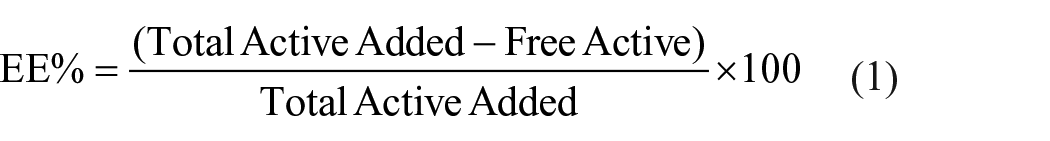Abstract
In this study, biocomposite membranes were developed by incorporating resveratrol (RSV)-loaded PCL-PEG composites, modified with graphene oxide (GO) and hydroxyapatite (HAP). The aim was to enhance hydrophilicity with GO and improve bioactivity with HAP. The release kinetics of RSV was evaluated by using Franz diffusion cells and compared with various kinetic models, including Korsmeyer-Peppas, Higuchi, and Baker, all of which showed high correlation coefficients (R²) close to 0.99. Mechanical tests was performed to determine the suitability of these membranes for tissue engineering applications. The composite membrane modified with GO and HAP exhibited tensile strength of 105.2 ± 5.8 MPa, tensile modulus of 3895 ± 159 MPa, elongation at break of 8.4 ± 0.9%, and toughness of 5.88 ± 0.46 MJ/m³. In vitro cell adhesion studies, visualized using DAPI fluorescence staining, demonstrated increased cell adhesion to the composite membranes over periods of 1, 3, 5, 7, and 14 days. These findings highlight the potential of the RSV-loaded PCL-PEG membranes, enhanced with GO and HAP, for applications in bone tissue engineering.
Keywords
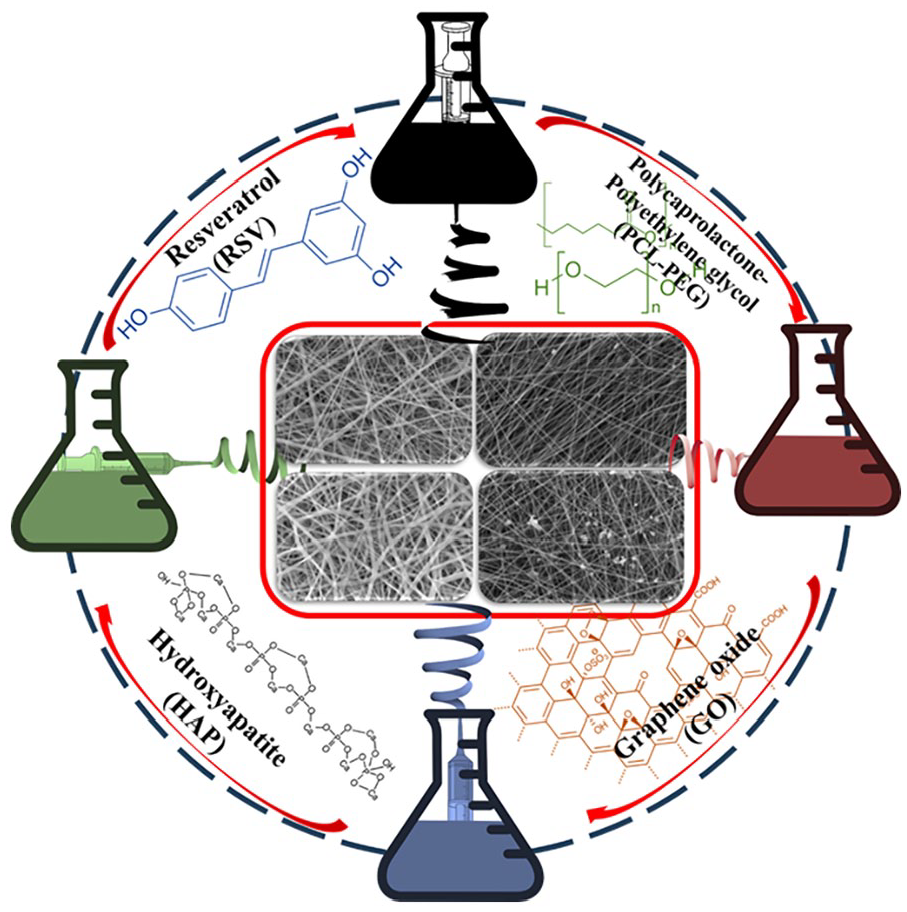
Graphical abstract of resveratrol (RSV) loaded PCL-PEG, PCL-PEG-Graphene oxide (GO), PCL-PEG-Hydroxyapatite (HAP) and PCL-PEG-HAP-GO biocomposite membrane produced by electrospinning method.
