Abstract
Nanofibrous scaffolds have emerged as promising candidates for localized drug delivery systems in the treatment of cutaneous cancers. In this study, we prepared an electrospun nanofibrous scaffold incorporating 5-fluorouracil (5-FU) and etoposide (ETP) for chemotherapy targeting melanoma cutaneous cancer. The scaffold was composed of polyvinyl alcohol (PVA) and chitosan (CS), prepared via the electrospinning process and loaded with the chemotherapeutic agents. We conducted relevant physicochemical characterizations, assessed cytotoxicity, and evaluated apoptosis against melanoma A375 cells. The prepared 5-FU/ETP co-loaded PVA/CS scaffold exhibited nanofibers (NFs) with an average diameter of 321 ± 61 nm, defect-free and homogenous morphology. FTIR spectroscopy confirmed successful incorporation of chemotherapeutics into the scaffold. Additionally, the scaffold demonstrated a hydrophilic surface, proper mechanical strength, high porosity, and efficient liquid absorption capacity. Notably, sustained and controlled drug release was observed from the nanofibrous scaffold. Furthermore, the scaffold significantly increased cytotoxicity (95%) and apoptosis (74%) in A375 melanoma cells. Consequently, the prepared 5-FU/ETP co-loaded PVA/CS nanofibrous scaffold holds promise as a valuable system for localized eradication of cutaneous melanoma tumors and mitigation of adverse drug reactions associated with chemotherapy.
Introduction
Skin cancer is widely recognized as the predominant form of malignancy worldwide. Over the past few decades, there has been a significant increase in the occurrence and prevalence of both melanoma and non-melanoma skin cancers. 1 In 2024, the American Cancer Society projects an estimated 100,640 newly diagnosed cases of melanoma in the United States. 2 Skin cancer is characterized by the excessive growth of abnormal skin cells. This phenomenon is primarily attributed to unaddressed DNA damage in the skin cells, leading to genomic abnormalities and rapid cell proliferation, ultimately culminating in the development of cancerous tumors.3,4
Skin cancer can be classified into two primary types based on cellular origin: non-melanoma and cutaneous melanoma. Non-melanoma skin cancer (NMSC) originates from cells derived from the epidermis and can be further categorized into two main subtypes: cutaneous squamous cell carcinoma and basal cell carcinoma. Together, these two subtypes account for 99% of all cases of NMSC. 5 While melanoma tumors originate from melanocytes resident in the epidermis and represent the most significant malignant lesions in human skin. Most skin malignancies arise from exposure to sunlight, which can occur either through prolonged exposure over time or brief, intense episodes resulting in sunburn. Exposure to ultraviolet (UV) radiation present in sunlight has been shown to have detrimental effects on the DNA of skin cells.4,6
A diverse range of treatment interventions have been used in order to eliminate lesions of the skin and tumors. Presently, numerous therapies are used to relieve the symptoms of skin cancer. These forms of therapy include chemotherapy, radiation therapy, immunotherapy, and surgical removal of the tumor. The choice of therapeutic mode depends on factors such as the tumor’s accessibility, stage of advancement, borders, and size. 7 The primary approach to skin cancer management includes surgical interventions to achieve the complete removal of the lesion to prevent its growth and spread, as well as preserve its aesthetic appearance and functional capabilities, while its recurrence and complete containment are often not possible. 8
Furthermore, various chemotherapeutic agents are supplied by topical, oral, or parenteral routes to eradicate malignant cells, particularly in the case of high-grade cancers. 9 The challenge of effectively treating potentially dangerous tumors in skin cancer persists despite the approval of numerous pharmaceuticals by the Food and Drug Administration (FDA), as their efficiency has been shown to be limited. One potential explanation for this phenomenon is the complex structure of both the skin layers and cancerous tissue that hinders the effective administration and penetration of medications into the skin or tumors. Consequently, this impedes the accumulation and sustained presence of pharmaceuticals at the desired site.9,10 Moreover, the lack of precise drug targeting and selectivity towards tumor-specific locations, the development of drug resistance, and limited bioavailability at the intended site of action contribute to significant adverse consequences and inadequate effectiveness as a remedy. 11 Therefore, numerous attempts are underway to find targeted and specialized medications. In the last few decades, nanomedicine has enhanced therapeutic tactics employing bio-distribution, bioavailability, stability, pharmaceutical kinetics, and specific delivery. These qualities prompted curiosity about employing nano-based techniques for managing skin cancer.12–14
Various drug delivery strategies have been created to enhance the efficacy and decrease the adverse outcomes of topical therapeutic pharmaceuticals.15,16 The intriguing solid fibers, known as NFs, possess sub-micrometric dimensions and attractive characteristics, including a high surface-to-volume ratio, significant porosity, appropriate mechanical strength, and ease of production.17,18 Due to its simplicity of use and ability to regulate the features of NFs, electrospinning is the most often used technique of producing NFs.19,20 Natural polymers, such as cellulose, CS, collagen, starch, and silk, or synthetic polymers like PVA, polycaprolactone (PCL), poly (lactic-co-glycolic acid), polyethylene oxide, polyvinylpyrrolidone, etc. are commonly utilized to create electrospun NFs.21,22 Hence, composite NFs with enhanced mechanical properties can be created by combining naturally occurring polymers with biodegradable synthetic ones.18,23 Of the interesting feature of NFs is their capacity to carry high loads of various therapeutic agents, preventing their degradation and enabling controlled release to achieve maximum combined therapeutic effects.
The anticancer drug 5-fluorouracil (5-FU) is widely used in chemotherapy for treating melanoma skin cancer. Both oral and intravenous methods of administration are available for this medication. However, conventional administration of 5-FU poses two notable challenges. Firstly, it leads to pronounced systemic side effects due to its cytotoxic impact on healthy cells. Secondly, its biological half-life is short because it is rapidly broken down by the enzyme dihydropyramidine dehydrogenase. Additionally, it is not completely or uniformly absorbed when taken orally. 24
Etoposide (ETP) is a semisynthetic derivative of podophyllotoxin. It has a lactone, which is a cyclic ester, and a glycosidic connection that connects a glucose molecule. The inclusion of the glucose moiety in the drug is crucial for enhancing its solubility and facilitating cellular penetration. 25 Nevertheless, the conventional formulation of ETP has many drawbacks, such as inadequate solubility, restricted bioavailability, and non-specific toxicity. 25
The incorporation of several therapeutic agents to selectively target tumor-promoting or cell-maintaining mechanisms is a critical strategy of tumor treatment.26,27 Combination therapies are increasingly popular in cancer treatment as they can synergize to combat cancer more effectively, reduce drug toxicity, and mitigate the development of multi-drug resistance through various mechanisms. The efficacy of this approach lies in its ability to simultaneously target multiple biological pathways.28–30 In this regard, employing nano-based techniques in delivering combinations of drugs to cancerous cells has emerged as a promising approach. Utilizing multiple chemotherapy drugs in a treatment regimen has been demonstrated to improve the effectiveness of cancer therapy while minimizing the adverse side effects commonly associated with single-drug use or monotherapy. This approach involves the sustained, controlled, and precise delivery of chemotherapy agents.31–33
several studies34,35 have investigated drug delivery approaches in the context of skin cancer. For instance, Fakhri et al. 34 reported Fe3O4-Ag2O quantum dots (QDs) incorporated into cellulose fibers as carriers for anticancer drugs. These nanocomposites exhibit remarkable targeting potential toward melanoma cells, along with enhanced scavenging properties. This unique approach highlights their potential as nanocandidates for future antitumor drug delivery systems in skin cancer. In another study Zhu et al. 35 developed core-shell NFs comprising a CS-loaded PCL shell and a 5-FU-loaded Poly(N-vinyl-2-pyrrolidone) (PVP) core for synergistic therapy in melanoma skin cancer. The NFs, with an average diameter of 503 nm, demonstrated high drug-encapsulating efficiency and favorable mechanical properties. Notably, the burst release of 5-FU effectively inhibited melanoma cells (B16F10 cells), while sustained CS release mitigated adverse effects on normal skin cells (L929 cells). These results suggest a promising strategy for cancer treatment with minimal impact on healthy cells.
