Abstract
Hemicelluloses are promising candidates for synthesizing nanosystems for potential biomedical and photocatalytic applications. Glucuronoxylan (hemicellulose)-capped manganese oxide nanoparticles (GX-MnO NPs) were synthesized from quince (Cydonia oblonga M.) seed hydrogel. Ultraviolet-visible spectroscopic analysis revealed a distinct surface plasmon resonance peak at 310 nm for MnO NPs, with an estimated band gap energy of 2.60 eV. The interactions between MnO NPs and the functional groups of hydrogel were characterized using Fourier-transform infrared spectroscopy, while the cubic structure was evident from X-ray diffraction results at 2θ location. Scanning electron microscopy showed that the NPs had a roughly spherical shape with an average size of 38.5 nm. Energy-dispersive X-ray spectrum indicated the sample’s composition, highlighting a significant presence of manganese (39.29%), oxygen (29.3%), and minor elements from hydrogel. The NPs displayed noteworthy in vitro antibacterial and antibiofilm activities against Bacillus licheniformis, Escherichia coli, and Aeromonas. An in vivo wound healing experiment illustrated that wounds treated with GX-MnO NPs healed entirely within 10 days in albino mice. Further, GX-MnO NPs served as an excellent photocatalytic system in the sunlight-assisted degradation of methylene blue (90.5%) and methyl orange (89.7%). Intriguingly, degradation efficiencies of 47.6% and 45.7% were achieved, respectively, when the NPs were operated in the dark. Thus, the study suggests GX-MnO NPs as versatile and promising agents to address biomedical and dye-contaminated wastewater concerns.
Keywords

Introduction
In the last decade, the biosynthesis of nanomaterials has attracted researchers’ interest as a reliable, safe, and environmentally friendly route. 1 Nanotechnology has developed a diverse range of nanoscale materials with desired properties and has revolutionized many industries, such as mechanical, electronics, energy harvesting, environmental remediation, and medical sectors. 2 The physiochemical techniques to produce nanoparticles (NPs) face challenges due to using expensive, energy-intensive, and toxic materials. 3 In light of the potential risks associated with the environmental impacts of conventional methods and the need for eco-friendly approaches, green processes are critical in synthesizing NPs. 4
Antibiotic resistance and biofilm formation are pressing issues in medical research, leading to increased financial burden, disease spread, and death. 5 Despite the ongoing expansion of the pharmaceutical landscape, multidrug-resistant bacteria are becoming more prevalent and presenting severe threats to public health worldwide. 6 Rapid progress in developing and implementing new approaches is needed to treat human infections and eliminate biofilms. In this context, metal and metal oxide NPs have demonstrated excellent antimicrobial potential due to their quantum size effect, huge surface-area-to-volume ratio, and high chemical reactivity. 7 These nanomaterials exert bactericidal effects by generating reactive oxygen species (ROS), disrupting membrane integrity, denaturing protein, damaging DNA, and releasing cellular contents, ultimately causing structural damage and cell death. 8
Chronic non-healing wounds represent a social and economic burden for patients and are associated with devastating consequences, including prolonged wound closure, increased risk of infection, and reduced quality of life. 9 In the quest for newer and innovative therapeutic regimens, nanoformulations are the focus of significant attention in wound care, helping reduce side effects and improve overall efficacy. 10 This surge is attributed to the physiochemical and biological properties of nanomaterials, making them ideal for wound dressings with enhanced patient outcomes. 11
Excessive discharge of organic dyes from textiles, food processing, and pharmaceutical industries into natural water bodies results in severe deterioration of aquatic ecosystems. These colorants are hazardous, recalcitrant, and non-biodegradable, raising serious concerns regarding their detrimental effects on nature and living organisms. 12 Traditional treatment methods exacerbate various environmental issues by generating hazardous secondary pollutants. 13 Therefore, it is crucial to utilize straightforward and environmentally safe strategies for water purification. Photocatalysis, leveraging simultaneous adsorption and photodegradation properties, stands out as a compelling alternative with excellent removal efficiency against persistent organic pollutants. 14
Amongst various oxide NPs, MnO NPs have stimulated intense research owing to their affordability, charge transportation characteristics, tunable morphologies, enhanced stability, and minimal toxicity. 15 MnO NPs possess potent antimicrobial properties and can be used for applications like catalysis, biosensing, diagnostic imaging, and photocatalytic wastewater treatment.16–20 Numerous preparation techniques are available for creating MnO-based nanomaterials; typical examples include self-reacting microemulsion, sol-gel, deposition, solid phase reaction, microwave-hydrothermal, and thermal decomposition.21–26 Most of these preparation methods use toxic solvents and chemicals, which may present significant environmental and biological hazards. Thereby, the scientific community has been actively exploring cost-effective and eco-friendly approaches for producing MnO NPs.27–29
Recently, polysaccharides have gathered great interest in fabricating NPs. 30 Cydonia oblonga Miller (quince), a medicinal plant from the Rosaceae family, is believed to have originated mainly in Iran and Turkey and has also been cultivated in several other regions.31,32 The proximate composition of quince seed hydrogel indicated higher contents of carbohydrates (75.04 ± 2.87%) and uronic acid (13.16 ± 1.73%), and a weight-average molecular mass of around 9.61 × 10 6 g/mol. 33 Following hydrolysis, it appeared to consist primarily of sugar units such as xylose, glucose, galactose, arabinose, and mannose with a ratio of 46:32:10:8:4, respectively. 34 As already elucidated, 1 H and 13 CNMR spectroscopy revealed a highly branched glucuronoxylan (GX) polysaccharide that is, 4-O-methyl-D-glucurono-D-xylan as its main component.34,35 Quince seed hydrogel is well known for its diverse therapeutic properties, including bactericidal, fungicidal, free-radical scavenging, cough suppressing, bone regeneration. and wound healing.36–40 As a biotemplate, it exhibited minimal potential for generating NPs,41–44 thereby, presenting an important direction for future research.
Herein, green, environmentally sustainable, and cost-effective GX-MnO NPs were developed by photoreduction, employing GX-hydrogel from quince seeds as a bioreductant and encapsulant. The synthesized NPs were characterized using advanced spectroscopic, diffractometric, microscopic, and elemental analysis techniques to confirm their formation, chemical structure, crystallinity, morphology, and elemental composition. Additionally, the NPs were explored for multifaceted applications, mainly focusing on their antibacterial, antibiofilm, wound healing, and photocatalytic properties, thus addressing the unmet healthcare and environmental challenges. Ultimately, this study seeks to establish GX-based NPs as promising pharmaceutical formulations and environmentally viable photocatalysts.
Materials and methods
Materials
Quince seeds (also called Bahi Dana) were bought from the herbal store in Lahore, Pakistan. Potassium permanganate (KMnO4), methylene blue (MB), and methyl orange (MO) were received from Sigma-Aldrich. Mueller agar solution, 0.5 McFarland Turbidity standard, Luria-Bertani/Lysogeny broth (LB), and the isolates of bacterial cultures were provided by the Microbiology Lab, Department of Zoology, GC University, Lahore, Pakistan. Male albino mice were purchased from the Institute of Biochemistry and Biotechnology, University of Veterinary and Animal Sciences, Lahore, Pakistan. All other solvents and reagents employed during experiments were also obtained from Sigma-Aldrich. The solvent utilized for preparing solutions and dispersions was distilled water.
Extraction of GX-hydrogel and biosynthesis of GX-MnO NPs
GX-hydrogel was typically isolated using the hot water maceration method. Approximately 100 g of seeds were precisely weighed and rinsed with distilled water multiple times to eradicate extraneous substances. After, the seeds were drenched in distilled water (500 mL) for about 30–45 min. The aqueous mixture was heated (Simmerstat CH2090-001 PTFE plate) at 60–70°C for 120 min under gradual and continuous agitation. The hydrogel was squeezed from seeds using a nylon cloth and then washed with n-hexane and distilled water. It was then oven-dried (IRMECO GmbH incubator model B-53) at 55°C for 48 h. The hydrogel was ground to desired particle size and stored in a container provided with a screwed lid. In KMnO4 solution (20 mM, 10 mL), GX-hydrogel suspension (1%, 10 mL) was added under continuous stirring to ensure homogeneous mixing. The biosynthetic solution was left exposed to sunlight for 20–30 min before it turned blackish brown from dark purple. The synthesized NPs were separated by high centrifugation (Model M-800, 4000 rpm, 20 min) and washed with distilled water thrice. After, the residue underwent drying in an electric oven at 55°C for 5–6 h and carried out for analyses. Figure 1 depicts a preparative scheme for the biological production of GX-MnO NPs.

