Abstract
Objectives:
Silk fiber is difficult to degrade in vivo, which limits its application in tissue engineering materials such as artificial nerves. Therefore, in this study aim to promote its degradation in vivo by chemical treating silk fibers in vitro.
Materials and methods:
Sulfate-polyacrylamide gel electrophoresis (SDS-PAGE), scanning electron microscopy (SEM) observations, mechanical test, Fourier transform infrared spectroscopy (FT-IR) measurements were used to investigate the degradation effect of chemicals (hydrochloric acid, phosphoric acid, acetic acid, sodium hydroxide, calcium hydroxide, sodium bicarbonate, and calcium chloride) on silk fiber in vitro. Immunofluorescence staining and transcriptome analysis were used to investigate the effect of inflammatory factors on the degradation of chemically treated silk fiber in rats.
Results:
(1) Silks were separated into finer fibers in each group. (2) FT-IR absorption peaks of amides I, II, and III overlap in each group. (3) Silk degradation degree in each group was higher than that in an untreated group. The calcium chloride-treated group was completely degraded. (4) Fibronectin, collagen I, collagen III, integrin α and CD68 were immunofluorescence positive in all vegetation section. (5) There were no significant differences in the expressions of collagen I, collagen III, and fibronectin in the vegetations formed on the 14th day of subcutaneous implantation, while integrin α, CD68, TNF-α, IL-1b, and IL-23 express at higher levels with IL-10 at lower levels.
Conclusions:
All chemicals could completely degrade silk; however, their degradation products were not the same. The chemicals change the mechanical properties of silk by separating it into finer fibers, which increase the contact surface area between the silk and tissue fluid, accelerating the degradation of monofilaments in vivo by promoting inflammation and macrophage activity through the increased and decreased expressions of pro- and anti-inflammatory factors, respectively.
Keywords
Introduction
Silk is composed of two monofilaments wrapped in sericin. 1 Sericin,2–4 silk fibroin,5–18 and degummed silk (monofilament)19–22 were used in biomedical applications.23–26 Sericin and silk fibroin extracted from raw silk or cocoons are water-soluble. However, in some cases, such as artificial nerves, 19 artificial bones, 20 and artificial blood vessels, 21 it is necessary to reduce their water solubility to maintain their morphology and structure in vivo.
Owing to the different strains of Bombyx mori 3 and silk-protein preparation procedures, the components of the prepared sericin and silk fibroin vary. During sericin preparation, raw silk or cocoons were usually treated in aqueous solutions of sodium carbonate, 27 citric acid, 3 and urea. 2 Alternatively, heat degradation was performed to degrade the sericin recovered from the solutions. 2 The molecular weight of sericin obtained from such procedures included 15–75, 3 50–150,3,4 10–225, 3 25–150, 3 250, 4 and 5–400 kDa. 4 Additionally, sericin contained a mixture of at least 15 different polypeptide chains ranging in size from 20 to 220 kDa. 3 The relative amino acid content varied among sericin proteins,2,3 although their amino acid species were consistent.
For silk fibroin preparation, raw silk or cocoons were usually treated with sodium carbonate,12,14,15,17,28–30 sodium lauryl sulfate, 13 sodium metasilicate, 31 sodium bicarbonate, 32 citric acid, 33 and papain. 9 Thereafter, soap–soda ash 33 and heat degradation 11 methods were adopted to obtain silk fibers, which were then treated with urea, 2 citric acid, 3 sulfuric acid, 14 hydrochloric acid, 16 formic acid,17,34 sodium carbonate,2,3 calcium chloride, 17 lithium bromide,12,35–38 [Bmim][Cl], 39 calcium nitrate-methanol, 13 CaCl2/ethanol/water, 8 and enzymes (trypsin, chymotrypsin, proteinase K, subtilisin, pepsin, AspN, proteinase) 40 to produce silk fibroin. Alternatively, cocoons were directly treated with LiBr, 41 LiSCN, 15 and CaCl2–ethanol–H2O solution 10 to produce silk proteins. The molecular weights of the obtained silk fibroin were approximately 25,12,40 200, 10 35–100, 18 100–150, 40 30–200, 12 20–200, 16 less than 35, 18 and more than 100 kDa. 18 Thus, different times of treatment with the same reagent result in products with different ranged of molecular weight distributions. 18 Furthermore, the relative contents of the products including fibroin light chain, fibrohexamerin, fibroin heavy chain, and sericin1 obtained from silk degummed with different enzymes, such as trypsin, chymotrypsin, proteinase K, subtilisin, pepsin, AspN, proteinase, differ. 40 Therefore, acids, alkalis, salts, and enzymes could all degrade silk, such that these degradations occurred not only in the mutual separation of silk protein molecules but also in the breaking of peptide bonds and amino acids. Hence, the type, concentration, and treatment duration of chemical compounds were directly related to silk degradation.
Degummed silk was in artificial nerve, 19 bone, 20 blood vessel, 21 and ligament 22 grafts owing to its excellent mechanical properties and biocompatibility. Mechanical and in vivo degradation properties were separate requirements for different implants. As silk fibers were required to retain the morphology and mechanical properties of artificial bones, blood vessels, and ligaments in vivo for a long time, they were not expected to disintegrate in vivo. However, artificial nerves required silk with poor mechanical properties and predictable complete disintegration capabilities in vivo over time. Therefore, new methods to regulate the degradation rate in vivo, as indicated by the aforementioned two aspects, required explorations.
In present study, chemical treatments were expected to promote in vivo degradation while retaining the mechanical strength of silk. For this purpose, HCl, NaOH, NaHCO3, H3PO4, Ca(OH)2, CaCl2, and CH3COOH were used for the treatment of silk fibers in vitro, and their solubility and effects on the morphology, structure, and mechanical properties of the silk fibers were investigated. Based on these results, the in vivo degradation properties were explored by subcutaneous implantation in rats.
