Abstract
Background:
Bioactive cerium-doped (Ce-BGs) glasses with proven antioxidant properties, which may reduce post-implant oxidative stress, were studied in vitro and in vivo to evaluate their application in bone regeneration. Based on the Kokubo (K) composition, they contain 3.6 and 5.3 mol% cerium (referred to as K3.6 and K5.3, respectively).
Methods:
Ce-BGs were synthesized by melting and sieved to produce granules (size range = 200–500 µm). In vitro studies were conducted against MLO-Y4 cells using direct Neutral Red (NR) and indirect Bromo-2-deoxyUridine (BrdU) assays to assess cell viability and proliferation respectively. In vivo studies were carried out using a New Zealand white rabbit model to evaluate bone healing potential.
Results and discussion:
NR results showed a significant increase in cell viability for Ce-BGs: 77% for K and 79 and 85% for K3.6 and K5.3, after 24 h. After 72 h, cell viability decreased for K to 58% and increased for K3.6 and K5.3 (76% and 116% respectively). Cerium inhibits cell proliferation in BrdU assay as explainable by the increased durability of Ce-BGs. In vivo studies, after 30 and 60 days, revealed a delayed degradation for Ce-BGs that can stimulate the osteo-regeneration without inflammatory or degenerative effects. Moreover, the new bone area (NBA) was higher for Ce-BGs compared to control; after 60 days 32% for K5.3 versus 21% for K.
Conclusions:
Ce-BGs granules show improved direct cytocompatibility in vitro and enhance the long-term bone remodeling process in vivo, contributing to a more controlled and effective bone healing compared to the K granules. This improved behavior can be linked to the antioxidant and anti-inflammatory properties of cerium, that can assist bone regeneration and reduce implant-associated inflammation, and to their slower dissolution rate that supports the controlled release of ions. These results suggest Ce-BGs as a promising device for therapeutic applications on hard tissues.
Introduction
Bioactive glasses (BGs) are biomaterials capable of stimulating a favorable biological response by binding to the host body tissues. 1 Due to their ability to bind quickly to the bone, stimulating osteoblastic proliferation and bone matrix neo-apposition, 2 BGs are used in the regenerative treatment of bone defects resulting from oncological resection, fractures, osteoporosis or severe atrophy of the jaws. 3
Two mechanisms underpin the bioactivity of BGs: binding to the host bone and osteoconduction, 4 a crucial step in the bone healing process. The extraordinary ability of BGs to bind the bone is explained by the formation on its surface of a hydroxyapatite (HA) layer structurally similar to the natural bone mineral matrix after the initial dissolution. HA then interacts with collagen fibrils to integrate with the bone tissue. 5 Of interest, some BGs have been shown to bind to soft connective tissues as well. 6 Moreover, BGs present hold osteoinductive properties which depend on the release of their constituent ions such as silicon, phosphate and calcium. This process stimulates osteoblasts adhesion, proliferation and activity and is a crucial phase of the regenerative process.7 –9 In addition, in vitro tests have shown that the ions release during glass degradation in a simulated body fluid (SBF) promotes neoangiogenesis.10,11 45S5Bioglass®, a biodegradable glass of the quaternary system Na2O-CaO-SiO2-P2O5, was discovered by Hench in 1969. 12 Later, Kokubo et al.13,14 developed phosphate-free glass comparable to 45S5Bioglass® in terms of bioactivity, proving that an adequate compositions of the Na2O-CaO-SiO2 ternary system is sufficient for formulation of biologically active glasses and that P2O5 is not essential to this end.
Given the diffusion of musculoskeletal pathologies that require bone grafting or prosthetic surgery, 15 BGs have been optimized in their chemical composition to improve their characteristics. Therapeutics inorganic ions (TIIs) have been added to BGs to obtain specific biological effects, for example increased osteogenesis and angiogenesis, and antibacterial or antioxidant activity.8,16,17 These TIIs include cerium, 18 zinc, 19 copper, 20 gallium, 21 magnesium, and strontium. 22 Cerium in particular presents multiple pharmacological properties, such as antiemetic, bacteriostatic and potentially antitumor activities. 18 Furthermore, the Ce3+/Ce4+ redox couple has a remarkable enzyme-mimetic properties and cerium is able to recapitulate the activity of enzymes such as superoxide dismutase (SOD) and catalase (CAT) in the scavenging of reactive oxygen species. 18 These antioxidant properties can be effectively exploited to reduce post-implantation oxidative stress and accelerate patient convalescence. Moreover, the osteogenic properties of cerium compounds are well-known and linked to the ability of cerium to activate specific cellular pathways such as tumor necrosis factor (TNF) and sucrose nonfermentable (SNF). 18
We previously profiled the bioactivity and cytocompatibility of cerium-doped BGs (Ce-BGs with cerium at 1.2, 3.6, and 5.3 mol%) and based on Kokubo composition. 23 In this study, we demonstrated that Ce-BGs disks (1 mm of thickness and a surface of about 2 cm2) with higher amounts of cerium (3.6 and 5.3 mol%) preserved bioactivity and improved cytocompatibility with respect to non-doped BGs. Here we present the in vivo evaluation of those Ce-BGs in the form of granules, underpinned by the corresponding cytocompatibility studies.
