Abstract
Atomization is a treatment method to make inhaled liquids into aerosols and transport them to target organs in the form of fog or smoke. It has the advantages of improving the bioavailability of drugs, being painless, and non-invasive, and is now widely used in the treatment of lung and oral lesions. Aerosol inhalation as the route of administration of therapeutic proteins holds significant promise due to its ability to achieve high bioavailability in non-invasive pathways. Currently, a great number of therapeutic proteins such as alpha-1 antitrypsin and Dornase alfa are effective. Recombinant humanized collagen type III (rhCol III) as a therapeutic protein is widely used in the biomedical field, but atomization is not a common route of administration for rhCol III, presenting great potential for development. However, the structural stability of recombinant humanized collagen after atomization needs further investigation. This study demonstrated that the rhCol III subjected to atomization through compressed air had retained its original molecular weights, triple helical structures, and the ability to promote cell adhesion. In other words, the rhCol III can maintain its stability after undergoing atomization. Although more research is required to determine the efficacy and safety of the rhCol III after atomization, this study can lay the groundwork for future research.
Graphical abstract
Introduction
Atomization is the process of converting the liquid into droplets or fine sprays under the influence of the tension on its surface and other internal or external factors. Specifically, the speed of the liquid reaches the desired level through the nozzle, causing the liquid to extend into jets or sheets. Under the effects of interior or exterior elements, the surface of the liquid ligament is broken by the surface tension into small droplets, which are decomposed into small and uniform droplets when they pass through the gas channel. 1 Atomization is now a common way of administering drugs to the upper and lower respiratory tracts in cases of pulmonary and nasal diseases. Compared with traditional administration methods, its theoretical advantages lie in being a non-invasive and painless treatment system. 2 Atomized inhalation of liquid preparations is a beneficial method for local or systemic drug administration, as it facilitates the dispersion of drugs in appropriate media and enables their delivery to target organs in the form of aerosols. It can avoid the first-pass effect of the liver by inhalation thus increasing the drug concentration and the duration in the target organ. Furthermore, inhalation drastically lowers the amount of drugs entering the blood circulation, which can mitigate the major side effects caused by intravenous injection and lower safety risks. 3
Inhalation of atomized proteins has great potential as a route of administration and its bioavailability was reported to be the highest in any non-invasive pathway. Currently, some proteins have been confirmed to remain stable during nebulization.4,5 For example, the evaluation of the recombinant human deoxyribonuclease I (rhDNase) after atomization in all four devices by Cipolla et al.’s team showed a slight loss in enzymatic activity and no rise in aggregation. Circular dichroism spectra showed that neither the secondary nor the tertiary structure of rhDNase had changed after atomization. 6 When utilizing an eFlow vibrating membrane nebulizer, the researchers discovered no discernible impact on the structure or function of the therapeutic protein. 7
Recombinant humanized collagen type III (rhCol III) was prepared by genetic engineering techniques to reverse transcribe specific gene fragments of human collagen into cDNA, then bound to the vector and transferred to yeast, bacteria, or animals for fermentation, isolation, and purification. 8 rhCol III plays a key role in cosmetics, promoting wound healing (especially in oral ulcer healing 9 ), and the human cardiovascular system.10,11 There are few studies on the restorative application of atomization rhCol III in the nasal cavity or oral mucosa, and therefore, this study aims to atomize rhCol III.
Nevertheless, it should be noted that not all proteins can retain their stability throughout the process of atomization. While decomposition into tiny inhalable droplets, liquids may experience prolonged exposure to gas-liquid interfaces, solvent evaporation, heating, or aerosol recycling and recovery, during which stress causes some proteins to degrade. 12 These instabilities depend mainly on the atomizer, drug (including concentration, viscosity, density, etc.) and temperature. 13 Therefore, a compressed air atomizer will be used to atomize the rhCol III and evaluate its structure to investigate whether the rhCol III structure may stay stable after atomization and to give a theoretical framework for further investigations.