Materials and methods
Ethics statement
This study was approved by the Animal Care and Ethics Committee of the Chongqing Academy of Animal Sciences (protocol number cqaa2021006).
Materials
Silkworm (Suju × Minghu) cocoons and raw silk were provided by the Institute for Silk and Related Biomaterials Research, Chongqing Academy of Animal Science. Sprague–Dawley rats (200 ± 10 g) were purchased from the Third Military Medical University of the Chinese PLA. Antibodies of collagen type I, collagen type III, fibronectin, CD68 and integrin α were produced by Gene Tax Co., Abcam, Abcam, BOSTER Biological Technology Co., and Santa Cruz Biotechnology, respectively. Nuclear immunofluorescence kit (DAPI), pentobarbital sodium, SDS-PAGE kits, pre-stained protein ladder, and dialysis membranes (YA1049-0.5M, 1KD) were obtained from Biosynthesis Biotechnology (Merck, Beyotime, Thermo Fisher Scientific, and Shanghai Acmec Biochemical Co., respectively). Anhydrous sodium carbonate, sodium bicarbonate, sodium hydroxide, calcium hydroxide, phosphoric acid, hydrochloric acid, acetic acid, and anhydrous calcium chloride were purchased from Chongqing Chuandong Chemical (Group) Co., Ltd.
Degumming of silkworm cocoons and raw silk
The cocoons were cut into pieces, while raw silk was prepared as bundles. The experimental methods previously used by our research group were employed to remove sericin from the cocoons and silk. 19
In vitro degradation of fibroin fibers (FF) by chemicals
Approximately 0.05 g of degummed cocoons pieces were weighed and put into 10 mL aqueous solutions of HCl (6 mol/l), NaOH (27.25 mol/l), NaHCO3 (0.97 mol/l), H3PO4 (14.65–17.24 mol/l), Ca(OH)2 (0.0218 mol/l), CaCl2 (6.71 mol/l), and CH3COOH (17.5 mol/l), respectively. The specimens were placed on a shaking table and left to react for 54 h at room temperature. A centrifuge (cence, TGL-20 M, China) at 3000 rpm for 10 min was employed, and the obtained specimen precipitates were dried (40°C) and weighed to calculate the degradation rate. Supernatant was considered for dialysis (1 KD), which was dried to obtain silk fibroin powder. Samples and 5% or 12% stacking or separating gel were prepared according to the instruction manual for SDS-PAGE reagents, using a pre-stained protein ladder as a marker.
Mechanical test
The degummed silk bundles were treated with 0.05, 0.1, and 0.5 mol/l aqueous solutions of HCl, NaOH, NaHCO3, H3PO4, CaCl2, CH3COOH, and 0.02 mol/l of Ca(OH)2 for 24 h at room temperature. Then, the bundles were rinsed with deionized water for 1 h, followed by removal and drying at 40°C. A tensile test with an equipment (CTM 2100, Xie Qiang instrument manufacturing Co., Ltd. Shanghai, China) was conducted.
Scanning electron microscopy (SEM) observations and Fourier transform mid-infrared spectroscopy (FT-IR) measurements
The degummed silk bundles were treated in 0.05 mol/l aqueous solutions of HCl, NaOH, NaHCO3, H3PO4, CaCl2, CH3COOH, and 0.022 mol/l Ca (OH)2 for 24 h at room temperature. Then, the bundles were rinsed with deionized water for 1 h, removed and dried at 40°C, and subjected to SEM (ZEISS, EVO LS10010102) and FT-IR (Thermo Scientific Nicolet iN10) for measurements.
Subcutaneous implantation of chemically treated silk
Sprague–Dawley rats were randomly divided into eight groups. The degummed silk bundles treated in 0.05 mol/l aqueous solutions of HCl, NaOH, NaHCO3, H3PO4, CaCl2, CH3COOH, and 0.02 mol/l Ca(OH)2 for 24 h at room temperature were implanted under the rat skin, following a previously reported method, 42 with an untreated degummed silk bundle as a control.
In vivo degradation rate and immunofluorescence staining observation
On the 7th, 14th, and 21st day of subcutaneous implantation, the vegetation was removed and reweighed to calculate the degradation rate. Paraffin sections (LEICA, RM2235) were taken from the vegetations on day 7. Immunofluorescence staining observations (Nikon, ECLIPSE80i) were performed with DAPI, fibronectin, collagen I, collagen III, integrin α, and CD68, respectively.
Transcriptome analysis
Transcriptome analyses were performed on the vegetation generated on days 7th, 14th, and 21st days after subcutaneous implantation in the NaOH-treated group (Biomarker Technologies).
Results
Fibroin fiber degradation ability of chemicals
Highly concentrated chemicals were used to investigate their ability of degrading degummed silk (FF). The concentrations of NaOH, Ca (OH)2, NaHCO3, and CaCl2 were saturated, with 27.25, 0.022, 0.97, and 6.71 mol/l, respectively, as their molar concentration. Liquid chemicals such as CH3COOH, H3PO4, and HCl were used without dilution, and their molar concentrations were 17.50, 14.48, and 6.00 mol/l, respectively.
Figure 1(a) showed, H3PO4 completely degraded FF within 36 min while NaOH took 54 h to completely dissolve FF. In 54 h, a 85.20% degradation rate of silk fibers treated with HCl was attained and the others were generally similar with an order as: Ca (OH)2 > NaHCO3 > CaCl2 > CH3COOH.