Compared to disks, granules possess greater reactivity due to a greater exposed surface area, and they can also be purposed as injectable materials, which could simplify the regenerative treatment of small bone defects, particularly in anatomically complex regions such as the craniofacial area.
This study is the first to critically evaluate the performance of the Ce-BGs in vivo, providing valuable insights for their progress toward clinical applications.
Materials and methods
Synthesis of Ce-BGs granules
The molar compositions of the Ce-BGs granules and their corresponding abbreviations are reported in Table 1. The parent glass is the phosphate-free glass by Kokubo et al., 14 named K, and was used as a reference to compare the behavior of Ce-BGs. The glasses were prepared by the melting method 24 and from a mixture of powders precursors (from Carlo Erba, Milan, Italy): SiO2, Na2CO3, CaCO3, and CeO2 in about total 100 g. The powders were melted in a platinum crucible at 1350°C for 2 h by using 2 heating rates: 5°C/min in the range 20°C–1000°C and 15°C/min above 1000°C. The melt was then quenched on a graphite plate mold and sieved to produce granules of 200–500 µm in size.
Nominal composition (mol%) of Ce-BGs granules.

In vitro assessment of cytocompatibility for Ce-BGs granules
The assessment of cytocompatibility of Ce-BGs granules was evaluated through in vitro tests in accordance with ISO10993 guidelines. 25
The granules were subjected to both the cell viability test, Neutral Red (NR), and the proliferation test, 5-bromo-2′-deoxyuridine (BrdU).
The assessment was carried out with MLO-Y4 cells derived from murine long osteocytes and from Istituto Zooprofilattico Sperimentale (IZS) Brescia, Italy. The cells were cultured in DMEM (Euroclone, Milan, Italy) supplemented with 10% fetal bovine serum (Euroclone, Milan, Italy), 100 µg/mL pen-streptomycin (Invitrogen-Thermo Fisher Scientific Corporation, Waltham, MA, United States), and sodium pyruvate 1 mM (Euroclone, Milan, Italy) at 37°C in a humidified atmosphere of 5% CO₂ in air.
To ensure sterilization, the granules were autoclaved at 121°C for 20 min. before the tests.26 –28
NR
Before performing the NR assay, morphological analysis of MLO-Y4 cells in direct contact with granules was carried out after 24 and 72 h. Morphological assessments are an essential part of cytocompatibility testing in biomedical research, particularly in the field of tissue engineering. 29 As per the ISO10993 guidelines, the morphological parameters observed included signs of cellular distress such as lysis, swelling and intracytoplasmic thickening.
The NR assay is a test that measures the ability of cells to incorporate the vital NR dye in the absence of membrane lesions, particularly in lysosomal membranes. The test was carried out in a 24 well plate. MLO-Y4 cells were seeded at a concentration of 1.5 × 105/mL DMEM in multiwell, Ce-BGs granules (40 mg/mL) were added to each well. All samples were incubated for 24 and 72 h at standard conditions of 37°C, 90% humidity, and 5% CO2/air. After incubation, 150 μL of NR solution (NR N2889 Sigma-Merck, Darmstadt, Germany) was added to each well, and the plates were placed in the incubator for 3 h. After this incubation, the supernatant was discarded, and each well was rinsed with Dulbecco’s phosphate buffered saline (D-PBS- Sigma-Merck, Darmstadt, Germany). A 150 μL mixture of ethanol/acetic acid was added to extract the NR dye from the cells.
The absorbance was measured for each well by spectrophotometer at a wavelength of 540 nm. The measured optical density is directly proportional to the ability of the lysosomal membrane to retain NR dye, so a high density indicates no cytotoxicity in undamaged cells.
Experiments were conducted in triplicate using serum-free DMEM (CTRL−) and phenol (CTRL+) as reference terms.30,31 The results are reported as cell viability percentage.
BrdU
The BrdU assay is commonly used to quantify the proliferation of cells by measuring the incorporation of the thymidine analog, BrdU, during DNA synthesis.
