Abstract
Background
The aim of this study was to develop a novel thermoresponsive material suited for tissue engineering and investigate the growth and harmless detachment of cells cultured on the surface of thermoresponsive tissue culture polystyrene (TCPS).
Methods
Thermoresponsive N-isopropylacrylamide (NIPAAm) and biocompatible chitosan (CS) were grafted onto the surface of TCPS by ultraviolet (UV)–induced graft polymerization. The chemical composition, surface morphology and thermoresponsiveness of the modified TCPS were investigated by X-ray photoelectron spectroscopy (XPS), atom force microscopy (AFM) and contact angle (CA), respectively. Furthermore, the growth and detachment behaviors of mouse fibroblast cells (L929) on the surface of the modified TCPS were studied by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay.
Results
The modified TCPS exhibited good hydrophobic/hydrophilic property alterations in response to temperature. The cytocompatibility of the materials was improved due to the introduction of CS. Cells could be spontaneously detached from the surface without any damage, by controlling environmental temperature. The viability of cells obtained by temperature induction was higher than that obtained by enzymatic digestion.
Conclusions
This study developed a simple and economical method to fabricate thermoresponsive cell culture dishes and provided new thoughts and experimental bases for exploring novel material applied in tissue engineering.
Introduction
Due to the direct contact with human tissue, the safety requirements of biomaterials are higher than for other materials. Although there are several biomaterials with enough mechanical strength and suitable degradation rates, some of them have poor biocompatibility and lower anticoagulant ability. Surface modification of these materials can endow them with better compatibility, anticoagulant properties and responsiveness between materials and cells. Therefore, surface modification of materials is the preferred method to develop new clinical materials (1, 2). The purpose of surface modification is to create a good artificial extracellular matrix environment to meet the requirements of cell growth and to ensure the normal function of cells. Hatakeyama et al and Nishi et al (3, 4) immobilized bioactive materials such as protein and polypeptide on the surface of materials to function as ligands or receptors in order to strengthen the interaction between cell and materials. Yamaguchi et al (5) treated highly porous poly(L-lactic acid) (PLLA) membranes with a low-temperature oxygen plasma to improve the cell adhesion and proliferation. The results indicated that the plasma treatment not only accelerated cell adhesion but also enabled cells to proliferate in the form of sheets resembling biological tissue. However, the methods used were not satisfactory, due to the harsh and expensive experimental conditions. It is necessary to develop an economical and effective method to modify and functionalize inert surfaces.
Chitosan (CS) is the deacetylation product of chitin in which the N-acetyl pyran structure is similar to the glycosaminoglycan of extracellular matrix. Owing to its good biodegradability, biocompatibility and the presence of available active groups, CS has been widely used in tissue engineering (6–7–8). Gümüşderelioğlu et al (9) prepared a CS-based biomaterial by the freeze-drying method and then linked heparin onto it to investigate the morphology, proliferation and differentiation of the preosteoblast cell line MC3T3-E1 on the material. The result showed that the CS-based biomaterial could promote the proliferation and differentiation of preosteoblasts. Huang et al (10) mixed collagen, CS and thermoplastic polyurethane to prepare uniform nanofiber by electrostatic spinning. The survivability of endothelial tissue cells and Schwann cells on the nanofiber proved that the nanofiber had good biocompatibility and that well-balanced fiber can regulate the morphology of cells by inducing the orientation of cells. Therefore, these inert surfaces modified with CS are expected to enhance the adhesion and proliferation of cells on the materials.
In addition to the biosafety and biocompatibility, promising biomaterials are also expected not to induce immune rejection when they are implanted into the body. To this end, Okano proposed a cell sheet method to repair tissue and regenerate organs (11). The cell sheet method is not based on a single cell but on a cell sheet. Cells are incubated on the surface of thermoresponsive materials to form cell sheets. After that, the intact cell sheet can be detached from the surface by temperature induction. Cell sheets have been overlaid to form 3-dimensional structures for tissue repairing and organ regeneration (11, 12). For example, using the cell sheet method, cornea (13), myocardial tubes (14), trachea (15) and esophagus (16) were reconstructed or repaired by Okano's group. By this method, only cell sheets were implanted onto the impaired site, to avoid any immune rejection or tissue fibrosis caused by the materials.
