Abstract
Cerium oxide nanoparticles (CeO2 NPs) have unique physicochemical properties that make them suitable for various applications, particularly in biomedicine. To enhance their compatibility with biological systems, we functionalized these nanoparticles with selected amino acids, which significantly improved their potential for biomedical use, as our findings show. Cerium oxide has a fluorite-type structure and can exist in both trivalent and tetravalent states. Notably, cerium oxide also serves as an effective radioprotectant, offering selective protection to normal cells over tumor cells. The present embodiment provides functionalized cerium oxide nanoflakes with a plurality of amino acids. The surface modification by amino acids has been studied and characterized with various techniques. The cytotoxicity and the biological activity of nanoceria and the functionalization of nanoceria were evaluated. The result of this investigation shows the change in morphology structure and optical properties. The anticancer activity of the samples shows significant results against the MCF-7 and SCC-29B cell lines overall. In the case of comparing pure and functionalized nanoceria in the microbial study, the functionalization of nanoceria shows better significance by improving biological activity in some cases than synthesized/pure nanoceria and becomes suitable for biomedical applications. Therefore, the results show that the functionalization of nanoceria plays an important role in manufacturing and the possibility of using it as a therapeutic compound in the future to improve the public health of society.
Introduction
The field of nanomaterials has attracted significant attention from researchers due to their exceptional physicochemical properties and their potential for diverse applications across various disciplines. 1 Among these materials, cerium oxide (CeO2) nanoparticles stand out due to their high surface-to-volume ratio and their ability to cross biological barriers, making them suitable for both medical and industrial use. Cerium, a lanthanide and one of the most abundant rare earth elements, forms cerium oxide, a compound with a fluorite-type crystal structure (space group Fm3m). Nanostructured cerium oxide (CeO2) has become a focus of considerable interest due to its versatility. It has been engineered for use in a wide range of applications.2–4 CeO2 has one of the most distinctive properties: its ability to reversibly switch between Ce3⁺ and Ce4⁺ oxidation states. This redox behavior underpins its antioxidant capabilities, making it highly valuable in biological applications such as biosensing, 5 drug delivery, 6 and protection against oxidative damage. 7 Studies have demonstrated that cerium oxide nanoparticles can scavenge reactive oxygen species (ROS), thereby reducing oxidative stress, 8 minimizing inflammation, and preventing conditions such as cardiovascular myopathy 9 and retinal degeneration caused by intracellular peroxides. 8 Additionally, CeO2 exhibits superoxide dismutase-like activity, 10 further enhancing its biomedical relevance. Moreover, CeO2 nanoparticles serve as promising radio-protective agents, selectively shielding normal tissues while offering minimal protection to tumor cells. 11 According to Khorrami et al., 12 cerium oxide nanoparticles have shown biocompatibility and safety for medical applications, particularly in mitigating oxidative stress. Numerous studies have explored the anti-cancer potential of cerium oxide nanoparticles (CeO2 NPs) and their cytotoxicity on various cell lines. Esmail et al. investigated the cytotoxic effects of CeO2 NPs on mouse fibrosarcoma cell lines and found promising evidence supporting their use in fibrosarcoma treatment. 13 Similarly, Babu and colleagues reported that CeO2 NPs are cytocompatible and may be used both as contrast agents in imaging and for cancer therapy. 14 Recent investigations have also emphasized the need to evaluate both cytotoxic and genotoxic effects across different cell lines, including their potential applications in wound healing and topical therapies.15,16 Another study demonstrated the ability of nanoceria to protect the gastrointestinal epithelium from radiation-induced damage, highlighting its value in both nanomedicine and radioprotection. 17 In addition, cerium oxide has shown selective cytotoxicity, effectively reducing the viability of cancer cells while remaining non-toxic to healthy cells. 11
Several physicochemical properties influence the biological activity and performance of nanomaterials, including their shape, size, crystalline structure, chemical composition, surface functionalization, degree of aggregation, surface charge, and hydrophobic or hydrophilic nature.18,19 Among these factors, surface functionalization plays a pivotal role in enhancing the performance of cerium oxide nanoparticles compared to their non-functionalized or pristine counterparts. Functionalized nanoparticles demonstrate improved interaction with biological systems, better dispersion, and enhanced cellular uptake. Surface functionalization of nanoceria is critical in optimizing its interactions with biological systems. Functionalization strategies enhance the nanoparticles’ stability, dispersion, and compatibility within biological environments, which is particularly important in biotechnology and biomedical applications. These modifications can significantly improve the performance of nanoceria in areas such as targeted drug delivery, imaging, and biosensing. Moreover, the surface modifications offer new insights into the mechanisms underlying nanoceria-induced cytotoxicity.20,21
By modifying the surfaces of these nanomaterials, we seek to find future solutions for cancer cells. That is considered the second cause of death worldwide, and it affects people of all ages. About 9.6 million deaths in 2018 in the world were due to cancer, according to WHO. 22 Cancers of the lung and female breast are the top cancer types in terms of incidence, accordance to the World Health Organization. In India, there are six types of cancer, according to the World Health Organization (WHO). Breast cancer and oral cancer are the highest types of cancer from the number of patients, and then cervical cancer, lung cancer, stomach cancer, and colorectal cancer. There are different methods used for the treatment of cancer, like Surgery, chemotherapy, and radiotherapy. These treatments are almost expensive and have side effects. So, researchers and scientists try to find urgent solutions for these people who suffer from this disease. Nanomaterials (1–100 nm) are rapidly developing in the field of medical imaging and drug delivery, and it is one of the solutions that have biological activity and can interact with cells, and have minimal side effects. Therefore, it may be useful in the diagnosis and treatment of cancer cells. There are several methods to assay cancer or tumor cell lines. One of these assays is the Sulforhodamine B colorimetric. Sulforhodamine B (SRB) colorimetric assay, which was developed by Skehan in 1990, is widely used to investigate the cytotoxicity of drugs/ compounds used in tumor studies because of its highly cost-effective screening. 23 The SRB method is a highly sensitive method that is used to investigate cytotoxicity against cancer and non-cancerous cell lines due to the independence of cell metabolic activity,24,25 and it is also considered a method with high cost-effectiveness.