Introduction
Bone tissue engineering (BTE) has gained significant attention in recent years due to the increasing prevalence of bone-related disorders 1 and the limitations of current treatment options. BTE has emerged as a transformative approach to addressing complex challenges in the repair and regeneration of damaged bone tissue. The traditional methods for bone repair, including autografts, allografts, and synthetic bone substitutes, have constituted a pivotal aspect of clinical practice. 2 Autografts, which involve the harvesting of bone from the patient’s own body, are regarded as the gold standard due to their biological compatibility and osteogenic potential. However, these approaches are limited by donor site morbidity, surgical complexity, and the limited availability of donor bone. 3 Allografts, derived from cadaveric donors, do not require a second surgical site but are associated with risks of immune rejection, disease transmission, and variable incorporation into the host bone. 3 Synthetic bone substitutes, such as calcium phosphate ceramics and bioactive glasses, provide a convenient and reproducible alternative but often lack the biological stimuli necessary for optimal bone regeneration. 4 These traditional approaches, while demonstrating efficacy to varying degrees, are constrained by significant limitations. The major challenges encompass the necessity for optimized drug delivery systems to facilitate bone healing, the development of scaffolds with tailored mechanical and biological properties, and the provision of sustained release of therapeutic agents to enhance regenerative outcomes. Consequently, there is an unmet need for advanced materials that can address these limitations and promote more effective bone regeneration.
The development of scaffolds that are capable of effectively supporting bone regeneration is of great importance for the successful implementation of BTE applications. Among the numerous materials that have been explored, polycaprolactone (PCL), polyethylene glycol (PEG), graphene oxide (GO), and hydroxyapatite (HAP) have demonstrated particular promise as candidates for the creation of biocomposite membranes that can enhance the mechanical and biological properties of scaffolds.5,6 PCL is a biodegradable polyester that exhibits excellent mechanical properties rendering it a suitable candidate for bone tissue engineering applications. Its slow degradation rate allows for sustained support during the healing process, which is essential for large bone defects. 7 The integration of PEG into PCL scaffolds can enhance hydrophilicity and facilitate cell adhesion and proliferation, which are pivotal for efficacious bone regeneration. 8 The incorporation of PCL and PEG has been demonstrated to generate a conducive microenvironment for osteoblasts, stimulating their growth and differentiation. 9
GO has also attracted considerable interest in the field of BTE due to its distinctive properties, including a high surface area, mechanical strength, and biocompatibility. Moreover, GO has been demonstrated to enhance the osteogenic potential of scaffolds by providing a conducive environment for cell attachment and proliferation 10 GO can also enhance the mechanical properties of PCL scaffolds, rendering them more suitable for load-bearing applications. 7 The release of GO is beneficial for cells in some biomedical applications but may lead to toxicity in the absence of a controlled release mechanism. In addition, the release of GO at high concentrations may inhibit proliferation and trigger apoptosis in cells. This is particularly important for bone cells (osteoblasts) and cartilage cells (chondroblasts).11,12 Moreover, the integration of GO into PCL/PEG scaffolds has been demonstrated to enhance the release of bioactive molecules, which can further stimulate osteogenic differentiation. 13 HAP, a naturally occurring mineral form of calcium apatite, is also a crucial component in bone tissue engineering because of its structural and compositional similarity to bone mineral. HAP has the capacity to enhance the bioactivity of scaffolds, thereby promoting mineralization and integration with surrounding bone tissue. 14 Although HAP is considered an ideal material for bone regeneration, its excessive release during scaffold degradation can disrupt the balance of calcium and phosphate in the surrounding tissues, leading to mineral deposits (e.g., calcification). HAP particles can trigger an inflammatory response in surrounding tissues. This can impair tissue integrity and negatively affect the healing process.15–17
The combination of HAP with PCL and GO has been demonstrated to yield a composite scaffold that not only mimics the mechanical properties of bone but also facilitates osteoconductivity and osteoinductivity. 18 The incorporation of HAP into the scaffold markedly enhances calcium deposition, which is a vital process in bone regeneration. 9 Recent advances in the fabrication techniques for these biocomposite membranes, including electrospinning and 3D printing, have enabled the creation of scaffolds with tailored architectures and porosities that mimic the natural bone structure.19,20 The incorporation of hierarchical structures within the scaffolds can further enhance their mechanical properties and biological performance. 21