The most promising thermoresponsive material for the cell sheet method is poly(N-isopropylacrylamide) (PNIPAAm), which has a hydrophilic acylamino group and hydrophobic isopropyl group. The lower critical solution temperature (LCST) of a PNIPAAm aqueous solution is 32°C. Below that temperature, the molecular chain of PNIPAAm presents a stretch state, and the hydrophilic group is exposed to the water, which results in hydrophilicity. When the temperature is above 32°C, the PNIPAAm chain will shrink, and exposure of the hydrophobic group will lead to hydrophobicity of the molecular chains (17). Thermoresponsive tissue culture polystyrene (TCPS) is prepared by grafting PNIPAAm onto the surface of TCPS. Cells proliferate and spread on the thermoresponsive TCPS at 37°C. After the formation of the cell sheet, the thermoresponsive TCPS is transferred to a low-temperature environment (lower than LCST) for intact cell sheet detachment (18). Varghese et al prepared thermoresponsive surfaces by coating TCPS with the copolymer of N-isopropylacrylamide and methyl methacrylate solution in isopropanol. L929 cells cultured on the thermoresponsive copolymer coated–surface could be transferred to polyvinylidene difluoride (PVDF) membrane by incubating below LCST (19).
Further, Okano's group has developed various technologies to promote the applications of thermoresponsive polymer-grafted surfaces in cell sheet tissue engineering, such as enabling the coculture of different species of cells with a patterned thermoresponsive surface, immobilizing bioactive molecules on a thermoresponsive surface for serum-free culture and fabricating a high-orientational cell sheet on a micropatterned thermoresponsive polymer brush surface (20).
In this study, PNIPAAm was grafted onto the surface of TCPS by ultraviolet (UV) irradiation to endow the surface with thermoresponsiveness. Furthermore, CS was covalently linked onto the surface to reinforce the bioactivity of the material. The growth and detachment behaviors of mouse fibroblast (L929) on the thermoresponsive TCPS were also investigated.
Materials and Methods
Modification of TCPS with CS and PNIPAAm
N-Isopropylacrylamide (NIPAAm) 1.14 g (Tokyo Chemical Industry Co., Japan) and 0/1 mL acrylic acid (AAc) were dissolved in dimethyl sulfoxide (DMSO) with 0.32 g anthraquinone-2-sulfonic acid sodium salt monohydrate (AQS; Sinopharm Chemical Reagent Co., China) as photosensitizer. TCPS pieces were put into the above solution and exposed to a UV lamp (360 nm, 1,000 W) for 0.5, 1.5 and 3.0 hours under N2 atmosphere. After UV-induced graft polymerization, the samples were dipped into absolute ethanol and distilled water in turn to remove unreacted monomers. Subsequently, carboxyl-functionalized thermoresponsive TCPS was immersed in 3 mL 1% HCl solution with 30 mg CS (Mr = 50,000, 85% degree of deacetylation; Qingdao Medical Corp., China) and 3 mg N-hydroxysulfosuccinimide sodium salt (sulfo-NHS; Shanghai Medpep Co., China) to react for 3 days at 25°C. N-(3-Dimethylaminopropyl)-N-ethylcarbodiimide hydrochloride (EDC·HCl; 30 mg) was added into the reaction system every day. The thermoresponsive TCPS, carboxyl-functionalized thermoresponsive TCPS and CS-based thermoresponsive TCPS were abbreviated as XSN, XSNA and XSNAC, respectively, where X represents irradiation time. The chemical structures of CS and PNIPAAm are shown in Figure 1.