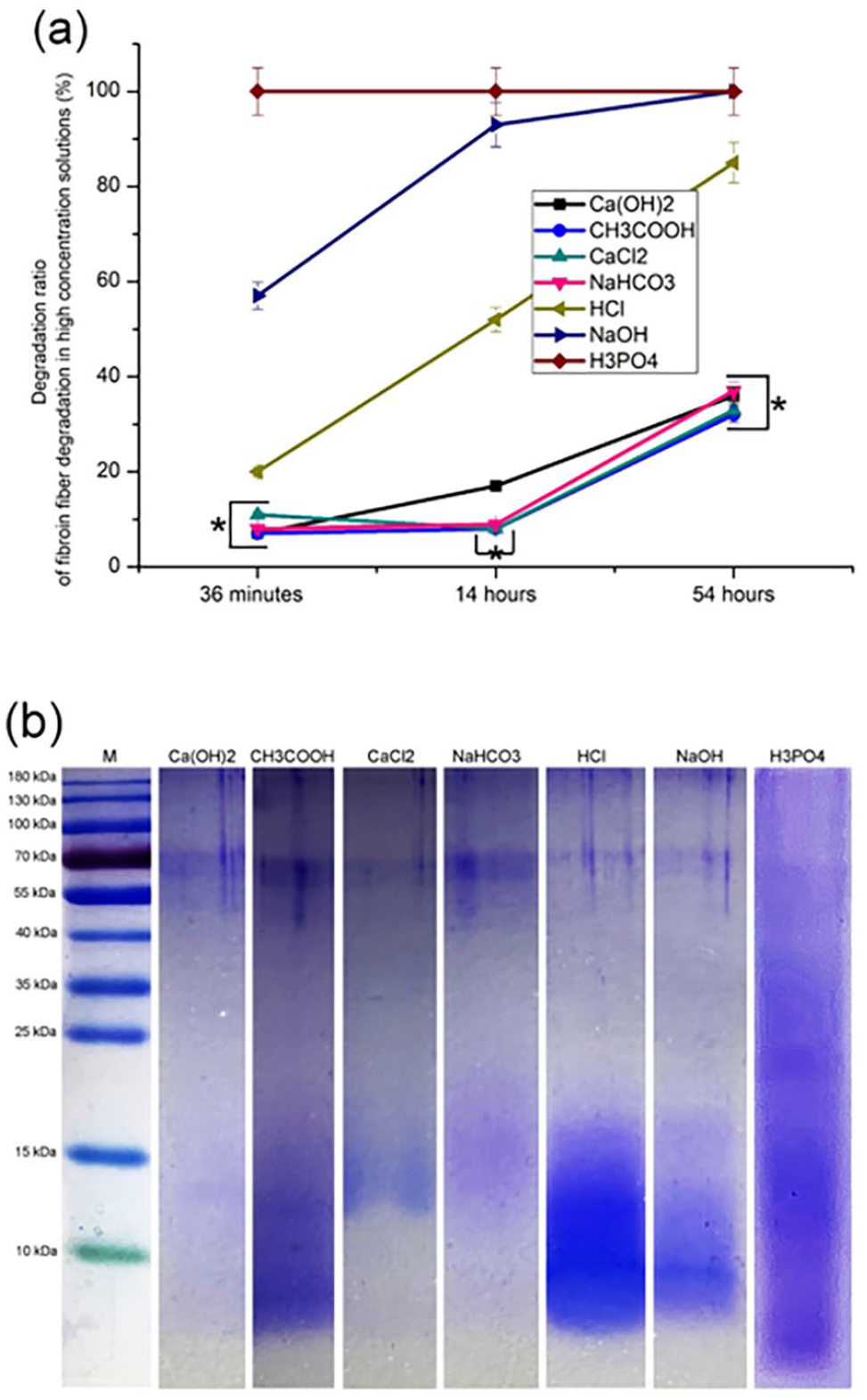
Fibroin fiber degradation in high concentration solutions: (a) degradation ratio (%) and (b) molecular weights of the degradation products (SDS-PAGE).
The degraded products were dialyzed (1000 Da) and subjected to sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE). In Figure 1(b), each group had bands around 180 and 55–70 kDa, while their maps were different in other regions, which suggested that there were significant differences in the molecular weight distribution of degradation products among each group.
Effect of chemical concentration on mechanical properties of fibroin fibers
To investigate the effects of chemical concentrations on the mechanical properties of the fibroin fibers, three gradient concentrations were set: 0.05, 0.1, and 0.5 mol/l. As the concentration of saturated lime water was only 0.022 mol/l, the same concentration of Ca(OH)2 was used during the experiment. The fibroin fibers were treated for 24 h with aqueous solutions of the aforementioned chemicals, and the obtained materials were collected for mechanical testing.
The value of breaking strength is positively related to material strength. In Figure 2(a), compared with untreated fibroin fibers (control), the breaking strength of fibroin fibers was reduced by varying degrees after treatment with different chemicals, while fracture strength in the same chemical treatment group was negatively correlated to concentration. Breaking strength value in the NaOH, HCl, H3PO4, and Ca(OH)2 treatment groups were lower than that in the CH3COOH, NaHCO3, and CaCl2 treatment groups. These results indicated that chemical treatment reduced the mechanical strength of silk.