Stages of biosynthesis of GX-MnO NPs from GX-hydrogel and KMnO4 precursor.
Characterization
Conformation of initially synthesized NPs was found by analyzing the optical density through an ultraviolet-visible (UV-Vis) spectrophotometer (Shimadzu-1700 Japan). The transmittance response of the diluted sample (1 mL) dispersed in a quartz cuvette was obtained between 200 and 800 nm. Fourier transform infrared (FTIR) spectroscopic screening was performed using a Nicolet iS50 spectrophotometer (Thermo Scientific, United Kingdom). Powdered hydrogel and GX-MnO NPs were pelleted individually with potassium bromide (KBr), and their interference patterns were acquired in the 400–4000 cm−1 region. The study was undertaken to determine the functional groups and biomolecules of GX-hydrogel capable of reducing metal ions and stabilizing NPs. The information on crystalline structural characteristics of the biosynthesized NPs was collected by a Proto AXRD LPR X-ray diffractometer in reflection mode with copper (λ = 1.54 Å) radiation source. The pattern was measured at an accelerated 30 kV voltage, 30 mA current, and a scanning rate of one step per second in a 2θ region from 5° to 80°. Morphological analysis was conducted using the scanning electron microscopy (SEM) technique (Nova Nano-SEM 450) by dispersing a small amount of diluted sample onto the carbon-coated grids and then drying under an infrared lamp. SEM micro- and nano-graphs were taken by adjusting the sample height and magnification. An energy-dispersive X-ray (EDX) attachment equipped with SEM apparatus provided information about the chemical composition of the synthesized NPs. The size distribution of the particles was estimated using ImageJ software.
Antibacterial assay
The antibacterial potential of as-synthesized GX-MnO NPs was evaluated following the agar diffusion bioassay. The bacterial strains, such as B. licheniformis, E. coli, and Aeromonas were inoculated and standardized using 0.5 McFarland standard. The fresh overnight-grown bacterial suspensions were spread on the sterilized LB agar plates (32 cm diameter and 2 cm thickness). After the media setting, five equally spaced wells of 3 mm diameter were created on the entire agar surface using a sterile cork borer. The positive control (PC) and the negative control (NC) wells received rifampicin (50 µg/mL) and distilled water, respectively. The remaining wells were introduced with the aqueous solutions of 100, 75, and 50 µg/mL concentrations of the NPs. The incubation was done overnight at 37°C, and the clear circular areas around wells were measured as zones of inhibition (ZOI) using a meter ruler.
To access the minimum inhibitory concentration (MIC), various concentrations (30, 40, 50–180 µL/mL) of GX-MnO NPs were added into sterile test tubes containing LB growth medium (3 mL) and 24 h old bacterial strains (30 µL) adjusted equivalent to 0.5 McFarland turbidity standard. The test tubes were left in a 37°C incubation chamber all night. After one whole day of incubation, absorbance values were checked at 523 nm, and the concentration corresponding to the least absorbance was considered MIC for a particular strain. The minimum bactericidal concentration (MBC) was estimated by spreading dilution (100 µL) that did not show growth under MIC determination on agar plates and incubating them at 37°C. The plates were observed, and the concentration with the least/non-visible growth was taken as MBC. The same procedure was repeated with all three bacterial isolates to determine MIC and MBC. Experiments of standard drug and NP samples were performed in triplicate, and the results are represented as mean ± standard deviation (SD).
Anti-biofilm assay
The activity of GX-MnO NPs against the bacterial biofilms was examined using the tube method for B. licheniformis, E. coli, and Aeromonas with slight modification. 45 Fresh LB broth (3 mL) and microbial suspensions (30 µL) were inoculated in each labeled test tube and kept at 37°C for up to 5 h. The medium containing bacterial suspension was replaced with the NPs (100 µL) without touching the film and maintained further at 37°C for 1 day. After this period, media was carefully removed, and freely floating non-adherent cells were discharged by rinsing twice with phosphate-buffered saline (100 µL). The tubes were then air-dried for 20 min. After drying, adherent “sessile” isolates in the tubes were treated with 30 mL of crystal violet stain (0.1% w/v), and the tubes were rinsed thoroughly with autoclaved distilled water to eliminate excess stain. Afterward, the test tubes were supplemented with glacial acetic acid (33%), and absorbance at 590 nm was checked spectrophotometrically. The tubes holding bacterial specimens without GX-MnO NPs were taken as control/blank. The percentage of biofilm inhibition was recorded by finding the difference between the optical densities of control and tested samples.
Wound healing activity
A total of 18 (8–10 weeks old) male albino mice with weights typically ranging between 20 and 30 g were fed a regular diet and housed in individual cages. The hairs on the dorsal surface of mice were shaved and depilated using an electrical trimmer and a hair removal cream (Veet), respectively. Mice received a combination of xylazine (5–7 mg/kg) and ketamine (35–50 mg/kg) before the wound application. In the next step to create open excision wounds, an 8 mm biopsy punch was pressed hard enough over the shaved area to remove circular parts of the skin tissue. Afterward, the animals were categorized into the NC, PC, and GX-MnO NPs formulation groups. NC mice remained without treatment, while PC mice were exposed to a standard treatment, that is, neomycin. The experimental group was treated with a formulation containing GX-MnO NPs (1.0 mg) in Vaseline (1.0 g). In both PC and experimental groups, treatments were applied topically on alternative days. Photographs were taken every other day for each wound, and the mean sizes of wound area were calculated using a simple ruler. The wound healing potential was determined as a percentage of wound contraction by comparing the initial measurement with subsequent measurements using the formula:
Dye degradation activity
The degradation performance of GX-MnO NPs was investigated using MB and MO as model pollutants under dark and sunlight stimulation. For photocatalytic experiments, as-prepared NPs (1–5 mg) were dispersed into 50 mL of 10 ppm dye solutions diluted from stock solutions of 5–10 mg/100 mL concentrations and exposed to solar irradiation for different time intervals (0, 20, 40, 60, 80, 100, and 120 min). The control samples were maintained in a dark setting without adding NPs to establish a comparative baseline. Throughout the irradiation process, concentrations of dyes were evaluated by taking out 3 mL of each treated solution every 20 min for 2 h. Similar experiments under identical reaction conditions were performed in the dark for comparative analyses. The disintegration percentages were tracked by monitoring the absorbance spectra between 300 and 800 nm and calculated using the given expression:
Herein “
Additionally, the kinetic mechanism of the dye degradation phenomenon was studied as per the following formula:
Where “Co” and “Ct” align with the amount of the dyes at initial and after exposure to sunlight for a certain period t, “
Results and discussion
Biosynthesis of GX-MnO NPs
The biological synthesis of NPs is superior to conventional physical and chemical approaches because of its eco-friendly, non-toxic, and biocompatible nature, thus favorable for biomedical and environmental applications. 46 Tons of NPs are being produced using biological entities such as polysaccharides and phytochemicals. 47 Quince seed hydrogel comprises a natural, high molecular weight GX-polysaccharide, 4-O-methyl-D-glucurono-D-xylan.35,48 The functional groups of GX chains are believed to be capable of reducing precursor salt and capping the surface of the NPs. 49 In this investigation, sunlight exposure of KMnO4 solution mixed with GX-hydrogel suspension for about 20–30 min led to a complete reduction of [Mn(GX)]2+ complex into [Mn(GX)]0 and subsequent capping of GX-MnO NPs. The preliminary evidence of the NP’s synthesis was obtained by a gradual color change from dark purple to blackish-brown. The finding correlates with the already-reported result where blackish-brown MnO NPs were produced using onion bulb extract. 50 Figure 2 represents the possible mechanism by which GX-polysaccharide residues synthesize MnO NPs in the reaction mixture of quince seed hydrogel and KMnO4.

General mechanism of reduction and capping of MnO NPs by GX-enriched quince seed hydrogel.