Materials
The lyophilized recombinant humanized collagen type III (rhCol III) was provided by Shanxi Jinbo Bio-Pharmaceutical Co., Ltd; Bovine serum albumin standard (BSA) was purchased from Solarbio Life Sciences; Bovine collagen type I reference material (bCol I)was purchased from National Institutes for Food and Drug Control; Sodium chloride, Disodium Hydrogen Phosphate and Sodium Dihydrogen Phosphate were purchased from Sinopharm Chemical Reagent Co., Ltd.; Acetonitrile was acquired from Sigma Aldrich; Formic acid was purchased from Tokyo Chemical Industry Co.; Ltd. Pure water was all derived from the Millipore.
Methods
Atomization and sample collection
The rhCol III was used as a sample, while BSA and bCol I were designated as control groups. These were all subjected to atomization using a 403E compressed air atomizer under varied parameters, encompassing different durations of atomization and sample concentrations. Briefly, the samples were injected into the atomizing cup to start atomizing after rhCol III and BSA were diluted in pure water to 0.5, 0.75, and 1 mg/mL. When it reached a certain atomization time (as shown in Table 1), the atomization was stopped for collection and then the collected samples (The diameter of the liquid below five microns accounts for more than 60%) were stored in a 4°C refrigerator.
Parameters of atomization.
The bCol I which was diluted in acetic acid (0.01 M) to 0.5, 0.75, and 1 mg/mL (The concentration gradient was eventually selected for atomization for the reason that the solution was too viscous at concentrations higher than 1 mg/mL, which led to a very low flow rate of atomized gas), then atomized as above and subsequently stored in a −20°C refrigerator for storage.
Molecular weight detection
The rhCol Ⅲ and the control groups were diluted triple with water, mixed with the 5X sample buffer at a ratio of 4:1 respectively, centrifuged, and then loaded onto the SDS-PAGE gel (gel percentage 20%). A total of 5 µL treated samples were loaded per well. After running for 60 min at 120 V, the gels were removed and stained with an eStain™ LG protein chromatography, stained for 10 min, after which the gels were photographed and analyzed.
Protein content determination
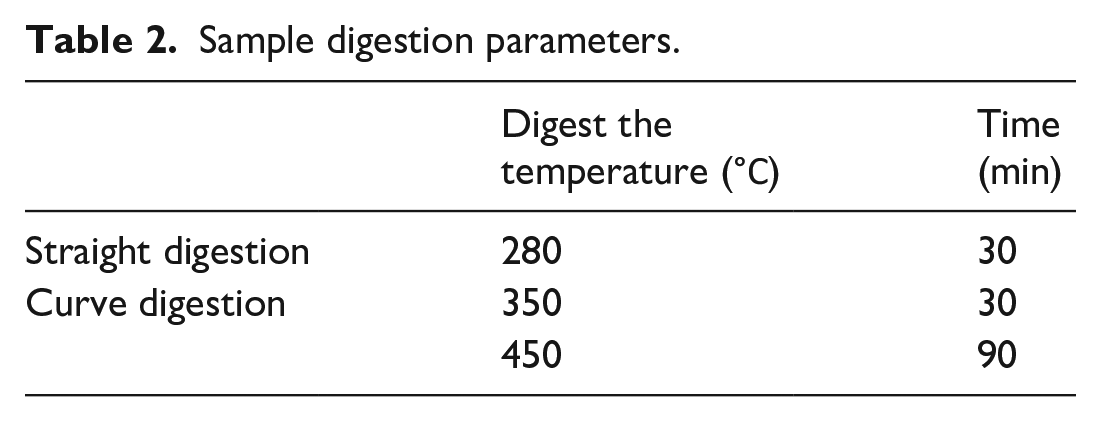
The content of protein was determined by the Kjeldahl method in China Pharmacopeia (2020 edition) with a slight adjustment. The rhCol Ⅲ and the control groups were weighed respectively and placed in dry digestive tubes with 0.2 g copper sulfate, 3 g potassium sulfate, and 10 mL sulfuric acid. According to the different samples, different temperature lines and curves were set for digestion (details present in Table 2). Cooling was performed after the sample digestion, and the protein content of both groups was determined using an Automatic Kjeldahl Analyzer (K9860, Hanon).