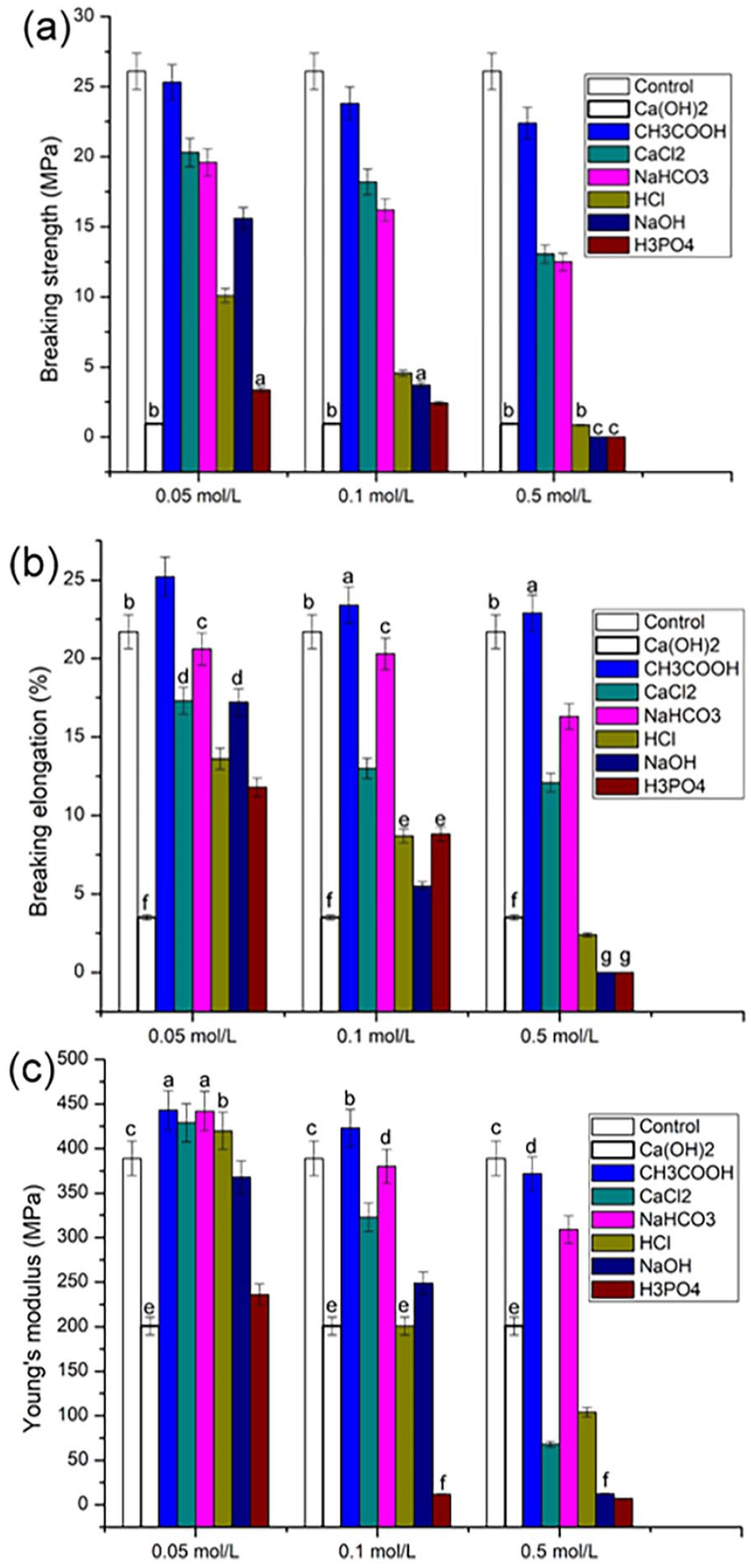
Mechanical properties of fibroin fibers after chemical treatment: (a) breaking strength (MPa), (b) breaking elongation (%), and (c) Young’s modulus (MPa). The values symbolized with the same characters (a–g) indicated no significant difference (p > 0.05), while the others indicated significant difference (SD = 5% mean ± SEM, n = 3, t-test).
The value of elongation at break is positively correlated with the material ductility. In Figure 2(b), the elongation at break of the fibroin fibers in all groups treated with chemical reagents excepting the CH3COOH group decreased by varying degrees compared to the control group, and in the same chemical treatment group, the elongation at break was negatively correlated with concentration. Compared with the CH3COOH, NaHCO3, and CaCl2 treatment groups, the NaOH, HCl, H3PO4, and Ca(OH)2 treatment groups decreased the elongation at break of fibroin fibers by a greater extent. These results indicated that chemical treatment reduced the ductility of fibroin fibers.
The value of the Young’s modulus is positively related to material hardness. Figure 2(c) showed that, within the same chemical treatment group, the material hardness was negatively correlated with concentration. Compared with the control group for a condition of 0.05 mol/l concentration, the hardness of fibroin fibers after CH3COOH, CaCl2, NaHCO3, and HCl treatment increased. Similarly, under the condition of 0.1 mol/l concentration, the hardness in CH3COOH group increased. The hardness of other samples decreased by varying degrees. These results suggested that higher concentrations of chemical treatment could reduce the hardness of the fibroin fibers, while lower concentrations could improve its hardness.
In sections following, based on the mechanical test results (Figure 2) and the saturation concentration of Ca(OH)2 being 0.0218 mol/l, the concentration of Ca(OH)2 group was 0.02 mol/l, while the others were 0.05 mol/l.
Effects of 0.05 mol/l chemicals on structure and degradation of fibroin fibers
The degummed silks were treated in aqueous solutions of 0.05 mol/l of CH3COOH, H3PO4, NaOH, CaCl2, HCl, NaHCO3, and 0.02 mol/l Ca(OH)2 at room temperature for 24 h, rinsed with deionized water, and dried for the experiment. Degummed silks served as controls.
After treatment, the monofilament was split into fine fibers having diameters of 0.2–1.2 μm, as shown in Figure 3(a). Based on different diameters, the obtained fibers were divided into two types: fibers with diameters ranging 0.2–0.4 μm (blue arrows) and 0.6–1.2 μm (green arrows). The number of fine fibers could be varied among different groups.