Characterization
UV-Vis absorption spectroscopy is a vital tool for assessing the optical response of a sample across different wavelengths, potentially providing information about its concentration and the energy band gap. The analysis was carried out to obtain the absorbance maxima of MnO NPs over the scan range of 200–800 nm. A prominent peak at around 310 nm was observed (Figure 3(a)), corroborating with absorption peaks reported previously.51,52 The peak location and blackish-brown color observed during the biosynthesis of MnO NPs corresponded to the localized surface plasmon resonance (LSPR) when surface electrons underwent collective coherent oscillation upon electromagnetic excitation. The size, morphology, and interparticle spacing are distinct properties of the NPs primarily governed by the LSPR phenomenon. 53 Band gap energy (Eg), also known as the energy needed to induce the movement of electrons from the valence to the conduction band upon excitation, was estimated by extending the linear portion of the UV-vis absorption band. Tauc’s method was considered to obtain the Eg value of GX-MnO NPs using the following relation.

(a) UV-Vis absorption peak and (b) Tauc’s plot representing Eg of GX-MnO NPs.
Herein,
FTIR method is an effective and non-destructive technique for identifying the presence of biochemical bonds within a sample over an extensive spectral range. The study was undertaken to reveal the formation of MnO NPs and corresponding peaks related to biomolecular constituents of GX-hydrogel linked with the NPs. Several prominent bands near 3282, 2924, 2858, 1731, 1599, 1416, 1245, and 1035 cm−1 were noted for the hydrogel. A broad band centered around 3282 cm−1 was tied to the presence of phenolic (O-H) groups, while two weak-to-moderate signals between 2800 and 3000 cm−1 were related to C-H stretching of alkanes.56,57 Multiple absorption peaks at 1731, 1599, 1416, and 1245 cm−1 represented the esterification of the carboxylic (COOH) group, C = O stretching of carbonyl group, C-H bending of alkanes, and C-N stretching of amines, respectively.33,58–60 The most prominent peak of the glycosidic association (C-O-C) of polysaccharides was visible at wave number 1035 cm−133, suggesting the existence of GX residues within the hydrogel network. After the formation of MnO NPs, the absorption bands shifted to 3232, 2942, 2354, 1595, 1331, and 1032 cm−1. The Intensities of bands at 3282, 1599, and 1035 cm−1 appeared to reduce, while the peaks at 2858, 1731, and 1416 cm−1 disappeared. New peaks at 2354 and 1331 cm−1 can be seen because of the C-H bending in polysaccharides.61,62 Vibrational modes detected around 545, 503, and 466 cm−1 confirmed Mn-O stretching, demonstrating the existence of as-synthesized NPs in the sample. The characteristic MnO peaks are compatible with the previously observed band. 52 All peak changes approved the function of GX-hydrogel in the reduction and efficient encapsulation of MnO NPs. FTIR spectra of the hydrogel and GX-MnO NPs are exhibited in Figure 4(a) and (b).
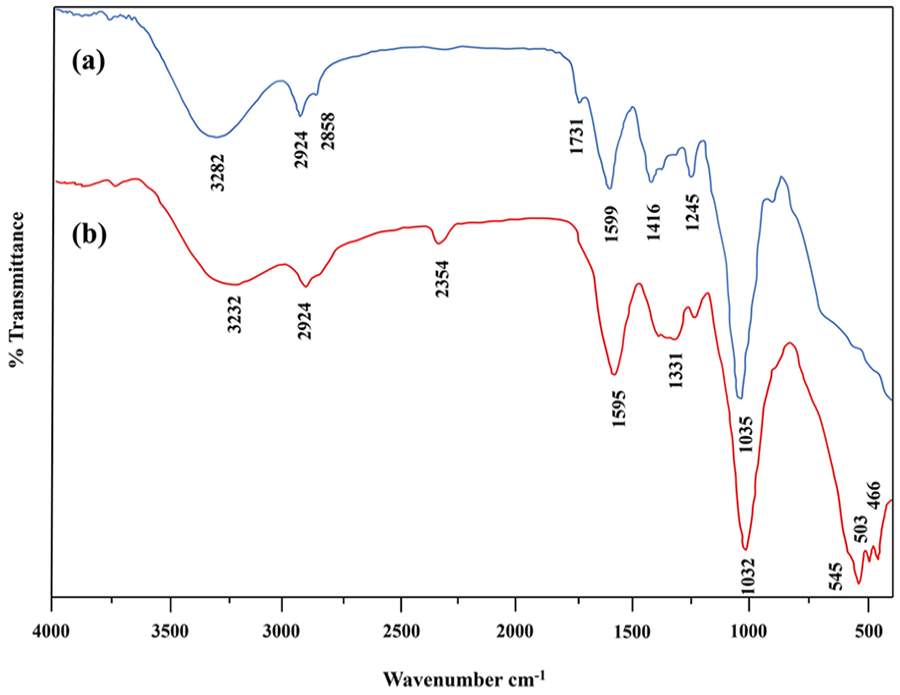
(a) FTIR patterns of quince seed hydrogel and (b) GX-MnO NPs.
XRD is a frequently employed technique for the investigation of crystallinity and phase composition of powdered/solid/liquid samples. XRD examination was performed on GX-MnO NPs, and multiple distinctive peaks were observed at 2θ values of 20.3°, 31.5°, 34.1°, 38.0°, 51.9°, and 65.8°, corresponding to (100), (110), (111), (200), (220), and (311) planes of the cubic structure (PDF#07-0230; Figure 5). Likewise, a similar scattering pattern was recorded for MnO NPs synthesized using green tea leaf extract. 16 The degree of crystallinity of the NPs correlates with the existence of highly intense characteristic peaks. Nanomaterials with ordered atomic arrangement can have unique properties such as enhanced electrical conductivity, catalytic activity, and antimicrobial properties. 63

The X-ray diffraction pattern of GX-MnO NPs.
The morphological dimension and the average size of GX-MnO NPs were revealed through the SEM technique, and micrographs were taken over a range of magnifications. The particles appeared roughly spherical, randomly dispersed, and aggregated, as presented in Figure 6(a) and (b). An inadequate reaction time may be the cause of aggregation/agglomeration, reducing the amount of capping agents on the surface of the NPs and preventing controllable growth. 64 The size distribution curve estimated using ImageJ software claimed an average particle diameter of 38.5 nm (Figure 6(c)). The morphological characteristics closely matched the earlier research work, where MnO NPs fabricated using Dittrichia graveolens extract exhibited a spherical morphology with a mean diameter of 38 nm. 65 Similarly, egg white liquid-mediated MnO NPs possessed a spherical shape with an average diameter of 29 nm. 66

SEM micrographs of GX-MnO NPs taken at: (a) 500 nm and (b) 1 µm magnifications, and (c) size distribution curve.
EDX scan was carried out to analyze the relative contents of compositional elements of the given sample, and the corresponding map is illustrated in Figure 7. The appreciable percentages of manganese (Mn: 39.29%) and oxygen (O: 29.3%) were identified in the spectrum, validating that MnO NPs have been correctly synthesized. The analysis also showed the distribution of other elements, including carbon (12.08%), sodium (0.69%), magnesium (1.04%), silicon (0.82%), chlorine (0.62%), potassium (14.23%), and calcium (1.92%). The existence of additional elemental signals could be related to the involvement of macromolecules of GX-hydrogel, playing an important role in the green production of the NPs. Recently, a study unveiled Mn (38.27%) and O (36.91%) as expected significant elements of MnO NPs produced from the filtrate of Bacillus subtilis. 67 In another study, the EDX spectrum displayed the mass percentages of Mn and O as 26.4% and 23.3%, respectively, 68 consistent with the current research. Thus, all characterization findings provided valuable insights into the ability of GX-enriched quince seed hydrogel to effectively synthesize MnO NPs under sunlight stimulation.

EDX spectrum of the green-synthesized MnO NPs.