Sample digestion parameters.
Purity analysis
The rhCol Ⅲ and the control groups were centrifuged for several minutes, and which supernatant was separated on an ACQUITY UPLC Protein BEH C4 Column (300 A, 1.7 µm, 2.1 mm × 150 mm). The mobile phase A was composed of 0.1% trifluoroacetic acid in water, and mobile phase B was 0.1% trifluoroacetic acid in acetonitrile. The column was used to separate the sample at a flow rate of 0.3 mL/min and the column temperature was 55°C after the autosampler had loaded it with 5 μL, and the detection wavelength was 214 nm.
Structure identification
The rhCol Ⅲ and the control groups were processed overnight at −20°C and melted prior to circular dichroism analysis. The scanning wavelength of 180–260 nm was set for blank buffer baseline testing, and then the circular dichroism absorption of the sample solution in the range of 180–260 nm was collected. The circular dichroic meter parameters were set to the bandwidth of 1.0 nm, steps of 1.0 nm, scanning speed of 50 nm/min, three repetitions and measurement temperature of 4°C.
Cell adhesion
HFB was employed in the cell adhesion assay, with bovine type I collagen diluted to 1 mg/mL in D-PBS serving as the positive control, labeled as the PC group. The NC group represented the negative control, consisting of the D-PBS buffer solution alone.
The rhCol Ⅲ group, the control group, the PC group and the NC group were added to an enzyme labeling plate at 100 μL per well, and the supernatant was discarded after 4°C incubation overnight. Next, 100 μL 1%BSA (inactivated at 56°C for 30 min) was added to the enzyme labeling plate, and incubated at 37°C for 60 min. The supernatant was discarded, and all groups were cleaned third with D-PBS. A total of 100,000 HFB cells, previously cultured, were seeded into each well and further incubated at 37°C for 120 min. After washing the wells with D- PBS, 100 μL of CCK-8 solution, prepared by mixing with DMEM medium containing 5% serum-free medium, was added to each well and left to incubate for 2 h at 37°C. The absorbance at 450 nm was detected and recorded using a multifunctional enzyme marker (H1M, BioTek).
Statistical analysis
All data are presented as mean ± standard deviation (n ⩾ 3). Unless otherwise specified, one-way ANOVA was used for statistical analysis. The following differences were deemed significant: *p < 0.05, **p < 0.01, ***p < 0.001, and ****p < 0.0001.
Results
Effect of atomization on the molecular weight
The results of the SDS-PAGE analysis of rhCol III and the control groups, collected under various atomization conditions, were illustrated in Figure 1. In the BSA group, distinct protein bands ranging from 60 to 75 kDa were observed, which broadened progressively as the BSA concentration increased, consistent with the anticipated molecular weight range for BSA. 14 In the bCol group, distinct protein bands appeared within the 100–140 kDa range, representing the α1 and α2 chains of type I collagen individually; these bands widened over time with prolonged atomization. For the rhCol III group, clear protein bands were observed at approximately 45 kDa, which broadened as the collagen concentration increased, indicating that the atomization process did not affect the molecular weight of rhCol III.

The SDS-PAGE profiles of the rhCol III and the control groups (M is marker; a is 0 min; b is 10 min; c is 20 min; d is 40 min).
