Abstract
Collagen type XVII is a unique transmembrane protein that plays a crucial role in anchoring epithelial cells to the underlying basement membrane. However, the low natural abundance of collagen type XVII in the human body poses significant challenges for large-scale extraction and production. With a development in biosynthesis technology, type A recombinant humanized collagen type XVII (rhCol XVII) has emerged as a promising and innovative alternative. In this study, rhCol XVII was successfully synthesized and expressed using genetic engineering techniques. The amino acid composition of rhCol XVII was analyzed, and its molecular weight was determined to be consistent with the theoretical design. Key physicochemical properties, including isoelectric point, particle size, and thermal stability, were evaluated. The experimentally measured pI was approximately 9.00, a particle size ranges from 820 to 857 nm, and thermal stability up to 23.60°C. The structure of rhCol XVII was characterized using infrared spectroscopy and circular dichroism. Furthermore, the cytocompatibility of rhCol XVII was assessed in vitro, and its systemic safety was evaluated in vivo. This research provided a solid foundation for the potential application of rhCol XVII in tissue engineering and regenerative medicine.
Introduction
Collagen type XVII (Col XVII) is a unique transmembrane protein, with its N-terminus located intracellularly and its C-terminus extending into the extracellular space. Structurally, it comprises an intracellular domain, a transmembrane domain, and an extracellular domain. The extracellular domain contains 15 collagen-like domains (Col domains) interspersed with 16 non-collagenous domains (NC domains), forming an interrupted triple-helical structure. This structure arrangement confers both flexibility and functional versatility to Col XVII. 1 As a central structural component of extracellular domain in hemidesmosomes (HDs), Col XVII is essential for anchoring epithelial cells to the underlying basement membrane. Its extracellular domain directly binds to basement membrane components, while its intracellular domain interacts with cytoskeletal proteins, serving as a critical bridge between the extracellular matrix and the cytoskeleton. 2 Col XVII is also implicated in various dermatological and autoimmune diseases, highlighting its potential as a therapeutic biomaterials. 3 The expression level of Col XVII decreases during skin aging, 4 corneal aging, 5 and hair loss, 6 resulting in basement membrane dysfunction and a weakened dermo-epidermal junction. 7 Additionally, it interacts with keratin intermediate filaments and the actin cytoskeleton through its intracellular domains, modulating cytoskeletal reorganization and contributing to cell migration. 8
Col XVII not only serves as a structural anchoring protein to maintain the stability of tissue structure, but also acts as a bioactive component that regulates the biological behaviors of surrounding cells, such as cell adhesion, proliferation, migration, phenotype maintenance and differentiation. Qiao et al. 9 demonstrated that knocking down Col XVII expression in HaCaT cells using small interfering RNA (siRNA) inhibited cell migration through the p38 MAPK signaling pathway. The MAPK pathway plays a crucial role not only in cell migration but also in cell adhesion, gene transcription, and stress responses. These findings have indicated that Col XVII is essential for keratinocyte adhesion and transmitting signals from the extracellular matrix to intracellular pathways during cell migration. In addition, Tuusa et al. 10 had reviewed that Col XVII plays a pivotal role in regulating the adhesion, separation, development and differentiation of epithelial cells, which is crucial for keratinocyte differentiation and regeneration.
Col XVII is found in the basement membranes of the skin, cornea, and other epithelial tissues. Animal-derived Col XVII faces significant challenges, including low extraction efficiency, potential immunogenicity, and batch-to-batch variability, all of which compromise its safety and biological function. Recombinant humanized collagen as its amino acid sequence is entirely derived from human-specific functional domains and a tandem arrangement of these functional regions, which has favorable safety and effectiveness. Therefore, the ability to mass-produce type A recombinant humanized collagen type XVII (rhCol XVII) through genetic engineering is of great significance. As a novel biomaterial characterized by a favorable safety profile and defined biological functions, rhCol XVII holds significant promise for future medical research and development. Wang et al. 11 discovered that rhCol XVII could promote the growth and adhesion of primary human keratinocytes (HPKCs). In clinical applications, it had been demonstrated to accelerate wound healing in patients with bullous diseases (BD), thereby reducing the risk of subsequent secondary infections. Hao et al. 12 demonstrated that rhCol XVII dissolved in a carbomer spray could be formulated into a drug-loaded dressing. Using a rat oral ulcer model, they evaluated the therapeutic effects of the rhCol XVII containing spray and found that it significantly accelerated ulcer healing. Additionally, in vitro studies showed that rhCol XVII reduced cellular inflammation in human gingival fibroblasts while promoting cell proliferation and migration. These findings suggest that rhCol XVII-containing sprays hold promise as a novel approach for oral ulcer treatment. Wang et al. 13 established a skin injury model by exposing human HaCaT keratinocytes to ultraviolet B (UVB) irradiation. Their findings demonstrated that rhCol XVII protected skin basement membrane integrity by inhibiting the MAPK and Wnt signaling pathways, indicating its therapeutic potential for UVB-induced skin damage.