Resveratrol (RSV), a natural polyphenolic compound, has been shown to possess significant osteogenic properties. Moon et al. 22 demonstrated that RSV can enhance mitochondrial biogenesis and osteogenic differentiation in human periosteum-derived MSCs, suggesting its potential as a therapeutic agent in BTE. Similarly, Wajima et al. 23 reported that RSV treatment improved bone density and healing in type 2 diabetic rats, indicating its role in modulating osteogenic and osteoclastogenic mechanisms during bone remodeling. Moreover, Ozturk et al. 24 reported that RSV showed the preventive effects on bone quality deterioration caused by ovariectomy-induced osteoporosis in rats. Bo et al., 25 found that RSV supplementation positively affected bone health in type 2 diabetic patients, indicating its potential as an anabolic agent for enhancing bone density. These findings cumulatively suggest that incorporating RSV into scaffolds could provide additional benefits in promoting bone regeneration, particularly in patients with metabolic disorders. In addition to mechanical and biological enhancements, the use of RSV-loaded biocomposite membranes may also address challenges related to inflammation and oxidative stress in the bone healing process. RSV has also gained attention for its antioxidant and anti-inflammatory properties. 26 For instance, RSV has been shown to exert antioxidant effects, which can mitigate oxidative stress and inflammation, common issues in diabetic patients and those with compromised bone healing. 27 This property could further enhance the healing environment provided by the scaffolds, promoting better integration with host tissues. Although RSV is known for its antioxidant and anti-inflammatory properties, some problems can arise when it is released in large quantities from the scaffold. For example, sustained high doses of RSV may lead to unwanted stimulation or inhibition of cells surrounding the skeleton, potentially affecting the balance of cellular activities and interfering with bone regeneration processes.28,29 Therefore, the controlled release of RSV from the scaffold can provide a sustained therapeutic effect, further supporting bone regeneration. One of the primary challenges in incorporating RSV into scaffolds is achieving controlled and sustained release of the compound to maximize its therapeutic efficacy. Electrospinning has emerged as a versatile technique for fabricating nanofibrous membranes that offer high surface areas and controlled drug release properties.30,31 This method allows for the creation of scaffolds with precise control over fiber diameter and morphology, which are critical for drug delivery and tissue integration. 32
The development of RSV-loaded PCL-PEG/GO/HAP biocomposite membranes may represent a new promising approach in BTE. Accordingly, this study aimed to develop and characterize RSV-loaded biocomposite membranes using PCL-PEG, GO, and HAP for BTE applications. Towards that end, the primary objectives of this research were to evaluate the in vitro release kinetics of RSV from these biocomposite membranes and to assess their impact on cell performance.
Materials and methods
Polycaprolactone (PCL, Mw = 80.000 g/mol), PEG (Mw = 2000) and Resveratrol (RSV) were purchased from Sigma-Aldrich (Munich, Germany). Dichloromethane (DCM) was purchased from Merck (Darmstadt, Germany). All materials are chemically stable and biocompatible. Sulfuric acid (H2SO4), potassium permanganate (KMnO4), phosphoric acid (H3PO4) and hydrogen peroxide (H2O2) were purchased from Sigma-Aldrich to synthesis of Graphene oxide. Calcium nitrate tetra-hydrate (Ca(NO3)2·4H2O), ammonium di-hydrogen phosphate ((NH4)H2PO4) and ammonia solution (NH4OH) were supplied from Sigma-Aldrich/Germany to synthesis of Hydroxyapatite (HAP) and sodium tripolyphosphate (Sigma-Aldrich/Germany) was selected as the surfactant.
Synthesis of hydroxyapatite nano-powder
HAP nano-powder were synthesized via wet chemical precipitation method using previously described by Ozder et al. 33 HAP precipitate and water were formed by the reaction of acid and alkaline solutions (ammonium di-hydrogen phosphate and calcium nitrate tetra-hydrate, respectively) which were used to synthesize HAP nano-powder as precursors. The experimental temperature was set to 25°C and the pH of reaction medium was set to above 10 to achieve desired features of HAP. Briefly, 0.1 mole of calcium nitrate tetra-hydrate and 0.02 mole ammonium di-hydrogen phosphate were separately dissolved in 100 mL distilled water. Then, pH of the two solutions was adjusted by ammonium solution by 10 and ultrasonicated for 10 min. When these two solutions were dropped into one another, HAP nano-powder formed as a precipitate. The precipitated HAP nano-powder was washed with distilled water and centrifuged at 4100 rpm for 4 min and this process was repeated six times.