The aim of our study was to develop nanofibrous scaffolds composed of PVA and CS co-loaded with both 5-FU and ETP, to enhance their cytotoxic effects on melanoma A375 cells. To our knowledge, this work has not been performed previously. We investigated key physicochemical characteristics of the NFs, including morphology, porosity, water absorption rate, and drug release profile. Additionally, in vitro experiments were conducted to assess lethality features and quantify apoptosis levels using flow cytometry.
Materials and methods
Materials
5-Fluorouracil (5-FU, MW: 130.1 g.mol−1) and etoposide (ETP, MW: 588.6 g.mol−1) were bought from Sigma-Aldrich (Germany). PVA (MW = 72,000 Da, Hy = 99.5%) and chitosan (CS, MW: 190,000 Da, deacetylation degree: 75%–85%) were acquired from Sigma-Aldrich (Germany). Acetic acid, and ethanol were acquired from Merck Company in Germany. The cell culture materials used in this study were obtained from Gibco (France). These materials included Dulbecco’s modified Eagle’s medium (DMEM) with high glucose, fetal bovine serum (FBS), 0.05% trypsin/EDTA, 3-(4,5-dimethylthiazol-2-yl-2,5-diphenyltetrazolium bromide) (MTT), and phosphate buffered saline (PBS). All other chemical solutions were of standard analytical quality.
Preparation of PVA/CS NFs loaded with 5-FU/ETP
The preparation PVA/CS NFs loaded with ETP and 5-FU was performed using the electrospinning machine of Fanavaran Nano Meghyas (Fanavaran Nano Meghyas Ltd., Co., Tehran, Iran). Initially, PVA/CS NFs were prepared to achieve the desired diameter and shape, after which both ETP and 5-FU drugs were loaded into the NFs. A combination of PVA (10% w/v dissolved in distilled water) and CS (2% w/v dissolved in 1% acetic acid) in a mixed polymer blend at a ratio of 80:20 v/v was used to identify the most suitable PVA/CS NFs. To load the drugs, ETP and 5-FU at a 50:50 ratio, with a concentration of 5 wt%, were introduced into a polymer solution of PVA/CS and then subjected to electrospinning device. The electrospinning parameters are available in Table 1. After preparing the desired thickness, the fiber mats were cross-linked with glutaraldehyde (GA) vapor and separated from the aluminum sheet. Subsequently, the mats were transferred to a vacuum desiccator and left there for 12 h at 25°C to remove of GA before being preserved for further analysis and investigation. It should be noted that GA is preferred due to its affordability, wide availability, high efficiency, high binding capacity, and easy operation. Although it is toxic in its liquid form, its toxicity decreases after crosslinking. During the crosslinking process, residual GA content can be minimized. Typically, polymer crosslinking with GA is carried out at a low concentration of both GA and polymer.36,37
Parameters used in the electrospinning method.
Characterization of the morphology and diameter
The diameter and surface morphology of the NFs were measured using scanning electron microscopy (SEM) at an accelerating voltage of 25.0 kV. The specific instrument used for this analysis was the HITACHI S-4700 (Japan). The dried scaffolds were collected and then subjected to a gold plasma sputter coating prior to imaging. The NFs’ diameter distributions and average diameter were determined using the Image J program applied to the SEM pictures.
Evaluations of water absorption, porosity, and wettability
The water absorption capacity of PVA/CS NFs and PVA/CS scaffolds loaded with 5-FU/ETP was determined. The scaffolds were accurately weighed as their dry weight and then submerged in distilled water with a pH of 7.4 at a temperature of 37°C. After immersion for 0.5, 1, 2, 3, 6, 12, 24, 48, and 72 h, the samples were removed from the medium and put on filter paper to remove any residual water. Afterward, the scaffolds were measured for their wet weight. The water absorption percentage (W) of the scaffolds was determined by using the provided Equation (1).
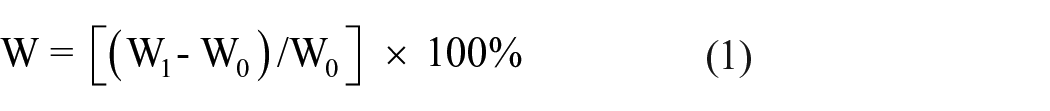
W0 and W1 represent the weights of dry and wet scaffolds, respectively.
Additionally, the liquid displacement technique was used to determine the porosity of both the free PVA/CS and the 5-FU/ETP loaded PVA/CS scaffolds. 38 The scaffolds were immersed in pure ethanol for 1 h, following which the porosity was determined using the following Equation (2):
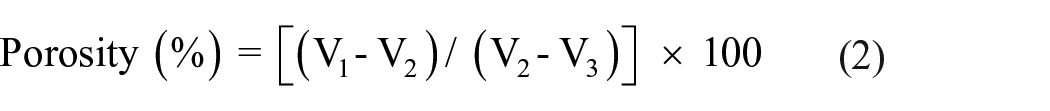
V1 represents the starting volume of ethanol, V2 represents the volume after immersion, and V3 represents the volume of the ethanol after removing the mat after 1 h.
The wettability properties of PVA/CS scaffolds, both unloaded and loaded with 5-FU/ETP, were assessed using contact angle measuring equipment. About 4 µL of distilled water was pipetted onto the surface of each sample, and photographs were taken of the droplets over time at 25°C.
FTIR spectroscopy
The 5-FU/ETP loading and the interaction between the fiber elements was examined using Fourier transform infrared (FTIR) spectroscopy. The spectroscopic evaluation was conducted using a Shimadzu IRAffinity-1S spectrophotometer, covering a wavelength range of 400–4000 cm−1 at 25°C.
Mechanical properties
The mechanical properties of the PVA/CS scaffold and the 5-FU/ETP loaded PVA/CS scaffold were assessed using a Universal Testing Machine (Santam, Iran). The scaffolds were prepared for testing by cutting them into dimensions of 3 cm × 0.5 cm. They were then securely fastened in the grips and subjected to stretching at a speed of 2 mm/min using a load cell with a capacity of 10 N. A slower rate (e.g., 2 mm/min) enables accurate measurement of material properties, striking a balance between precision and efficiency. This loading rate is commonly employed in mechanical investigation of nanofiber properties.39–42 The mechanical parameters were measured three times for each sample, and the stress-strain curves were documented.
In vitro drug release
The whole immersion approach was utilized to determine the release profile of encapsulated 5-FU and ETP from the prepared 5-FU/ETP nanofibrous scaffolds. 43 The scaffolds were cut into a specified form (5 cm × 4 cm) and submerged in a beaker filled with 100 mL of PBS with 0.2% tween 80 (pH 7), then placed in an orbital shaker at 100 rpm and the temperature stayed at 37 ± 0.5°C. At predefined time intervals (1, 3, 6, 12, 24, 36, 48, and 72 h), 1 mL of the release medium was taken from the beaker and replenished with fresh PBS. 44 The absorbance of the sample was determined at 266 nm (λmax) for 5-FU and at 283 nm (λmax) for ETP by UV-Visible spectroscopy by using a quartz cuvette. 45 A graph depicting the percentage of drug release over time was created for each medium.