Given the significant biological and structural activity of rhCol XVII in clinical applications, we successfully designed and produced rhCol XVII using genetic engineering. The full-length amino acid sequence of rhCol XVII consists of three tandem repeat units of Gly683-Ala745 derived from the native collagen type XVII amino acid sequence. In the present investigation, we analyzed the amino acid composition and molecular weight of rhCol XVII to verify its consistency with the theoretically designed sequence. The physicochemical properties, including isoelectric point (pI), particle size, and thermal denaturation temperature, were also measured. Moreover, the advanced structure of rhCol XVII was characterized using infrared spectroscopy and circular dichroism. The effects of rhCol XVII on the biological behavior of human skin fibroblasts (HSFs), including proliferation, migration, adhesion, and cytoskeletal morphology, were evaluated. Furthermore, an enzyme-linked immunosorbent assay (ELISA) was performed to investigate its effect on extracellular matrix protein expression. Systemic toxicity and pyrogenicity tests were conducted in accordance with the national standard GB/T 16886.8 to evaluate the biosafety of rhCol XVII. This research provided a solid foundation for the potential application of rhCol XVII in tissue engineering and regenerative medicine.
Materials and methods
Materials
Lyophilized rhColXVII was provided by Shanxi Jinbo Biological Pharmaceutical Co. Ltd. The structural sequence of rhCol XVII refers to the National Center for Biotechnology Information (NCBI), with its NCBI reference sequence being: Q9UMD9.3 (SEQID No.9). HSFs were obtained from the National Authentic Cell Culture Bank in Shanghai, China. Fetal bovine serum (FBS, Gibco, Australia origin) was bought from Thermo Fisher Scientific Corporation (USA). Dulbecco’s Modified Eagle Medium (DMEM, high glucose) and phosphate buffered saline (PBS) powder was purchased from Servicebio (Wuhan, China). Sigma-Aldrich (USA) provided fluorescein diacetate (FDA), and 4′6-diamidino-2-phenylindole (DAPI). The preparation of bovine type I collagen (bCol I) was conducted following the methodology described in the research by Wang et al., 14 which met the requirements of this study.
Characterization of rhCol XVII
Analysis of amino acid content
Based on prior research, the amino acid composition of rhCol XVII was analyzed using an amino acid analyzer (L-8900, Tokyo, Japan). 15 Briefly, 2.00 mg of rhCol XVII was dissolved with 1 mL of 6 M hydrochloric acid, purged with nitrogen gas for approximately 3 min, then sealed and placed in a dry block heater for hydrolysis at 110°C for 24 h. After hydrolysis, the samples were vacuum-dried to remove the acid, and the hydrolysates were dissolved in sodium citrate buffer (pH = 2). The chromatographic column was selected to separate different amino acid and the detection wavelengths were 570 and 440 nm. Samples were then analyzed according to the instrument’s manual instructions.
Molecular weight and purity analysis
Sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE)
The molecular weight of rhCol XVII was characterized using a gel electrophoresis system (Power Pac HV Power Supply, BIO-RAD). The rhCol XVII was dissolved in double-distilled water at room temperature to a concentration of 1.00 mg/mL. SDS-PAGE was performed using a 15% separating gel and a 4% stacking gel. The samples were prepared with loading buffer (100 mM Tris-HCl, pH = 6.8, 4% SDS, 20% glycerol, and 0.2% bromophenol blue) in a 1:1 ratio, then boiled for 15 min. A 10 μL aliquot of each sample was loaded onto the gel and electrophoresis was performed at 60 V until the samples entered the separating gel, after which the voltage was increased to 120 V. After electrophoresis, the gel was stained with a dye solution 0.1% (w/v) containing Coomassie Brilliant Blue R-250, 50% methanol, and 10% acetic acid) for 25 min, followed by destaining with a solution containing 25% (v/v) methanol and 7.5% acetic acid solution until the bands were clearly visible.
High performance liquid chromatography mass spectrometry (HPLC-MS)
The molecular weight and distribution of rhCol XVII were determined by HPLC-MS. rhCol XVII was dissolved in ultrapure water to a final concentration of 1 mg/mL. The rhCol XVII solution was separated on a BioResolve RP mAb column (450 A, 2.7 μm particle size, 3 mm × 100 mm). Mobile phase A consisted of a 0.1% TFA solution in double-distilled water, and mobile phase B was an acetonitrile solution containing 0.1% TFA. A 2 μL aliquot of the sample was loaded using an autosampler and separated on a column at a flow rate of 0.3 mL/min with a UV detection at 210 nm. The samples were analyzed using an XevoG2-XS QT of mass spectrometer, operating in positive ion scanning mode within the range of 500–4000 m/z. The raw data were processed by using UNIFI (1.8.2, Waters) software, for specific parameter settings refer to Table 1.
Software parameters.
Isoelectric point and particle size analysis
The rhCol XVII solution was prepared at a concentration of 1 mg/mL using deionized water and placed in a beaker. The pH of the rhCol XVII solution was then titrated to approximately 3.00 using 1 mol/L HCl. Subsequently, the pH of rhCol XVII solution gradually increased to basicity by adding 0.2 mol/L NaOH. The pH of the rhCol XVII solution was continuously monitored using a pH meter (FE20K, Mettler Toledo). During the titration process, the Zeta potential and hydrodynamic diameter of the rhCol XVII were measured using a laser particle sizer (NANO ZS9, Malvern, UK) at each pH point. The Zeta potential and diameters were averaged over three scans for each measurement. After the measurement was completed, under neutral conditions, the sample was serially diluted, and its particle sizes were determined at concentrations of 1.000, 0.500, 0.250, and 0.125 mg/mL, following the same method as described previously.
Thermal stability analysis
The thermal denaturation temperature of recombinant human collagen XVII (rhCol XVII) was determined using a differential scanning calorimeter (DSC 3+, Malvern). After weighing the sample, it was dissolved in phosphate-buffered saline (PBS) to form a 1 mg/mL solution. PBS was slowly added to the measurement cell and the pressure cap was tightened prior to scanning. Once the baseline response value was low and repeatability was good, the sample measurement was performed. The solution was removed from the sample cell and the treated sample was slowly added to the cell. The pressure cap was then tightened and the scan proceeded. After the scan is complete, repeat the above procedure for the next sample. Set the temperature program to start at 15°C and increase at a rate of 90°C/h to 60°C.