Effect of atomization on protein purity
Protein breakdown during the atomization process could interfere with the protein’s purity. First, the content of proteins was analyzed as shown in Figure 2, in the BSA group, the protein content decreased during the atomization in the low concentration group (0.5 mg/mL), while the maximum protein content was reached at 20 min of atomization time in the medium concentration group (0.75 mg/mL) and high concentration group (1 mg/mL); In the bCol group, the maximum protein content was achieved at 40 min in the low concentration group (0.5 mg/mL), while the maximum protein content was reached at 10 min of atomization time in both the medium concentration group (0.75 mg/mL) and the high concentration group (1 mg/mL). Therefore, it can be concluded that both the level of concentration and the length of atomization time altered the protein content of the control group, resulting in drug instability. In contrast, in the rhCol III group, most of the atomization process led to a decrease in the protein content of the samples regardless of the concentration level, while the length of the atomization time did not have much effect on the protein content of the samples. Therefore, it can be assumed that the atomization process causes the loss of a part of the sample protein. This situation may originate from the following reasons: due to the long duration of the atomization process, the temperature inside the atomization device increases, and the increased temperature may lead to the degradation of protein components; studies have shown that freezing may also lead to the degradation of proteins 4 ; in addition, the selection of the atomization parameters may lead to the instability of the proteins, which may result in chemical or physical degradation.
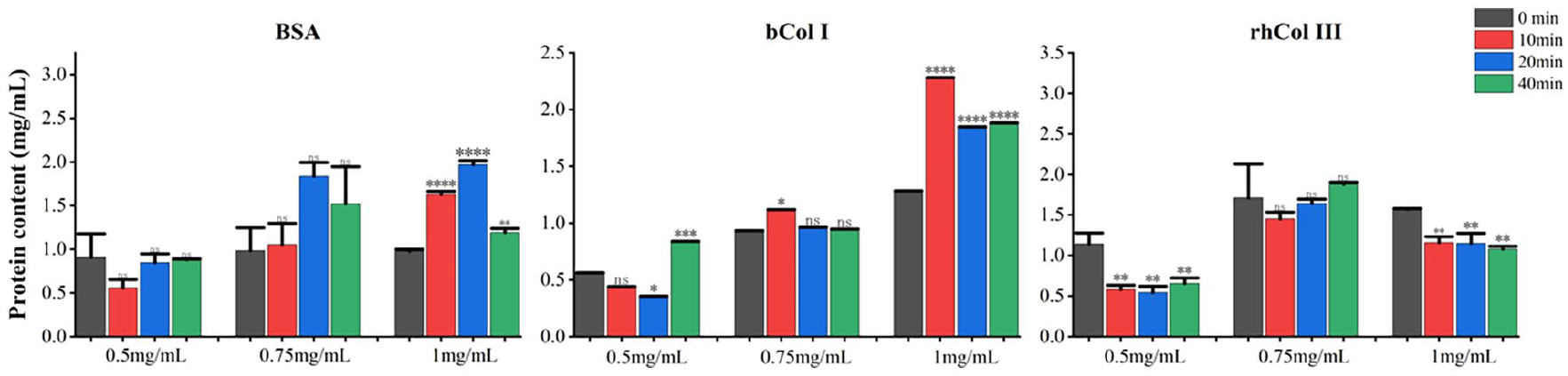
Protein content of the rhCol III and the control groups.
Next, the rhCol Ⅲ and the control groups of various atomization conditions were examined by UPLC to determine whether there were extra impurities of the products in the atomization process. Figure 3 illustrates that the retention time of rhCol Ⅲ and the control groups were consistent respectively and that the chromatographic peaks were completely separated and no other impurity peaks were present. Through the processing and integration of these chromatographic peaks using Empower3 software, it was revealed that the atomization process led to a reduction in the proportion of the main peak area of BSA at low concentrations and an increase at medium and high concentration groups, with the opposite of bCol I. In the rhCol III group, the proportion of the main peak area increased in the atomization process. The results showed that the purity of rhCol III component increased during the experimental concentration, with the percentage of the main peak area above 90% in all groups.