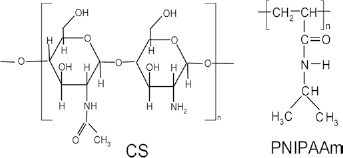
The chemical structures of chitosan (CS) and poly(N-isopropylacrylamide) (PNIPAAm).
Characterization of the modified TCPS
The chemical composition of the modified TCPS was analyzed by X-ray photoelectron spectroscopy (XPS; Perkin-Elmer 5300; Perkin-Elmer, USA). The content of carboxyl and CS on the modified TCPS was determined by toluidine blue O (TBO) staining method. The surface topology of the modified TCPS was observed by atom force microscopy (AFM; AFM/STM 550; Agilent Co., USA). The thermoresponsiveness of the modified TCPS was analyzed using the sessile drop method with a contact angle meter (OCA20; Dataphysics Instrument GmbH, Germany) at 20°C and 37°C. Five different positions on each sample were selected, and a mean average value was obtained.
Cell experiments
Samples were dipped in 75% ethanol for 10 minutes and then rinsed with sterile phosphate-buffered saline (PBS) buffer and Dulbecco's modified Eagle's medium and Ham's F12 (DMEM/F12; Shanghai Sangon Biological Engineering Technology & Services Co., China) for 3 times each. After that, they were sterilized by UV irradiation (15 W) for 20 minutes and then preincubated in medium with 10% fetal bovine serum (FBS) and double antibody at 4°C for 1 day. L929 cells were seeded onto the surfaces of the modified TCPS at 3 × 104cells/mL and incubated at 37°C under a humidified atmosphere of 5% CO2.The activity of the cells was monitored with the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) method every 2 days to investigate the growth of cells. The morphology of cells cultured on the modified surfaces was observed by phase contrast microscope. After incubation for 4 days, all samples were transferred into a incubator to induce cell detachment at 20°C. Detached cells from each surface were collected and counted with a blood counting chamber. Cell detachment rate was calculated from the ratio of the number of detached cells and the total number of seeded cells. Cells obtained by temperature induction and trypsinization from the same modified surface were seeded respectively onto tissue culture plate to investigate the reproduction of cells by MTT assay.
Statistic analysis
SPSS version 13.0 software was used for the analysis of variance, and statistical significance was determined, and set at a p value <0.05.
Results
Characterization of the chemical composition of the modified surface
The chemical composition of modified surfaces was analyzed with XPS, and the results are shown in Table I and Figure 2. It can be seen from the data in Table I that the amount of N on all of the modified surfaces was increased significantly compared with that on the unmodified TCPS. The amount of N on the surfaces of the original TCPS and the modified TCPS increased in the order of TCPS (0.7%) <0.5SN (2.1%) <0.5SNA (2.5%) <0.5SNAC (4.3%). As shown in Figure 2, a new peak appeared at the bonding energy of 288.5 eV from N—C = O on the modified surfaces compared with the unmodified TCPS surface. The peak intensity increased in the order of TCPS (0%) <0.5SN (1.35%) <0.5 SNA (2.04%) <0.5SNAC (4.32%). However, the intensity of π* from the base polystyrene substrate on modified surfaces was lower than that on the original TCPS surface.
Atomic concentration of carbon, oxygen and nitrogen on the surface of TCPS, 0.5SN, 0.5SNA and 0.5SNAC
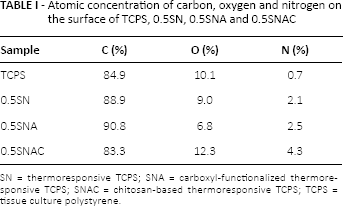
SN = thermoresponsive TCPS; SNA = carboxyl-functionalized thermoresponsive TCPS; SNAC = chitosan-based thermoresponsive TCPS; TCPS = tissue culture polystyrene.