Cytotoxicity study on melanoma cell
The A375 human melanoma cells were supplied by the Iranian Biological Resource Center. The cell line was cultured in 25 cm² tissue culture flasks and maintained in DMEM medium supplemented with 10% FBS, 1% penicillin, streptomycin, and amphotericin B antibiotics. The cells were cultured in an incubator containing 5% carbon dioxide. The cytotoxicity effect of samples on A375 cells was assessed using a direct method called the MTT colorimetric test. In the MTT cell viability experiment, A375 cells were seeded into a 96-well plate at a density of 10,000 cells per well. The cells were then incubated for 24 h to facilitate cell attachment. After a sterilization process using UV light for a duration of 2 h, the prepared scaffolds including PVA/CS NFs, 5-FU loaded PVA/CS NFs, ETP loaded PVA/CS NFs, 5-FU/ETP loaded PVA/CS NFs, free 5-FU, free ETP and the combination of both were added to the wells and incubated for 24 and 48 h. Following the incubation times, the cells were rinsed with the PBS (pH 7.4) and then exposed to MTT solution (5 mg/mL) for a duration of 3 h. Next, the MTT was removed and the resulting formazan crystals were dissolved in 100 μL of dimethyl sulfoxide. The absorbance was read using the ELISA plate reader at a wavelength of 570 nm. Cell cytotoxicity was calculated using the following Equation (3). For each experiment, three identical samples were assessed. 46 .

Measuring the cell apoptosis by flow cytometry method
The quantification of apoptosis was conducted using Annexin V-FITC and propidium iodide (PI) dyes for staining (Apoptosis Detection Kit, Multi Sciences, Hangzhou, China). The formulations included PVA/CS NFs, 5-FU loaded PVA/CS NFs, ETP loaded PVA/CS NFs, 5-FU/ETP loaded PVA/NFs, free 5-FU, free ETP, and their combination. The scaffolds were disinfected with UV light for 2 h. A375 skin melanoma cells (3 × 105) were cultured in each 12-well plate well and treated with the samples for 24 h. After PBS washing, trypsinization was performed. The resulting mixture was centrifuged at 1200 rpm for 2 min, cells were suspended in PBS, then mixed in binding buffer and stained with Annexin V-FITC and PI dyes for 20 min. The labeled cells were analyzed using flow cytometry with the Calibur BDFACS instrument.
Analytical statistics
The data was analyzed using Graph Pad Prism 9. The statistics were provided as mean ± SD. ANOVA and Tukey tests were used to examine variations between groups. Variations with a p-value less than 0.05 were considered statistically significant.
Results and discussion
In this study, we produced nanofibrous scaffolds composed of PVA/CS, which were loaded with both 5-FU and ETP using the electrospinning technique. The goal was to create a localized co-delivery system for treating skin melanoma. The premise was that developing suitable nanofibrous mats containing 5-FU and ETP could potentially improve the management of skin tumors while reducing the adverse effects associated with systemic chemotherapy. To this end, electrospun PVA/CS NFs were first prepared and subsequently loaded with 5-FU and ETP. The resulting scaffold underwent morphological characterization and an assessment of its anti-melanoma efficacy.
Scaffolds’ characteristics of morphology
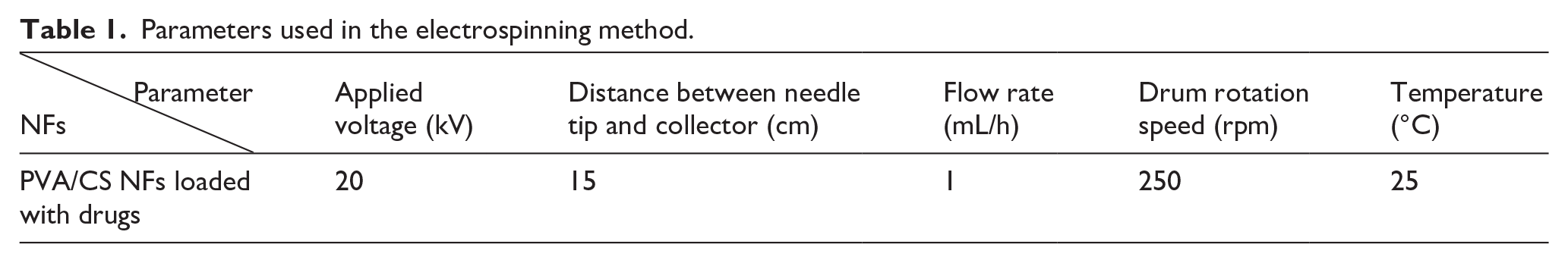
Figure 1 displays the SEM images and size distribution histograms of four types of scaffolds: PVA/CS scaffolds, PVA/CS with 10 wt% 5-FU, PVA/CS with 10% ETP and PVA/CS scaffolds with 10 wt% 5-FU/ETP. The free scaffold had a consistent morphological feature in terms of their diameter, sleek surface, and absence of beads, along with a porous structure. Furthermore, the mean diameter of PVA/CS NFs was determined to be 201 ± 37 nm, as seen in Figure 1(a). By adding 10 wt% of 5-FU, the NFs were made that had a consistent structure and an average diameter of 246 ± 44 nm (Figure 1(b)). Furthermore, the SEM image revealed that incorporating 10 wt% of ETP resulted in NFs that exhibited a consistent shape, with a diameter distribution of 260 ± 48 nm (Figure 1(c)). The 5-FU/ETP loaded PVA/CS scaffold exhibits a non-bead, homogenous, and uniform structure, indicating successful incorporation of 5-FU and ETP into the scaffold. It was observed that the addition of 10 wt% of 5-FU/ETP to PVA/CS resulted in an increase in the average diameter of the NFs to 321 ± 61 nm (Figure 1(d)). The observed increase was considered insignificant and attributed to an elevation in the solution’s viscosity. As depicted in Figure 1(b), the incorporation of 10 wt% 5-FU and ETP had no discernible effect on the morphology of the NFs, which remained smooth and bead-free. Alavarse et al. 47 reported that CS can be used as a thickener for electrospinning PVA solutions to improve fiber uniformity. Lv et al. 48 also demonstrated that a polymeric mixture of CS and PVA possessed excellent electrospinnability.