Antibacterial assay
Pathogenic bacteria are a leading cause of long-term morbidity or even mortality worldwide and, thus, are one of the most urgent public health concerns. Traditional treatment modalities face complications as bacteria vigorously acquire resistance against the detrimental effects of multiple classes of antibiotics. 69 Therefore, developing alternative, unconventional, and more effective strategies to control/prevent infections is crucial. In recent years, MnOx NPs have drawn particular interest as promising candidates to cope with bacterial growth.16,70 In this study, the agar diffusion bioassay was used to appraise the antibacterial potential of GX-MnO NPs against Gram-positive and Gram-negative bacteria compared with a standard drug, that is, rifampicin. Clear zones around aqueous solutions of the NPs (100, 75, and 50 µg/mL) were observed against B. licheniformis (20.33 ± 0.33, 17.00 ± 0.58, and 12.33 ± 0.33 mm), E. coli (25.17 ± 0.17, 20.33 ± 0.33, and 16.50 ± 0.29 mm), and Aeromonas (20.33 ± 0.33, 16.33 ± 0.17, and 12.83 ± 0.44 mm; Figure 8). On the other hand, PC inhibited three bacterial strains with maximum zones of 15.33 ± 0.33, 17.83 ± 0.44, and 16.00 ± 0.29 mm, respectively, and no activity was noticed using NC (Figure 8). Consistent with this study, MnO NPs obtained by employing a cell-free supernatant of Bacillus subtilis ATCC6633 displayed impressive activity against Bacillus cereus (18, 20, and 23 mm) and E. coli (20, 23, and 25 mm) at concentrations of 50, 100, and 150 μg/mL, respectively. 71 Similarly, MnO NPs synthesized from green tea extract impeded the growth of E. coli, Klebsiella pneumoniae, and Pseudomonas aeruginosa with inhibition zones of 12, 14, and 18 mm, respectively. 16 The functionalization of MnO NPs with curcuminaniline biomaterial enhanced the antibacterial effectiveness against Staphylococcus aureus (18 mm), B. subtilis (19 mm), E. coli (10 mm), and Salmonella typhi (19 mm). 72

Antibacterial effects of GX-MnO NPs (100, 75, and 50 µg/mL) and positive control on the adherence of (a) B. licheniformis, (b) E. coli, and (c) Aeromonas.
The microbe-fighting capacity of the NPs was further screened via the MIC and MBC determinations. After 24 h of incubation, the tested strains, including B. licheniformis, E. coli, and Aeromonas, were inhibited by MnO NPs at minimum concentrations of 106.67 ± 3.33, 65.00 ± 2.89, and 108.33 ± 1.67 µg/mL, respectively, suggesting their role in effectively curtailing bacterial growth. The concentrations that were able to kill the selected isolates were recorded to be 156.67 ± 3.33, 137.67 ± 2.33, and 136.00 ± 2.08 µg/mL, respectively, confirming the bactericidal action of MnO NPs. One study demonstrated that the MIC value of biosynthesized-MnO NPs was lower for E. coli (15 µg/mL) than B. cereus (20 μg/mL). 71 Another study verified the MIC of Mycoendophyte-synthesized MnO NPs at 210 µg/mL. However, the MBC values were recorded between 200 and 280 µg/mL against a range of pathogenic bacteria. 73 The disparity in findings compared to the literature may be ascribed to the use of diverse synthetic methodologies and variable diameters of the NPs. 74 Bar diagrams of mean ZOI and MIC/MBC can be seen in Figure 9(a) and (b).

Bar graphs representing: (a) mean ZOI and (b) mean MIC/MBC against B. licheniformis, E. coli, and Aeromonas.
The ZOI values were more prominent at the highest applied concentration of MnO NPs (Figure 9(a)). Overall, GX-MnO NPs revealed superior antibacterial activity than rifampicin. It can be noticed from Figure 9(b) that the NPs imparted more excellent growth inhibition effects than bacterial killing, thereby representing a new perspective in inhibiting bacterial infections. This study documented a more potent inhibitory effect toward Gram-negative bacteria followed by Gram-positive bacteria. The assay results are related to previous studies, where MnO NPs prevented Gram-negative bacteria from multiplying more efficiently than Gram-positive bacteria.71,75 The increased resistance of Gram-positive bacteria might be due to a thicker cell wall, hindering the NPs from penetrating the cell. 76 In the case of Gram-negative bacteria, the increased negative charge may cause Mn2+ to bind more tightly to the cell wall, inducing more excellent antibacterial effects. 77 It is believed that the metal cations generated from the aqueous solution of metal oxide NPs are involved in disrupting DNA replication, damaging cell walls, generating ROS, producing oxidative stress, and inhibiting biofilm formation, protein, and essential cell structures, ultimately reducing bacterial cell viability.78,79 Figure 10 illustrates the possible action mechanism of the antibacterial potential of GX-MnO NPs.
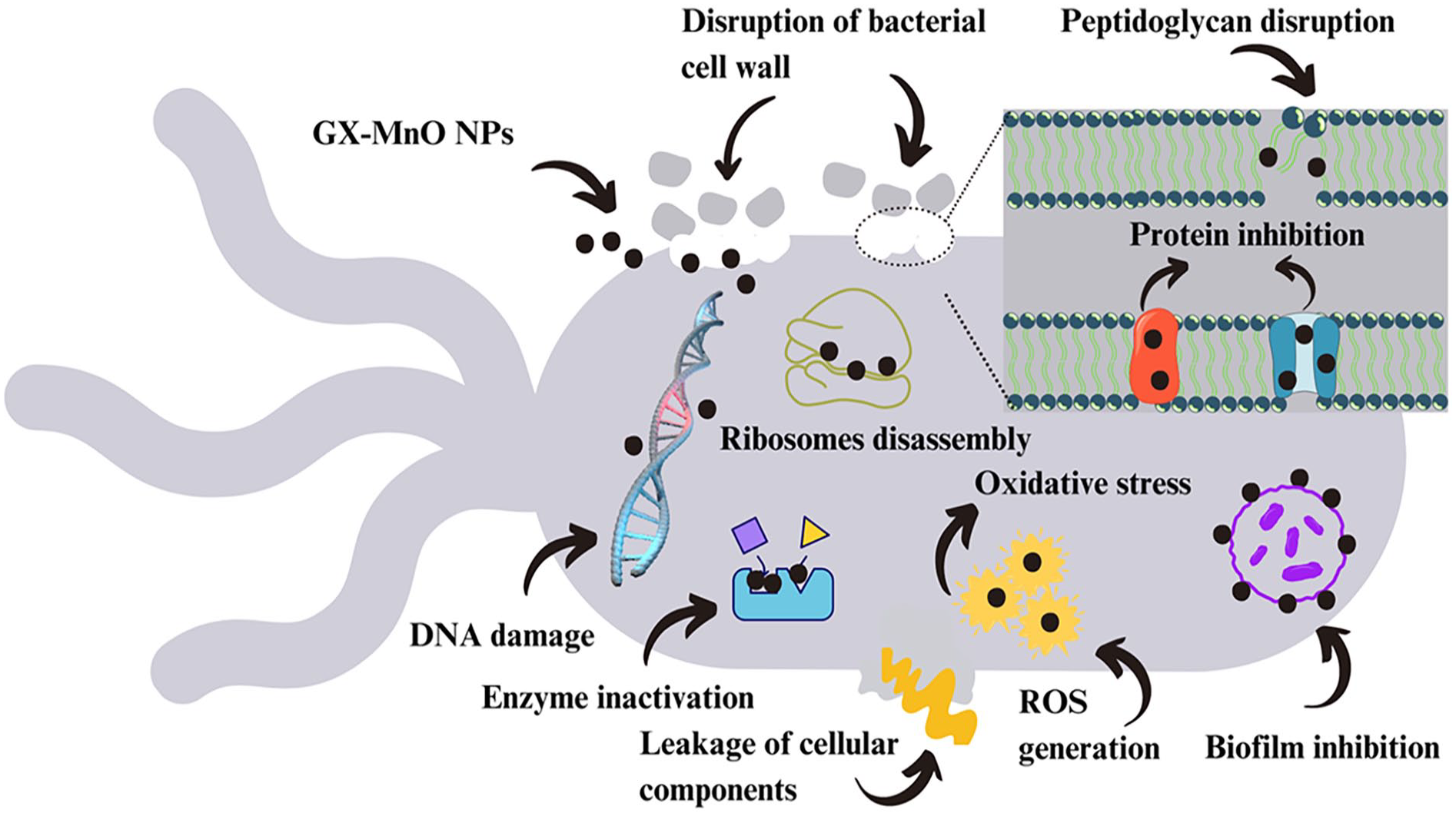
The general mechanism underlying bacterial cell inhibition by GX-MnO NPs.