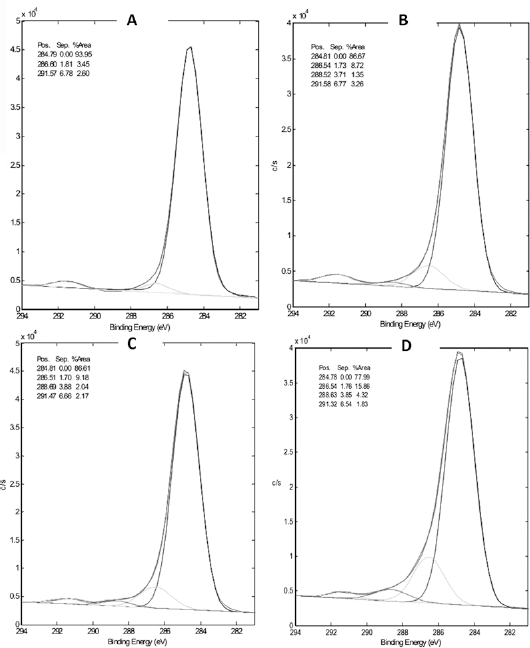
C1s core-level spectra of tissue culture polystyrene (TCPS) (
The content of carboxyl and CS groups on the modified surfaces was determined with the TBO staining method, and the results are shown in Figure 3. It can be seen that the content of carboxyl groups increased with increasing irradiation time. For example, the content of carboxyl groups was 6.77 nmol/cm2 for 0.5SNA, 25.33 nmol/cm2 for 1.5SNA and 65.34 nmol/cm2 for 3.0SNA, respectively (Fig. 3A). The content of CS was 0.615 nmol/cm2 for 0.5SNAC, 4.48 nmol/cm2 for 1.5SNAC and 6.76 nmol/cm2 for 3.0SNAC, respectively (Fig. 3B).
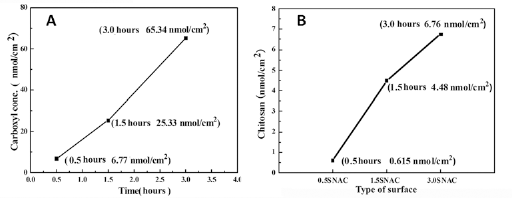
(
Morphology of the modified surfaces
The morphology of the different surfaces was observed by AFM. As shown in Figure 4, the original TCPS was relatively smooth, with a roughness of 1.2 nm. After modification with NIPAAm and CS, the roughness was increased to 65.1 nm for 0.5SN and 54.2 nm for 0.5SNAC. Moreover, there were many peak-like protrusions on the 0.5SN surface and a cloudy protrusion on the 0.5SNAC surface.
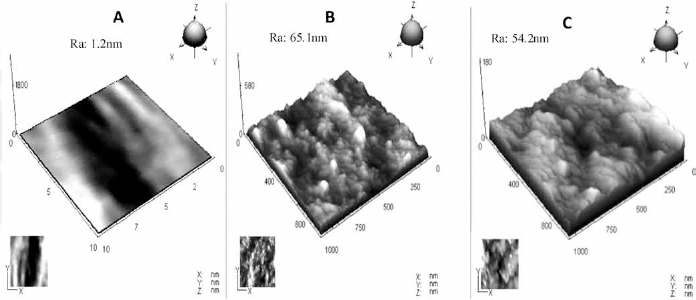
Atom force microscopy (AFM) images of tissue culture polystyrene (TCPS) (
Thermoresponsiveness of the modified surfaces
The thermoresponsiveness of the modified surfaces was investigated by measuring the water contact angle at 20°C and 37°C. As shown in Table II, the contact angle of the original TCPS did not change dramatically when the external temperature was changed. However, for all of the modified surfaces, the contact angles increased when the environmental temperature was changed from 20°C to 37°C. Besides, the contact angles of 0.5SNA and 0.5SNAC surfaces were smaller than that of the 0.5SN surface. The difference of contact angles between under and above 32°C on the 0.5SN surface was higher than that on other surfaces.