Fourier transform infrared spectroscopy analysis
Fourier transform infrared (FTIR) spectra were obtained using an infrared spectrophotometer (Nicolet iS50, Thermo Fisher USA). The infrared spectra of rhCol XVII were measured over a wavenumber range of 4000–500 cm⁻¹, with data collected at a resolution of 2 cm⁻¹ per point. The spectra dates were analyzed using Origin.2025 software.
Circular dichroism analysis
The rhCol XVII was dissolved in a 10 mM phosphate buffer solution (pH7.4) at a concentration of 10 mg/mL, incubated at 4°C for 4 h and then diluted to 1 mg/mL prior to circular dichroism spectral analysis (Chirascan TMV 100). CD spectra were recorded with three accumulations in the wavelength range of 190–260 nm, with a path length of 0.1 cm and a scanning speed of 50 nm/min. The baseline correction was performed using a PBS buffer. The rhCol XVII was incubated at 4°C for 24 h and the change in ellipticity was monitored at 221 nm across a temperature range of 4°C to 24°C at a rate of 1°C/min.
Scanning electron microscope
Scanning electron microscopy (ProX, Feiner) was used to characterize the microscopic morphology of rhCol XVII. rhCol XVII lyophilized fibers were gently torn to expose the internal structure and placed on the carrier holder, which were then coated a thin layer of gold via sputter coating.
Transmission electron microscopy
A 1 mg/mL rhCol XVII solution was deposited onto a carbon-coated copper grid (200 mesh) for observation using a Tecnai G2 F20 S-TWIN (FEI, USA). After incubating for 3–5 min, excess liquid was aspirated, and the samples were stained with phosphotungstic acid. After 30s of staining, the excess staining solution was removed, and the samples were air-dried for 5 min. The observations were performed at an accelerating voltage of 5 kV.
In vitro assessment of rhCol XVII
Cell adhesion
Before the experiment, 1.00 mg/mL rhCol XVII and 1.00 mg/mL bCol I solutions and PBS solution (blank control group) were prepared. A 100 μL aliquot of the prepared sample solution was added to high-adhesion 96-well plate (CLS9018, Corning, USA) with five replicate wells per group and incubated overnight at 4°C. The wells were washed 2–3 times with PBS to remove any unbound proteins on the bottom of the plate. Next, 100 μL of heat-denatured 1% BSA-PBS solution was added to each well to cover the nonspecific binding sites, and the plate was incubated at 37°C in a 5% CO2 incubator for 1 h. After each well was washed 2–3 times with 200 μL PBS, and 100 μL of HSFs suspension (1 × 106 cells/mL) was added and incubated at 37°C for 1 h to allow cell adhesion. After incubation, the wells were washed 4 times with 200 μL PBS to remove unadhered cells. Then, 100 μL of a mixture (DMEM culture: CCK-8 = 9:1) was added to each well and incubated for 2 h at 37°C in a 5% CO2 incubator. The absorbance (OD value) of each well was measured at 450 nm using a microplate reader (Varioskan Flash, Thermo Flasher Scientific, USA).
F-actin fluorescence staining
HSFs were inoculated into 6-well plates with 1 × 106 cells/well and cultured overnight. Afterward, the wells were washed 2–3 times with PBS to remove any detached cells. In the blank group, 2 mL of serum-free basal medium was added, while in the experimental groups, serum-free basal medium containing 0.04, 0.40, and 4.00 mg/mL rhCol XVII was added. On days 1, 3, and 7, HSFs were labeled with F-actin fluorescent and its morphology and spreading were observed using laser scanning confocal microscopy (LSCM). Specifically, HSFs were first fixed in 4% (w/v) paraformaldehyde solution for 30 min at room temperature, followed by permeabilized with 0.5% (v/v) TritonX-100 solution for 5 min. After three washes with PBS, F-actin was stained with a diluted cyclic peptide solution (5 μg/mL) for 1 h at room temperature, and the samples were then stained with DAPI (10 μg/mL) for 5 min. Finally, the HSFs were washed three times with PBS and observed using a LSCM 880 (Zeiss, Germany).
Cell migration
The concentration of HSFs was adjusted to 1 × 105 cells/mL, and 50 μL of cell suspension was seeded into culture chambers of Culture-Insert inserts (Ibidi, Gräfelfing, Germany), with five replicate wells for both the blank and experimental groups. HSFs sufficiently adhered on the wells and reached 85% confluence after overnight incubation. Following the removal of the silicone barrier in the center of the Culture-Insert, the wells were washed 2–3 times with PBS to remove detached cells. In the blank group, 2 mL of serum-free basal medium was added, while in the experimental group, serum-free basal medium containing 0.40 and 4.00 mg/mL rhCol XVII was used. The scratch area was photographed at 0, 12, 24, 48, and 72 h using an inverted microscope (BX60, Olympus, Japan). The area was subsequently measured using ImageJ 8 software, and cell migration rate was calculated.
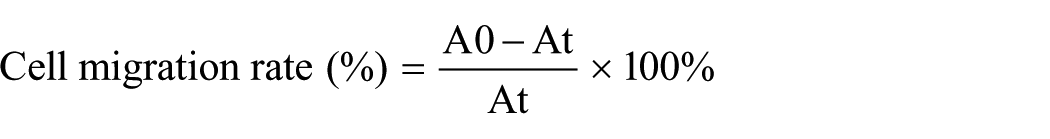
where A0 represented the initial scratch area, and At meant the remaining scratch area at given time.