Anti-biofilm assay
Biofilm, also known as microbial aggregation on a surface, has hindered the treatment of bacterial infections owing to its high tolerance rate toward many different antibiotics, 80 thus calling for urgent action to identify novel antibiofilm therapies. In this regard, GX-MnO NPs have been assessed for biofilm inhibition activity against B. licheniformis, E. coli, and Aeromonas. The individual bacterial suspension was introduced in a labeled test tube and maintained for 24 h, and then treatment of MnO NPs (100 µL) was added to each tube. The biosynthesized NPs reduced the biofilm formation by 20% ± 0.03% (B. licheniformis), 30% ± 0.03% (E. coli), and 21 ± 0.04% Aeromonas. Paenibacillus polymyxa strain-mediated MnO2 NPs exhibited a comparable antibiofilm potential (24.9% against Xoo strain GZ 0006), 81 similar to the results obtained in this study.
Wound healing activity
An open excision wound model in male albino mice assessed the healing efficacy of GX-MnO NPs. The dorsal wounds were created on the back of mice, and the NPs formulation and standard drug were topically applied every other day. Figure 11 discloses the images of untreated, neomycin-treated, and NPs formulation-treated wounds, providing a record of the healing process across different time points.
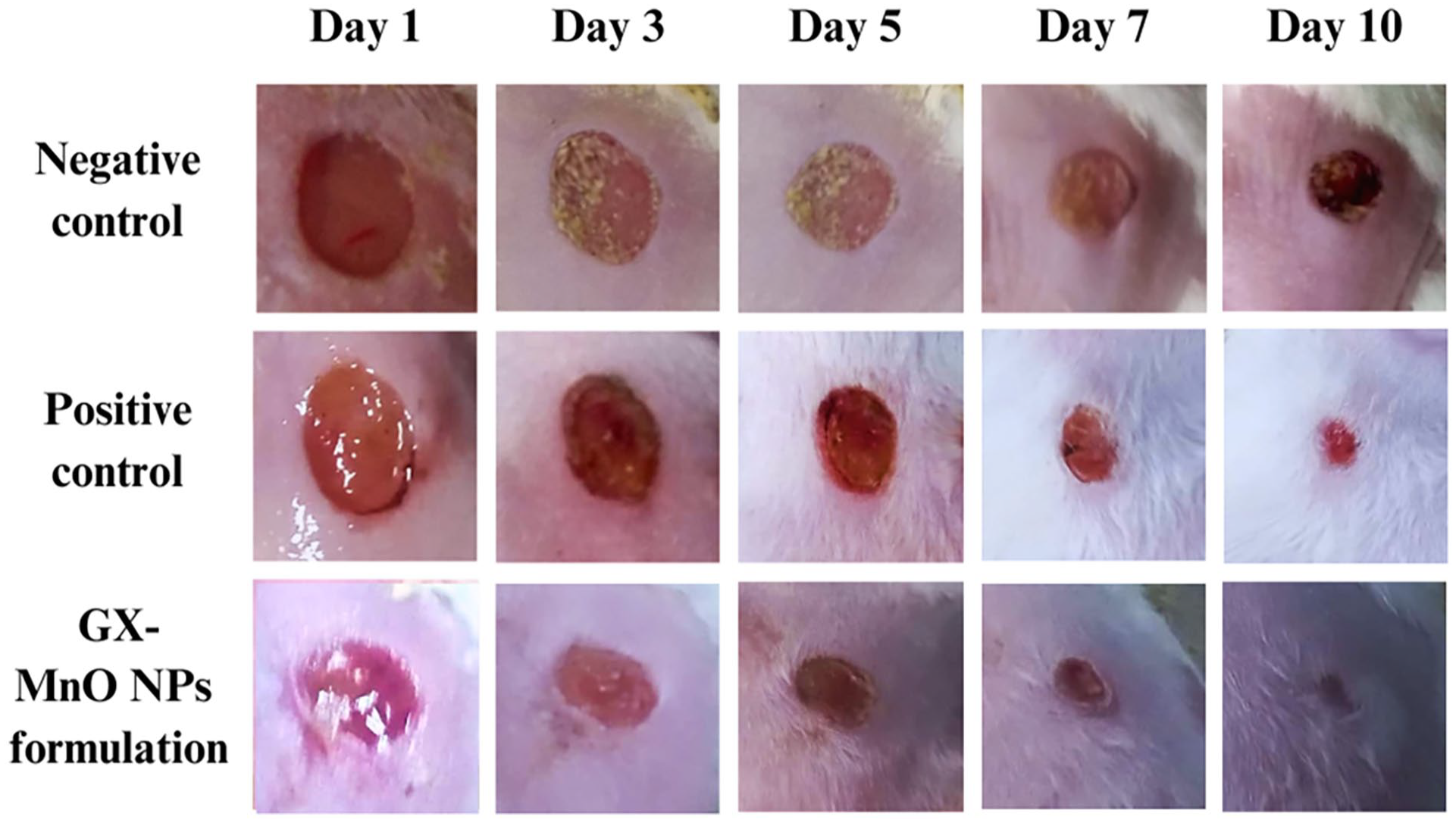
Photographs showing: (a) untreated, (b) neomycin-treated, and (c) GX-MnO NPs formulation-treated open excisional wounds.
The excisional wounds treated with the NPs formulation expressed an increased wound recovery rate from day 1 onward than those in the PC and NC groups. On the 10th day after the injury, the wound closure in the experimental group was 100%, whereas 83.2% and 62.6% reductions in the wound areas were observed in the PC and NC groups, respectively (Figure 12).
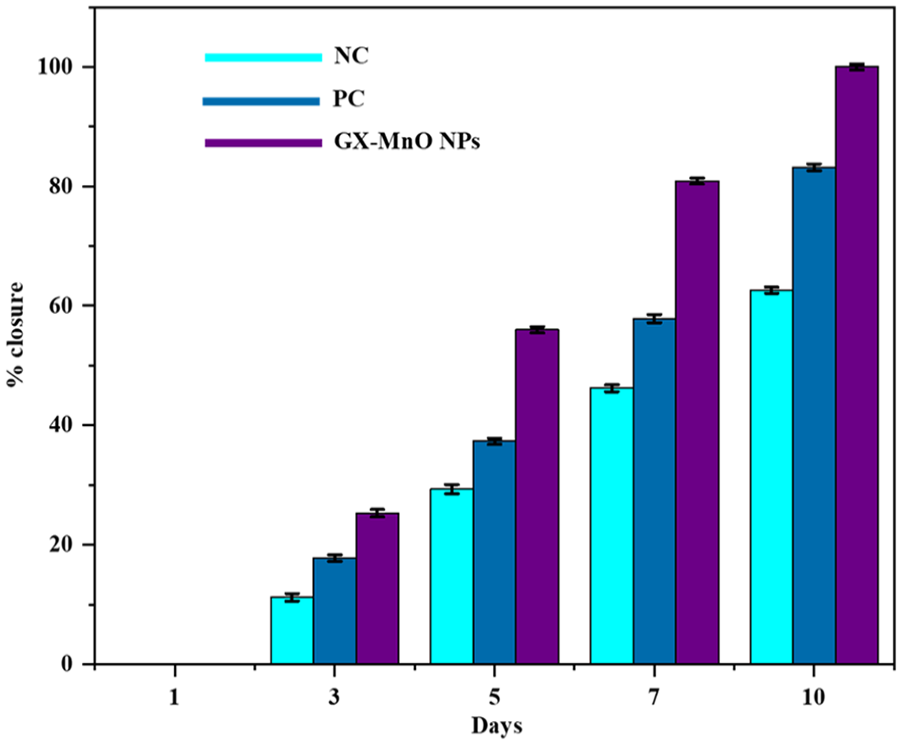
Comparison of wound healing between the studied groups over time.
