concern is the maximum storage modulus of LGnate. The addition of alginate results in improved resistance towards shear stress as well as higher stiffness and shape fidelity of LGnate in regard to LG-dECM hydrogels. 12 This is crucial to enable dLG based bioprinting in the first place. Though considering alginate the biomechanically dominant component in LGnate, the maximum storage modulus of LGnate was about 30% smaller compared to 2% alginate hydrogels. The storage modulus describes the elastic response to shear stresses in viscoelastic materials and a lower value might therefore indicate reduced mechanical stability. 28 One reason might be the composition of the LGnate hydrogel. The chosen 1:1 ratio was determined empirically to balance printability with biofunctionality in accordance with results published in De Santis et al. 15 Preliminary trials with higher dLG fractions resulted in poor extrusion fidelity and loss of structural stability. The concentrations of alginate and dECM need to be in balance to enable microscale phase separation. 15 While the concentration of dLG in LGnate did result in the desired shear thinning properties, too high of a concentration might lead to macroscale phase separation possibly causing mechanical instabilities. 15 In line with observations from de Santis et al., the tested cell loading density of alginate and LGnate bioinks prior to gelation did not cause significant alteration of their bioprinting properties. 15 However, in regard to the stress-crossover point, which is the most critical parameter, LGnate showed no inferiority to 2% alginate and thus can be considered just as resistant towards applied shear stress. One major benefit of LGnate over 2% alginate is observed in the comparison of LG cell viability. Higher values of metabolic activity in LGnate on day 3 and 7 indicate higher survivability rate of the printing process as well as more favourable growth conditions and thus faster recovery of the cells. Representatively, this increase in viability was utilized as a marker for the biofunctional benefits introduced by the dLG component. Although the present study demonstrated cell viability and mechanical stability for up to 7 days, longer-term data on matrix stability, degradation kinetics, and remodeling. Indeed, alginate and LGnate hydrogels crosslinked with Ca²+ exhibit gradual degradation in line with other alginate based bioinks. 29 In designing LGnate, this degradability was viewed as advantageous, but it also limited the feasible culture duration. Alternative crosslinking strategies such as using barium ions are known to reduce degradation rates, but result in cytotoxic effects, as evidenced in Souza et al. 30 Functionalization of alginate enables cell-matrix interactions and has been proven as a feasible improvement to generate alginate scaffolds for long-term culture systems omitting the need for removal of alginate components down the line. 31 A similar functionalization of LGnate will thus be subject of future investigations. Further, regarding pre-gel bioinks, cell sedimentation plays a vital role when considering recent advances in bioprinting towards increasingly complex technical setups and scaffold designs. With the associated increase in printing duration, maintaining homogenous cell distributions over long periods of time is a key property of bioinks. Thus, the significance of the excellent observed sedimentation coefficient in LGnate is underlined. With regard to scalability, LGnate has been proven stable for printing small constructs and viability analysis has shown sufficient diffusion properties at that scale. Larger models will require strategies to overcome diffusion limitations. Perfusion bioreactors 32 or pre-vascularization15,33 have been shown as successful options in dECM based models and will be accommodated in future approaches.
In summary dLG hydrogels comprise many favourable biofunctional properties which render these scaffolds suitable for 3D cell culture in LG research.10,12 Their inherent biomechanics however are not compliant with bioprinting. Contrarily, alginate bioinks offer excellent biomechanical properties, but no enhancement of biofunctionality. 14 The presented results showed that LGnate bioink combines advantages of both materials. Inferring from the aforementioned limitations though, further experiments or modifications should be conducted in order to establish LGnate based 3D in vitro models. The aim was to show that alginate as an additive to dLG hydrogel, and thus also potential similar dECM hydrogels derived from glandular tissues, is able to induce the biomechanical properties necessary for 3D bioprinting and provides a valuable building block for in vitro based LG studies. As another advantage, alginate and dLG components also allow for plentiful modification and adaption to overcome the presented issues. Taking into account that dLG solution in its unmodified state is not suitable for use in bioprinting, this study accomplished to develop a viable bioink. Reinforcing the pre-gel solution of dLG with 2% alginate yields a bioink that can successfully be used for printing complex, cell laden 3D structures. Due to a higher viscosity of the dLG component compared to 2% alginate pre-gel, cells show minimal sedimentation in the hybrid LGnate supporting even cell distribution during printing. In a time period of 7 days, cell viability of LG associated cells was retained and promoted. With regard to the rapid emergence of dECM based hydrogels in tissue engineering, LGnate provides an exemplary solution to frequently observed limitations in dECM applications. Concluding, LGnate bioink represents a promising building block for 3D bioprinting and LG associated research.
Footnotes
ORCID iDs
Ethical considerations
This article does not contain any studies with human or animal participants
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.