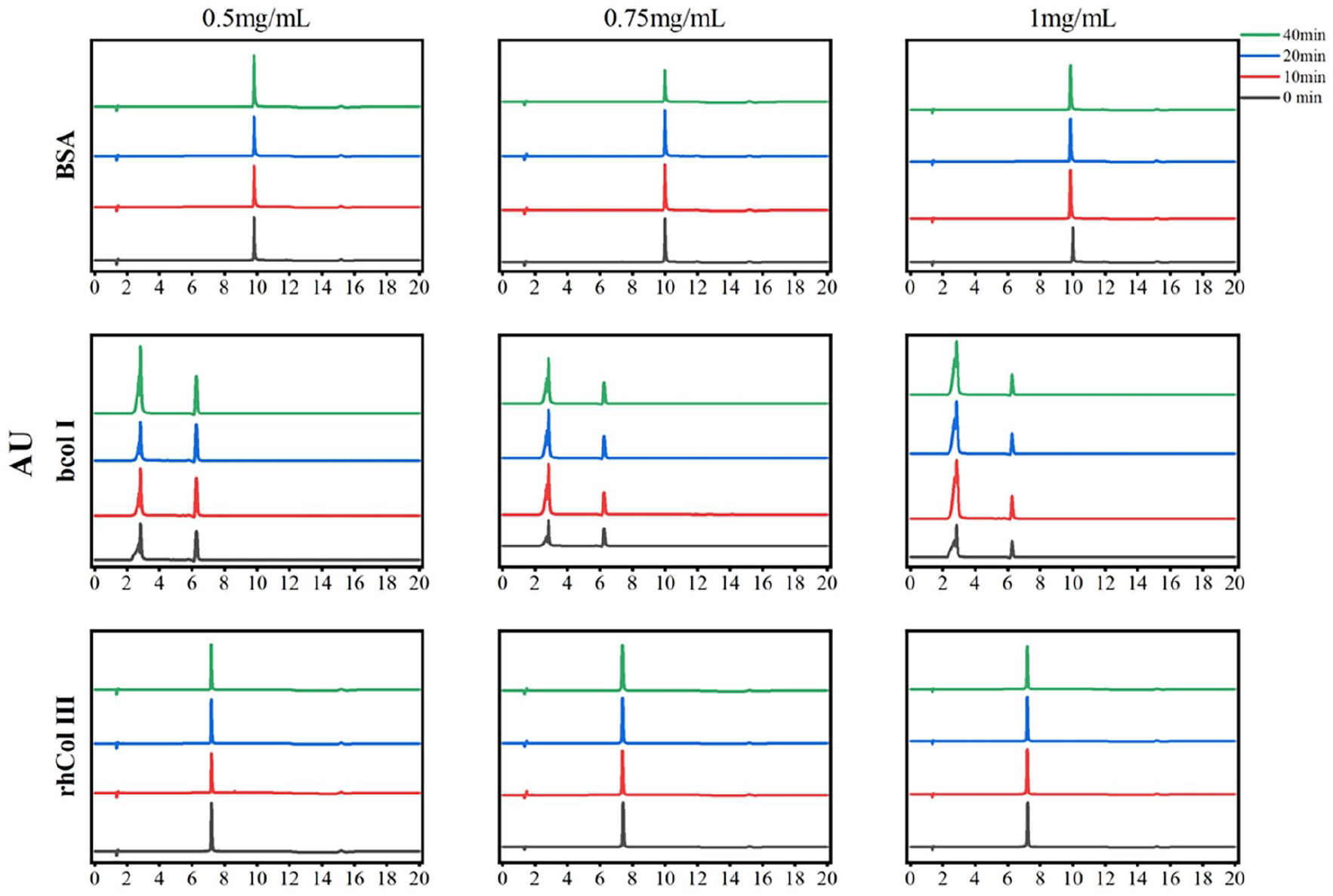
Liquid chromatography analysis of the rhCol III and the control groups.