In our work, we study the functionalization of nanoceria by amino acids. Amino acids are a diverse class of molecules that have one identifying property. The amino acids possess the same basic structure: a carboxylic acid group and an amino group. It can be divided into categories depending on the side chain which is attached to the a-carbon. Some of this group are polar and others non-polar, the third one is uncharged, and the fourth one has a charge. Cells in human/animals, or plants use amino acids to build proteins. 26 AAs are used in various applications such as antimicrobial and drug Delivery. 27 In the current investigation, we used alanine (Ala), serine (Ser), valine (Val), cysteine (Cys), aspartic (Asp), glutamine (Gln), glutamic (Glu), methionine (Met), and arginine (Arg). We evaluate how morphological, structural, physicochemical properties, and antimicrobial and anti-cancer activities can be affected by introducing an amino acid that can interact or contribute to the surface of nanoparticles. Then it may help to improve some properties of pure nanomaterials. Therefore, we selected different types with various properties of amino acids to surface modify nanoceria to evaluate the presence of bioactivity and anticancer activities in vitro study of CeO2 and functionalized CeO2. Furthermore, we study the antimicrobial activity against Aspergillus niger and Aspergillus fumigatus. The Aspergillus is considered a pathogenic fungus, especially for people who have a deficiency of immunity and who suffer from respiratory disease, and those who suffer from Covid 19 (Coronavirus disease 2019), which has spread around the world and more than 6 million people have died till May 2, 2023. 28 The anti-cancer screening was also studied against various cell lines such as Human Breast Cancer Cell Line (MCF-7), Human Prostate Cancer Cell Line (PC-3), Human Colon Cancer Cell Line ((HT-29), Human Lung Cancer Cell Line (A-549), and Human Oral Cancer Cell Line (SCC-29B). The nanoceria and functionalized nanoceria were submitted to a primary in vitro anticancer screening at the Advanced Centre for Treatment, Research & Education in Cancer (ACTREC), Mumbai, India.
To our knowledge, this is the first study to use functionalized cerium oxide nanoparticles with nine amino acids with different properties to assay as anticancer treatments.
Material and methods
In the current work, we used various materials, such as L-serine, L-valine, L-cystine, L-aspartic, L-glutamine, L-glutamic, L-arginine, and dimethyl sulfoxide were purchased from Alfa Aesar. L-methionine was purchased from Molychem Company. While L-alanine, Agar-Agar, dextrose monohydrate, Methylene blue, and ethanol were purchased from Molychem. All chemicals were used without further purification. Figure 1 displays details of materials, molecular weight, and molecular structures of selected amino acids.

Chemical structure of selected amino acids grouped according to side chain.
Synthesis and preparation of functionalization of cerium oxide nanoparticles
The chemicals were commercially used with AR purity and without any purification. Initially, 3.73 g (0.01 M) of CeCl3⋅7H2O was dissolved in 100 mL of distilled water and stirred at 80ºC, and then 1 mL of polyethylene glycol 400 was added. 1.68 g (0.03 M) of potassium hydroxide was dissolved in 100 mL of distilled water. The second solution was added to the first dropwise and maintained pH to 10 with continuous stirring for 4 h. The formed solution was filtered and washed with ethanol and distilled water, and dried. Then the dry sample was annealed in the oven for 2 h at 400ºC. The formation of cerium oxide nanoflakes (CeO2 NFs) (light yellow) was achieved by grinding a mortar and pestle to obtain a fine powder of nanoparticles. 29
The surface-functionalized nanoceria was prepared by magnetic stirring. The functionalization of CeO2 NFs was prepared by mole: mole of nanoceria and amino acid. Then, the ratio of 1:1 of nanoceria with amino acid was put in a round-bottom flask. 50 to 60 mL of ethanol was added. The previous solution was kept under constant stirring at 1000 rpm for 12 h at 60ºC. After that, the solution was flirted by using standard Whatman filter paper No.1. The filtered solution was subjected to washing with double-distilled water and kept to dry overnight. The dried functionalized nanoceria was crushed and ground in a ceramic mortar using a pestle to ensure the production was fine and powdered functionalized nanoceria nanoflakes (FCeO2 NFs). The fine powder was isolated for further characterization. We repeated this work for each amino acid used in this study.
X-ray diffraction technique
The functionalization of cerium oxide nanoparticles was studied using an XRD – BRUKER D8 diffractometer – with Cu-K radiation to confirm the structure of functionalization CeO2 in the range of 2ϑ from 10º to 80º. Calculated the size of the crystal using the Debye-Scherrer equation.
Field emission scanning electron microscopy and energy-dispersive X-ray spectroscopy
The structure morphology of functionalization CeO2 was characterized by FESEM-FEI Nova Nano SEM 450, combined with EDS-Quantax-200; Bruker, Berlin, Germany, to get information about chemical composition.
FTIR spectroscopy and Raman microscopy
To confirm the functional groups of functionalized CeO2 by using FTIR-BRUKER ALPHA II, from 4000 to 500 cm−1, and Raman spectroscopy with an Olympus microscope with green from 200 to 1000 cm−1 with a resolution of 4 cm−1 at room temperature.
UV-VIS spectrophotometer
To study the optical properties of functionalized CeO2, a UV-VIS spectrophotometer, ELICO, SL 210 double beam, in the wavelength range from 190 to 900 nm, with a path length of 10 mm and accuracy ±0.5 nm, was used. The spectra were recorded for all samples in absorbance mode at room temperature.
In vitro cytotoxicity evaluation
Sources of cancer cell lines
Advanced Centre for Treatment, Research & Education in Cancer (ACTREC), Mumbai, India
Human cancer cell lines
The selected human cancer cell lines were the human Breast Cancer cell line (MCF-7), human Prostate Cancer cell line (PC-3), human Colon Cancer cell line (HT-29), human Lung Cancer cell line (A-549), and human Oral Cancer cell line (SCC-29B).