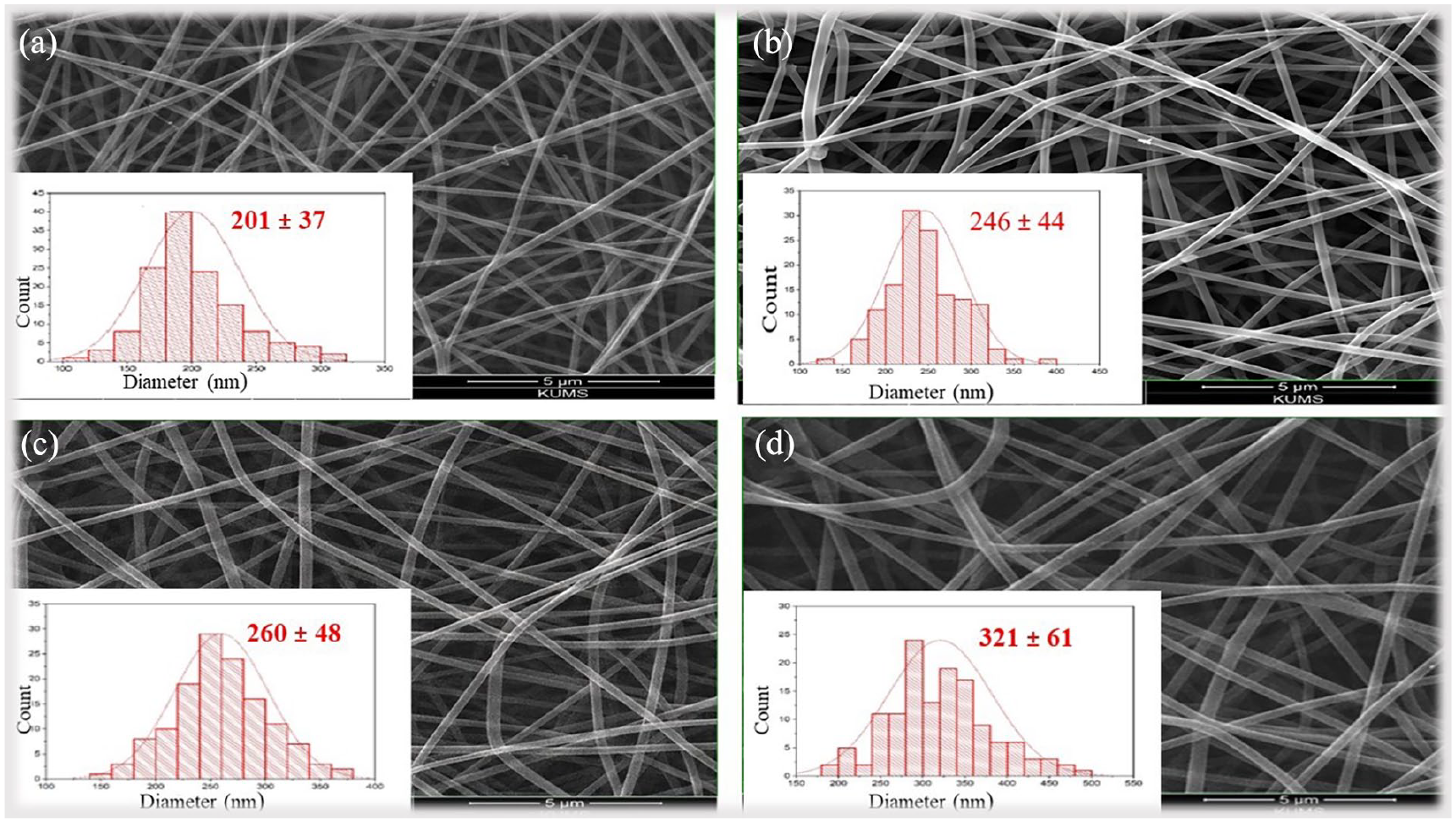
SEM micrograph of electrospun PVA/CS NFs (a), and the NFs incorporated with 10 wt% 5-FU (b), 10 wt% ETP (c) and 10 wt% 5-FU/ETP (50:50) (d) accompanied by the size distribution histograms.
Water absorption, porosity, and wettability
The water absorption capacity of the scaffolds determined through the immersion approach is shown in Figure 2. During the 30-min immersion, the free PVA/CS scaffold and the 5-FU/ETP loaded scaffold exhibited an average water absorption percentage of 206% and 238%, respectively. After 72 h of immersion, the water uptake percentages increased to around 640% and 680% for the two scaffolds, respectively. There was no notable disparity between the two scaffolds, indicating that the addition of 5-FU/ETP did not affect the water absorption capacity of the PVA/CS NF. In addition, these findings show that the scaffolds can quickly absorb water. This might be because they are porous and have hydrophilic functional groups, like hydroxyl and amino groups in CS and PVA chains. 49 The water absorption capacity of 5-FU/ETP PVA/CS scaffold was found to be greater than that of free PVA/CS NFs. This suggests that the high swelling ratio in the 5-FU/ETP loaded PVA/CS NFs is attributed to the formation of an enhanced flexible network due to intra-polymer chain reactions, increased flexibility, and the presence of additional hydrophilic structures.50,51 Abdulkhani et al. 52 studied water absorption properties of cellulose/PVA films. They found that cellulose/PVA films had higher water absorption (73%–78%) compared to cellulosic films (33%–42%). The presence of hydroxyl groups in PVA contributes to its water absorption capacity. Chen et al. 53 reviewed stimuli-responsive electrospun NFs for drug delivery. Electrospun NFs, due to their high specific surface area and porosity, are ideal for drug delivery systems. They can efficiently load drugs and respond to specific conditions for controlled release.
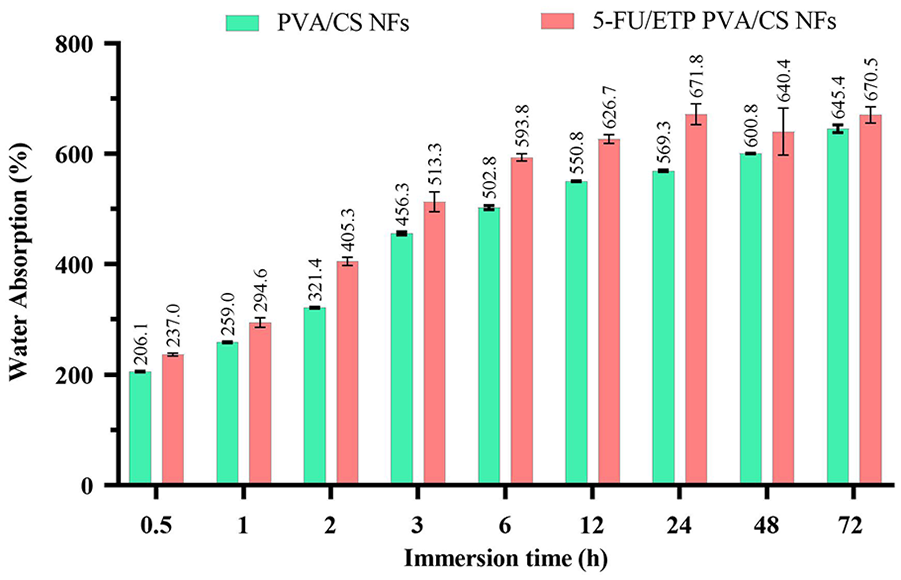
Water absorption capacity of free PVA/CS and 5-FU/ETP loaded nanofibrous scaffolds.