Synthesis of graphene oxide
Initially, a 1 L beaker was prepared, and into it, a mixture of 360 mL concentrated sulfuric acid and 40 mL phosphoric acid was added. To facilitate thorough mixing, a large stir bar was introduced, and the beaker was positioned in an oil bath, ensuring that the oil level surpassed the liquid level within the beaker. The oil bath was set to maintain a temperature of 60°C, while a thermometer was inserted into the synthesis volume to monitor and control the temperature. Simultaneously, the stir plate was adjusted to a stirring speed of 200 rpm. Subsequently, a gradual addition of 3.0 g graphite and 18.0 g potassium permanganate was carried out within the acid mixture and the reaction mixture was carefully poured into the ice-filled beaker. Additionally, 3–5 aliquots of 3 mL of 30 wt.% hydrogen peroxide were introduced. This induced a transition in the suspension from a purple hue to a vibrant yellow coloration with discernible bright white specks. To further purify the resultant product, it underwent a thorough washing process consisting of six successive rinses with distilled water, followed by centrifugation at 3000 rpm for a duration of 45 min. The acid component was subsequently disposed down the drain, accompanied by a copious amount of running water. The product was further subjected to washing with pure water, wherein each centrifuge tube was filled with pure water and a glass rod was utilized to disrupt the pellet. Once again, centrifugation was conducted at 3000 rpm for 45 min. This washing procedure was repeated three times, this time utilizing 35 wt.% concentrated hydrochloric acid. The pellet was gently disrupted using a glass stir rod, and the tubes were gently agitated to promote better mixing. Centrifugation was then repeated at 3000 rpm for 45 min. Following this, the product was washed three times with anhydrous ethanol and subjected to centrifugation at 3000 rpm for 45 min, yielding graphene oxide in an ethanol solution (GO).
Preparation of biocomposite nanofibers
PCL was dissolved with various percentages as 1, 3, 5, and 7 wt.% in Dichloromethane (DCM) was stirred with the magnetic stirrer at room temperature (23°C) for 1 h. After that step 0.005 g PEG and RSV were added separately in 3 wt.% PCL solution and mixed with magnetic stirrer for 1 h.
The GO and HAP materials were fully dissolved using three cycles of ultrasonication lasting for 2 h each, followed by 30 min of stirring to create dispersions. Cold water was periodically added to the ultrasonic bath to maintain a constant temperature. Next, the GO and HAP dispersions, each at a concentration of 1 wt.%, were mixed with 10% PCL-PEG in continuous magnetic stirring overnight at room temperature. A 10% PCL solution was selected due to its ideal properties for Electrospun materials.
A conventional Electrospun equipment (Inovenso Co., Istanbul, Fatih Sultan Mehmet Vakıf University, BIORGINE Lab, Turkey) was used to prepare pure PCL-PEG, PCL-PEG-RSV, PCL-PEG/GO, PCL-PEG/GO-RSV and PCL-PEG/GO/HAP/RSV composites. The mixture was introduced into a 10 mL glass syringe equipped with a 19-gauge stainless steel needle and subsequently subjected to the electrospinning process under constant conditions.
The selection of the electrospinning process parameters such as flow rate, needle-to-collector distance, voltage, temperature, and humidity are critical for controlling the morphology and properties of the resulting biocomposite fibers. 34 The parameters used were as follows: a flow rate of 1 mL/h, a needle-to-collector distance of 15 cm, a high voltage of 15 kV, and a controlled temperature of 25°C with a relative humidity of 40%. The resulting fibers were collected on an aluminum foil-covered, grounded rotary drum with a diameter of 100 mm. The drum rotated at a speed of 5 rpm, and the collection process lasted for 90 min. After collection, the biocomposite membranes were carefully dried for a minimum of 2 days under a fume hood to effectively eliminate any residual solvents. 31 The prepared biocomposite membrane (BCM) groups were named PCL-PEG, PCL-PEG-RSV; BCM1, PCL-PEG/GO; BCM2, PCL-PEG/GO-RSV; BMC3 and PCL-PEG/GO/HAP/RSV; BMC4.