Cell culture and experiment design
The cell lines were grown in RPMI 1640 medium (Roswell Park Memorial Institute Medium, which was developed by George Moore et. al. in 1967, at Roswell Park Memorial Institute, used in cell culture) containing 10% fetal bovine serum and 2 mM L-glutamine. The cells were inoculated into 96-well microtiter plates. After that, the microtiter plates were incubated at 37°C, 5% CO2, 95% air, and 100% relative humidity for 1 day before the addition of experimental drugs. The nanoceria and functionalized nanoceria (FAla, FSer, FVal, FCys, FAsp, FGln, FGlu, FMet, Farg), and the standard compound Adriamycin (ADR) were solubilized in Ethanol (EtOH) at 100 mg/mL and then diluted to 1 mg/mL with water and stored frozen before use. Adriamycin was used as the positive control compound in the cytotoxic assay. At the time of adding the drug, the frozen concentrate (1 mg/mL) was dissolved and diluted to (100, 200, 400, and 800) μg/mL with EtOH. Aliquots of 10 µL of these different drug dilutions were added to the suitable microtiter wells, which already contained 90 µL of the medium. Then the final drag concentration becomes (10, 20, 40, and 80) μg/mL. The plates were incubated at standard conditions for 2 days (48 h), and the assay Sulphorhodamine B (SRB) was terminated by adding cold Tri-chloro-acetic acid (TCA). The SRB assay was developed in 1990 by Philip Skehan and his colleagues. This method is used to measure the cell proliferation and cytotoxicity of drugs and is used for large-scale drug screening applications. 30 The cells were fixed in situ by the gentle addition of 50 µL of cold TCA 30% (w/v) and incubated for 1 h at 4°C. The plates were washed five times with tap water to discard the supernatant and air-dried. The 50 µL solution of SRB at 0.4% (w/v) in 1% acetic acid was added to each of the plates and incubated for 20 min at room temperature. After that, the unbound and residual dye was removed by washing five times with 1% acetic acid and letting the plates air-dry at room temperature. To determine the optical density, the bound dye was subsequently eluted with 10 m Mtrizma base solution (pH 10.5) to each well of the plate, and the plate was shaken on an orbital shaker for 10 min to solubilize the protein-bound dye. The absorbance was read on a microplate reader at a wavelength between 490 and 530 nm to avoid nonlinear measurement and at 690 nm for reference.30,31
Data analysis
Percent Growth was expressed as the ratio of the average absorbance of the test well to the average absorbance of the control wells × 100
Percentage cell growth inhibition was determined as (100 − percentage cell growth in test well).23,32
Criteria for the determination of activity
The test samples show growth inhibition of ⩾50% when the solution concentration is 80 μg/mL, then it is considered to be active.
Statistical analysis
Statistical calculations were carried out using Microsoft Excel 2021.
In vitro study, antifungal assay
Potato dextrose agar (PDA) media is typically used for mycelia growth inhibition assays. PDA media was prepared and sterilized in an autoclave for 15 min at 115°C and poured into petri dishes. Each Petri dish contains 10 mL of PDA media. The two types of pathological fungi, Aspergillus niger (A. niger), and Aspergillus fumigatus (A. fumigatus) were cultured from rot fruit, and the isolation and identification of fungi were explained in Saif et al. 33 The fungi were grown on PDA petri dishes (90 mm diameter) and incubated at room temperature, 28°C ± 2, for 5 days. Petri dishes were observed, and even the fungal colonies grew and consisted of mycelia and conidia. After that, the fungal colonies were ready to be cultured again on PDA media. Antifungal activity of nanoceria and functionalized nanoceria were screened against A. niger and A. fumigatus. The antifungal screen activity of the CeO2 and functionalized CeO2 was carried out by the diffusion method with poisoned food, as described in Balouiri et al. 34 A stock solution for each sample was prepared by adding 3 mg of nanoceria/functionalized nanoceria to 10 mL of dimethyl sulfoxide (DMSO). The dispersion of nanomaterials was sonicated to break the agglomerations and to make them uniform for 30 min. The 50 μg/mL of these stocks (minimal inhibitory concentration (MIC)) was incorporated into Petri dishes containing 10 mL molten agar of PDA and mixed well. The plates were cooled for a sufficient time to solidify the agar medium. A mycelia disc (2 mm) was cut from the zone of overgrowth of 5-day-old cultures and inoculated in the center of Petri dishes. In the control plate, PDA media was used without treatment. The Petri dishes were incubated at 28 ± 2ºC under suitable conditions. After 5 days, colony development was observed, and the diameter was measured. The antifungal activities of growth inhibition were measured by using a standard scale. The measurement was replication three times for each treatment, and the mean value and the standard deviation (SD) were obtained. The diameter in millimeters (mm) of zone inhibition ± Standard Division (SD) was reported as mean ± SD.
Percent (%) inhibition
Inhibition of mycelia growth was determined based on the radial growth of mycelia in mm on the PDA media plate and expressed in terms of percent inhibition, a parameter calculated using the following formula 35 :
Results and discussion
XRD study
XRD was employed to ascertain the crystalline structure of functionalized nanoceria, and the previous work shows the crystalline structure of nano ceria. 36 Figure 2 shows the XRD patterns of the functionalization of nanoceria with various amino acids.
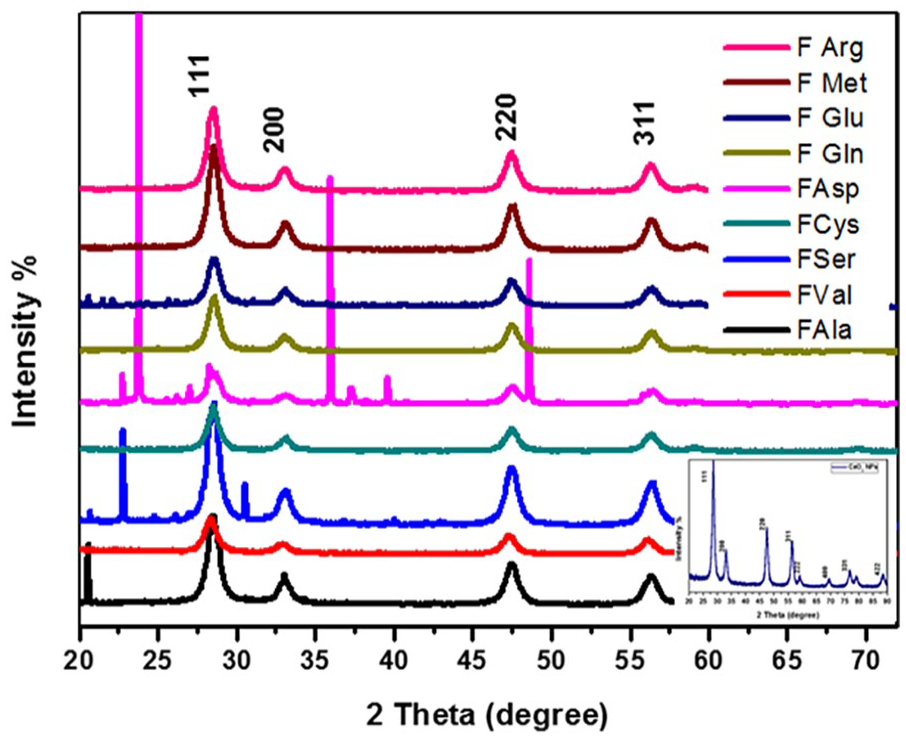
XRD of functionalization of CeO2 NFs.