Furthermore, the scaffold’s porosity plays a crucial role in facilitating cell proliferation on the scaffold and its subsequent degradation.54,55 Porosity analysis indicated that the PVA/CS scaffold without drug loading (free PVA/CS) had a porosity of 91.7% ± 4.9% of the total scaffold volume, while those loaded with 5-FU and ETP exhibited a slightly lower porosity of 87.9% ± 4.7% (Figure 3(a)). The drug loading resulted in a decrease in porosity; however, the percentage increase was not statistically significant (p > 0.05). Overall, the reduction in porosity of PVA/CS NFs due to drug loading depends on factors such as the drug’s solubility and release behavior. 56 Zolghadri et al. 57 reported that the porosity typically ranges from 75% to 82% in PVA/CS scaffolds. These highly porous structures allow for cell attachment and nutrient exchange, making them suitable for tissue engineering applications. The results suggest that the scaffold mat has a significant amount of pores, which allows for efficient flow of nutrients and gases and absorbing fluids.54,58
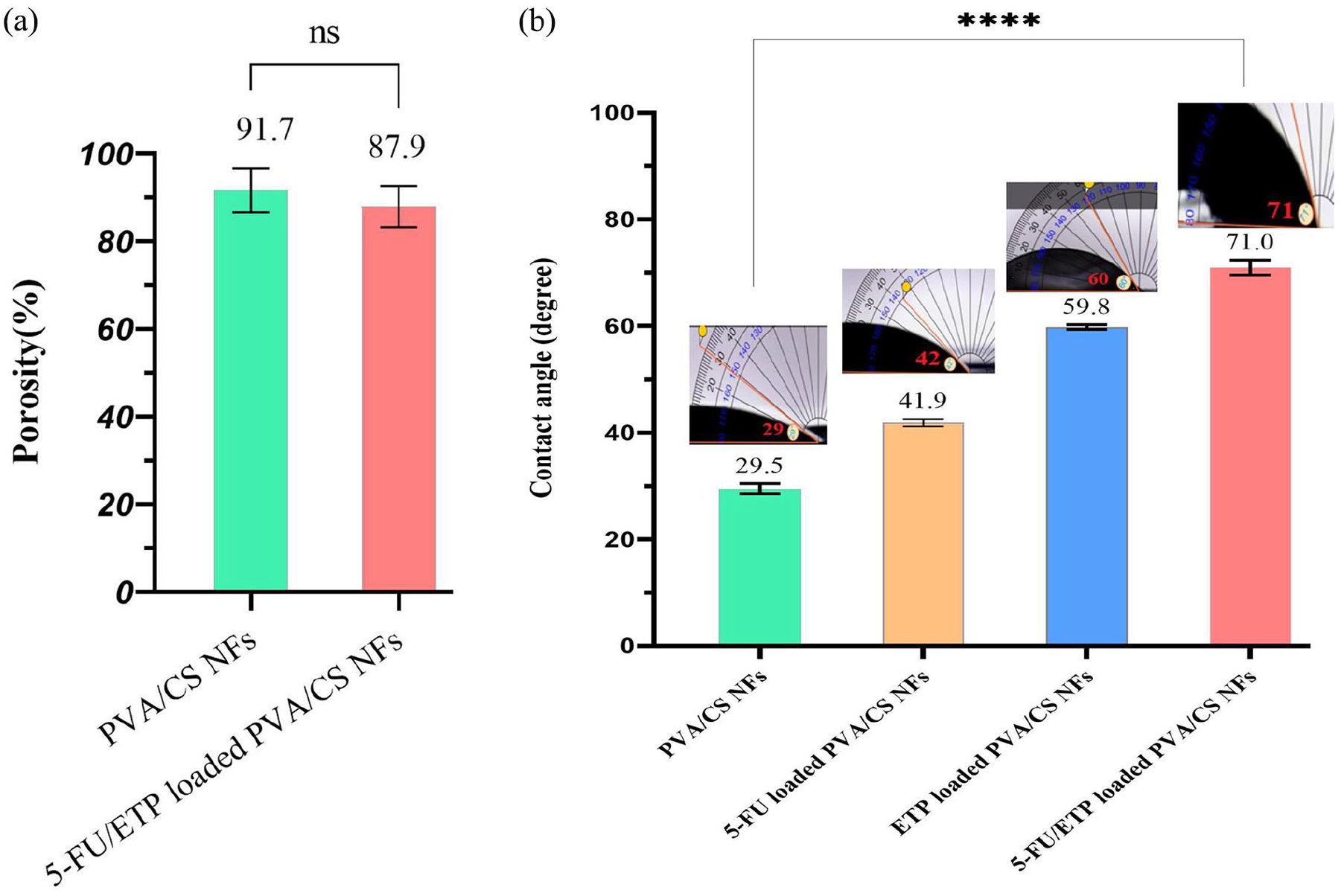
Porosity diagram (a) and water contact angle (b) for fabricated NFs.
The water contact angle was used to assess the hydrophilicity of the scaffold surface. This property may impact the maintenance of moisture levels as well as the adhesion and proliferation of cells on the scaffolds.55,59 Consequently, the hydrophilicity of the scaffolds was quantified. The average contact angles for free PVA/CS NFs, PVA/CS NFs loaded with 5-FU, PVA/CS NFs loaded with ETP, and 5-FU/ETP loaded PVA/CS NFs were approximately 29°, 42°, 60°, and 71°, respectively (Figure 3(b)). The results revealed a highly hydrophilic and wettable surface for the mats. In addition, the incorporation of 5-FU and ETP into the PVA/CS scaffold led to a slight increase in the contact angle. In a research, 60 electrospun PVA/CS nanofibrous mats showed a mean contact angle of 57.8° for PCL-PVA and 68.3° for PVA-PCL-CS. These values demonstrate the varying hydrophilicity of different polymer blends and their potential impact on drug delivery and tissue regeneration.
FTIR studies
FTIR spectroscopy was used to examine the functional groups and interactions between the components of NFs and to verify the existence of pharmaceuticals in the resulting NFs. The spectra that were obtained are shown in Figure 4. The PVA/CS NF spectrum exhibited peaks at 1088 cm−1 and 1258 cm−1, corresponding to the stretching of ether groups (C-O-C). Additionally, a peak at 1746 cm−1 was observed, indicating the elongation of the carbonyl group (C=O) in the remaining vinyl acetate repeat units of PVA and the remaining acetyl groups in CS. 61 The -CH2 group induces both asymmetrical and symmetrical elongation vibrations, which contribute to the absorbing pattern at 2946 cm−1.61,62 The elongation of OH bonds in PVA and CS is associated with a broad absorption band at 3325 cm−1, which overlaps with the vibrating of NH2 groups in CS primary amines.61,63
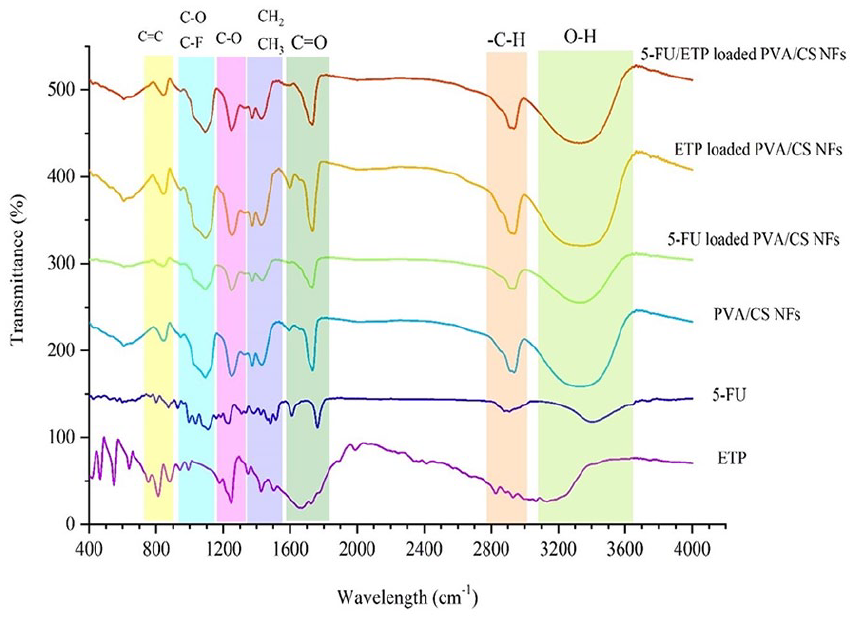
FTIR spectrum of free drugs, synthetic free NFs, and NFs loaded with drugs simultaneously and independently.
The ETP spectrum exhibited distinct bands corresponding to specific molecular vibrations. These included the carbonyl stretch vibration of the lactone ring at 1730 cm−1, the OH stretch vibration of the phenolic and sugar OH groups at 3200 cm−1, the aromatic band at 1600 cm−1, and the C-O stretch vibration at 1260 cm−1. 64 Moreover, 5-FU is composed of functional groups such as C=O bonds, aromatic rings (C=C), and C-F bonds, which are apparent in the absorption spectra at wavelengths of 1760 cm−1, 860 cm−1, and 1100 cm−1, respectively. 65 The spectra of ETP loaded PVA/CS NFs and 5-FU loaded PVA/CS NFs exhibit distinct peaks associated with 5-FU and ETP, respectively, confirming the successful loading of the drugs. The spectrum of the 5-FU/ETP loaded PVA/CS NFs reveals the presence of both drugs in the NF structure. This is evident from the unique peaks of 5-FU and ETP drugs that are seen in the prepared NFs. Additionally, the typical bands of PVA and CS polymers are clearly demonstrated. Nevertheless, several peaks experienced a minor displacement towards a higher or lower wavelength and/or were superimposed as a result of physical reactions between molecules.