In a related study, GX-Au NPs synthesized using GX-hydrogel extracted from quince seeds showed rapid wound recovery (90%) within 5 days in the murine model. 41 Similarly, the excisional wounds treated with Mimosa pudica seed hydrogel-mediated GX-Ag NPs-based dressing demonstrated 100% wound contraction on day 15 in male albino rabbits. The results of histological studies revealed remarkably accelerated re-epithelialization and tissue granulation in the healing region on the same day. 49 Moreover, MnNPs@ZC produced from an aqueous extract of Ziziphora clinopodioides Lam leaves enhanced the wound healing process (72.5% ± 2%) by promoting fibroblast/fibrocyte ratio, blood vessels, hexosamine, hydroxyproline, and hexuronic acid contents and reducing the number of lymphocytes, neutrophils, and total cells. 82 The wound healing property of the NPs has been attributed to a multifaceted mechanism that involves increasing collagen deposition, accelerating hemostasis and angiogenesis, regulating inflammatory responses, and imparting antimicrobial and free radical scavenging effects. 83
Dye degradation activity
Photocatalytic degradation is the most widely accepted and convenient technique for decomposing complex pollutants efficiently. The current work explores the catalytic activity of GX-MnO NPs under dark and solar irradiation using MB and MO as representative dyes. The gradual decolorization of suspensions indicated positive outcomes. The removal tests were carried out in batch systems, and the residual dye concentrations were determined spectrophotometrically. At optimum concentrations of dye solution (5.0 mg/100 mL diluted into 10 ppm, 50 mL) and GX-MnO NPs (5.0 mg), the removal of MB (47.6% and 90.5%) and MO (45.7% and 89.7%) reached its maximum within 120 min of incubation in the dark and sunlight, respectively. Higher production of photogenerated ROS, accelerated adsorption, and improved charge distribution are some of the crucial factors that contribute to relatively higher degradation efficiencies under sunlight exposure. 84 Figure 13(a)–(d) showcase dark and solar-driven decomposition spectra of MB and MO as a function of time by employing GX-MnO NPs.
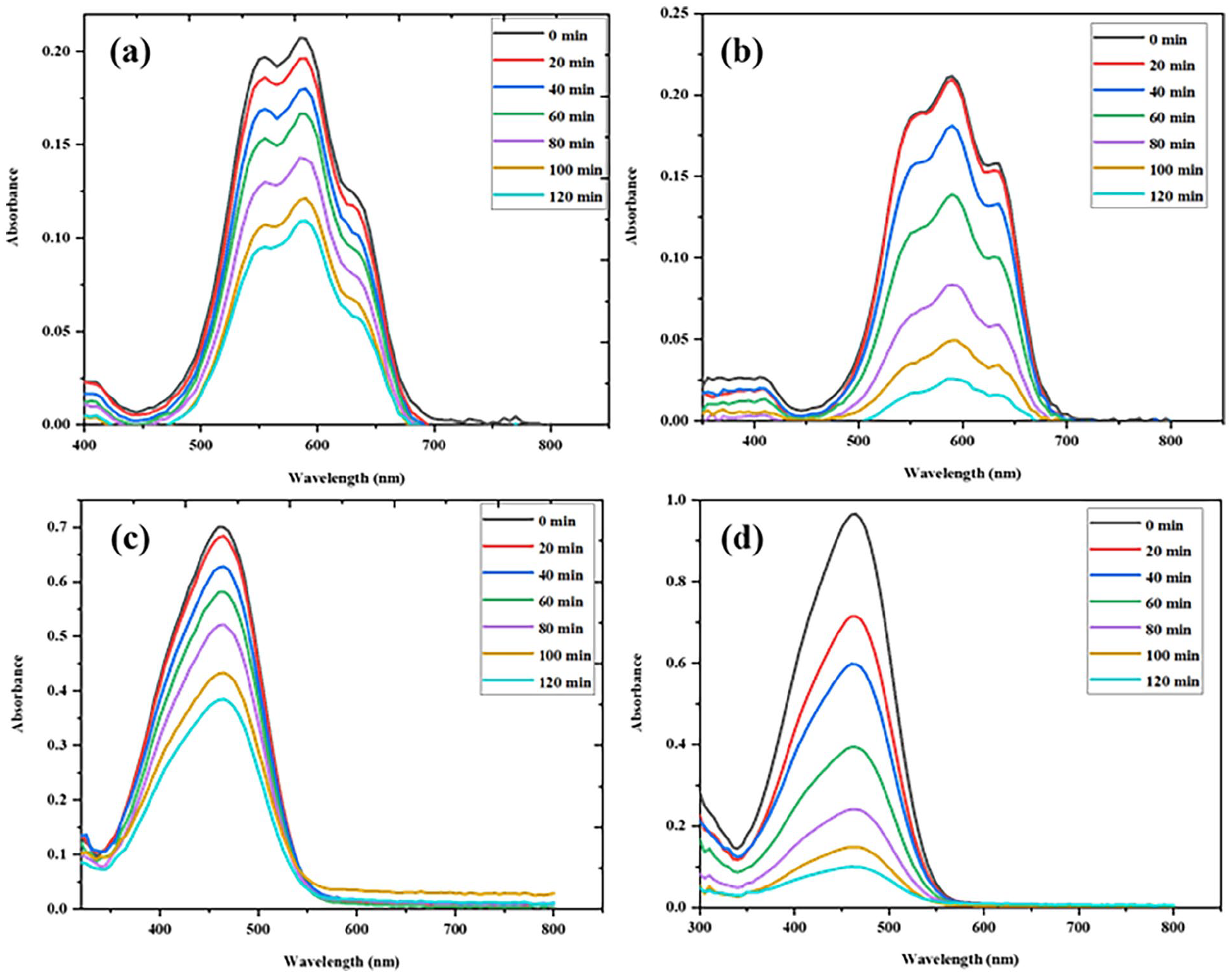
UV-vis absorption spectra of: (a) MB degradation in the dark, (b) MB degradation under sunlight, (c) MO degradation in the dark, and (d) MO degradation under sunlight in the presence of GX-MnO NPs.
Initially, λmax values of the MB and MO absorption spectra were around 590 and 464, respectively. It is noteworthy that the degradation potential of GX-MnO NPs peaked after 120 min across all cases (Figure 14(a) and (b)), thus, the reaction time can be regarded as another essential parameter to be considered in this phenomenon. The rise in catalytic activity concerning time is probably because of the activation of a greater number of active sites during prolonged interaction, contributing to an enhanced dye removal rate. 85
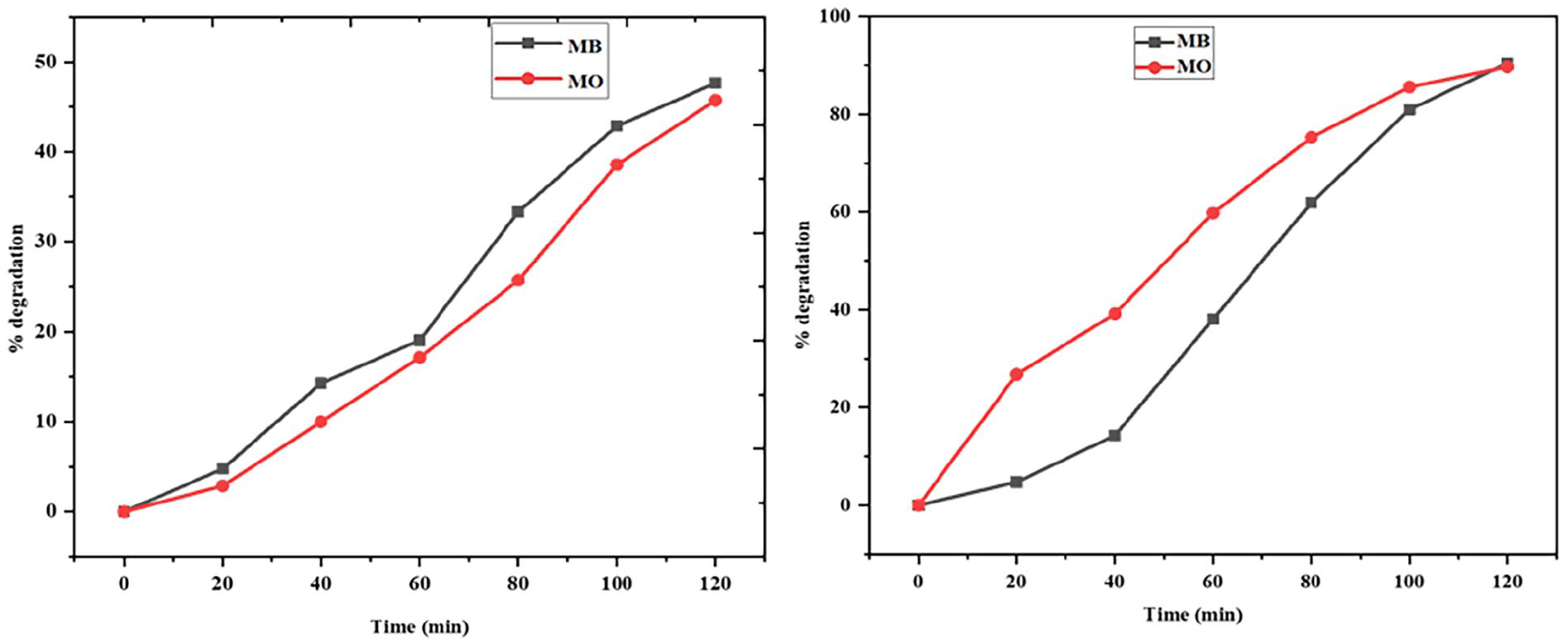
Time-dependent degradation rates of MB and MO: (a) in the dark and (b) under natural sunlight.