Cell proliferation
HSFs were seeded in 96-well plates at a density of 5000 cells per well and cultured overnight in DMEM. Subsequently, the medium was replaced with a DMEM containing rhCol XVII at different concentrations (0.04, 0.40, and 4.00 mg/mL), and the medium was changed every other day. After 1, 3, and 7 days of culture, 100 μL of DMEM containing 10% CCK-8 was added to each well, and the cells were incubated in 37°C for 1 h. Then, the absorbance of the well plate was measured at a wavelength of 450 nm using an enzyme marker to evaluate the cell proliferation rate. At the same time, a parallel group without rhCol XVII was set as a blank control. Calculate the relative cell proliferation rate according to the following formula:

Determination of hyaluronic acid and Collagen type I collagen contents
HSFs were cultured in DMEM medium containing 15% fetal bovine serum (Hyclone, UT, USA), 1% penicillin-streptomycin (Hyclone, USA), and rhCol XVII at concentration at 0.04, 0.40, or 4.00 mg/mL at 37°C in a 5% CO2 atmosphere incubator for 3 days. The supernatant was collected, and ELISA (Zhuocai Biotechnology Ltd, Shanghai, China) was used to measure the expression levels of hyaluronic acid (HA) and collagen type I (Col I). Finally, the absorbance at 450 nm was measured using a microplate spectrophotometer.
Systemic acute toxicity and pyrogen test
Systemic acute toxicity
Twenty male BALB/c mouses (weighing approximately 20.00 g) were housed in a standardized animal facility (22°C–24°C, 50% humidity) with a 12 h light/dark cycle and allowed to acclimatize for 7 days before the experiment. On day 8, the mouses were randomly assigned into experimental group and blank group. The body weight of each animal in both groups was recorded. rhCol XVII was dissolved in saline to a concentration of 4.00 mg/mL and administered orally to the experimental group at a dose of 1 mL. Vital signs, feeding, and drinking behavior were monitored, and body weights were recorded at 24, 48, and 72 h. After 72 h, the mouses were euthanized, and their hearts, livers, spleens, lungs, and kidneys were collected, fixed in 4% paraformaldehyde (Biosharp, China), and stained with hematoxylin-eosin (H&E).
Pyrogen test
Three New Zealand Large White Rabbits (weighing about 2.00 kg) were housed in a standardized animal facility (22°C–24°C, 50% humidity) with a 12 h light/dark cycle and allowed to acclimatize for 7 days prior to the experiment. The rabbits were fed with the same diet, and their body temperatures were monitored during this period to ensure that there was no loss of body weight, and that their overall condition, including spirit, appetite, and excretion was normal. rhCol XVII was dissolved in saline at a concentration of 4.00 mg/mL and injected into the marginal ear vein of the rabbit. The body temperature was monitored every 30 min following the injection.
Statistical methods
Data were analyzed using Graph Pad Prism6 (Graph Pad Software, Inc, USA) and OriginPro 2021 (OriginLab Corporation, Northampton, MA, USA). All data are expressed as mean ± standard deviation (n ⩾ 3). Unless otherwise specified, statistical analyses were performed using one-way ANOVA. Significant differences between groups were defined as *p < 0.05, **p < 0.01, *** p < 0.001, and ****p < 0.0001.
Result
Characterization of rhCol XVII
The rhCol XVII employed in this study was produced using recombinant technology, from the Gly683-Ala745 segment of the native collagen XVII protein sequence (Figure 1). Following fermentation in Escherichia coli (E. coli), the recombinant collagen was purified through steps such as centrifugation, filtration, and chromatography. The complete amino acid sequence of rhCol XVII consists of three repeated units Gly683-Ala745, with each repetitive unit having the amino acid sequence: GLQGLRGEVGLPGVKGDKGPMGPPGPKGDQGEKGPRGLTGEPGMRGLPGAVGEPGAKGAMGPA.

The theoretical amino acid sequence of rhCol XVII.
The amino acid content analysis of rhCol XVII was presented in Table 2. Twelve distinct amino acid species were detected in rhCol XVII, with compositions closely matching theoretical amino acid content. Based on previous work 14 and accounting for the instrument’s inherent errors, 16 we maintained the deviation between experimental and theoretical values within 0.10%, which is considered reasonable. In addition, the arithmetic mean value for each amino acid content was calculated, and these values were less than 12%, which is complied with the accuracy guideline for amino acid analysis results specified in Chinese national standard GB5009.124-2016. Furthermore, histidine (His) was not detected in rhCol XVII, indicating that the protein tag had been completely removed. 17
Amino acid content of rhCol XVII.
The SDS-PAGE results (Figure 2(a)) showed a single target protein band, which indicated that rhCol XVII had relatively high purity. Drawing from the design of rhCol XVII amino acid sequence, the theoretical molecular weight of rhCol XVII calculated to be 17,789.37 Da. The molecular weight band of rhCol XVII appeared at approximately 18 kDa, which was consistent with the theoretical value. HPLC-MS (Figure 2(b)) presented a sharp peak with no impurity peaks, further confirming the high purity of rhCol XVII. The measured molecular weight of 17,788.8 Da matched the theoretical value, validating the alignment between the actual and theoretical molecular weights of rhCol XVII.