Characterization
Morphological analysis
The morphological characteristics of the biocomposite membranes were examined using scanning electron microscopy (SEM) with an EVO LS 10 (ZEISS) microscope. The SEM was operated at an accelerating voltage of 5 kV (5 KX magnification) in secondary electron mode to capture high-resolution images of the membrane surface. To overcome the non-conductive nature of the polymer samples, all BCM specimens were sputter-coated with a thin layer of gold-palladium alloy to ensure adequate conductivity and image clarity. Fiber diameter distribution was quantitatively assessed using the Diameter J plugin for ImageJ software, which enables automated analysis of fiber diameters from SEM images. Diameter J processes the images by evaluating the diameter of nanofibers at each pixel along the fiber axis, generating a histogram to represent the diameter distribution. The tool also provides summary statistics, including the mean fiber diameter, offering a comprehensive characterization of the membrane’s morphological features.
Transmission electron microscopy (TEM) was employed to analyse the morphology of the synthesized GO. For this analysis (Hitachi HF-2000 TEM), 10 mg of GO powder was dispersed in 20 mL of ethanol. The suspension was then dropped onto carbon-coated copper grids and allowed to air-dry for 10 min. The prepared grid was subsequently positioned in the instrument chamber, and imaging was conducted at an accelerating voltage of 200 kV.
FTIR analysis
Fourier Transform Infrared Spectroscopy (FTIR) was performed using a Shimadzu spectrometer to identify the functional groups present in the biocomposite membranes. The analysis was conducted in the wavenumber range of 4000–400 cm−1, providing a comprehensive assessment of the molecular vibrations corresponding to various functional groups. 35 The samples were prepared and mounted appropriately to ensure optimal signal acquisition. The resulting spectra were collected and analyzed to determine the characteristic peaks, which were subsequently assigned to specific functional groups present within the biocomposite membranes.
XRD analysis
X-ray diffraction (XRD) analysis was conducted using a Panalytical XPERT-PRO diffractometer to determine the crystal structure of the biocomposite membranes. The measurements were performed using a Cu Kα radiation source, with an internal wavelength of 1.54 Å, powered by a 40 mA current and a 45 kV voltage generator. 36 The XRD patterns were recorded over a 2θ range from 10° to 70°, providing detailed information on the crystallographic phases and degree of crystallinity within the biocomposite membranes. The collected diffraction data were analyzed to identify and quantify the crystalline phases present in the samples.
Mechanical analysis
The mechanical properties of the BCM and PCL-PEG material were evaluated using a tensile mechanical test, following the standard procedure outlined in ASTM D882. 37 The samples were processed into rectangular strips with dimensions of 100 mm × 20 mm × 0.1 mm. The test was conducted using a universal testing machine, where the samples were subjected to a uniaxial tensile load at a constant crosshead speed of 5 mm/min until the fracture point was reached. The force and elongation were recorded throughout the test to calculate the tensile strength, elongation at break, toughness and Young’s modulus of the tested materials. Toughness was calculated as the energy absorbed by the BCM samples before the fracture by the area under the stress-strain curve.
Encapsulation efficiency and RSV loading efficiency
The experiment was designed to assess the release profile and loading efficiency of BCM samples loaded with RSV. The BCM samples were immersed in a phosphate-buffered saline (PBS) solution at pH 7.4, maintained at a concentration of 1 mg/mL. The RSV-loaded samples in the PBS solution were subjected to centrifugation at predetermined time intervals (15, 30, 45, 60, 120, 240, 300, 360, 420, and 480 min) for a total duration of 8 h. 38 After each centrifugation period, the process was halted, and the supernatant was carefully collected for further analysis. The UV absorption spectra of the collected supernatants were measured using a UV-visible spectrophotometer (UV-1280, SHIMADZU, Japan) to determine the concentration of RSV released into the PBS. Fresh PBS was used for each experiment to maintain consistent conditions throughout. The encapsulation efficiency (EE) and RSV loading efficiency (LE) were calculated using standard formulas to quantify the amount of RSV encapsulated within the PCL-PEG polymeric composites. The RSV loading in the composites was determined using equation (1), which relates the mass of RSV loaded to the total mass of the composite.
For determining loading efficiency, dried samples weight measured and loading efficiency were determined according to equation (2).