This assay was performed using sample (K, K3.6, K5.3) eluates prepared according to the ISO 10993-12 32 guidelines. To ensure sterile conditions, the eluates were filtered through a 200 µm filter prior to the assay.
The eluates were then added to 96 well plates containing MLO-Y4 cells and incubated for 24–72 h. The incorporation of BrdU was detected using specific monoclonal antibodies, and color development was measured by absorbance readings at 370 nm using a UV-visible spectrophotometer. The amount of detected signal is proportional to the intensity of newly synthesized DNA or the number of proliferating cells in the culture.
Experiments were conducted in triplicate using serum-free DMEM (CTRL−) and phenol (CTRL+) as reference terms.9,33 –35 The results are reported as cell proliferation percentage.
In vivo tests of Ce-BGs granules
Animals and surgery
We have evaluated the effectiveness of Ce-BGs granules for bone tissue regeneration and repair using, as in vivo animal model, the New Zealand white rabbit. This model is suitable for such experiments due to the high rate of bone remodeling and the similarities of bone tissue between it and human tissue33 –35 The rabbits weighing 3.5–4.5 kg (Harlan Laboratories S.r.l., Correzzana MB, Italy) have been maintained and acclimatized according to Italian law (Legislative Decree no. 26/2014) and European legislation (EEC no. 63/2010). All the experiment was carried out in accordance with the Bioethical Committee of the Italian National Institute of Health and licensed with Decrees of the Italian Ministry of Health (Protocol Number: 210/2013-B). The surgical protocol was implemented, which included the creation of a defect, defined as “critical size defect,” in the rabbit’s femur bone and subsequent filling with granules; the protocol involved the fabrication of a defect in the bilateral femoral lateral condyle of rabbit (Figure 1).

Photographs of the surgical session in the rabbit model: creation of a defect in the femoral condyle (a) and filling the defect with BGs granules (b).
Animals were fasted prior of anesthesia. The surgical session included general anesthesia using a mixture of xylazine (4 mg/kg body weight – Sedaxylan, Dechra Veterinary Products S.r.l., Turin, Italy) and ketamine (30 mg/kg body weight – Imalgene 1000, Merial Italia S.p.A., Milan, Italy).
The area on the lower limb to be operated on was shaved and cleansed with topical antisepsis medication, following the administration of anesthesia. A 3 cm long skin incision was made on the lateral skin of the distal femur; after blunt dissection of the muscles, an incision was made on the periosteum with a scalpel to expose the cortex of the femur. Bilateral 5 mm diameter and 10 mm in depth bone defects were surgically created in the distal third of the rabbit femur using piezosurgery® unit with insert tip IM4A (Mectron, Carasco, Genova, Italy) under continuous saline irrigation. Then, using a compressed gauze for 5 min, the holes were plugged and prepared to accommodate about of 100 mg of K3.6 and K5.3 granules in the left femur while the right femur defect was filled with the same amount of K granules. Following the insertion of the materials, the surgical flaps were closed in layers and sutured using 4.0 glycolide/L-lactide copolymer (Vicryl®, Ethicon, Johnson & Johnson Spa, Pomezia, Rome, Italy) and 3.0 silk (Perma-hand® Silk Suture, Ethicon, Johnson & Johnson Spa, Pomezia, Rome, Italy) for the skin. Iodine-based solution was applied to the sutured area in order to avoid post-surgical infection. A wide spectrum antibiotic (enrofloxacin, 10 mg/kg body weight – Baytril® 5%, 50 mg/mL, Bayer S.p.A., Milan, Italy) was administrated intramuscularly jointly with analgesic medication (buprenorphine, 0.05 mg/kg body weight – Temgesic®, Indivior Italia S.r.l., Milan, Italy).
To evaluate the repair process we split the rabbits into two groups (Scheme 1, Paragraph 2.3.2): with an observation period of 30 and 60 days before euthanasia.36 –38 All rabbits were euthanized by a lethal injection of veterinary euthanasia solution embutramide/mebezonium iodide (0.3 mL/kg body weight – Tanax 50 mg, MSD Animal Health S.r.l. Italy, Segrate MI, Italy) under general anesthesia with a mixture of xylazine (4 mg/kg body weight; Sedaxylan®, Dechra Veterinary Products Srl, Turin, Italy) and ketamine (30 mg/kg body weight; Imalgene 1000®, Merial Italia SpA, Milan, Italy).