Effect of atomization on the structure
The distinctive effects of collagen were also due to its triple-helical shape, which made it fundamentally different from other proteins. The triple helix structure of the rhCol III was susceptible to deterioration because of environmental factors. 15 Furthermore, the position of the absorption peak will alter with changes in the amino acid sequence and length. 16 The triple helical structures of the rhCol III, BSA, and bCol I groups were examined using a circular dichroism spectrometer to assess their structural stability under various atomization conditions. As shown in Figure 4, the BSA had two negative peaks at 209 and 220 nm, which were the main feature of the α helical structure. At the same time, regardless of the concentration, the intensity of the two negative peaks reached the maximum at 20 min, which indicated that the atomization did not affect the BSA structure. Both the rhCol III and bCol I groups had the triple helical structure represented by the positive absorption peak typically around 221 nm and the negative absorption peak around 197 nm. In the bCol I group, the characteristic peak intensity of the triple helix in the low concentration group gradually increased with the atomization time. In the medium and high concentration groups, the characteristic peak intensity of the triple helix increased throughout the whole atomization process, but the characteristic peak intensity reached the maximum at 10 min, and then gradually decreased. In the rhCol III group, regardless of the concentration, the characteristic peak intensity decreased. This decrease may result from the increased temperature of the instrument during the atomization process, which destroyed the hydrogen bonds in the triple helix of part of the collagen and caused structural modifications. At the same time, in the low and medium concentration group, compared with the untreated sample, the peak value of the atomized sample decreased obviously. In the high concentration group, the peak value will decrease obviously only when the atomization time is 40 min. It can be inferred that minimal structural damage occurs when the atomization concentration is set at 1 mg/mL and the atomization process lasts for 10–20 min. In conclusion, the emergence of the positive peak near 220 nm indicated that the collagen still maintained the triple helical structure, which meant that the atomization under different conditions of rhCol III still conserved its inherent triple helical.
Circular chromatogram of the rhCol Ⅲ and the control groups.
Effect of atomization on the cell activity
Cell adhesion is one of the fundamental phenomena taking place in living organisms and influences many other procedures like proliferation, differentiation, migration, or cell viability. 17 At the same time, cell adhesion, the initial stage of cell-material contact, is the prerequisite for cell anchoring, diffusion, and multiplication on the surface of materials. 18 As protein adhesion increases, it can facilitate greater cell attachment to the culture plate. CCK-8 can be oxidized and reduced to water-soluble yellow products by adherent cells, and the quantity of yellow products is proportional to the degree of color intensity. The absorbance of collagen-coated cells on a cell culture plate was detected by an enzyme-labeled instrument, and the relative cell adhesion of collagen samples was evaluated. To determine the cell adhesion of the rhCol III, BSA, and bCol I groups, the absorbance at 450 nm was measured utilizing a microplate reader. As shown in Figure 5, compared with the NC group, the PC group had a significantly enhanced cell adhesion effect, with a statistically significant gap. From the bCol I group, it can be seen that the cell adhesion capacity of all three concentration groups reached the maximum at 0 min, indicating that excessively high or low concentrations would lead to the weakening or even disappearance of the cell adhesion capacity during the atomization process; in the BSA and rhCol III groups, the samples possessed significant cell adhesion capacity before and after atomization. Specifically, in the rhCol III group, the maximum cell adhesion capacity was reached at 10 min in the low concentration group, whereas the medium and high concentration groups reached maximum adhesion immediately at 0 min. However as the atomization time increased, the cell adhesion decreased slightly in the medium and high concentration groups. Consequently, although rhCol III maintained the cell adhesive capability within the given atomization conditions, the process was found to modify its specific affinity to cells when concentrations reached excessively high levels.
Cell adhesion of the rhCol III and the control groups in different atomization parameters.