The pattern of XRD of functionalized nanoceria exhibits a very strong peak at 2θ = 28.548°, 28.528°, 28.379°, 28.539°, 28.539°, 28.564°, 28.580°, 28.583°, 28.519° corresponding to the crystal plane (111) for FAla, FSer, FVal, FCys, FAsp, FGln, FGlu, FMet, and FArg, respectively. In comparison with the Gayathri et al. 37 study, it shows that the characteristic peak for functionalized nanoceria at the angles 28.548°, 28.528°, 28.379°, 28.539°, 28.539°, 28.564°, 28.580°, 28.583°, and 28.519° indicates the crystalline structure of FCeO2. That confirms our XRD results for FCeO2. While the remaining peaks show weak or modulated intensity. The previous peaks can be indexed to a cubic fluorite structure corresponding to a face-centered cubic and in agreement with JCPDS file number 81-0792, which confirms the crystalline structure. 37 The peak positions of different types of functionalized nanoceria showed no critical change due to guest materials. The grain size (crystallite size (D)) of FAla, FSer, FVal, FCys, FAsp, FGln, FGlu, FMet, and FArg was calculated at the high-intensity peak of line broadening of the reflections (111) using the Debye-Scherrer equation (1):
where D is the crystallite size, λ is the wavelength of Cu kα (0.15406 nm), θ is the Bragg’s angle (peak position), and β is the line broadening at full width at half maximum (FWHM). Supplemental Table 1S shows that the diffraction peaks were slightly shifting in the peak position 2θ and slightly broadening in the peak observed after CeO2 capping by amino acids, which confirmed the presence of capping/dopant of CeO2 surface. It was noted from the XRD pattern that some additional peaks were observed when functionalized CeO2 by some amino acids, such as Aspartic and Serine amino acids, which indicated the strong effect of amino acids on the lattice of nanoceria. Additionally variety in the intensity and broadening in some peaks after inducing AAs on the surface of CeO2 as shown in the inset of Figure 2.
That is also interpreted in the form of the rearrangement of atoms in the nanoceria or distorted structure of CeO2 NFs. The increased value of FWHM than synthesized CeO2 indicated to change in the surface due to stress-strain, grain distortion, or other 38 and reduced grain size. Moreover, the change in lattice parameters after introducing the AAs matrix can prove that some sites of Ce atoms may be replaced by atoms of AAs and enter into the lattice of CeO2, 39 also we can observe gradual variation in the intensity peak by the decrease in the molecular weight of AAs after functionalization as shown in Figure 2. Therefore, from the XRD pattern could be observed that the coated nanoceria by AAs did not change the crystal structure of the nanoceria core. From XRD data can be calculated the microstructure parameters for distinguishing the role of introducing amino acids matrix on the surface of nanoceria, such as the lattice constant, dislocation density, and micro-strain (ε). 40 The lattice constant, dislocation density (δ), and micro-strain (ε) were calculated using equations (2) to (4), respectively.
Where a is the lattice constant,
where d111 is the inter-planar distance between the crystallographic plane at (111).
where D is the grain size, and δ is the dislocation density.
where ε is the micro-strain, β is the full width half maximum (integral breadth), and θ is the Bragg angle. The values of grain size (D), lattice constant (a exp.), micro-strain (ε), d-spacing, and dislocation density (δ) were reported in Supplemental Table 1S, and a small change in lattice constant was observed after functionalization by AAs. This variation in a value may be due to a change in the type of AAs, which have different molecular weights and are slightly different in structure. That is also dependent on the oxygen vacancy in the structure of nanoceria and depends on the chemical surface of nanoceria. 41
FESEM study
The morphology and microstructure of cerium oxide (CeO2) nanoparticles functionalized with nine different amino acids were characterized using Field Emission Scanning Electron Microscopy (FESEM) and Energy Dispersive X-ray Spectroscopy (EDS). As depicted in Figures 3 and 4, the functionalized samples exhibit aggregated clusters composed of thin, irregularly shaped platelets, which appear to be interconnected through a self-assembly mechanism. Both open and closed porosity were observed across all samples, indicating the potential for high surface area and accessible active sites. The observed agglomeration or aggregation in the amino acid-functionalized CeO2 nanoparticles is primarily attributed to the presence of carboxylic acid groups in the amino acid structures. During the surface modification process, these groups facilitate electrostatic interactions between the amino acids and the nanoparticle surfaces, promoting nanoparticle clustering. Furthermore, X-ray diffraction (XRD) analysis confirmed a reduction in nanoflake size upon functionalization, suggesting that the presence of amino acids may influence crystal growth dynamics. This aggregation behavior is likely governed by multiple factors, including the ionic strength of the solution and the modified surface chemistry of nanoceria. Specifically, the carboxyl groups and hydrogen bonding interactions between the amino acids and the nanoflake surfaces are key contributors to the formation of agglomerates. These findings are consistent with previous studies highlighting the critical role of surface functional groups in modulating nanoparticle assembly and dispersion behavior.42,43 The EDS analysis of the amino acid-functionalized cerium oxide (CeO2) nanoparticles confirmed the absence of extraneous impurities on the nanoparticle surfaces. The EDS spectra revealed distinct energy peaks corresponding to elements such as cerium, oxygen, carbon, nitrogen, and, in some cases, sulfur, which are consistent with the presence of surface-bound amino acids. These findings validate the successful surface modification of nanoceria with the selected amino acids. The elemental composition data for all samples are illustrated in Figure 4.

FESEM of functionalization of CeO2 NFs.