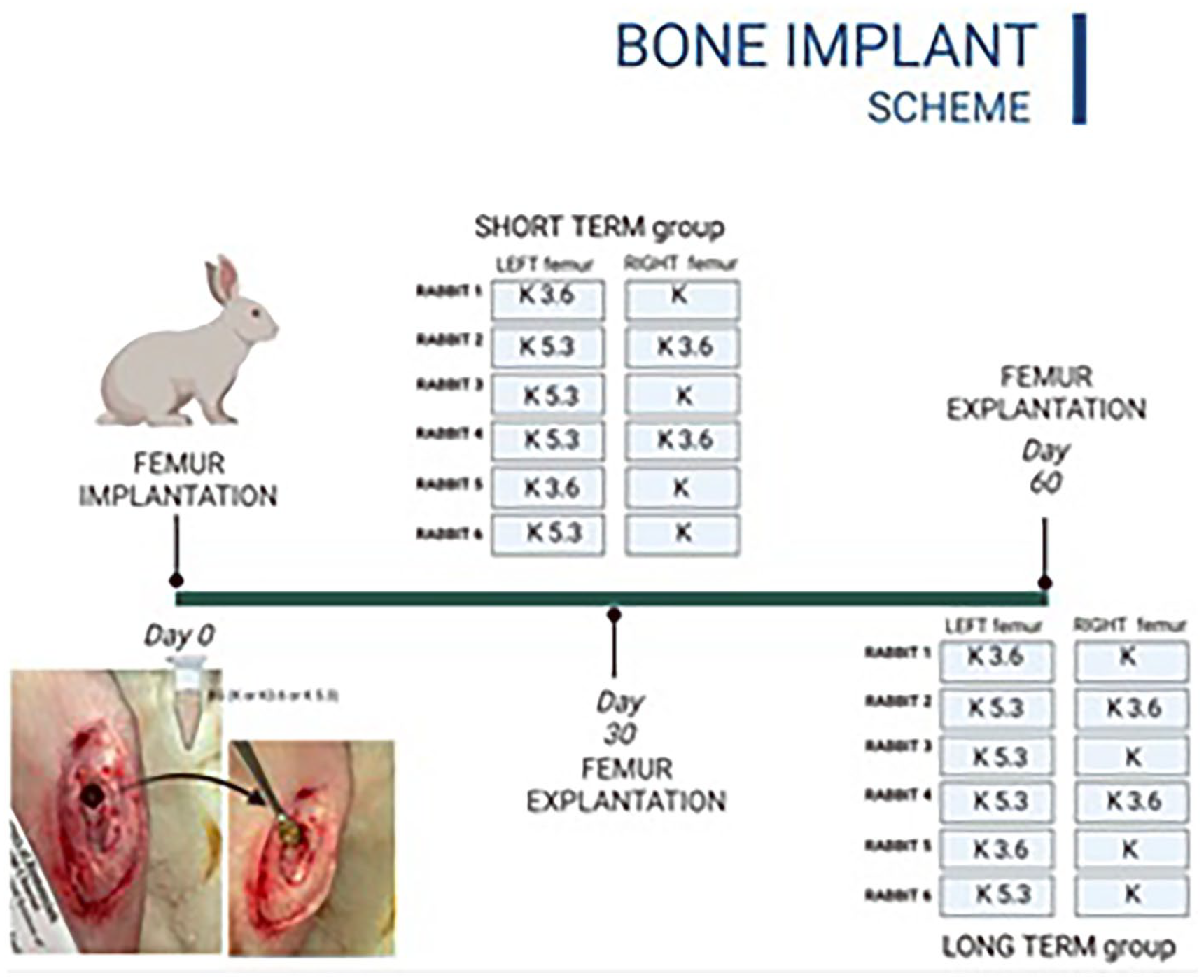
Experimental design for two sets of investigations in rabbits. The first group, labeled as “Short Term group,” shows the implantation of granules in the left and right femurs of rabbits, followed by their sacrifice 30 days after surgery. The second group, labeled as “Long Term group,” shows the granules implanted in the left and right femurs of rabbits, followed by their sacrifice 60 days after surgery.
Experimental design
The study was designed for short-term (30 days) and long-term (60 days) evaluation: six rabbits were sacrificed after 30 and another six after 60 days of implantation. In each rabbit, granules were implanted in both femurs according to Scheme 1 made in such a way as to obtain an adequate amount of samples containing several replicates of the same material.
Post implant characterization
Within the scope of the investigation, a comprehensive macroscopic evaluation of the defect site and its contiguous tissue was conducted for each animal. Subsequently, the femurs were disarticulated and the surrounding soft tissue was meticulously dissected; the femurs were immersed in a 4% solution of formaldehyde in phosphate-buffered solution (PBS) for histological fixation. Defect sites were dehydrated in ethanol, infiltrated with methyl methacrylate (PMMA), and incorporated using a 4°C water bath. After polymerization, the PMMA blocks were sectioned (thickness of 200 µm) using a saw microtome (Leica SP 1600 diamond, Leica SpA, Milan, Italy).