For better understanding, the trends of the experimental data were estimated according to the Langmuir-Hinshelwood expression. From the results, both dark and photodecomposition reactions represented pseudo-first-order kinetics with correlation coefficient (R 2 ) values greater than 0.9. Under solar illumination, GX-MnO NPs demonstrated the ability to degrade MB and MO at maximum rate constants of −0.017 and −0.019 min−1, respectively, while the rate constants of −0.006 and −0.005 min−1 were obtained when the tests were performed in the dark. The degradation kinetics of MB and MO obtained by plotting lnCt/C0 versus time have been illustrated in Figure 15.
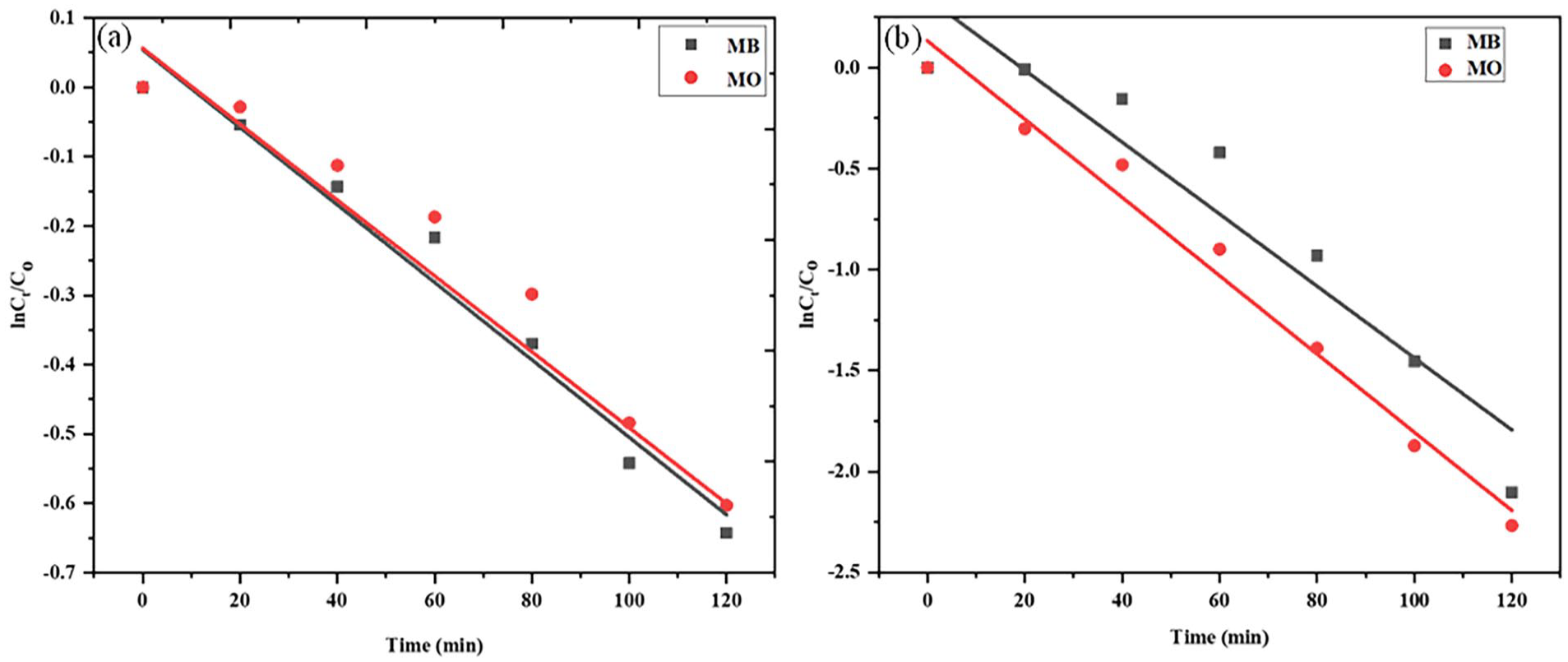
Pseudo-first order kinetic plots for GX-MnO NPs-mediated degradation of MB and MO: (a) in the absence and (b) presence of sunlight.
When exposed to sunlight of the desired/sufficient wavelength, semiconductor nanomaterials can generate an electron-hole pair. The fate of a photocatalytic reaction is decided by the relative rates of recombination and excitation of as-formed electron-hole pair and the photoredox processes. 86 Several researchers mentioned that the predominant moieties behind the degradation of organic pollutants are mostly ROS including photo-generated holes (h+), superoxide radicals (O2.−), and hydroxyl radicals (OH.).87,88 The surface adsorbed water molecules are oxidized due to the action of generated holes, while the excited electrons cause the reduction of adsorbed oxygen into O2−. radicals. 86 After that process, the dyes are wholly decomposed and converted into H2O, CO2, or inorganic ions, as reported by earlier experimental studies. 89 Figure 16 provides a mechanistic insight into the photocatalytic disintegration of MB and MO by GX-MnO NPs.
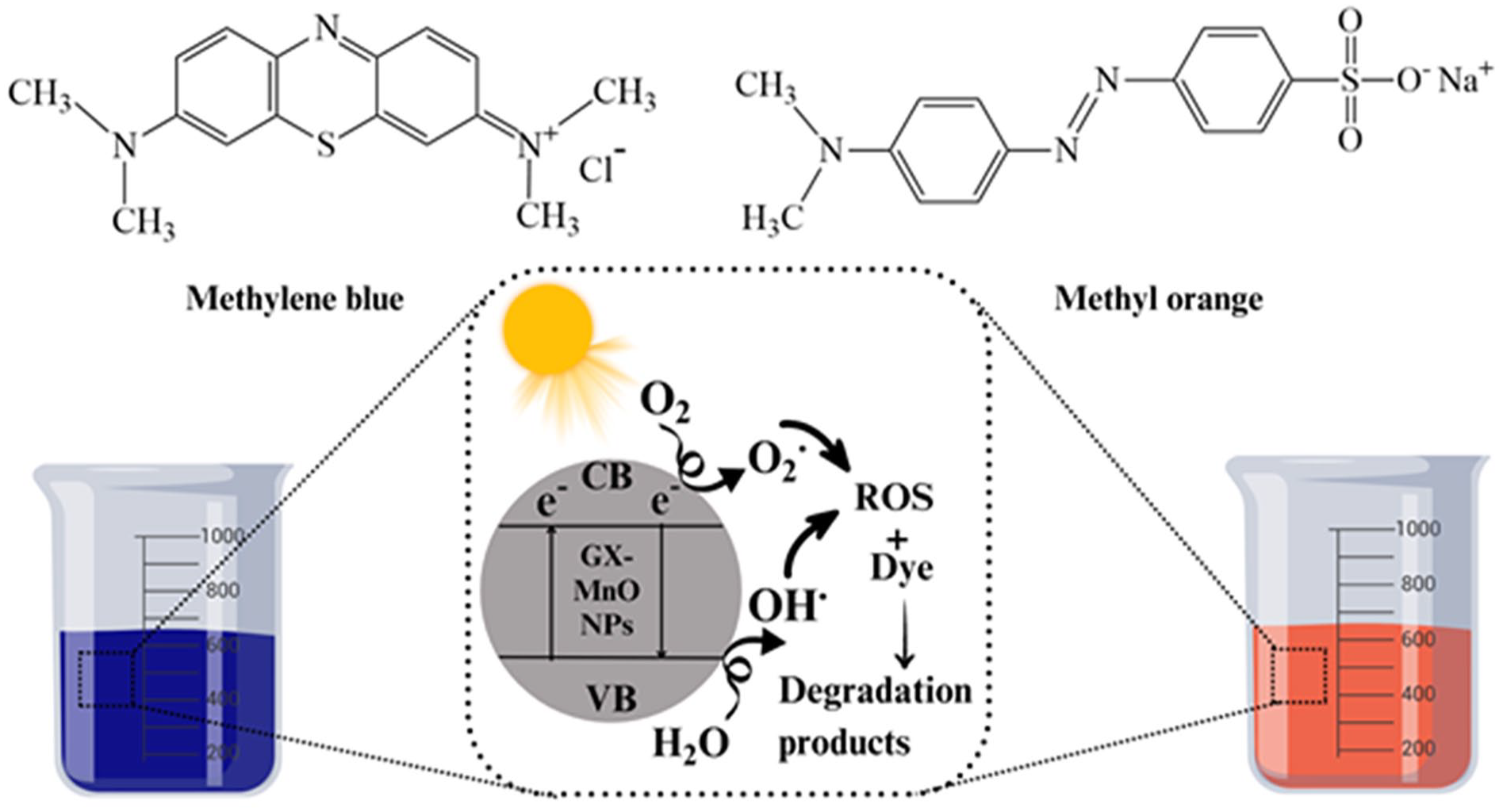
A plausible mechanism of action of photocatalytic activity of GX-MnO NPs.