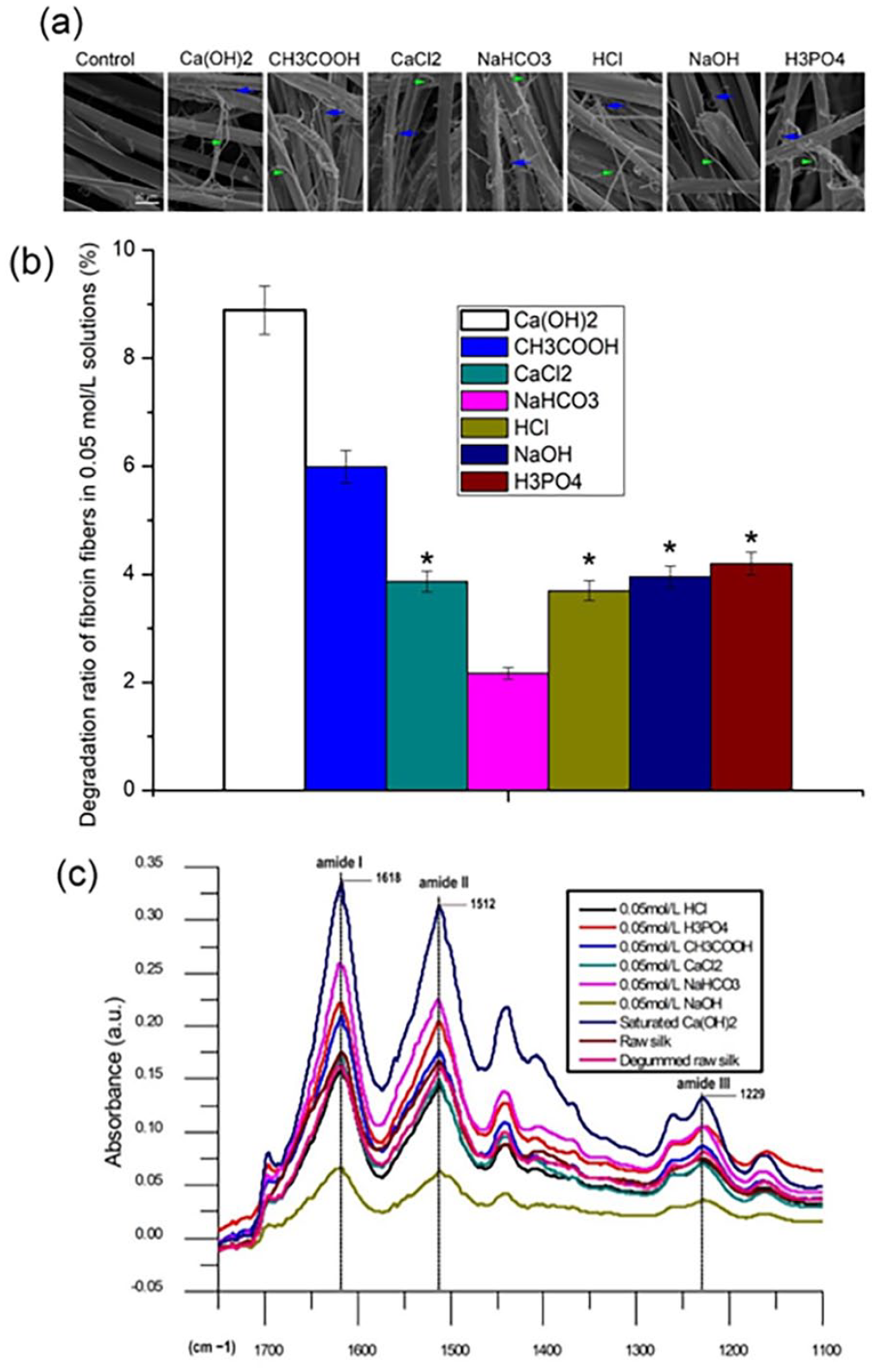
Fibroin fibers after chemical treatment for 24 h: (a) structure of fibroin fibers, (b) degradation ratio (%), (c) FTIR spectra.
In Figure 3(b), the order of degradation rate in groups from high to low was profiled as Ca(OH)2, CH3COOH, H3PO4, NaOH, CaCl2, HCl, NaHCO3, among which the differences in the H3PO4, NaOH, CaCl2, and HCl treatment groups were not significant.
In Figure 3(c), the absorption peak positions of amide I, amide II, and amide III of each group overlapped and were located at 1618, 1512, and 1229 cm−1, respectively.
In vivo degradation of fibroin fibers treated with 0.05 mol/l of chemicals
In this section, based on the mechanical test results (Figure 2) and the saturation concentration of Ca(OH)2 being 0.0218 mol/l, the concentration of Ca(OH)2 group was 0.02 mol/l, while the others were 0.05 mol/l.
Silk fibroin treated with different chemicals was implanted subcutaneously in rats, using untreated fibroin fibers as the control. At 1, 2, and 3 weeks, the vegetation was gathered to weight. The fresh weights of the vegetation formed in each treatment group varied but were lower than those in the control group, as shown in Figure 4. Fibroin fibers treated with the same chemical showed an overtime decrease in the vegetation weight. The degradation rates for each treatment group followed: CaCl2, NaOH, H3PO4, HCl, CH3COOH, Ca(OH)2, and NaHCO3.