These cross sections were then treated to analyze the bone tissue and surrounding structure.
Scanning electron microscopy (SEM) evaluation
To evaluate bone regeneration, the sections were polished to 100 µm thick (LS2; REMET, Italy) then were sputter-coated with a 10 nm gold-palladium layer using an Emitech K550 (Emitech Ltd in Ashford, United Kindom) and analyzed with SEM (SEM-QUANTA 200, FEI Company, Heindhoven, Netherlands) under high vacuum, using a solid-state backscattered electron detector. SEM was equipped with energy dispersion X-ray spectroscopy (EDS INCA-350, Oxford instruments, Abingdon, United Kingdom).
Histological evaluation
Cross-sections were lapped (LS1 REMET Casalecchio di Reno, (BO), Italy) to a thickness of 40–50 µm and stained to highlight the different components of bone tissue by histomorphologic staining with Stevenel blue and Van Gieson’s picrofuchsin (SVG staining). SVG contain: 1% of methylene blue (Sigma-Aldrich, Milan, Italy), 1.5% potassium permanganate (Carlo Erba, Milan, Italy), 0.1 g fuchsin (Sigma-Aldrich, Milan, Italy) and 10 mL of saturated picric acid. The sections were stained 30 min at 37°C, differentiated and dehydrated with ascending scale of alcohols.
Microscopic observations were performed using a ZEISS Standard 25 R microscope (Zeiss, Oberkochen, Germany) and equipped with a polarized filter for enhanced contrast and detailed visualization of birefringent structures in bone tissue.
Mineralized bone tissue appears pink, bone marrow, osteoid matrix, and cellular components have different shades of blue and fibrous tissue appears pink.
Histomorphometric analysis
A histomorphometric analysis was carried out on the SEM micrographs acquired using ImageJ software.39,40 A defect region of interest (ROI) was identified and used to calculate the subsequent areas: total area (TA), new bone area (NBA), residual material area (RMA), and connective tissue area (CTA). Histomorphometric analysis, while robust in providing quantitative data, has certain limitations. First, it depends strongly on the accurate identification and segmentation of ROIs, which can introduce variability in the choice of area. Secondly, the analysis processes static two-dimensional sections and is therefore not a complete three-dimensional analysis.
Statistical analysis
The results obtained from the in vitro and in vivo studies were statistically analyzed using One-way ANOVA and Dunnett’s Multiple Comparison post hoc test was performed using GraphPad Prism. 41 The statistical significance was determined based on the p-value, with a p-value less than 0.05 being considered statistically significant.
Results
In vitro
The cytocompatibility of Ce-BGs granules was studied using MLO-Y4 cells to assess their effect on cell viability and proliferation.
Morphological evaluations (Figure 2) showed no signs of cell distress such as lysis, swelling, or intracytoplasmic thickening, indicating that the Ce-BGs granules did not cause any adverse effects on the MLO-Y4 cells. 42 The absence of adverse effects on cell morphology also indicated that the granules should present no risk once implanted as a bone substitute.

Optical microscope images (marker = 200 µm) of MLO-Y4 cells exposed to K(a,d), K3.6(b,e), and K5.3(c,f) granules for 24 (top) and 72 (bottom) h.
The cell viability and proliferation were assessed using NR (Figure 3) and BrdU (Figure 4) assays, for 24 and 72 h.

NR uptake of MLO-Y4 cells exposed to direct contact with K, K3.6, K5.3 granules for 24 (a) and 72 h (b).

BrdU tests of MLO-Y4 cells exposed to eluates of K, K3.6, K5.3 for 24 (a) and 72 h (b).
NR results after 24 h showed for K a cell viability of 77%; the addition of cerium increases the viability up to 79% and 85% for K3.6 and K5.3, respectively. After 72 h, the cell viability was significantly reduced to 58% for K, while in contact with K3.6 and K5.3, increased to 76% and 116%, respectively, highlighting how the presence of cerium induces an improvement in the cytocompatibility of Ce-BGs.
BrdU tests, Figure 4, indicated, after 24 h of contact with K eluate, a cell proliferation of 99%, in line with the control (CTRL−). Doping with cerium resulted in a significant reduction in cell proliferation, with values of 64% and 55% for K3.6 and K5.3, respectively; after 72 h, these values increased slightly for K3.6 (66%) and more significantly for K5.3 (74%), but still remained lower than for K (89%).