Static water contact angles (CAs) of TCPS, 0.5SN, 0.5SNA and 0.5SNAC at 20°C and 37°C

SN = thermoresponsive TCPS; SNA = carboxyl-functionalized thermoresponsive TCPS; SNAC = chitosan-based thermoresponsive TCPS; TCPS = tissue culture polystyrene.
Cytocompatibility of the modified surface
The growth of L929 on different modified surfaces was examined, and the results are shown in Figure 5A. In the early stage of cell culture (2 days), the viability of cells on XSN surfaces showed no obvious difference from that on XSNAC. After incubation for 4 days, the viability of cells on XSNAC was higher than that on XSN at each time point. With regard to the 2 saccharified surfaces, the viability of cells on 3.0SNAC was higher than that on 0.5SNAC. Further, the influence of CS on the cell culture medium was investigated. As shown in Figure 5B, the viability of cells was increased, with increasing concentration of CS in DMEM.
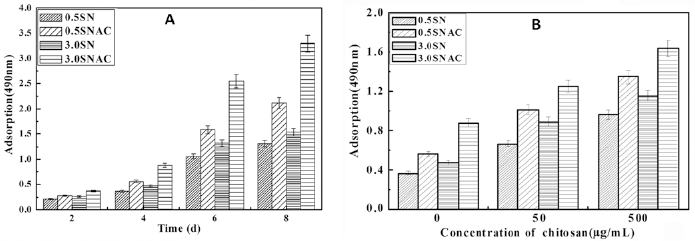
(
The morphology of L929 cells on different surfaces after 6 days of incubation at 37°C was recorded with a phase-contrast microscope. As shown in Figure 6, the cells spread out onto the modified surfaces and reached confluent after 6 days of incubation. Moreover, the number of cells on the surfaces of XSNAC was higher than that on the surfaces of XSN.

Phase-contrast photomicrographs of L929 cells cultured on the different surfaces after 6 days of incubation. Seeding density: 3×104 cells/mL. Scale bar: 100 μm. SN = thermoresponsive TCPS; SNA = carboxyl-functionalized thermoresponsive TCPS; SNAC = chitosan-based thermoresponsive TCPS; TCPS = tissue culture polystyrene.
Detachment of cells from the modified surface with temperature induction
The temperature-induced detachment of L929 cells was assessed by transferring them from 37°C to 20°C after incubation for 4 days at 37°C. Figure 7 shows the detachment curve of L929 from different surfaces. The original TCPS surface showed no thermoresponsiveness, and the detachment rate at each time point was lower than on other modified surfaces. However, it can also be seen that the detachment rate of cells on different modified surfaces increased in the order of XSN <XSNAC <XSNA (for same irradiation time). Moreover, the detachment rates of cells on surfaces with 3.0-hour irradiation time (Fig. 7B) were higher than those on surfaces with 0.5-hour irradiation time (Fig. 7A).
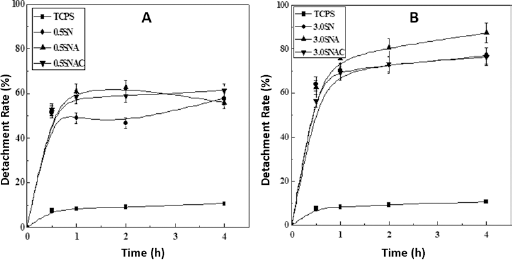
Detachment curves of L929 cells from different surfaces. Statistical significance of cell detachment from modified surfaces compared with that from unmodified surface was evaluated: p<0.001. SN = thermoresponsive TCPS; SNA = carboxyl-functionalized thermoresponsive TCPS; SNAC = chitosan-based thermoresponsive TCPS; TCPS = tissue culture polystyrene.
Reproduction of cells detached from the modified surface
The viability of cells obtained by temperature induction and by trypsin digestion was determined by the MTT method. As shown in Figure 8, the viability of cells obtained by temperature induction was higher than that obtained by trypsin digestion. The viability of cells obtained from 0.5SNAC was higher than that from 0.5SN.