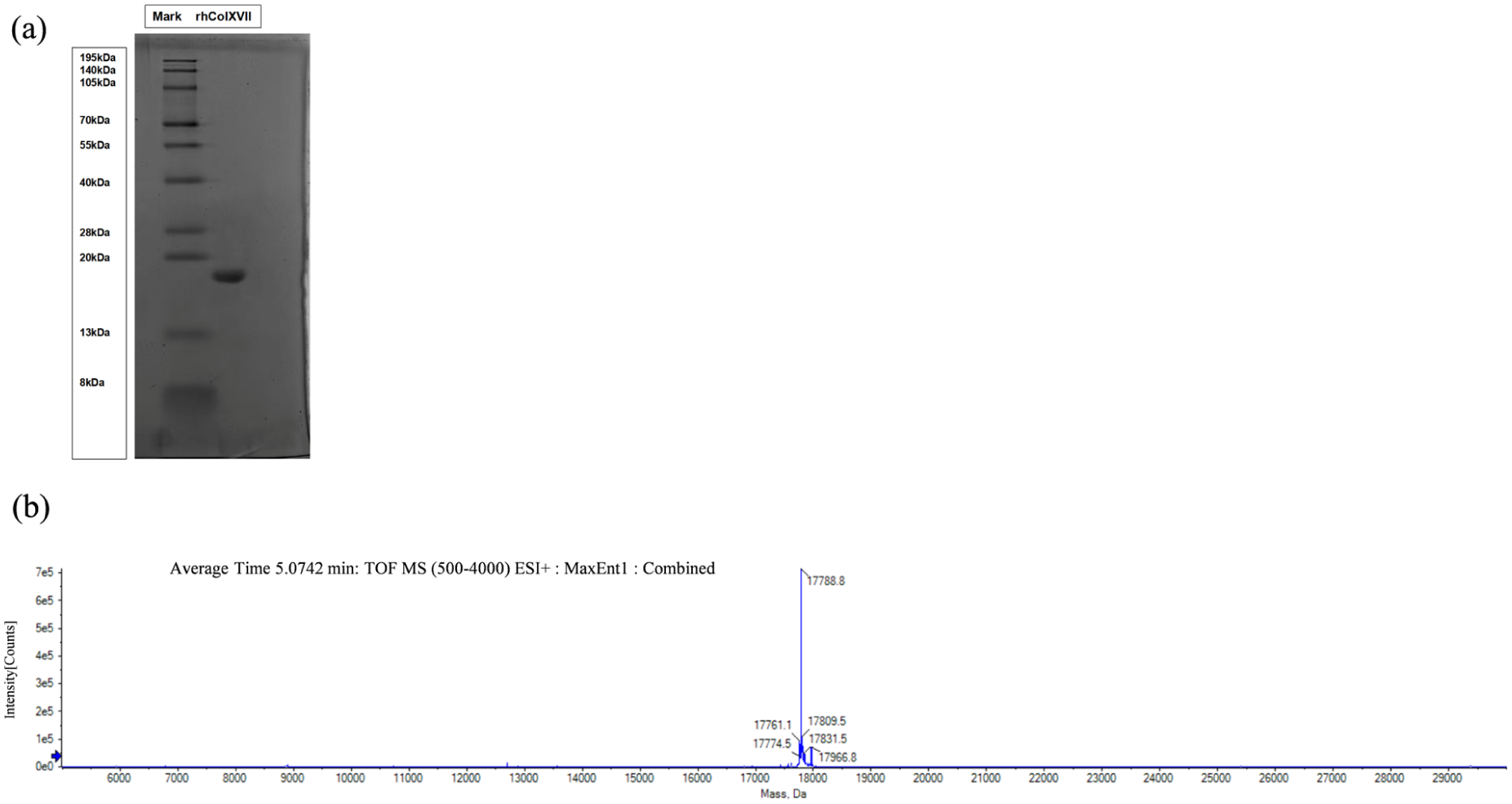
Molecular weight and purity analysis: (a) SDS-PAGE analysis of rhCol XVII and (b) mass spectrometry analysis of rhCol XVII.
The amino acid sequence of rhCol XVII was input into the peptide simulator (https://web.expasy.org/protparam/), yielding a theoretical pI of 9.69. As shown in Figure 3(a), the experimentally measured pI was approximately 9.00. rhCol XVII contained a higher proportion of basic amino acids (five lysines and three arginines) than acidic amino acids, increasing its propensity to bind protons (H+) and resulting in a higher, more basic pI. The pI deviation of 0.69 might be attributed to several factors. The proportions of charged residues in the α1 chain of rhCol XVII and natural type XVII collagen in the α1 chain were 22.22% and 18.30%, respectively. The higher proportion of charged amino acids in rhCol XVII might alter the surrounding hydration layer, hydrogen bonding, and other weak interactions, thereby slightly reducing its protonation state. More specifically, factors such as charge distribution, conformational changes, and measurement methodologies in the solution environment contribute to this discrepancy, which made it could be considered a reasonable error. 18

Characteristic of rhCol XVII: (a) isoelectric point in different pH, (b) particle size of rhCol XVII in different pH, (c) particle size of rhCol XVII, (d) DSC thermogram of rhCol XVII, (e) FTIR spectra of rhCol XVII, (f) circular dichroism spectra of rhCol XVII, and (g) Meadge of 221 nm on different temperature.