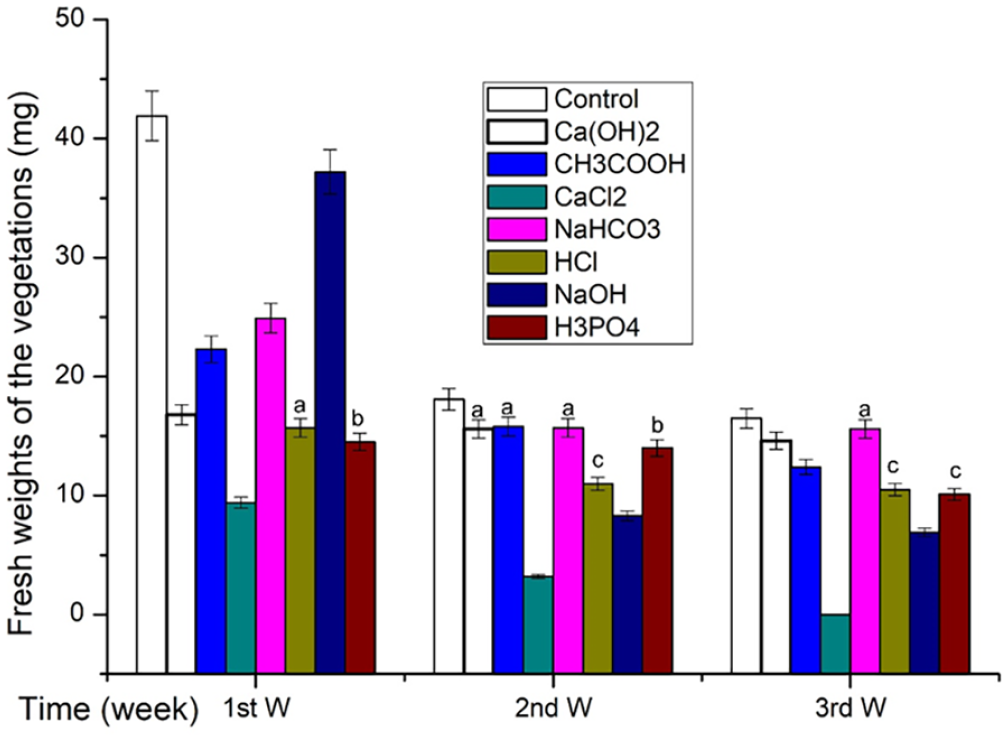
Fresh weights of the vegetations (mg). The values symbolized with the same characters (a–c) indicated no significant difference (SD = 5% mean ± SEM, n = 3, t-test, p > 0.05), while the others indicated significant differences.
Immunofluorescence staining
Antibodies of fibronectin, collagen I, collagen III, integer α, and CD68 were used as markers for fibroblasts, type I collagen, type III collagen, inflammatory response, and macrophages, respectively.
For a week, the vegetation formed by the subcutaneous implantation was observed by immunofluorescence staining of paraffin sections. Fibronectin, collagen I, collagen III, integrin α, and CD68 were all positive in all sections, as shown in Figure 5. The distribution of fibroblasts (Figure 5(a)), collagen I (Figure 5(b)), and collagen III (Figure 5(c)) were extensive and uniform, while integrin α (Figure 5(d)) and macrophages (Figure 5(e)) were distributed in clusters around the fibroin fibers in different stages of degradation.
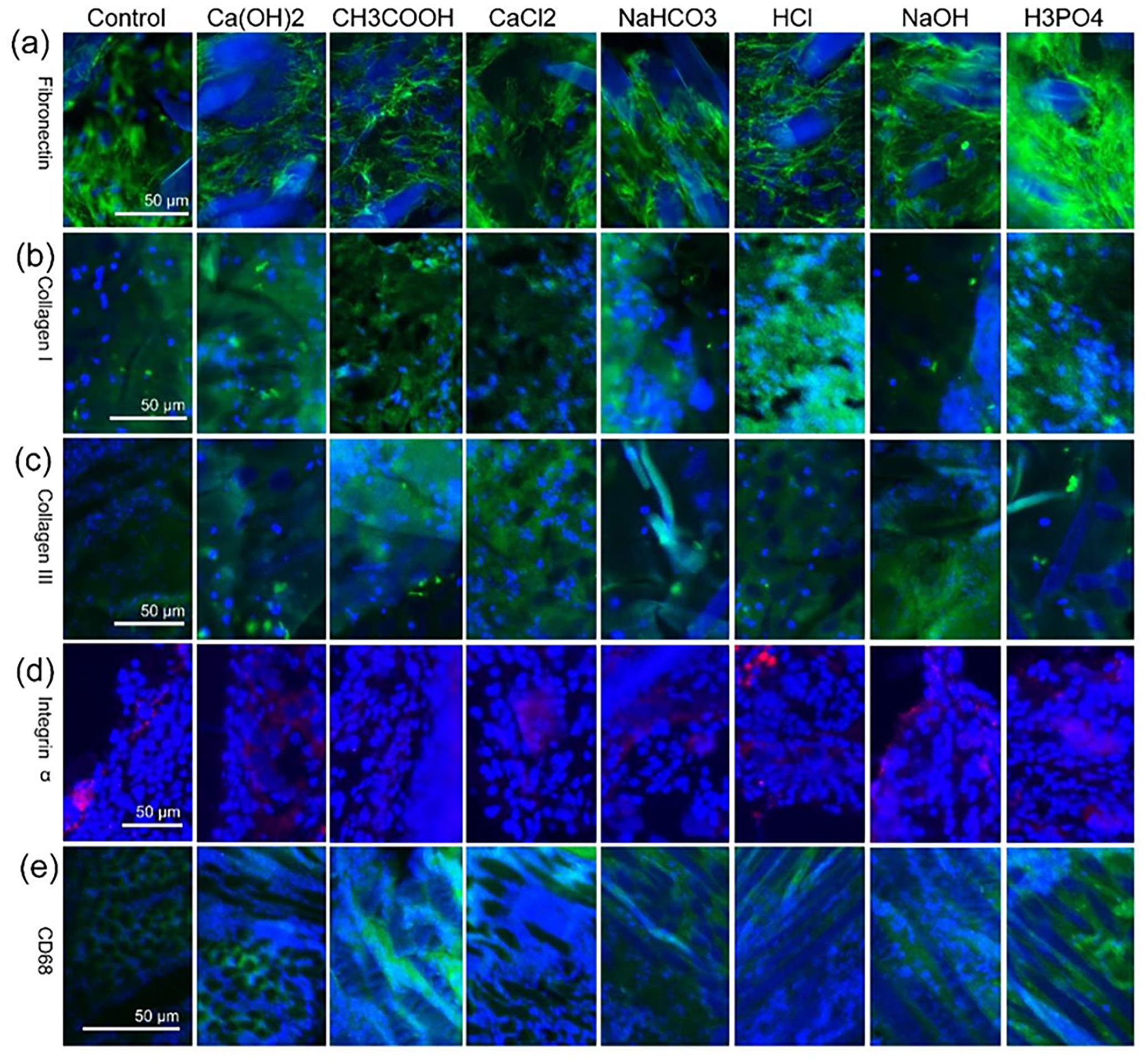
Immunofluorescence staining observation of vegetation sections: (a) fibronectin, (b) collagen I, (c) collagen III, (d) integrin α, and (e) CD68.