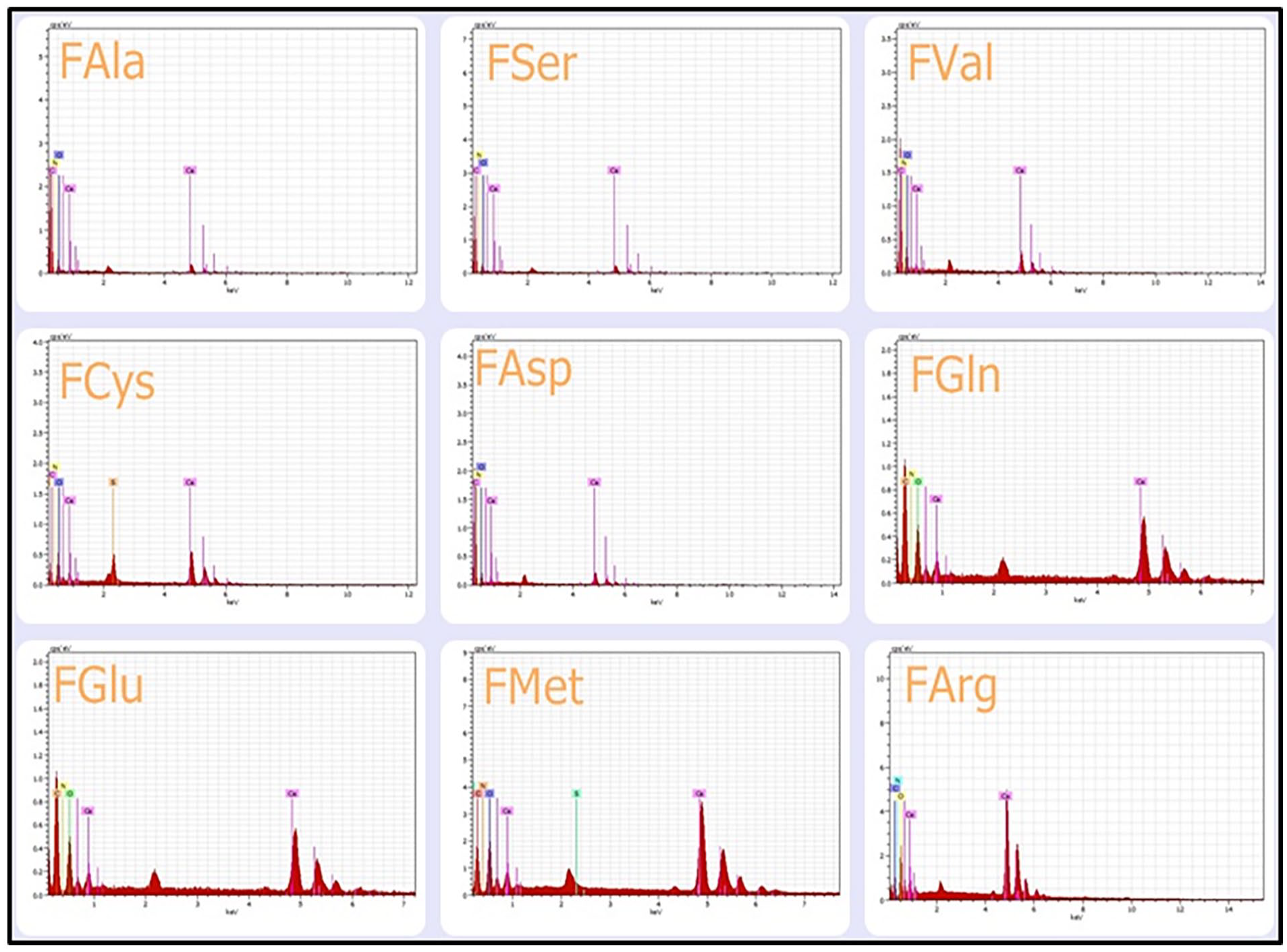
EDS of functionalization of CeO2 NFs.
FTIR study
Fourier Transform Infrared (FTIR) spectroscopy was employed to confirm the successful surface functionalization of cerium oxide nanoparticles (CeO2 NFs) with amino acids (CeO2-AA). This technique provided insights into the chemical interactions between the amino acid molecules and the nanoceria surface, as well as detailed information about the functional groups present. FTIR spectra were recorded in the range of 4000–500 cm−¹, with twenty scans per sample at a resolution of 4 cm−¹, all collected at room temperature.
The FTIR spectrum of the functionalized CeO2 nanoflakes (F-CeO2 NFs) displayed a prominent absorption band between 500 and 600 cm−¹, corresponding to the Ce–O stretching vibrations, which is characteristic of cerium oxide and aligns with previously reported findings. 44 A noticeable band near 2360 cm−¹ was attributed to the asymmetric stretching of carbonate species (CO3²−), likely formed from surface-adsorbed CO2 interacting with oxygen vacancies on the ceria surface. 45 Additionally, the peaks observed around 1600–1500 cm−¹ were associated with C=O stretching vibrations from carboxylic acid groups of the amino acids, 46 indicating their successful conjugation onto the nanoparticle surface.
Further, bands detected between 1450–1500 cm−¹ and near 700 cm−¹ were assigned to the bending vibrations of C–H bonds from methylene (-CH2-) groups. 47 Peaks appearing between 670–650 cm−¹ were ascribed to Ce–O–C stretching, implying the formation of coordination bonds between the cerium oxide surface and amino acid ligands. 48 The spectral region from 1600–1100 cm−¹ revealed NH2 and CH3 bending vibrations, while bands within 1000–650 cm−¹ were attributed to NH3 and CNH bending, as well as C–N stretching modes, all of which further corroborate the presence of amino acid moieties on the nanoceria surface.49–51 The complete FTIR spectra of all functionalized CeO2 samples, along with a reference spectrum of pristine CeO2 NFs, are presented in Figure 5.

FTIR spectra of functionalization of CeO2 NFs.
Raman study
Raman spectroscopy is a powerful technique for examining structural changes and surface modifications in nanomaterials, particularly in assessing the effects of functionalization on cerium oxide nanoparticles (CeO2 NFs). In this study, Raman spectra were recorded to evaluate the interaction between CeO2 and various amino acids (AAs). As shown in Figure 6, functionalization induced slight shifts in the vibrational peak positions and broadening of the characteristic Raman bands of CeO2, indicating structural modifications at the nanoscale.

Raman spectra of functionalization of CeO2.
The dominant Raman active mode of cerium oxide, typically observed around 465 cm−¹ and corresponding to the F2g symmetric stretching mode of the Ce–O bond in the fluorite-type lattice, exhibited a slight shift upon amino acid conjugation. The Raman peaks for the functionalized CeO2 samples appeared at 462.23, 459.44, 462.23, 459.44, 462.23, 462.23, 462.23, 462.12, and 462.96 cm−¹ for FAla, FSer, FVal, FCys, FAsp, FGln, FGlu, FMet, and FArg, respectively. This subtle variation in peak position and increased peak broadening suggests a change in the local lattice environment due to surface functionalization.