Figure 3(b) illustrated the changes in the size distribution of rhCol XVII particles at different pH. Near the pI, the particle size of rhCol XVII gradually increased, because its net charge approaches zero, reducing electrostatic repulsion and promoting molecular aggregation. 19 After dilution under neutral conditions, the particle size was remeasured. Figure 3(c) indicated that there was no significant difference in particle size was observed between 0.25 and 0.5 mg/mL, while concentrations of rhCol XVII below 0.25 mg/mL resulted in system errors that prevented accurate measurement. Therefore, 0.25 mg/mL may be the optimal concentration for particle size determination, while also potentially providing particle sizes that most accurately reflect the intrinsic behavior of rhCol XVII. Thus, the optimal collagen concentration is 0.25 mg/mL, yielding particle sizes that are 230–272 nm.
Figure 3(d) showed that rhCol XVII had exhibited two endothermic peaks. These peaks likely correspond to the thermal shrinkage temperature (Ts) of rhCol XVII at 18.43°C and the melting temperature (Tm) at 23.60°C, both of which were associated with collagen fibrillogenesis and the formation of intermolecular hydrogen bonds. 20 The stability of the triple-helical structure primarily relies on non-covalent interactions between polypeptide chains, such as electrostatic interactions and hydrogen bonds. 21 The results indicated that the thermal stability of rhCol XVII is lower than that of natural collagen. 14 Natural collagen possesses a highly ordered triple-helical structure and undergoes complex post-translational modifications, particularly the hydroxylation of proline and lysine residues. These modifications are crucial for the stability and thermal stability of collagen triple-helical structure. rhCol XVII was expressed in recombinant expression systems that often lack or only partially express the enzymes required for post-translational modifications, such as proline hydroxylation. These modifications are essential for collagen stability and biological functions, thus directly affecting its thermal stability. 22
Natural collagen forms a triple helix structure by twisting three α chains together, stabilized through inter- and intra-chain hydrogen bonds and inductive effects. 23 This structural organization results in distinct spectral characteristics in infrared spectroscopy, particularly in the amide A, amide I, and amide III regions. As shown in Figure 3(e), the infrared spectroscopic analysis of rhCol XVII had revealed significant features of amide A, B, and I, II, III bands, with characteristic peaks summarized in Table 3. The absorption peak of the amide A band in rhCol XVII was at 3307.31 cm−1, which correlates with the N–H stretching vibration of amino acids, indicating that there may be a considerable number of amino hydrogen bonds in rhCol XVII. The amide B band in rhCol XVII was observed at 2960.14 cm−1, which corresponds to the asymmetric stretching of C–H. 15 The absorption peak of the amide I band in rhCol XVII was at 1647.16 cm−1, arising from the stretching vibration of carbonyl groups on the peptide backbone and closely associated with the secondary structure of collagen, suggesting that rhCol XVII had stable C=O carbonyl bonds. The secondary structure of rhCol XVII is consistent with an α-helical conformation, as indicated by spectral analysis. The amide II and III bands were at 1535.28 cm−1 and 1234.18 cm−1, respectively. The amide II and III bands represented N–H bending vibrations and C–N stretching vibrations and C–H stretching vibrations, which were part of the peptide bonds in proteins, especially in recombinant collagen. 24
FTIR spectra characteristic peaks of rhCol XVII.

Circular dichroism (CD) spectroscopy was routinely used to evaluate the presence of triple-helical structures in collagen. We monitored the change in ellipticity at a wavelength of 221 nm across a temperature range of 4°C to 24°C. As shown in Figure 3(f), the CD spectrum of rhCol XVII exhibited a negative peak near 195 nm and a positive peak at 221–225 nm, confirming the formation of a collagen triple-helix structure in rhCol XVII (4°C–16°C). 25 The melting temperature of the triple helix measured by CD was approximately 20°C, lower than the Tm value (23.6°C) measured by DSC.
Figure 4(a) and (b) showed the freeze-dried fiber morphology of rhCol XVII observed at different magnifications using scanning electron microscopy (SEM). The SEM images revealed that rhCol XVII formed a loose fibrous network structure with interlaced fibers. 26 As shown in Figure 4(c), the average inter-fiber spacing was quantified at 9.32 ± 5.03 nm. During the production process of recombinant collagen, SEM can be utilized to evaluate whether the microstructure of the product meets the expected standards. This evaluation helps optimize preparation parameters, such as pH adjustment, crosslinking methods, and freeze-drying conditions, thereby improving the structural integrity and functionality of the recombinant collagen for biological applications. 27 Figure 4(c) and (d) show numerous interwoven fibrillar structures with fibrous morphology. These structures likely represent stacked or cross-linking of recombinant collagen fibers to form a network. However, it is also important to note that, unlike skin, tendons, and corneas—where Types I, III, and V collagens exhibit characteristic light–dark striations—rhCol XVII located at the epidermal–dermal junction do not display this periodic banding pattern. 7

Microscopic appearance of rhCol XVII: (a) SEM image at a scale plate of 500 μm, (b) SEM image at a scale plate of 40 μm, (c) histograms of diameter distribution measured from SEM images, (d) and (e) TEM image at a scale plate of 200 μm.
Evaluation of the biological effects of rhCol XVII on HSFs
As shown in Figure 5(a), both the rhCol XVII and bCol I group exhibited significantly higher cell adhesion efficiency compared with the PBS group (p < 0.0001). As an integral membrane protein, rhCol XVII likely promotes stable cell-matrix adhesion by mediating interactions with extracellular matrix (ECM) components, such as laminin and fibronectin, through its extracellular domain, thereby facilitating focal adhesion formation. Additionally, it may bind to integrins (e.g. α2β1 and α11β1) on the surface of HSFs, 28 further enhancing cell adhesion. 29 These interactions create an optimal extracellular environment that supports tissue regeneration and repair.