In vitro studies release kinetics model
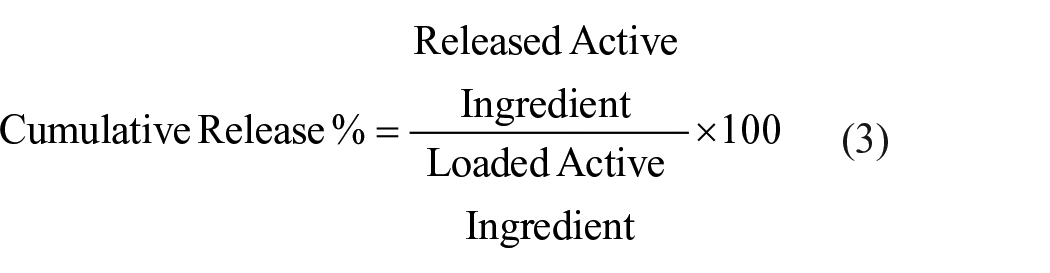
Release studies were performed with slight modifications of a procedure described in literature.39–41 Briefly, 10 mg of samples added into 50 mL of release media consisting of dH2O and EtOH in 3:2 volumetric ratio. Suspension mixed with a magnetic stirrer at 500 rpm, 1 mL of samples were taken hourly within the first 8 h and 24th h. Taken samples were centrifuged for 10 min at 9000 rpm and supernatants were analyzed at the proper wavelengths with Epoch II Multiplate Reader (Biotek Instruments). Released amount of active ingredients were determined with calibration curve calculations and cumulative release percentage was calculated depending on equation (3).
The release of RSV from the prepared biocomposite membranes were studied using Franz diffusion cells. The Franz diffusion device is a commonly used in vitro technique for measuring dermal absorption, where the active ingredient is applied to the surface of a skin sample. In the case of biocomposite samples, the most common method for dissolution testing involves using the Franz diffusion device. 26 In this study, the composites containing the active substance were placed in the membrane section of the Franz device. The experiment was conducted at a constant ambient temperature of 37°C with continuous mixing. At specific time points (15th and 30th min, 1st, 2nd, 4th, 6th, 8th, and 24th h), 2.5 mL of the sample was collected from the lower part of the diffusion cell and replaced with fresh medium preheated to 37°C. This process was repeated throughout the experiment. The collected samples were then analyzed using high-performance liquid chromatography (HPLC) to determine the amount of active substance present. The cumulative percentage of the active substance passing through the cells was plotted against time, and the cumulative percentage passed at the end of 24 h was calculated. Statistical analysis of the cumulative mass losses for the biocomposite membranes were performed using the Minitab program. The encapsulation efficiency and RSV loading were calculated using equations (1) and (2). The release kinetics of the RSV drug loaded into the BCM samples were analyzed by using different models; zero-order, first-order, Higuchi, Hixson, Korsmeyer–Peppas, and Baker mathematical model. The effectiveness of various release profiles was evaluated using the correlation coefficient (R2). It was determined that the mathematical model showing the highest correlation coefficient was the most appropriate representation of drug release kinetics. 26
Evaluation of cell line and adhesion
The BCM samples were subjected to sterilization before being cultured in DMEM, which served as the cellular nutrition source, resembling ECM. NIH 3T3 mouse embryonic fibroblast cells were utilized for the experiments and were cultured after isolation from a cell line. Each BCM sample was seeded with approximately 7 × 104 NIH-3T3 cell lines. After 72 h of cell culture, the cells were stained with DAPI (5 mM) dyes and subsequently examined using fluorescence microscopy to visualize their characteristics and behaviors.
Statistical analysis
The results were expressed as the mean ± standard deviation (SD) and were derived from three independent assays, each performed in triplicate to ensure reliability and reproducibility. Statistical analysis was conducted using one-way analysis of variance (ANOVA) to assess the significance of differences between groups. A p-value of less than 0.05 was considered to indicate statistical significance, ensuring that any observed differences were unlikely due to random variation. Post hoc analysis was performed, where applicable, to further evaluate pairwise comparisons between groups.
Result
Morphological analysis
As shown in Figure 1, SEM analysis demonstrated the successful fabrication of all the samples. The fiber diameter of the PCL-PEG composite was measured at 432.439 ± 150.946 nm (Figure 1(a)). The diameter of the BCM1 composite fibers, incorporating RSV into the PCL-PEG structure, was recorded as 466.64 ± 284.082 nm (Figure 1(b)). Comparison of the PCL-PEG and BCM1 composites indicated that the fiber diameter expanded upon drug loading. The HAP-incorporated PCL-PEG-RSV (BCM2) composite fibers exhibited a diameter of 623.74 ± 158.099 nm, attributed to the crystal structure and surface area of HAP (Figure 1(c)). In addition, the diameter of GO-incorporated PCL-PEG-RSV composite fibers was observed to be 517.85 ± 163.523 nm (Figure 1(d)). Further, the fiber diameter of the GO-HAP-co-incorporated PCL-PEG-RSV composite was observed at 1386.055 ± 355.193 nm (Figure 1(e)). A TEM image of GO (Figure 1(f)) showed that the samples possessed multiple layers due to their powdered form, with GO displaying an irregular shape, thick flat flake layers, a rough surface, non-wrinkled structure, and non-uniform particle size. Overall, the significant enlargement in fiber size of the drug-loaded PCL-PEG composite was attributed to the critical structure of HAP and the surface-wide architecture of GO.