These spectral changes can be attributed to several factors, including lattice strain, grain size reduction, and interactions between the amino acid molecules and the CeO2 surface. The observed broadening and downshift are consistent with phonon confinement effects and the formation of oxygen vacancies, which disrupt the long-range order of the crystal lattice. 52 Additionally, the incorporation of functional groups from AAs may lead to localized lattice distortions or inhomogeneous strain, further contributing to the spectral modifications.39,53 These findings are also supported by XRD results, which indicate changes in crystallite size and lattice parameters following functionalization.
UV-VIS study
The effect of surface-functionalized by different types of AAs dispersed in distilled water was recorded by absorption spectra of UV-Vis spectroscopy. The characteristic optical density of NFs and functionalized NFs is shown in Figures 7(a) and (b). There is a blue shift in the absorption peak by increasing the molecular weight of amino acids, if compared to pure cerium oxide, because of the reduction in particle size that is confirmed by XRD and FESEM. If the absorption peaks are located between 230 and 260 nm, the Ce3+ is rising, and for Ce4+, the absorbance is located between 300 and 400 nm. 54 In our work, all samples have an absorbance peak between 300 and 400 nm; therefore, by reducing the size of nanoflakes then there is a defect of oxygen vacancies which helps to raise Ce3+. The absorption peak of synthesized nanoceria and functionalized nanoceria by Ala, Ser, Val, Cys, Asp, Gln, Glu, Met, and Arg were 375, 357.8, 349.2, 344.5, 350.7, 353.5, 353.6, 345.6, 355.3, and 374.5 nm respectively which corresponding to blue shifting and increase Ce3+, and due to electronic transitions which referred to oxygen vacancies in nanoflakes structure. So, by an increasing amount of Ce3+, the size of the nanoflakes was reduced. 54 So the absorption of nanoceria in the UV region helps to use them in biomedical products. To estimate the energy band gap for each sample, equation (5) was used.

(a) UV-Vis spectra of pure CeO2 and (b) UV-Vis spectra of functionalization of CeO2.
where h is Planck’s constant, c is light speed, λ is the wavelength, and Eb.g is the energy band gap. By calculating the energy band gap for each sample using the above equation. We found the energy is 3.47, 3.55, 3.60, 3.54, 3.52, 3.51, 3.59, 3.49, and 3.31 for CeO2, FAla, FSer, FVal, FCys, FAsp, FGln, FGlu, FMet, and FArg, respectively. The value of energy for all samples was greater than the bulk of cerium oxide (Eg = 3.19 eV).
In vitro cytotoxic activity
Nanomaterials are a way to find a new treatment for cancer by creating novel antitumor therapies. Cerium oxide NFs exhibited an antioxidant effect due to their unique redox capabilities present on their surface, which have emerged as therapeutic anticancer agents as a single agent or in combination with conventional drugs for tumor treatments because of the activity of pro-apoptotic, and induction of autophagy.55,56 The metal oxide NFs’ inhibition efficacy may be attributed to the generation of reactive oxygen species (ROS).57,58 Also, ceria NFs generate ROS in cancer cells but scavenge it in normal cells depending on various factors of the environment, including pH. 59 Moreover, nanoceria was found to target mostly cancer cells compared to normal cells. 60
The pure nanoceria CeO2 (P-CeO2) and functionalization of CeO2 (F-CeO2) were screened for five cell lines using the SRB assay. Figures 8 and 9 show the effects of CeO2, FAla, FSer, FVal, FCys, FAsp, FGln, FGlu, FMet, and FArg against five types of human cell lines: human Breast Cancer Cell Line (MCF-7), human Prostate Cancer Cell Line (PC-3), human Colon Cancer Cell Line (HT-29), human Lung Cancer Cell Line (A-549), and human Oral Cancer Cell Line (SCC-29B). The P-CeO2 and F-CeO2 were studied in concentrations of 10, 20, 40, and 80 µg/mL and screened for anti-cancer activity by using the SRB assay, which is a sensitive method to measure the cytotoxicity of compounds. The morphology of the treated cells by P-CeO2 and F-CeO2 was observed in Figures 8(a) and (b). FSer, FVal, FAsp, and FGlu show a better significant effect on cytotoxicity and an increase in dead cells 61 against MCF-7 and SCC-29B than other F-CeO2 or P-CeO2 in a dose-dependent manner; these results suggest to development of these compounds for more inhibition and more cytotoxicity for cell lines. In Abdi Goushbolagh et al., 62 Atif et al., 63 and Barzegarparay et al., 64 explain the activity of nanoparticles against tumor cells (MCF-7) using radiation dose (dose-dependent). As shown in Figure 8, the image of control shows the main shape, morphology, and adherence to tissue culture, while the treatment cells show some differences in morphology and a decrease in the number of cell lines. Therefore, MCF-7 and SCC-29B show more sensitivity to the functionalization of nanoceria. This finding is similar to Zare-Bidaki et al.’s 65 and Al-Adilee’s 66 studies, which show the activity of Ag NPs and transition metal complexes, respectively, against the MCF-7 cell line. In another study, Kiani et al. show that LSE@Ag-doped ZnO NPs have significant cytotoxic effects against the HT-29 colon cancer cell line, further supporting the versatility of nanomaterials in cancer therapy. 67 These findings underscore the promise of nanotechnology in developing targeted, biocompatible cancer treatments with minimal effects on healthy cells. Moreover, nanoceria exhibits remarkable properties, including the ability to scavenge reactive oxygen species (ROS) and act as a free-radical scavenger. Its unique redox cycling between Ce³⁺ and Ce4⁺ states further enhances its potential as an anticancer and therapeutic agent for targeting tumor cells.68,69 The confirmed potential of these nanoparticles for antibiotic degradation, along with their demonstrated antibacterial and anticancer activities, highlights their value in biomedical applications. Moreover, their eco-friendly nanoparticles further support their suitability for safe and sustainable use in healthcare technologies.70–72

(a) Breast cancer cell line MCF-7 of CeO2 and functionalization of CeO2 (positive activity) and (b) oral cancer cell line SCC-29B of CeO2 and functionalization of CeO2 (positive activity).

(a) Lung cancer cell line A-549 of CeO2 and functionalization of CeO2 (negative activity), (b) human colon cancer cell line HT-29 of CeO2 and functionalization of CeO2 (negative activity), and (c) human prostate cancer cell line PC-3 of CeO2 and functionalization of CeO2 (negative activity).