Dunnett’s Multiple Comparison analysis (Table 2) was performed on NR and BrdU results to evaluate differences between the control group (CTRL−) and experimental groups (K, K3.6, K5.3). For NR, all experimental groups (K, K3.6, K5.3) showed increased cell viability compared to CTRL− at both 24 and 72 h. These differences reduced with increasing cerium concentrations, as indicated by lower mean difference (MD) and q-values for K3.6 and K5.3 relative to K. Notably, at 72 h, the K5.3 displayed a negative mean difference compared to CTRL−. For BrdU, K did not significantly differ from CTRL− at 24 h, whereas K3.6 and K5.3 exhibited significant increases in cell proliferation, with K5.3 showing the highest q-values. At 72 h, all samples demonstrated significantly greater BrdU values compared to CTRL−, with K5.3 again showing the most pronounced effect, as reflected in the highest q-value and mean difference.
Dunnett’s multiple comparison on NR and BrdU results.

These results confirmed an enhancement of cellular proliferation by cerium, particularly at higher concentrations and prolonged time points.
In vivo
Scanning electron microscopy (SEM) analysis
SEM analysis (Figure 5) revealed new trabecular bone around the residual granules. At 30 days, a different degradation of granules was observed; more pronounced for K (Figure 5(a)) and less for K3.6 and K5.3 (Figure 5(b) and (c)) due to the slowing of cerium-induced ionic dissolution. 18

SEM micrographs (marker = 500 µm) of examined sections with K (a and d), K3.6 (b and e), K5.3 (c and f) granules after 30 (top) and 60 (bottom) days. K granules are replaced by newly formed bone at 30 days (a); conversely, K3.6 and K5.3 granules are still observed, surrounded by newly formed bone (b and c). At 60 days, K3.6 and K5.3 granules are further consumed and almost completely replaced by bone tissue (e and f).
Figure 5(a) showed the presence of neo-deposited bone around K granules, of which only an imprint appears to remain. Their rapid ionic dissolution stimulated the formation of bone islands,8,43,44 which serve as the initial structure for new bone, supporting the soft callus and promoting the deposition of new bone matrix. 45 K3.6 and K5.3 granules, (Figure 5(b) and (c)), on the other hand, showed structural integrity with low degradation, accompanied by less neo-deposited bone compared to K; no significant differences were evident in relation to the amount of cerium. K3.6 and K5.3 showed excellent bone integration without the presence of fibrous capsules around the granules but rather with bone formation directly adjacent to the granules.
After 60 days, femurs containing K3.6 and K5.3 (Figure 5(e) and (f)) showed a more homogenous formation of new bone compared to K granules (Figure 5(d)).
Details of regeneration at 60 days ok K3.6 granules, Figure 6(a) and (b); indicated a new bone with a semi-concentric lamellar structure, demonstrating bone maturation. This was also evident from maps (Figure 6(c) and (d)) and EDS analysis (Figure 6(e)) which confirmed the presence of calcium phosphate-rich tissue adjacent to the granules.

Micrograph (a), (marker = 200 µm) of K3.6 granules after 60 days of implantation. Upon magnification, (marker = 50 µm) the granule (b) is completely surrounded by new bone, organized in a semiconcentric lamellar structure, with bone lacunae that demonstrate the presence of osteocytes and continuous bone regeneration. Maps (c and d), (marker = 80 µm), and EDS spectrum (e) highlight the different chemical composition and confirms the presence of calcium and phosphorus in the area adjacent to the granule.
Histology results
The histological analysis revealed a clear color differentiation between newly formed bone (orange-pink), granules (white, reflective), and area of granules dissolution (purple; Figure 7). Significant differences can be seen between K3.6, K5.3, and K granules in the area of the new bone near the residual granules.

SVG histomorphological staining (marker = 500 µm) of implants with K (a and d), K3.6 (b and e), K5.3 (c and f) granules after 30 (top) and 60 (bottom) days.
A light pink region is linked to the soft tissue that forms close to the implant is visible around the granules; this is an extracellular matrix composed of fibers and glycoproteins that will be mineralized by osteoblasts to form new bone. 46 Also visible is the process of calcification of extracellular matrix, where the cells have settled in non-mineralized ellipsoidal cavities (bone gaps), which will become osteocytes. This process provides the scaffold for new bone formation by depositing minerals like calcium and phosphate, which strengthen the matrix. Bone gaps are crucial in remodeling as they allow space for osteoclast activity (resorption) and subsequent osteoblast-driven formation of new bone, ensuring proper healing and adaptation to the implant and its integration with the surrounding tissue47 –49 (Alford, A. I., Kozloff, K. M. & Hankenson, K. D. Extracellular matrix networks in bone remodeling. The international journal of biochemistry & cell biology 65, 20–31 (2015).).