Mechanical results
The scaffolds should possess mechanical qualities, particularly tensile strength, that are sufficient to maintain structural integrity throughout use. 66 Figure 5 displays the stress-strain curves of the scaffolds. The PVA/CS scaffold had an average tensile modulus of 6.06 MPa, an ultimate tensile stress of 2.65 MPa, and an ultimate tensile strain of 43.9%. In contrast, the 5-FU/ETP loaded PVA/CS scaffold had a tensile modulus of 2.11 MPa, an ultimate tensile stress of 3.68 MPa, and an ultimate tensile strain of 79.6%. The presence of 5-FU and ETP in PVA/CS scaffold resulted in a substantial increase in the ultimate tensile strain, which proved to be suitable for the intended purpose. The incorporation of 5-FU and ETP into PVA/CS NFs leads to an enhancement in the tensile characteristics of the NFs. The ultimate tensile strain of the PVA/CS NFs loaded with ETP indicated a slight decrease; however, it still exhibited a suitable tensile strain for the intended use. The decrease in value may be attributed to a disruption in the polymeric structure inside the scaffold, resulting in a reduction in the interlocking and flexibility between the PVA and CS chains. It could also be brought on by a rise in the hydrogen bonding between the NH3+ groups of CS, the functional groups of ETP and the OH− groups of PVA.67,68 Introducing drugs may alter the physical and chemical characteristics of NFs, resulting in modifications to their mechanical properties. 69 Drug substances have the potential to interact with the polymer matrix of NFs, resulting in the creation of more robust connections and enhanced mechanical resilience. 70 Therefore, the results suggest that the fabricated scaffolds demonstrate excellent durability and flexibility across a range of tensile properties. 71
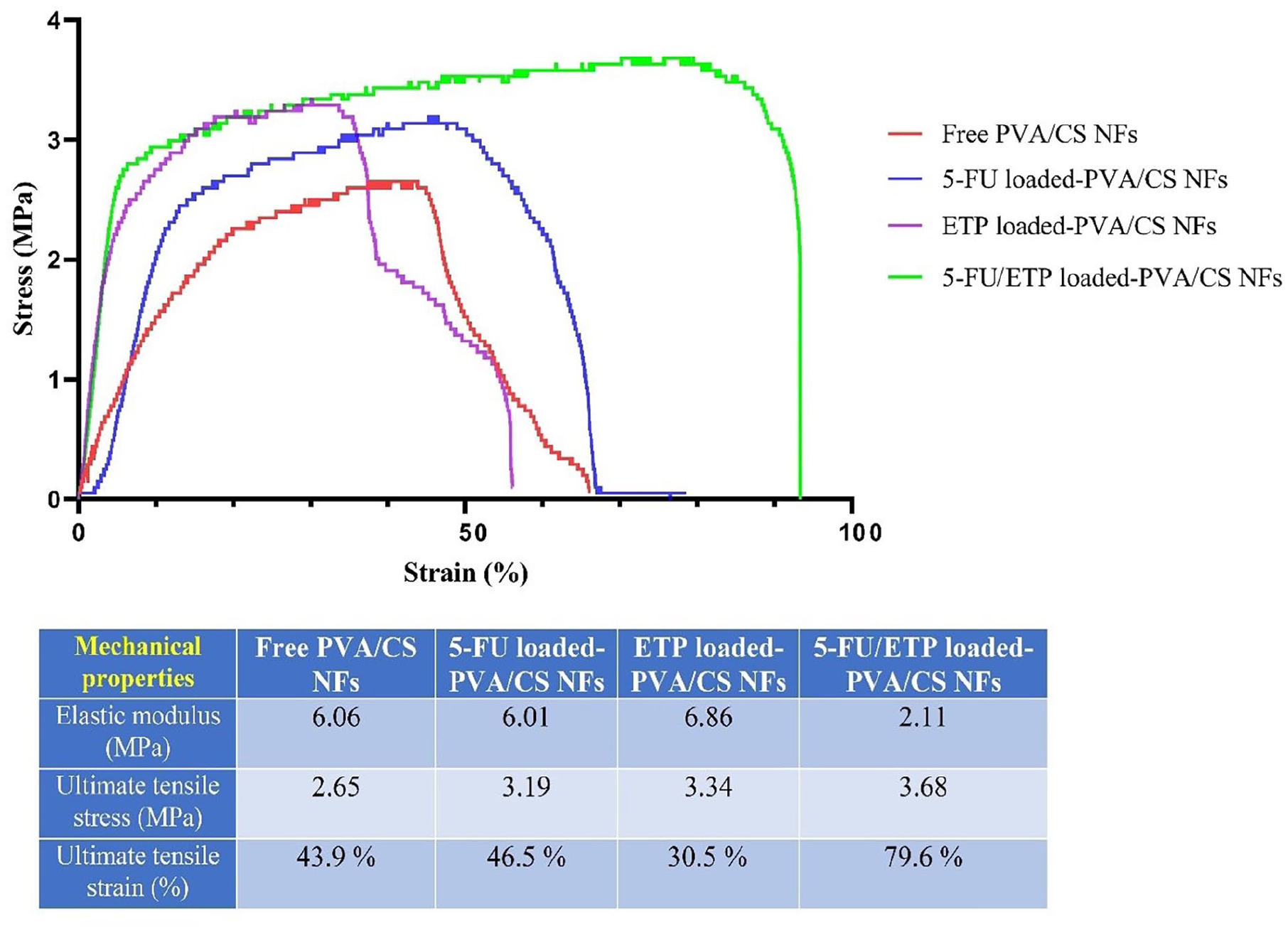
Strain stress diagram for free PVA/CS NF, 5-FU loaded PVA/CS NF, ETP loaded PVA/CS NF, and 5-FU/ETP loaded PVA/CS NF.
In vitro drug release studies
Figure 6 illustrates the release pattern of the drugs from 5-FU/ETP loaded PVA/CS NFs in a laboratory setting over a period of 72 h. Upon observation, it was noted that the release of the drugs depends on time. During 6 h, about 40.9% ± 6.1% of 5-FU and 23.9% ± 4.1% of ETP were released. After 24 h, the quantity released of 5-FU and ETP was 78.1% ± 3.7% and 63.2% ± 3.7%, respectively. Following a 72-h period, about 94.5% ± 1.9% and 87.4% ± 2.6% of 5-FU and ETP, respectively, were released from PVA/CS NFs loaded with 5-FU/ETP. The pattern of release of 5-FU and ETP from 5-FU/ETP loaded PVA/CS NFs indicated rapid release during the first several hours. The rapid release of the NFs can likely be attributed to their small size. After the initial burst phase, a slower release, similar to the delay phase, was observed. At this point, polymer degradation has a greater impact on the release process than NF degradation. 72 ETP exhibited a reduced rate of release from the matrix due to its limited solubility in water. As a result, the drug release profile from the NFs demonstrated that the pharmaceuticals were sustainably and continuously released from the polymer matrix for 72 h.