The remaining drugs (FAla, FCys, FGln, FMet, and FArg) did not elicit significant activity for the same cell lines. CeO2, FAla, FSer, FVal, FCys, FAsp, FGln, FGlu, FMet, and FArg show negative significance (no significance of cytotoxicity active) against PC-3, HT-29, and A-549, Figures 9(a) to (c).
Therefore, the result implies that the functionalization of nanoceria was efficient in two of cell lines from five cell lines. If the observed percentage of inhibition was 50% or more, the in vitro cytotoxicity was evaluated to be a positive effect 73 according to The National Cancer Institute NCI, 74 as appeared in Table 1 and Figure 10; otherwise was a negative effect as appeared in Table 2 and Figure 11.
Percentage cell growth inhibition of CeO2 and functionalization of CeO2 of MCF-7 and SCC-29B (positive effect).

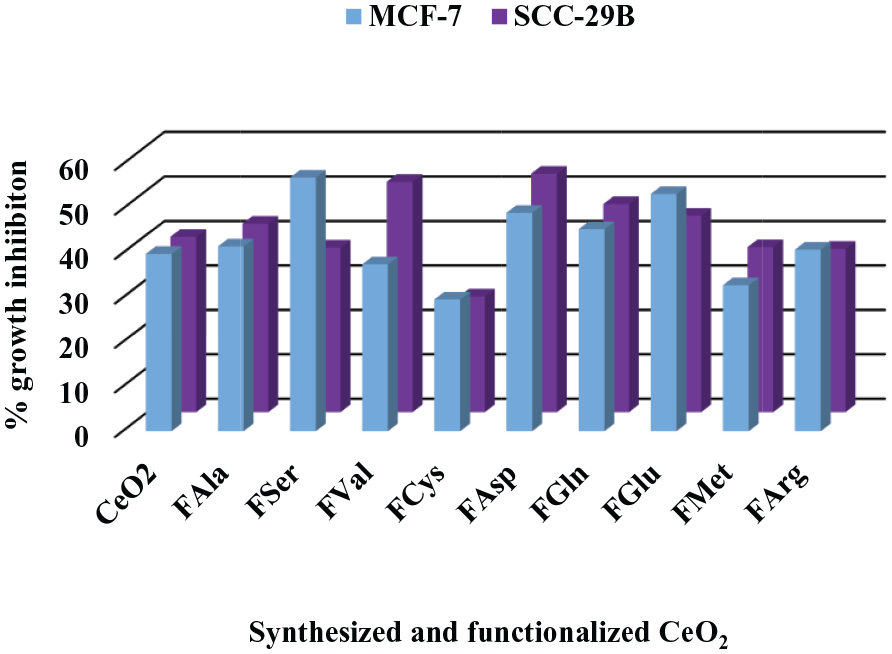
Percentage cell growth inhibition of CeO2 and functionalization of CeO2 of MCF-7 and SCC-29B (positive effect).
Percentage cell growth inhibition of CeO2 and functionalization of CeO2 against PC-3, HT-29, and A-549 (negative effect).
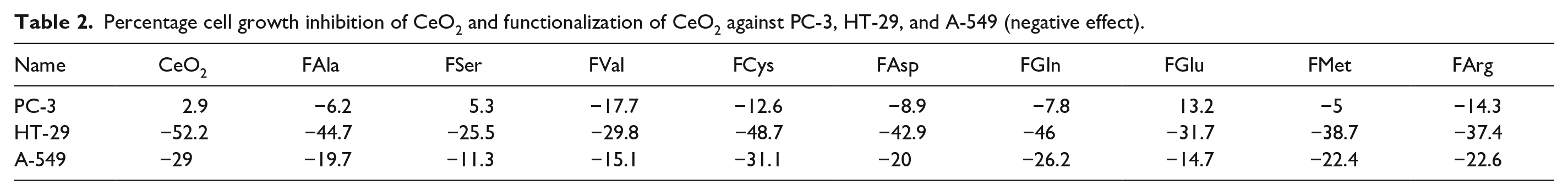

Percentage cell growth inhibition of CeO2 and functionalization of CeO2 against PC-3, HT-29, and A-549 (negative effect).
The results of the current study indicate that nanoceria exhibits anticancer activity against the tested cell lines, supporting its potential biocompatibility for cancer treatment. However, despite its promise, the biocompatibility and biosafety of nanoceria nanoflakes (NFs) must be carefully evaluated, as certain potentially toxic effects have been reported in vitro. These include inflammation, the formation of apoptotic bodies, lactate dehydrogenase (LDH) membrane leakage, chromosomal damage, and chromatin condensation.75,76
The observed cytotoxicity of nanoceria appears to depend on the nature of the surface-functionalized nanoflakes, which can influence intracellular interactions based on the specific functional groups introduced by amino acid (AA) capping. Additionally, cytotoxic effects are influenced by the dose-response relationship and the pH of the cellular environment, where variations in acidity or neutrality can significantly affect toxicity outcomes in both tumor and healthy cells.9,77
Antimicrobial activity
The antimicrobial activity of nanoceria and functionalized nanoceria were evaluated against pathogenic fungi species at 28 ± 2ºC. The pathogenic fungi were identified using the optical microscopy method, and DNA barcoding.
The two pathogenic fungi were identified by DNA barcoding as Aspergillus niger (Accession No. MZ435863) and Aspergillus fumigatus (Accession No. MZ435922) species by Sanger dideoxy sequencing at Paul Hebert Centre for DNA Barcoding and Biodiversity Studies (Dr Babasaheb Ambedkar Marathwada University, Maharashtra, India). The two pathogenic fungi can be identified by optical microscopy as Aspergillus niger: the mycelia of A. niger have a brown to dark brown color, and the darkish color of colonies with light yellow on the edges can be observed. The conidial head is brown to black. It is splitting into columns, and it has a diameter between 3.7 and 4.5 μm, 78 as shown in Figure 12(a). Whereas the Aspergillus fumigatus can be identified as colonies that have a gray-green color with a pale yellow color. The conidiophores are dome-shaped with a diameter of 20–30 µm. The conidial heads have a bluish to green color, columnar, and with a diameter between 2.4 and 3.5 μm, 79 as shown in Figure 12(b).

Antifungal activity of CeO2 and functionalization of CeO2: (a) Aspergillus niger and (b) Aspergillus fumigatus.