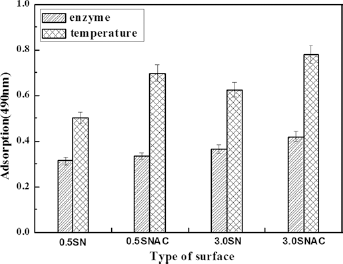
Cell viability of L929 detached with different methods using MTT assay. Statistical significance between cells recovered by lowering temperature and by trypsin treatment was evaluated: p<0.01. SN = thermoresponsive TCPS; SNA = carboxyl-functionalized thermoresponsive TCPS; SNAC = chitosan-based thermoresponsive TCPS; TCPS = thermoresponsive tissue culture polystyrene.
Discussion
The damage, loss and failure of tissue or organs are the most frequent, harmful and costly problems in human health care. Cell therapy and tissue engineering are promising alternatives to artificial permanent implants for repair of tissue lesions (21). Biomaterials occupy a very important position in tissue engineering due to their effects on the biological behaviors and culture efficiency of cells. In this study, a novel thermoresponsive material suited for tissue engineering was developed by a simple and economical UV-induced graft-polymerization method. XPS is the most appropriate technique to analyze the surface of materials. It can detect the elemental composition and chemical bonding state of surfaces. Our results suggested that NIPAAm and AAc were successfully grafted onto the surface of TCPS. Because the activity of the AAc monomer was higher than that of the NIPAAm monomer, macromolecular radicals could easily activate AAc. The presence of AAc radicals promoted the advancement of grafting reaction and resulted in the higher N content of 0.5SNA than of 0.5SN. The higher N content of the CS molecules resulted in the highest N content on 0.5SNAC surfaces. On the other hand, the grafting of NIPAAm with N—C = O onto the surface of TCPS resulted in the new peak appearing on the modified surfaces. The intensity of N–C = O on 0.5SNA increased due to the introduction of AAc, which promoted the advancement of the grafting reaction. The further increasing intensity of this peak in 0.5SNAC resulted from the formation of N—C = O between the amino groups of CS molecules and the carboxyl groups on the surface. The intensity of π* from the base polystyrene substrate decreased due to the covering of TCPS by the grafting layer, which hindered the transition from π to π*. The same phenomenon was also observed by Canavan et al (22).
TBO is a kind of cationic dye that can be mixed with a carboxyl group with a ratio of 1:1 and can also be detached by acetic acid. The absorbance of TBO in acetic acid can be used to measure the content of the carboxyl group. The content of CS on the modified surface (XSNAC) can be obtained by comparing the different content of the carboxy group before and after saccharification of XSNA. Compared with the results of other studies, higher amounts of CS in this present study were immobilized on the modified surface. For example, Hatakeyama et al reported that the density of RGDS (ArgGly-Asp) and INS (insulins) grafted onto TCPS by electron beam irradiation was approximately 0-390 pmol/cm2 and 0-40 pmol/cm2, respectively (3).
The modified surfaces exhibited different morphologies from that of original TCPS. The introduction of NIPAAm and CS improved the roughness of TCPS. CS was immobilized on the peak or in the gap between the peaks. The grafting layer was thickened, and the peak protrusions were connected to form cloudy protrusions (Fig. 4).
The measurement of water contact angles is often used to characterize the hydrophilic and hydrophobic properties of materials. Our results indicated that the introduction of PNIPAAm endowed TCPS with temperature responsiveness. This fact that the change of temperature affected the hydrophilic/hydrophobic property of the modified TCPS surface can be used to detach intact cell sheets by lowering environmental temperature. Furthermore, it could be concluded from the differences of contact angles on the same surface between under and above 32°C that 0.5SN was the most temperature-sensitive surface. The thermoresponsiveness of 0.5SNA and 0.5SNAC was less than that of 0.5SN, due to the introduction of nonthermoresponsive AAc and CS molecules. In addition, the introduction of hydrophilic AAc and CS improved the hydrophilicity of XSNA and XSNAC surfaces.