SEM images (5 KX magnification) of (a) PCL-PEG, (b) BCM1, (c) BCM2, (d) BMC3, (e) BMC4, and TEM image of (f) GO.
FTIR and XRD
FTIR spectra revealed distinctive peaks of various samples, including standard compounds, physical mixtures, and BCM materials (Figure 2(a)). Significant changes in the peaks were detected in the spectra of composite materials with and without RSV, indicating notable differences in their composition. In the FTIR spectra, the characteristic peak corresponding to the -OH stretching of PCL was identified at 3442 cm−1. An additional peak at 1732 cm−1 indicated the presence of the aliphatic ester group in PCL. Furthermore, bands at 2858 and 2923 cm−1 were observed, corresponding to the CH2 stretching of PCL. Characteristic peaks for PEG were found in the following vibrational regions. The bands at 2942.9 and 2849.5 cm−1 were related to the symmetric and asymmetric stretching of CH2 groups, respectively. An intense vibration band related to the carbonyl group C=O was confirmed at 1720 cm−1. In addition, the asymmetric and symmetric C-O-C stretching vibrations were shown at 1240 and 1169 cm−1. The peak at 3431 cm−1 exhibited characteristic bands for the phenolic -OH group, the band at 897.1 cm−1 is attributed to C-H bending vibration.33,42 The same bands were observed for HAP, PCL-PEG in BCM3 and BCM4 structure. In BCM3 and BCM4 composites, the bands at 1028 and 983 cm−1 assigned to PO4−3 deformation confirm the presence of HAP. The spectra of BCM2 and BCM4 composites were observed to have a broad and intense absorption band in the region 3600–2400 cm−1 corresponding to some OH stretching vibration and two recognizable absorption bands at 1723 and 1619 cm−1.43,44

(a) FTIR spectra of the samples and (b) XRD patterns of the samples.
As shown in Figure 2(b), BCM composite structures had their own characteristic pattern. For the PCL-PEG structure, peaks at 20.8° and 22.5° were observed in the (111) and (200) planes. When HAP was added to the PCL-PEG structures, the XRD curves of the BCM3 and BCM4 composites confirmed the presence of hydroxyapatite due to the presence of new peaks at 2θ of 32° and 33° referring to the (211) and (300) planes, respectively. The presence of GO structures was confirmed by the peak observed at 12° in the (002) plane. This peak was not detectable in the composite materials due to the low amount of GO loaded in the BCM2 and BCM4 structures.26,45–48
Mechanical properties
The stress-strain plot of materials (Figure 3) provided valuable insights into the mechanical properties of the PCL-PEG and BCM’s. The composites fractured directly from the yield point with an elongation of PCL-PEG; 4.9 (%), BCM1;7.2 (%), BCM2; 9.0 (%), BCM3; 9.3 (%), and BCM4; 9.8 (%), respectively.

Stress-strain curves of samples.
As shown in Table 1, the detailed values of the mechanical properties were obtained from the stress-strain curves for PCL-PEG and BCM materials. A gradual increase in toughness was observed from sample BCM1 to sample BCM4, corresponding directly to the increasing incorporation of GO, RSV, and HAP materials into the PCL-PEG composites.45,49
Tensile properties of samples.

In vitro release study
In vitro release kinetic curves of BCM1, BCM2, BCM3, and BCM4 were shown in Figures 4 and 5. The results of the release kinetic analysis showed that the release model of correlation coefficient (R2) values was 0.9845 (Baker-Londsdale mathematical model) (Figure 4(f)), 0.9913 (Higuchi mathematical model) (Figure 4(d)), 0.9725 (Baker–Londsdale mathematical model) (Figure 5(f)), and 0.9773 (Korsmeyer–Peppas mathematical model) for BCM1, BCM2, BCM3, and BCM4, respectively. As a result, it was determined that the release profile of RSV drug was concentration dependent. To understand the drug release mechanism, the release data were analyzed using the Baker–Londsdale, Higuchi and Korsmeyer–Peppas equations, which give an idea about the diffusion type, respectively.26,50–53 The corresponding graph generated through the mathematical model equations exhibited a high degree of linearity. As shown by the correlation coefficient greater than or equal to 0.90, this value indicated that the super state release into three different models. The release profile showed a combination of diffusion controlled and swelling controlled RSV release.