The results of screening against A. niger and A. fumigatus as the minimum inhibitory concentration (MIC) were summarized in Tables 3 and 4 and Figures 13 and 14. As shown in Figures 12(a) and (b), the antimicrobial activity of synthesized CeO2 and functionalized by a matrix of AAs shows moderate effectiveness. This toxicity/inhibitory activity of nanoceria and functionalized nanoceria against fungi is due to reactive oxygen species (ROS) and is affected by some factors such as pH, structure, morphology, size, surface functionalization of nanoflakes, and surface charge. 80 In addition to the ability of nanoparticles to penetrate fungal cells, where they bind to compounds containing sulfur and phosphorus, such as DNA, ultimately leading to the destruction of these microorganisms. 81 Biochemically, CeO2 NPs may exert their antimicrobial effect by interfering with the integrity and function of microbial cell membranes. The interaction between functionalized nanoceria by amino and the cell wall could lead to increased membrane permeability or disruption of lipoprotein structures, ultimately resulting in cell lysis. This mechanism mirrors the known effects of some antimicrobial agents that inhibit cell wall synthesis or destabilize membrane structures.82–84
Mean result of antifungal screening activity of pathogenic fungi Aspergillus niger and Aspergillus fumigates for p-CeO2 and F-CeO2, at 28 ± 1ºC.

“—” Negative screening result (separated in all petri dish).
Inhibition % of antifungal screening activity of pathogenic fungi Aspergillus niger and Aspergillus fumigates for p-CeO2 and F-CeO2, at 28 ± 1ºC.

“—” Negative screening result (separated in all petri dish).

Mean inhibition zone (mm) of antifungal activity of CeO2 and functionalization of CeO2.
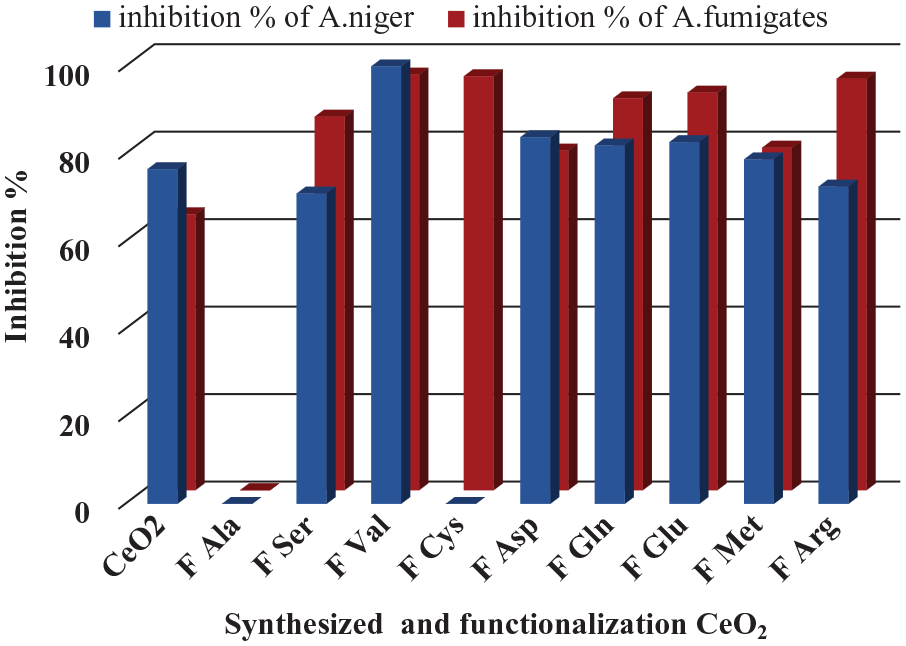
Inhibition % of antifungal screening activity of pathogenic fungi A. niger and A. fumigates for CeO2 and functionalization of CeO2, at 28 ± 1ºC.
The results indicated that nanoceria and capped nanoceria exhibit varying effective antifungal activity. Fval, FAsp, FGln, and FGlu show more activity than others against A. niger while FVal, FCys, FGlu, and FArg show more effectiveness than others against A. fumigatus. Therefore, these findings highlight the potential of CeO2 and FCeO2 as agents with distinctive antimicrobial capabilities (Figure 13).
Conclusion
This study highlights the biomedical potential of cerium oxide nanoflakes (nanoceria) functionalized with nine amino acids (Ala, Ser, Val, Cys, Asp, Gln, Glu, Met, and Arg). Structural characterization confirmed the successful synthesis and surface modification of nanoceria, with XRD verifying its fluorite structure and spectroscopic analyses supporting the presence of amino acid capping. The functionalization influenced morphology, surface chemistry, and biological behavior. Biological assays revealed that specific amino acid-functionalized nanoceria, particularly Ser- and Glu-capped CeO2, exhibited notable cytotoxicity against MCF-7 and SCC-29B tumor cell lines and strong antifungal activity against Aspergillus fumigatus. These effects are likely due to surface chemistry alterations introduced by amino acid capping, affecting redox activity, ROS scavenging, and cellular interactions. Conversely, other functionalized variants showed minimal or no biological activity, emphasizing the critical role of surface functional groups in therapeutic efficacy.
Overall, this work supports the potential of amino acid-functionalized nanoceria as a selective anticancer and antifungal agent. Future studies should focus on in vivo evaluations, mechanistic insights into cellular interactions, and dose optimization to further assess safety and therapeutic applicability in clinical settings.
Supplemental Material
sj-docx-1-jbf-10.1177_22808000251354888 – Supplemental material for Functionalization of nanoceria a dual approach to anticancer and antifungal applications
Supplemental material, sj-docx-1-jbf-10.1177_22808000251354888 for Functionalization of nanoceria a dual approach to anticancer and antifungal applications by Salama A. Yaseen, Faizaa A. Saif and Prabhakar B. Undre in Journal of Applied Biomaterials & Functional Materials
Footnotes
Acknowledgements
Dr. Jyoti Kode (Officer Incharge, Anti-Cancer Drug Screening Facility (ACDSF), Advanced Centre for Treatment, Research & Education in Cancer (ACTREC), Tata Memorial Centre, Mumbai) for the in-vitro screening of the drugs for Anti-cancer activity analysis of the sample. The authors are thankful to Deepak Gupta, Archaeological Survey of India-Aurangabad for providing Raman spectrometer facility.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors declare that this work was submitted as a patent in the Office of Controller General of Patents, Designs & Trade Marks (CGPDTM) Application Number: 202221045279.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.