It is known that cell attachment on biomaterial surface is affected by several factors, such as surface chemistry, surface roughness, surface free energy and surface hydrophilicity. The surface free energy of the modified surfaces increased in the order of 0.5 SN (35.5 × 10−3 J/m2) <3.0 SN (36.4 × 10−3 J/m2) <0.5 SNAC (40.9 × 10−3 J/m2) <3.0 SNAC (46.1 × 10−3 J/m2). The high surface free energy and roughness of XSNAC contributed to the attachment and growth of cells. On the other hand, CS has been widely used as a biomaterial due to its good biocompatibility. Our results demonstrated that the immobilization of CS on the surface of TCPS enhanced the cytocompatibility of the modified surfaces. In the early stage of cell culture, CS embedded in the network of PNIPAAm grafted on the surface failed to completely make contact with cells. However, with increasing culture time, more and more CS on the surface made contact with the cells, due to the movement of PNIPAAm and CS molecular chains, which greatly promoted the growth and activity of the cells. Furthermore, as described above, the content of CS on the surface of 3.0SNAC was the highest among all of the modified surfaces (Fig. 3B), which facilitated the spreading and proliferation of cells on the 3.0SNAC surface (Figs. 5A and 6).
After in vitro proliferation, cells are usually recovered using trypsin digestion. This traditional method often causes damage to nexin between cells and receptors expressed on the cell membranes. However, cells can be spontaneously detached from thermosensitive materials by changing the environmental temperature, to avoid the damage caused by trypsin digestion. PNIPAAm has been widely applied in the fields of tissue engineering, drug delivery systems, biological separation and enzyme immobilization due to its unique environmental responsiveness. In our experiment, all of the modified surfaces exhibited detachment behaviors to a different extent due to the transition of the PNIPAAm molecular conformation. When the environmental temperature was below the LCST (32°C) of PNIPAAm, hydrophilic groups in the PNIPAAm molecular chain were exposed to water, and their molecular character changed from hydrophobicity to hydrophilicity. The hydrophilic surface was not favorable for cell adhesion onto the surface. On the other hand, the extension and swelling of PNIPAAm at a temperature under 32°C weakened the interaction between cells and materials, which facilitated the spontaneous detachment of cells from the modified surfaces. More PNIPAAm was grafted onto the surface of TCPS due to the longer irradiation time, therefore, the detachment rate of cells on surfaces with 3.0-hour irradiation time was higher than that on surfaces with 0.5-hour irradiation time. The presence of larger amounts of carboxyl groups on XSNA resulted in a greater level of detachment of cells.
To verify that cells obtained by temperature induction still retained their activity, the viability of cells obtained by temperature induction and those obtained by trypsin digestion were compared. The results indicated that the detachment of cells by temperature induction could avoid the damage to cells caused by trypsin digestion. The introduction of CS not only reinforced the cytocompatibility of materials but also enhanced the biological activity of the detached cells. Thermoresponsive materials have huge advantages in tissue engineering.
Conclusions
In this study, the modification of TCPS with CS and the thermoresponsive material PNIPAAm, and the influence of modified surfaces on the cellular behaviors of L929 were shown. The results indicated that the modified surfaces underwent a transition between hydrophilicity and hydrophobicity near the LCST (32°C) of PNIPAAm. The introduction of CS promoted the growth of cells and reinforced the cytocompatibility of materials. By the lowering of the environmental temperature, cells can be spontaneously detached from the modified surfaces and the detached cells retain their original function. This method avoids the damage to cells caused by trypsin digestion and provides new ideas and an experimental basis for exploring novel materials in tissue engineering.
Footnotes
Financial support: This work was supported by Science and Technology Correspondent Project of Tianjin (Grant No. 15JCTPJC57500), Natural Science Foundation of Tianjin (Grant No. 16JCYBJC23800) and National Natural Science Foundation of China (Grant No. 51303129).
Conflict of interest: None of the authors has any financial interest related to this study to disclose.