The presence of bone gaps after 60 days (Figures 7(e), (f) and 8) suggests an ongoing remodeling and the deposition of new bone in areas adjacent to the material in close contact with its surface. The presence of white, reflective Ce-BGs granules after 60 days is an indication that they can play an active role in the promotion of osteogenesis.

Details of Figure 7(f) (magnification 40×, marker = 250 µm) showing the different areas of interest: newly formed bone (NBA) in pink, residual material (RMA) in purple and connective tissue (CTA) in light pink.
These results are consistent with the SEM/EDS analyses (Figure 6) highlighting the osteoinductive properties of Ce-BGs granules and their ability to stimulate the formation of new bone.9,34
Hystomorphometric analysis
After 30 and 60 days of implantations, the hystomorphometric analysis was carried out on specific ROI to select and identify the TA. TA values are crucial to calculate the different areas: RMA, CTA, and NBA (Tables 3, 4, and Figures 9–11).
RMA, NBA, CTA, and TA after 30 and 60 days of implantation.

Dunnett’s multiple comparison test on histomorphometry results.


Original image, (marker = 500 µm), (left) and correlated analysis (right) of ROI to calculate CTA (blue), RMA (green) and NBA (gray) with the ImageJ software.
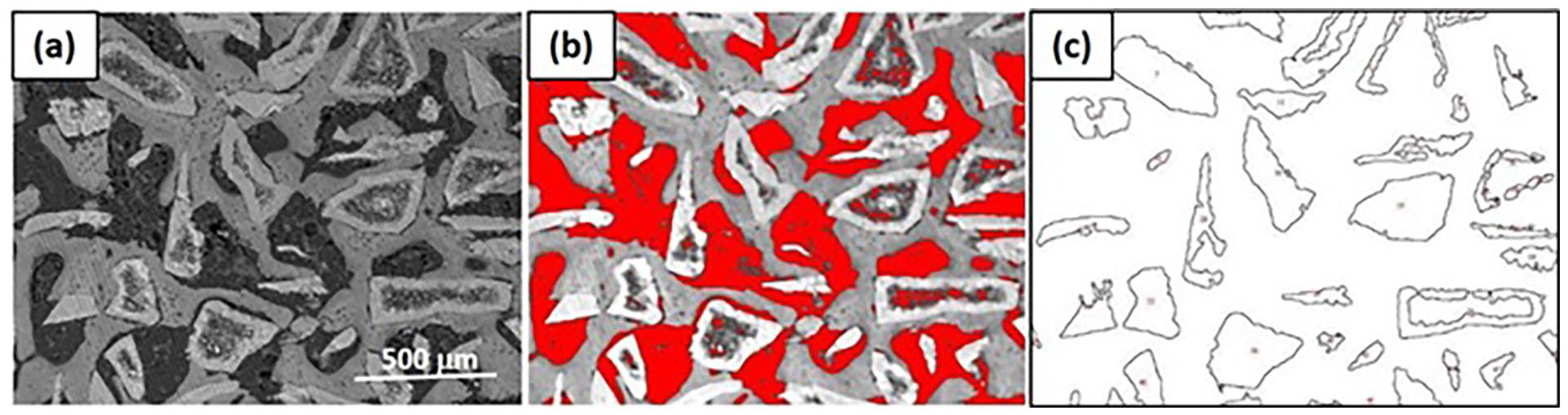
Histomorphometry showing how calculations were performed using ROI as TA (a), selecting the threshold function highlighted NBA (gray) and CTA (red) (b). RMA after selecting the threshold function was binarized in black/white (c) and subtracted from the previous step to calculate the NBA.

RMA, NBA, CTA (±1) calculated by histomorphometric analysis at 30 (top) and 60 days (bottom).
After 30 days, the analysis showed that the NBA for K granules was 29%, while K3.6 and K5.3 granules exhibited slightly lower values of 25% and 23%, respectively.
Conversely, RMA values were higher for Ce-BGs, with K3.6 and K5.3 having 30% and 31% of residual material, compared to 23% for the reference K. This difference shows slower degradation of Ce-BGs during the initial healing phase after implantation. CTA values were comparable across all analyzed BGs, reflecting a soft tissue integration.
At 60 days, significant differences emerged. The NBA for K5.3 granules increased to 32%, overcoming both K3.6 (28%) and the reference K (21%). This trend demonstrates the enhanced osteogenic potential of Ce-BGs, particularly at higher cerium concentrations. RMA values for K3.6 and K5.3 were 35% and 38%, respectively, compared to 30% for K granules, confirming the prolonged structural presence of Ce-BGs in the defect site. CTA percentages decreased in Ce-BGs (21% for K5.3 vs 27% for K), highlighting the transformation of soft tissue to mineralized bone over time.