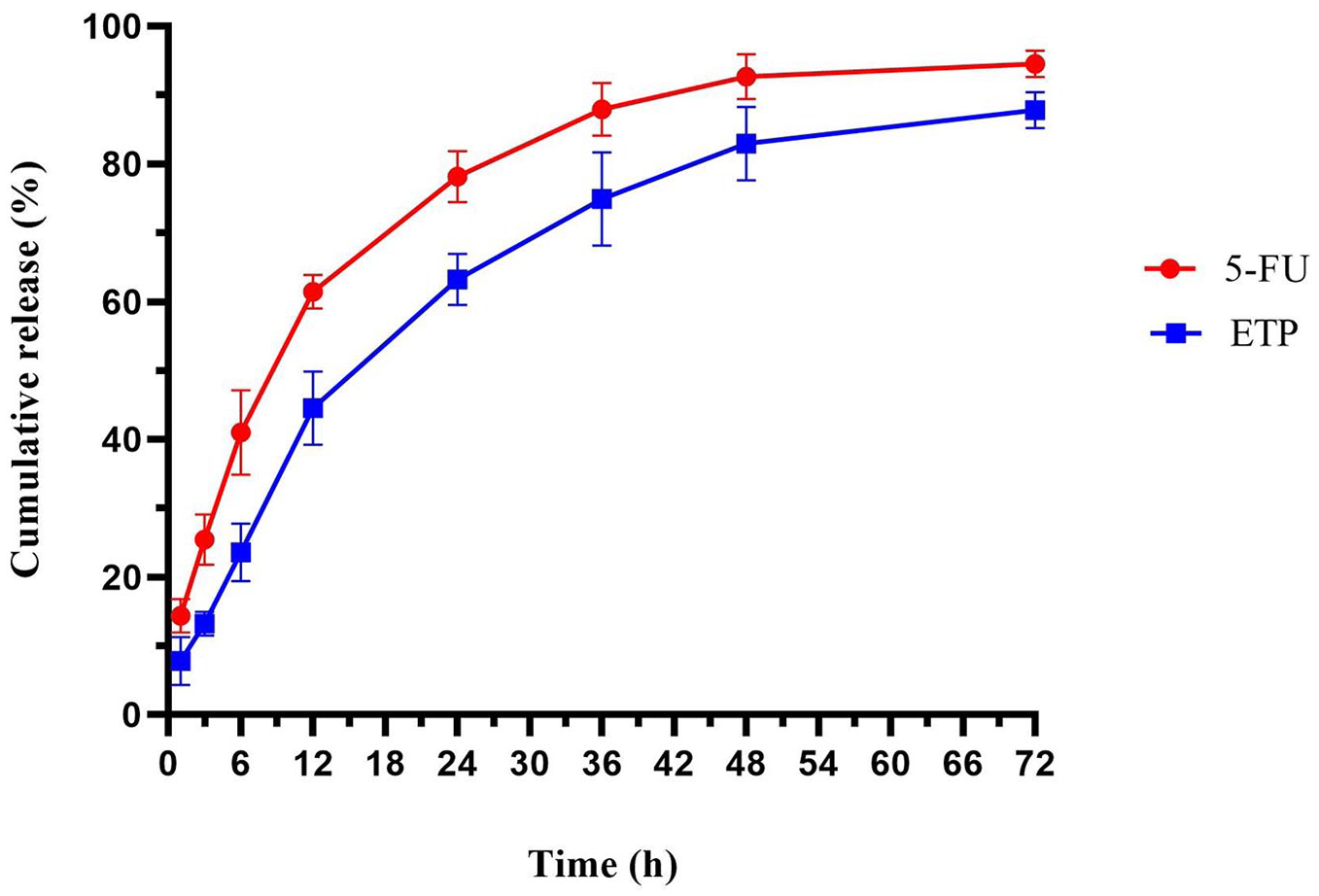
Cumulative release percentage of 5-FU and ETP released from 5-FU/ETP loaded PVA/CS NFs.
In vitro cytotoxicity studies
After exposing A375 cells to the scaffolds, cytotoxicity was evaluated using the MTT colorimetric method. As part of comparative therapies, we investigated the use of free drugs both individually and in combination. Consequently, the therapy, including 5-FU and ETP pharmaceuticals, resulted in the death of cancer cells. In contrast, the lethality of the groups treated with NFs loaded with ETP and 5-FU separately was greater than that of the free form Yadav et al.73,74 The cytotoxicity of treatments against A375 cells was about 4.9%, 51.5%, 57.5%, 11.6%, 36%, 58.5%, 71%, after 24 h of incubation, and after 48 h of incubation was 52%, 60.5%, 81.8%, 11.8%, 79%, 74.5%, 95%, for free 5-FU, free ETP, free 5-FU/ETP, PVA/CS NFs, 5-FU PVA/CS NFs, ETP PVA/CS NFs and 5-FU/ETP PVA/CS NFs, respectively (Figure 7). Cell viability decreased with increasing exposure time to free compounds and NFs from 24 to 48 h, and in other words, toxicity increased. Regarding the safety and biocompatibility of PVA/CS polymers, the cytotoxic effects of PVA/CS NFs were found to be insignificant after 24 and 48 h. 5-FU/ETP loaded PVA/CS NFs exhibited the highest death rate on A375 melanoma cancer cells compared to all other groups at both 24 and 48 h. This can be attributed to the synergistic anticancer effects, reduced drug-related toxicity, and stop multidrug resistance.
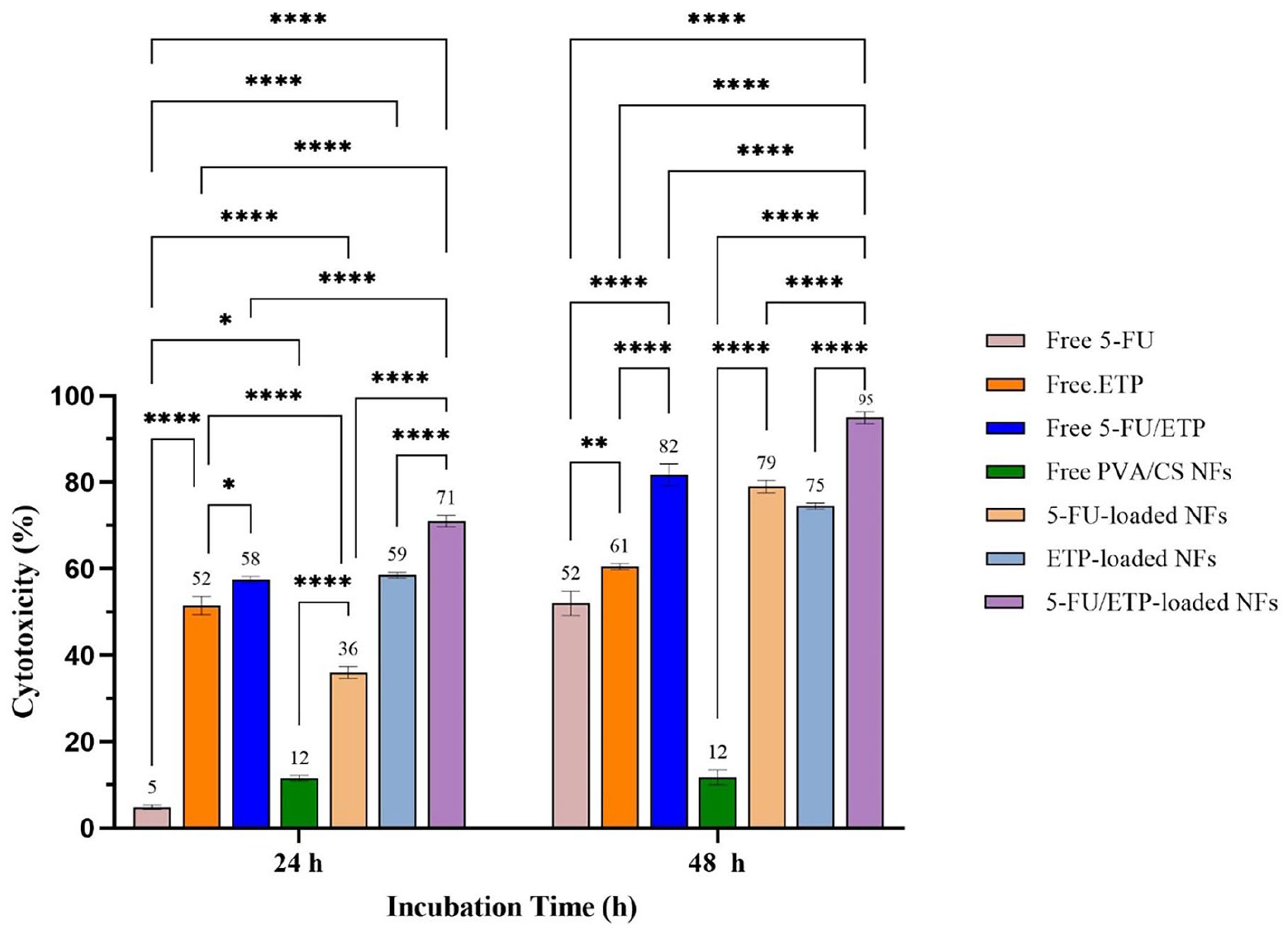
Cytotoxicity diagram against A375 melanoma cells after treatment with different formulations for 24 and 48 h.