Discussion
Pulmonary administration can be used as an alternative therapy to parenteral administration for the treatment of respiratory and systemic diseases and has great advantages such as large surface area of the lungs and rapid absorption; high volume of blood passing through the pulmonary circulation; avoidance of hepatic first-pass effect, low enzyme activity, thin epithelial barrier and high membrane permeability. Atomization can convert liquid preparations into tiny droplets that are delivered to the respiratory tract for the treatment of various diseases. 19 However, delivering proteins to the designated site is a great challenge to play its role, such as the fact that proteins may be subjected to various stresses during the atomization process, resulting in disruption of the structural integrity of the proteins and rendering them unable to function. In this study, the rhCol III after atomization was examined by SDS-PAGE, UPLC and circular dichroism, and the results showed that under the present conditions, the molecules of rhCol III after atomization were consistent with the theoretical molecular weights, which were all at 45 kDa, and the protein purity was basically unchanged. Meanwhile, during the CD structure analysis, the atomized rhCol III had a positive peak at 220 nm and a negative peak at 197 nm, still maintaining the triple helical structure; Moreover, cell adhesion tests indicated that rhCol III still could promote cell adhesion. Together, these results demonstrated that atomization did not disrupt the structural integrity of rhCol III.
However, even though the atomization process does not disrupt the structure of the proteins, delivering proteins to the site of action by means of atomized inhalation poses additional challenges. The effectiveness of atomization depends on the location of droplet deposition; the size, density, and shape of the released droplets affect the site of protein delivery, and only when droplet diameters are in the range of 1–5 μm can proteins be transported to the lower respiratory tract and alveoli for deposition and efficacy, thus avoiding upper respiratory tract obstruction. Droplets with diameters too small (<0.5 μm) result in approximately 80% of protein particles being expelled via exhalation due to Brownian motion, thereby hindering the proteins from exerting their therapeutic impact. Concurrently, these minute droplets, experiencing lesser influence from inertia and sedimentation, evade collection, potentially enhancing protein degradation rates. Conversely, droplets exceeding 5 μm in diameter tend to accumulate in the throat and upper respiratory tract, where they can be ingested, subsequently undergoing metabolism or elimination via the digestive and hepatic pathways. This detour significantly diminishes the proportion of the drug reaching systemic circulation.4,12,19 Furthermore, droplet particles may undergo degradation and pulmonary clearance mechanisms during transportation, limiting the availability and efficacy of the drug in the lungs. This leads to a rapid decline in the local drug concentration, thereby hampering the realization of optimal therapeutic benefits. Specifically, inhaled proteins are incessantly confronted with a myriad of metabolic enzymes and the ceaseless mucociliary clearance processes. Moreover, a large amount of pulmonary surfactant exists on the surface of the respiratory zone, which serves as a barrier to contact with the outside world, and passing proteins are adsorbed to the surface, resulting in reduced protein uptake.20–22
Therefore, it is also necessary to implement various strategies to increase protein uptake in the lungs, such as nanocarriers, which play a key role in drug delivery, can be modified by modifying the surface attachment of the nanocarriers to specify the delivery site of the drug and increase the concentration of the protein at a specific site. 23 The presence of protease inhibitors can attenuate protein degradation during delivery. 19 However, it is worth noting that nanocarriers may pose pulmonary toxicity and trigger lung damage 24 , the addition of protease inhibitors may induce metabolic disorders, the occurrence of various diseases, and other toxic side effects.25,26 Therefore, many challenges need to be gradually overcome in subsequent studies to efficiently deliver proteins to effective sites for action.
Conclusion
Therapeutic proteins have high bioavailability by non-invasive routes and hold great promise by atomized inhalation. However the structural stability of drugs after atomization needs to be further explored. This study showed that recombinant humanized collagen remained stable without disrupting its triple-helical structure in the presence of a compressed air atomizer.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