The slower degradation of Ce-BGs, reflected by their higher RMA, likely contributes to a sustained release of bioactive ions, creating a favorable microenvironment for osteogenesis. The increase in NBA observed with K5.3 granules at 60 days aligns with the antioxidant and osteogenic properties of cerium, which are known to reduce oxidative stress and enhance bone regeneration.
Statistical analysis (ANOVA and Dunnett’s post hoc tests) confirmed significant differences in NBA between K5.3 and K granules at 60 days (p < 0.05). These findings highlight the role of cerium doping in promoting sustained bone regeneration. As illustrated in Figure 9, the ROI segmentation and thresholding process highlight the spatial distribution of NBA, RMA, and CTA within the defect site. SEM and histological images further support these results, showing close integration of Ce-BGs with newly formed bone tissue (Figures 5–7).
Discussion
In vitro studies show that Ce-BGs granules improved the cell viability (NR) and inhibited the cell proliferation (BrdU). Specifically, at 24 h the NR was 77% for K and 79% and 85% for K3.6 and K5.3, respectively. After 72 h NR was reduced for K at 58% and increased for K3.6 and K5.3 (76% and 116% respectively). Conversely, cell proliferation results showed after 24 h of contact with eluates showed BrdU of 99% (K), 64% (K3.6), and 55% (K5.3); after 72 h, these values increased for both K3.6 (66%) and K5.3 (74%), but still remained lower than for K (89%).
As the NR assay involved direct contact with Ce-BGs granules, whereas the BrdU assay was performed on eluates, this discrepancy was likely due to the slower dissolution rate of Ce-BGs, 18 which results in less ionic release in eluates. Furthermore, the effects of cerium on cell behavior are complex and depend on several factors, including concentration, duration of exposure and cell type.16,50,51 The presence of cerium not only supports but may also improve long-term cell survival; this is consistent with previous studies that have reported the beneficial effects of cerium on cell viability and proliferation.9,23,52 –55
In vivo studies, after 30 and 60 days, revealed a delayed degradation for Ce-BGs that still stimulates osteo-regeneration without inflammatory or degenerative effects; moreover, new bone area (NBA) was higher for Ce-BGs, after 60 days (32% for K5.3 vs 21% for K).
The slower ion dissolution rate 18 in Ce-BGs granules likely promotes continuous bone regeneration leading to a more controlled release of ions over time, supporting continuous bone regeneration. Thus, doping with cerium not only supports initial bone formation, but also enhances the long-term remodeling process, contributing to more controlled and effective bone healing. The increased formation of new bone can be linked to the antioxidant and anti-inflammatory properties of cerium, which have been reported to improve bone regeneration and reduce implant-associated inflammation.51,56 –61
Conclusions
Ce-BGs granules with antioxidant properties were studied in vitro and in vivo to provide a comprehensive understanding of their promise as biomaterials for hard tissue applications.
In vitro studies showed excellent cytocompatibility with cerium promoting cell viability and significantly increases cell survival over time.
Importantly, in vivo studies indicated that the Ce-BGs granules are histocompatible and the effect of cerium on slowing down ionic dissolution promotes long-term bone regeneration. While K granules underwent rapid dissolution forming bone islands, K3.6 and K5.3 granules maintained structural integrity with excellent bone integration, and after 60 days K3.6 and K5.3 granules promoted more new bone formation than K granules.
In summary, the use of Ce-BGs granules in short- and long-term bone implantation showed a significant effect on the rate and quality of bone regeneration, with improved incorporation into the newly formed bone tissue and biocompatibility in line to that to that of mother glass. Furthermore, the granular form of Ce-BGs improves the adaptability of these materials to anatomically complex defects, suggesting promise in their clinical applications in craniofacial, dental and orthopedic surgery.
Further developments will involve loading the material with biomolecules and drugs to develop biomaterials able to act as in situ drug delivery systems.
Footnotes
Author contributions
Conceptualization (R.S, A.A, G.L), Methodology (R.S, A.A, G.L), Validation (R.S, A.A, G.L), Investigation (R.S., A.A, MDB, A.P) Data Curation (R.S, A.A, G.L., A.Z, C.C.), Writing - Original Draft (R.S, A.A, G.L), Writing - Review & Editing Preparation (R.S, A.A., L.C., G.L., A.Z, C.C.), Supervision (R.S, A.A, L.C, G.L).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