Apoptosis assessment
The results of apoptosis assessment are represented as the scatter plots in Figure 8. The apoptosis of A375 melanoma cells induced by 5-FU/ETP loaded NFs was 74.4%, which was the highest among all groups after 24 h of incubation. Free 5-FU and free ETP resulted in apoptosis rates of 37.6% and 41.3%, respectively. The combination of the two drugs resulted in an apoptosis rate of 61.6%, which was higher than that caused by free 5-FU and free ETP. The value of apoptosis for 5-FU loaded PVA/CS NFs and ETP loaded PVA/CS NFs was 62.7%, 67.2%, and 74.4%, respectively. The quantity of apoptosis induced by PVA/CS NF was significantly lower, which is expected due to the safety and biocompatibility of these polymers. 5-FU loaded PVA/CS NFs and ETP loaded PVA/CS NFs induced more apoptosis compared to these drugs when used in their free form. The results clearly showed that the co-delivery of the drugs increased the rate of apoptosis compared to treatment alone, possibly due to the concurrent induction of several pathways of apoptosis.
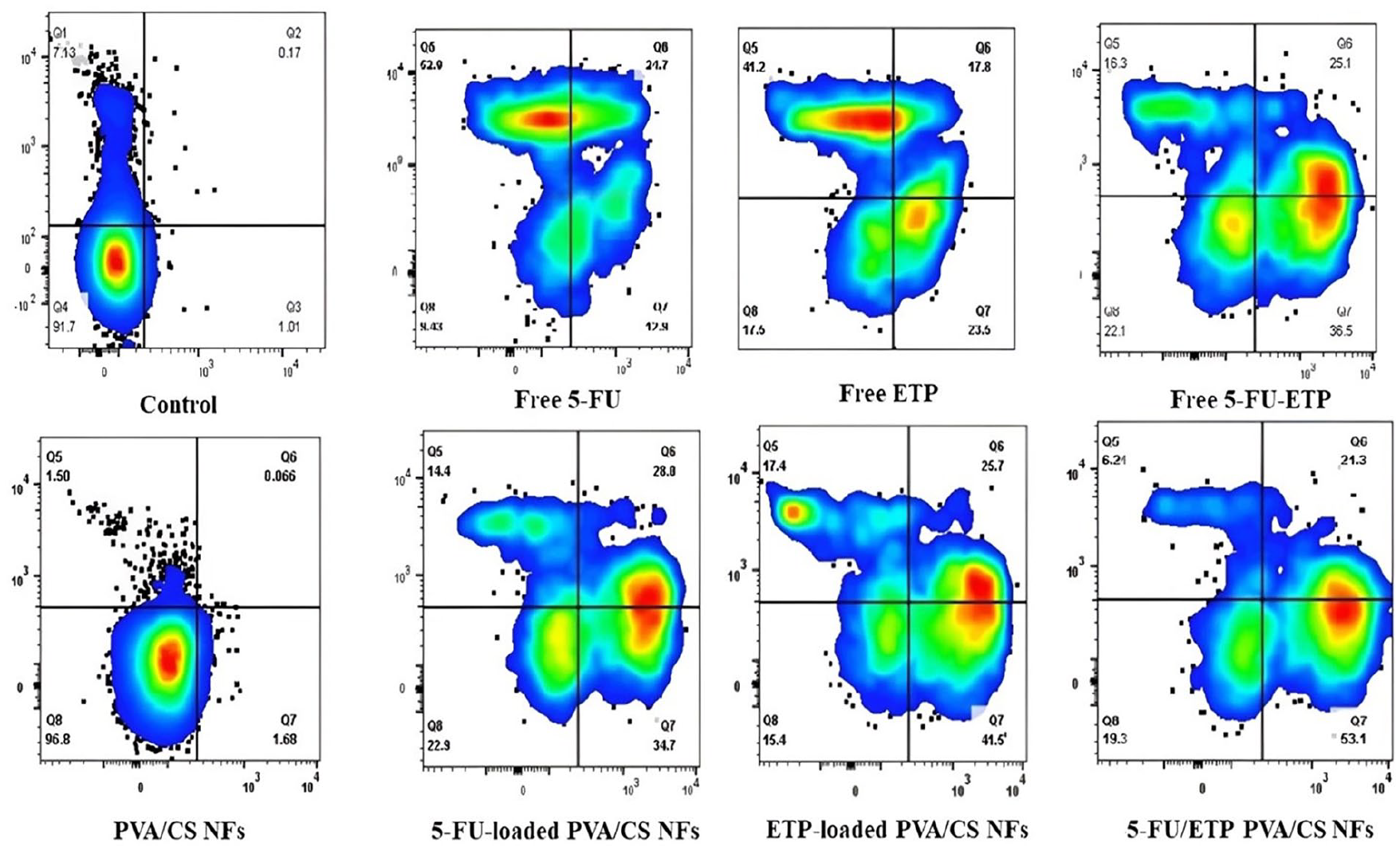
Flow cytometry analysis on apoptosis of A375 melanoma cells after treatment with various formulations for 24 h.
Conclusion
Our study successfully developed an electrospun nanofibrous scaffold incorporating 5-FU and ETP with an average diameter of 321 nm for chemotherapy in melanoma cutaneous cancer. The scaffold, composed of polyvinyl PVA and CS, demonstrated favorable physicochemical properties, sustained drug release, and significant cytotoxicity (95%) and apoptosis (74%) against melanoma A375 cells. This 5-FU/ETP loaded PVA/CS nanofibrous scaffold holds promise as a valuable system for localized eradication of cutaneous melanoma tumors while minimizing adverse drug reactions associated with chemotherapy. However, optimizing this formulation as well as a degradation study requires additional research. Specifically, investigating the impact of the crosslinking agent (GA) and non-toxic crosslinkers on 5-FU and ETP is crucial. It should be also investigated whether their properties change after crosslinking. Furthermore, we recommend further studies on these formulations, focusing particularly on normal cells such as fibroblasts and endothelial cells. Additionally, a comparative analysis of our NFs before and after crosslinking will provide valuable insights into the impact of crosslinking on the porous spaces within the NFs.
Footnotes
Acknowledgements
We would like to express my sincere appreciation and gratitude to Islamic Azad University, Tehran Medical Branch and Kermanshah University of Medical Sciences for their invaluable support.
Contributorship
Shirin Shojaei: Investigation, Methodology, visualization. Mahtab Doostan: resources, Writing – original draft. Hamidreza Mohammadi Motlagh: methodology, writing – review and editing. Seyedeh Sara Esnaashari: Conceptualization, Funding acquisition, Supervision, Project administration, Writing – review and editing. Hassan Maleki: Validation, Conceptualization, Supervision, Project administration, Writing – review and editing. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Islamic Azad University, Tehran Medical Branch, Grant No. 162597464.
Guarantor
HM.