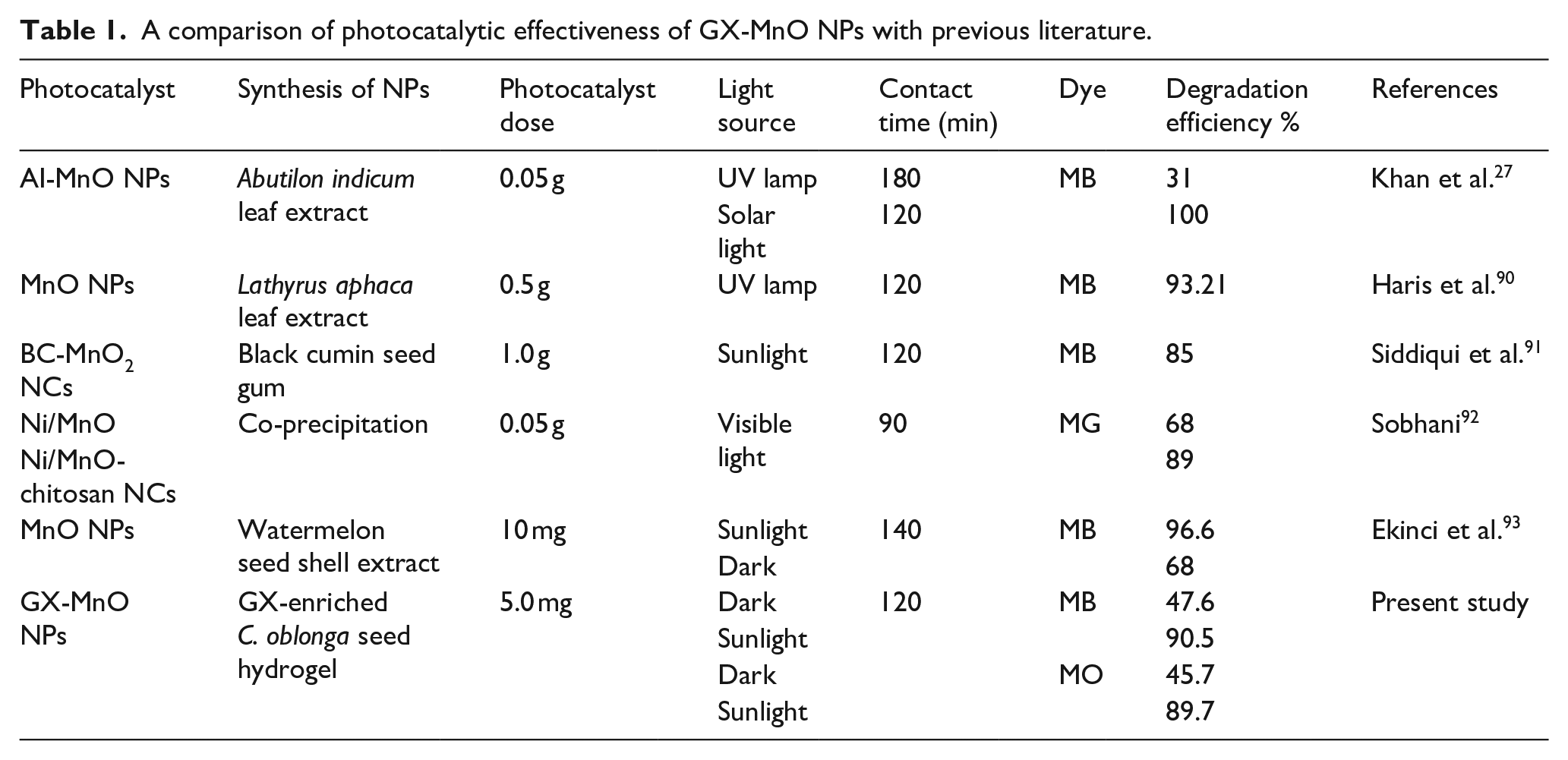
The photocatalytic breakdown of MB was undertaken by MnO NPs synthesized using the methanolic extract of Lathyrus aphasia leaves. The results indicated a promising degradation capacity of 93.21% under UV lamp irradiation. 90 In another study, black cumin seed gum-based Mn2O nanocomposite (BC/MnO2 NCs) showed 85% potential in removing MB from water under sunlight irradiation, 91 making them feasible and cost-effective candidates for wastewater treatment. When treated with Ni-mixed MnO and Ni/MnO-chitosan nanocomposite, malachite green (MG) dye experienced approximately 68% and 89% degradation, respectively. 92 In another study, MnO NPs produced from watermelon seed shell extract as a capping and stabilizing agent were employed to eliminate dye from aqueous media. The NPs were found to decompose MB up to 96.6% and 68% in the presence of sunlight and the dark, respectively, and followed the pseudo-first-order kinetics. 93 The effectiveness of Abutilon indicum leaf extract-mediated MnO NPs in removing MB was compared under the influence of UV lamps and solar light. In this study, the solar spectrum proved to have 100% efficiency in degrading MB within 120 min, making sunlight an excellent and renewable source to power photochemical reactions. On the other hand, the UV lamp induced only 31% disintegration of MB throughout 3 h. 27 This could be related to the narrower band gap property of semiconductor NPs, which allows them to absorb a larger portion of the solar spectrum, resulting in significant electron-hole pairs yield and, ultimately, an enhanced rate of disintegration. 94 Table 1 summarizes the different parameters (synthesis methodology, catalyst dose, light source, contact time, model dye, and percentage degradation) of degradation studies based on MnO NPs.
A comparison of photocatalytic effectiveness of GX-MnO NPs with previous literature.
Conclusion
This work employed an efficient and rapid approach for the biological production of GX-MnO NPs using GX-hydrogel extracted from quince seeds. The optical, structural, and morphological characteristics and chemical composition of as-developed NPs were studied through UV-Vis spectroscopy, FTIR, XRD, SEM, and EDX analyses. UV-Vis spectroscopy showed a strong absorption peak at 310 nm and a bandgap energy of 2.6 eV. FTIR technique reported the functional groups of GX-hydrogel involved in the reduction and capping processes, while XRD results confirmed a cubic structure for MnO NPs. According to the SEM analysis, the NPs were roughly spherical, with a mean diameter of 38.5 nm. At the same time, an EDX report identified the existence of elemental Mn and O in the fabricated nanomaterials. In the antibacterial study, GX-MnO NPs formed significant inhibitory zones against B. licheniformis, E. coli, and Aeromonas. Also, the biosynthesized NPs sufficiently eradicated biofilms formed by bacterial strains mentioned above. In vivo wound healing experiment displayed that GX-MnO-based nanoformulation showed 100% wound contraction on day 10 in male albino mice, representing an advancement in wound healing therapies. Under optimized experimental conditions, the NPs degraded MB (47.6% and 90.5%) and MO (45.7% and 89.7%) in the absence and presence of sunlight, respectively, therefore, offering a sustainable approach for wastewater treatment. Thus, in the near future, the NPs synthesized using GX-hydrogel as a biotemplate could have promising multifunctional potential in different biomedical and wastewater remediation industries.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical statement
The experimental design for animal use was approved by the Institutional Bioethics Committee (Ref. # GCU/IIB/270), GC University Lahore, Pakistan. It was conducted ethically following the ARRIVE guidelines and the U.K. Animals (Scientific Procedures) Act, 1986, and associated guidelines, EU Directive 2010/63/EU for animal experiments.