Transcriptome analysis
The subcutaneous vegetation formed from fibroin fibers treated with 0.05 mol/L NaOH and degummed silk were used as the experimental and control groups, respectively. On days 3, 7, and 14 after subcutaneous implantation, vegetation was harvested for transcriptome analysis.
The expressions of collagen type I, collagen type III, and fibronectin in the experimental and control groups at each instance were higher than those on the skin of healthy rats, as shown in Figure 6(a). On the third and seventh day, the expression levels in the NaOH-treated group were higher than those in the untreated group, while no significant difference in the expression levels between the two groups was observed on the 14th day. Similarly, no significant differences in the expression levels of collagen types I and III at each instant in the untreated group were observed. However, in the NaOH-treated group, the expression levels were reduced compared with the seventh day results. The expression of fibronectin in the experimental and control groups increased over time, and the expression in the NaOH-treated group was slightly higher than that in the untreated group at each instance.
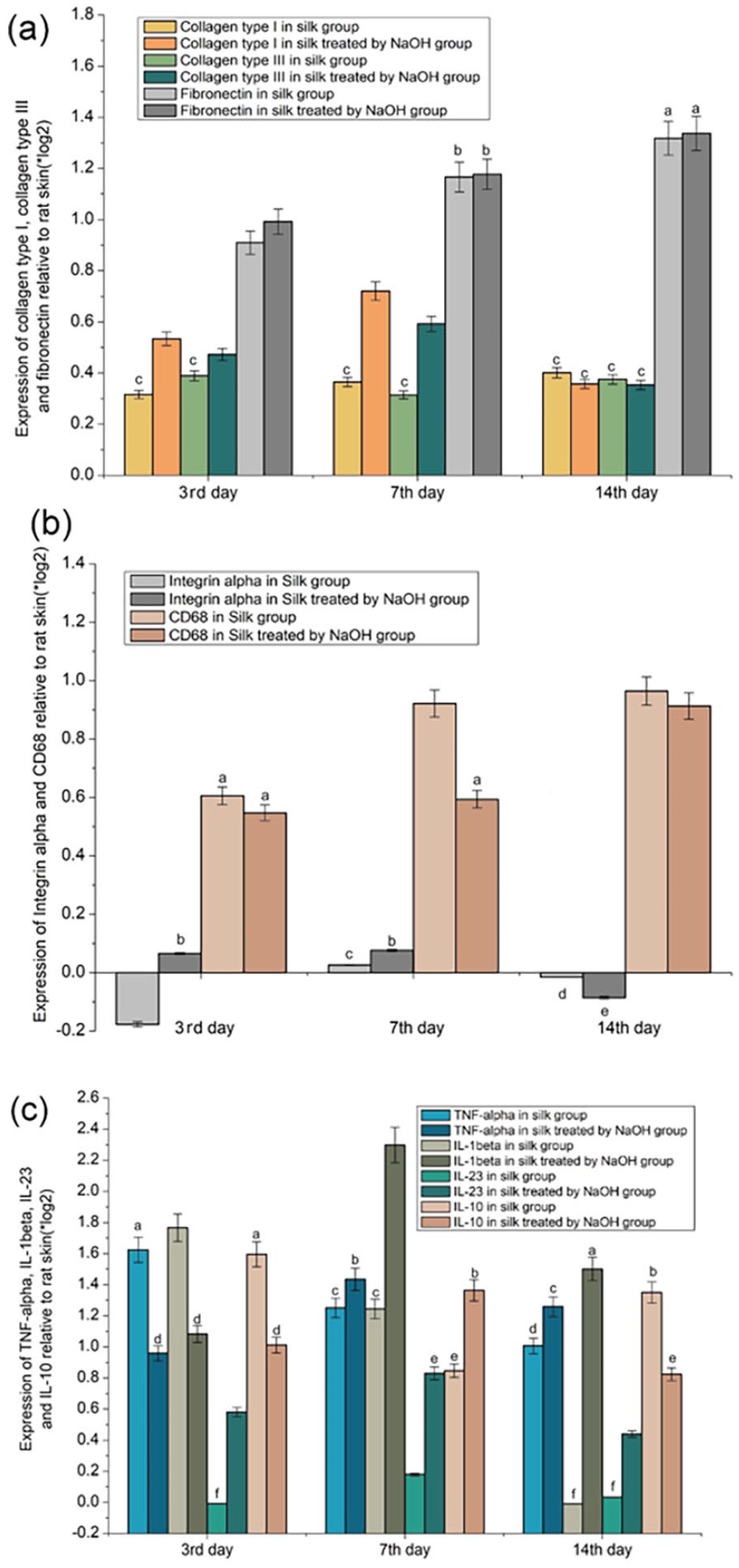
Transcriptome analysis, expressions relative to rat skin (*log2): (a) Collagen type I, collagen type III and fibronectin, (b) Integrin α and CD68 and (c) TNF-α, IL-1β, IL-23, and IL-10. The values symbolized with the same characters (a–f) indicate no significant difference (SD = 5% mean ± SEM, n = 3, t-test, p > 0.05), while the others indicate significant difference.
The expression of CD68 in the experimental and control groups at each instant was higher than that in the healthy rat skin, but the expression level in the NaOH-treated group was lower than that in the untreated group, as shown in Figure 6(b). In the untreated group, the expression of integrin α was lower on the 3rd and 14th day than in healthy rat skin with the exception on day 7. In the NaOH-treated group, the expression of integrin α on the third and seventh day was higher than that in the healthy rat skin and untreated group, while the expression on the 14th day was lower. The expression level of integrin α on the 14th day in both the NaOH-treated and untreated groups was lower than that on the seventh day. No significant difference in the expression levels of CD68 on the 7th and 14th day in the untreated group was observed. Instead, the expression level on the 14th day was higher than that on the seventh day in the NaOH-treated group.