Evaluation of the biological effects of rhCol XVII on HSFs: (a) HSFs adhesion in different groups, (b) F-actin staining image of HSFs, (c) relative migration rate of HSFs, (d) migration images, and (e) relative proliferation rates of HSFs.
Figure 5(b) illustrated the morphological changes of HSFs cultured with different concentrations of rhCol XVII at 1, 3, and 7 days. On the first day, compared to the control group, cells treated with rhCol XVII exhibited intact spindle-shaped morphology, with easily identifiable and normally structured actin filaments. Notably, the high-concentration group significantly promoted cell proliferation, resulting in higher cell density. By days 3 and 7, the cells continued to grow and proliferate leading to cell overlapping. Under the influence of concentration effect, the cell spreading state of the high-concentration rhCol XVII group was arranged more tightly packed and orderly.
Figure 5(c) and (d) showed the relative migration rate and representative images of HSFs at different time points. The results indicated that rhCol XVII promote cell migration. During the first 24 h, there was no significant difference between the relative migration rate between the low-concentration and blank groups. However, with the prolongation of time, rhCol XVII continued to stimulation with HSFs, which significantly increased the cell relative migration rate at 24 and 48 h. The high concentration group exhibited a significantly relative migration rate than that of the blank and low concentration groups at all time points (p < 0.0001). These findings had suggested that rhColXVII significantly promoted HSFs migration, which contributes to wound healing and facilitates the regeneration of new tissue. 7
Figure 5(e) illustrated the proliferation rates of HSFs compared to the blank control at 1, 3, and 7 days. The results of CCK-8 assay revealed that the cell proliferation-promoting effect of rhCol XVII became more significant with increasing concentrations. The high concentration group effectively promoted cell proliferation at all time points, with a significantly higher proliferation rate than the other groups (p < 0.0001). At medium concentrations (0.40 mg/mL) of rhCol XVII, cell proliferation rates were no different from the control group on days 1. By day 3, although there was no significant difference between the medium and low concentration groups, the medium concentration group exhibit higher proliferation rates than the blank control (p < 0.01). On the seventh day, the medium (p < 0.0001) and low concentration (p < 0.001) groups both significantly higher than the blank group. Meanwhile, the medium concentration group showed a significantly better effect on promoting cell proliferation (p < 0.05) compared to the low concentration group.
The effect of rhCol XVII on ECM expression
The effect of rhCol XVII on the HA and Col I secretion in HSFs was assessed by ELISA, with results were shown in Figure 6(a) and (b). Compared with the blank group, rhCol XVII with different concentrations could significantly increase the expression of HA and Col I after 3 days of culture with a concentration-dependent manner. Among the tested groups, the high-concentration rhCol XVII group was the most effective in promoting the synthesis of HA and Col I. In previous experiments, HA has been proven to promote cell division in HSFs through regulating the cell cycle. 30 Col XVII is a key component of the basement membrane, providing an extracellular scaffold and regulating cell behavior. In HSFs, rhCol XVII might enhance cell adhesion and matrix interactions, increase cell survival rate and metabolic activity, thereby promoting the synthesis of HA and Col I. These findings confirmed that rhCol XVII promoted ECM secretion at the protein level.

Effect of rhCol XVII on ECM expression level: (a) the relative expression of HA and (b) the relative expression of Col I.
Systemic toxicity and pyrogen test
As depicted in Figure 7(a), BALB/c mouses displayed normal weight gain without any signs of oral mucosa irritation following gavage. No mortality or systemic toxicity was observed in any of the animals. Moreover, HE staining results (Figure 7(b)) revealed no significant pathological changes in key organs, including the heart, liver, spleen, lungs, and kidneys, after treatment with rhCol XVII, indicating the absence of irritant or adverse reactions, nor fever and inflammation. These findings collectively demonstrated that rhCol XVII was non-toxic and non-pyrogenic in vivo, supporting its safety profile in animal models. Additionally, the body temperature of experimental animals fluctuates remained within ±0.8, meeting the GB/T 16886.11-2021. (Figure 7(c)).

Systemic toxicity and pyrogen test: (a) changes in the body weight of experimental animals, (b) H&E staining of organs, and (c) changes in body temperature of experimental animals.
Discussion
In this study, the rhCol XVII was selected based on the NCBI reference sequence Q9UMD 9.3. The Col 17 A1 sequence of human collagen type XVII was screened and optimized as follows (Figure 8). Initially, the DNA fragment corresponding to the helical region of human collagen type XVII was codon-optimized and recombined using PCR to obtain the target gene fragment. This fragment was then inserted into the PET-32a expression vector to construct a recombinant expression plasmid, which was subsequently transferred into Escherichia coli BL21 (DE3) competent cells for transformation and selection of positive E. coli strains. 31 Subsequently, a single colony of the optimized E. coli strain was selected and cultured for protein expression. The bacteria were resuspended in phosphate-buffered solution (40 mM NaH2PO3, 500 mM NaCl, pH = 7.8), sonicated, and centrifuged to collect the supernatant. The recombinant humanized collagen was purified using an Ni-NTA affinity column, with impurities removed by washing with 10 mM imidazole. On-column digestion with Tobacco Etch Virus (TEV) protease was performed at 4°C for 16 h to cleave the target collagen. Finally, the rhCol XVII solution was lyophilized via freeze-drying to obtain fibrous recombinant collagen sponge.

Production of rhCol XVI by genetic engineering.