As shown in Figure 6(c), the expression of IL-23 in the untreated group on the third day was slightly lower than that in the healthy rat skin, and the expression of each molecule at each instance in each group was higher than that in the healthy rat skin. In the NaOH-treated group, the expression levels of TNF-α, IL-1β, IL-23, and IL-10 were all higher on the seventh day than in the untreated group. Similarly, the expression levels of other molecules excluding IL-10 on the 14th day were also higher than those in the untreated group. TNF-α, IL-1β, IL-23 expressions in the NaOH-treated group were lower than those in the untreated group at each instant. IL-10 expression in the NaOH-treated group was higher than that in the untreated group on the third and seventh days, but lower than that in the untreated group on the 14th day.
Discussion
Preparation method of regenerated fibroin
Silk proteins obtained by decomposing raw silk or cocoons in chemical solutions are known as regenerated silk fibroin. Generally, the regenerated silk proteins prepared by various processes are polypeptide mixes having a wide molecular weight range. In this study, H3PO4, NaOH, and HCl completely degraded the silk fibers within a short period of time (Figure 1(a)), making them effective reagents for the manufacture of regenerated silk fibers.
The molecular weight distribution of silk fibroin degradation products was more concentrated in NaOH and HCl than in H3PO4. Additionally, the distribution was more concentrated in NaOH than in HCl, mainly around 10 kDa (Figure 1(b)). Thus, NaOH was more suitable for preparing regenerated silk fibroin than HCl and H3PO4.
Fibroin fiber structure
Fibroin fibers comprised fibers of widths 6 nm. 43 In present study, SEM observations revealed that silk fibroin fibers were cracked into fibers having 0.2–0.4 μm and 0.6–1.2 μm diameter after chemical treatment (Figure 3(a)). Therefore, silk fibroin fibers were formed by assembling nanometer-sized fibers into micron-sized fibers, followed by assembling micron-sized fibers into silk fibroin fibers (monofilaments). 44 FT-IR analysis (Figure 3(c)) showed that the absorption peaks of amides I, II, and III10,11,13,14,16,31,39 in silk fibers subjected to different chemical treatments were located at the same positions, confirming no significant alterations to the chemical structure of silk fibroin fibers during the chemical treatment. Therefore, the cleavage of the fibroin fibers into finer fibers by chemical treatment was the main reason for the change in their mechanical properties (Figure 2). Meanwhile, the produced fine fibers promoted degradation of fibroin fibers in vivo, as they increased the surface area of contact between the fibroin fibers, tissue fluids, and cells.
Degradation of chemically treated fibroin fiber in vivo
As shown in Figure 4, the fresh weight of vegetation formed at each interval in the untreated group (control) was higher than that in each chemical treatment group, and the weight of each treatment group decreased over time. In the third week, the CaCl2 treatment group was completely degraded, suggesting the promotion of fibroin fiber degradation in vivo by chemical treatment in vitro.
Fibronectin, collagen I, collagen III, and CD68 45 were the markers for fibroblasts, collagen type I, collagen type III, extracellular matrix and macrophages, respectively, while integrin α46,47 was the adhesion molecule involved in macrophage migration. Figure 5 shows that vegetation was composed of fibroin fibers, collagen I, collagen III, fibroblasts, and macrophages. The distribution of fibroblasts, collagen I, and collagen III (Figure 5(a)–(c), respectively) were extensive and uniform, while integrin α and macrophages (Figure 5(a) and (e), respectively) were distributed in clusters around the fibroin fiber in different stages of degradation. Thus, phagocytosis by macrophages was the main cause of fibroin fiber degradation, and fibroblasts encapsulated fibroin fibers by secreting collagens type I and III to form a cyst-like structure.
Transcriptome analysis (Figure 6) showed that the expression of collagen I, collagen III, and fibronectin at each instant in the experimental (fibroin fibers treated with NaOH) and control (fibroin fibers not treated with NaOH) groups was higher than that in the healthy rat skin. The result was consistent with the formation of subcutaneously implanted vegetation (Figure 5(a)–(c)). In both the experimental and control groups, the expression level in integrin α on the seventh day was higher than that in the healthy rat skin. This observation was consistent with those shown in Figure 5(d). At each instant, the expression of CD68 in the healthy rat skin was lower than that in the experimental and control groups, with the control group expression being lower than of the experimental group, indicating a lower macrophage activity in the control group than in the experimental group. On the 14th day of subcutaneous implantation, the expression of TNF-α,48,49 IL-1β,49,50 and IL-23 51 (pro-inflammatory cytokines) in the control group was lower than that in the experimental group. Similarly, IL-10, 52 an anti-inflammatory cytokine, on the 14th day of subcutaneous implantation had a higher expression in the control group than in the experimental group. Based on the differences in the expressions of CD68 and the above-mentioned inflammatory factors, NaOH treatment in vitro was speculated to compel fibroin fibers to promote inflammatory response and macrophage activity in vivo, thereby promoting their degradation in vivo owing to the separation of silk fibroin fibers into numerous fine fibers by NaOH treatment in vitro. Thus, the surface area of silk in contact with tissue fluid and cells increased.
Conclusions
The aqueous solutions of HCl, NaOH, NaHCO3, H3PO4, Ca (OH) 2, CaCl2, and CH3COOH all had degradation ability toward silk, which was positively correlated with their concentration, and their degradation products had different molecular weights. The chemicals affect the mechanical properties of silk by breaking it into finer fibers, making it “easy” for phagocytosis to promote its degradation in vivo. During its degradation process in vivo, inflammatory reactions and inflammatory cells played a significant role.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Chongqing Scientific Research Institution Performance Incentive Project (cstc2022jxjl0259, 22529 J, 22531 J, and 19537).