Amino acid analysis and HPLC-MS results confirmed the successful expression and purification of rhCol XVII. The triple-helical domain of natural collagen requires a repeating Gly-Xaa-Yaa sequence. 32 In rhCol XVII, glycine (Gly) constituted 33.33%, serving as a hallmark amino acid of collagen. Owing to its lack of a side chain, Gly minimizes steric hindrance, facilitating the formation of the triple-helical structure. 33 Additionally, proline (Pro) accounted for 15.87% of rhCol XVII. Pro is essential for the formation of the collagen triple helix and plays a crucial role in its structural integrity and thermal stability. 34
rhCol XVII was a basic protein containing a relatively high proportion of basic amino acids (Lys 7.94%, Arg 4.76%), while the proportion of acidic amino acids (Asp 3.17%, Glu 6.35%) is comparatively low. Therefore, the predominance of basic amino acids results in an increase in its pI, rendering the protein cationic under physiological conditions. 35 ECM is rich in proteoglycans and glycosaminoglycans, which contains a large number of negatively charged carboxyl groups and sulfonic acid groups. 36 The positive charge of basic proteins enhances its binding affinity to these components, thereby improving biocompatibility and stability. This property is advantageous for applications of rhCol XVII in wound healing and biomaterial integration. In a neutral environment (pH = 7.4), it carries a net positive charge and exhibits good water solubility.
The integrin binding sites of rhCol XVII include motifs such as GXXGEX (where X represents any amino acid), GEK, GPR, and GPP, which often exhibit excellent structural and functional properties and are more suitable for recombinant expression. 37 The GXXGEX motif serves as the minimal bioactive sequence for recognizing collagen-binding integrins α1β1, α2β1, α10β1, and α11β1. 38 This motif is implicated in collagen binding to cell surface receptors, particularly integrins. Integrins are transmembrane receptors that facilitate cell-collagen interactions within the ECM.14,39 Recombinant humanized type III collagen with a tailored amino acid sequence design derived from Gly 483–Pro 512 in human type III collagen, containing Gly-Glu-Arg (GER) and Gly-Glu-Lys (GEK) triplets, provides potent cell adhesion activity.14,40 Liu et al. 41 demonstrated that a collagen like peptide containing a GPR sequence can significantly promote the expression of the Co1A1, which plays a crucial role in the synthesis of ECM components. Additionally, the repetitive GPP and GPR unit provide a foundation for the triple helix of collagen, enabling the simulation of collagen biological activity by promoting cell adhesion, migration, and differentiation, thereby supporting tissue repair. 42 Collectively, through these repetitive motifs, rhCol XVII may form a triple helix similar to natural collagen, making it highly valuable in biomaterial design. However, rhCol XVII was expressed in Escherichia coli, which often lacks the necessary enzymes for post-translational modifications such as proline hydroxylation, which are crucial factor in collagen stability and biological function. 22 While the self-assembly conditions of rhCol XVII still need to be further explored, this lies outside the scope of our current investigation.
The above sequences endowed rhCol XVII with desirable biological properties, highlighting its potential as a promising biomaterial for modulating cell behavior. Previous studies have demonstrated that rhCol XVII can promote the proliferation and migration of human gingival fibroblasts (HGFs). 12 In this study, HSFs were chosen as the model cells due to their crucial role in connective tissue, where they actively contribute to extracellular matrix (ECM) synthesis, maintenance, and remodeling. 43 HSFs are also essential for tissue repair, wound healing, and overall organ function. 44 The rhCol XVII not only promote the proliferation, migration, and adhesion of HSFs but also could enhance the expression of hyaluronic acid and type I collagen. By interacting with integrin receptors on fibroblast surfaces, rhCol XVII increased ECM component synthesis, thereby improving ECM structure and function. Equally relevant is that rhCol XVII was fully humanized, non-toxic and non-immunogenic, as confirmed in animal experiments. 45
Recent research highlights the crucial role of Col XVII in maintaining skin barrier function, 1 regulating epidermal homeostasis, 3 and promoting skin regeneration. 46 The therapeutic potential of rhCol XVII in managing diverse cutaneous disorders, including hair follicle regeneration, 47 positions it as a promising candidate for dermatology and regenerative medicine research. Although we had characterized the structure of rhCol XVII and preliminarily verified its safety and efficacy, its specific application scenarios and mechanisms of action remain unclear. Future studies should be explored the molecular assembly mechanism of rhCol XVII, confirm the presence of its D-banding, and optimize its assembly efficiency and structural stability. By integrating materials science and biotechnological engineering, the physical and chemical properties of rhCol XVII can be further refined. Additionally, the efficacy and safety of rhCol XVII should be systematically evaluated in both in vitro and in vivo models for advancing its biomedical applications.
Conclusion
This study successfully expressed and purified rhCol XVII. rhCol XVII was enriched in basic amino acids, conferring a positive charge under neutral pH conditions. Meanwhile, the amino acid sequence of rhCol XVII conforms the Gly-Xaa-Yaa motif and contains a high proportion of proline, which supports the formation of correct collagen structure and the realization of the anchoring effect. Furthermore, its sequence contains a large number of cellular recognition sites, which is favorable for the execution of biological functions. Functional research demonstrated that rhCol XVII promoted the proliferation, migration, and adhesion of HSFs while enhanced their capacity to synthesize ECM components. rhCol XVII exhibited excellent biocompatibility, non-toxicity, and non-immunogenicity in animal models, reinforcing its potential as a promising biomaterial for tissue engineering and regenerative medicine.
Footnotes
Ethical considerations
The animals used in this experiment were approved by the Ethics Committee of Sichuan University (Ethics No.: WCHSIRB-D-2024-530), and meet ethical and moral requirements.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the National Key Research and Development Program of China (2023YFC2411205)
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