Release kinetics of RSV active substance contents in BCM1 and BCM2 via Franz Diffusion mathematical models; (a) Korsmeyer–Peppas, (b) first Order model, (c) zero order, (d) Higuchi, (e) Hixson–Crowell, and (f) Baker–Londsdale.

Release kinetics of RSV active substance contents in BCM3 and BCM4 via Franz Diffusion mathematical models; (a) Korsmeyer–Peppas, (b) first order model, (c) zero order, (d) Higuchi, (e) Hixson–Crowell, and (f) Baker–Londsdale.
Cell line
The evaluation of cell attachment and proliferation on the BCM constructs was achieved through DAPI fluorescence imaging of live cells on the BCM samples after 1, 3, 5, 7, and 14 days of incubation (Figure 6). The obtained fluorescence images revealed noteworthy observations. Cells cultured on the surface of BCM2 and BCM4 samples (Figure 6) displayed a notably higher affinity for the PCL-PEG component when compared to both the control group and BCM1 samples.

Fluorescence microscopy images of cells treated with various samples on days 1, 2, 3, 5, 7, and 14, with cells stained using DAPI following treatment.
Significant increases in cell numbers were observed on all BCM samples tested compared to the PCL-PEG control group (* in Figure 7, p < 0.05) (Figures 6 and 7). The highest observed cell numbers were observed on BCM3 and BCM4 samples at day 7 and 14 (p < 0.05) (Figures 6 and 7), which were statistically similar. It was observed that all samples did not negatively affect the cells and allowed cell attachment and proliferation during the in vitro test period.

Percentage (%) of L929 cells attachment on the different samples.
Discussion
The ultimate goal of this study was to enhance the mechanical, biological, and drug delivery characteristics of the scaffolds to optimize bone tissue engineering. Towards that end, RSV-loaded PCL-PEG membranes were developed by incorporating with GO and HAP. Especially, the simultaneous release of GO, RSV and HAP and their interaction with each other can cause potential problems. For example, the propensity of GO to generate ROS may conflict with the antioxidant properties of RSV. This can lead to a complex mechanism of action in the cellular environment and can have both positive and negative consequences on the cells. As these three components have different bioavailability times, the prolonged presence of GO may cause chronic toxicity in cells in case of rapid degradation of RSV and HAP. To address this issue, the PCL-PEG polymer composite were chosen to regulate the controlled release of these materials. The electrospinning method was used to improve the release kinetics and to achive slow degradation of the three materials.54–56 The PCL-PEG matrix utilized in current study capitalized on the advantageous properties of both polymers. PCL, is known for its biodegradability and mechanical stability, was chosen to provide necessary structural support and PEG was chosen to improve hydrophilicity, which is critical for facilitating cell adhesion and proliferation. 57 SEM analysis confirmed that loading RSV into the PCL-PEG matrix did not alter the composite’s morphology (Figure 1). The nanofiber composite structures were consistent with those reported in previous studies58,59 confirming the reproducibility of the observed structures. The fiber diameter distribution varied across the samples due to differences in solution viscosity and the electrospinning process. For instance, PCL-PEG fibers exhibited a uniform distribution of fiber diameters, while the diameters of BCM samples containing GO and HAP show a slightly broader range. Among the samples, BCM4, which contains the highest doping of GO and HAP, exhibited the thinnest fibers, with average diameters of approximately 1386.055 ± 355.193 nm. This reduction in fiber diameter might be attributed to the influence of GO and HAP on the electrospinning solution properties, specifically by altering its viscosity and electrical conductivity, which agrees with findings from recent studies.33,60 The fiber diameter distribution across samples was consistent with that of collagen fibrils in the ECM, which typically range between 50 and 500 nm. 61 This similarity is crucial for mimicking the ECM structure in tissue engineering applications, where fiber dimensions comparable to those of natural collagen fibrils play a pivotal role in supporting cell attachment and proliferation. In terms of fiber diameters, the observations are consistent with recent literature. For example, studies on electrospun PCL and PCL-based composites often report fiber diameters in the range of 200–600 nm,62,63 which aligns with the range observed in the PCL-PEG and BCM samples. The inclusion of nanoparticles such as GO or HAP in electrospun fibers has also been shown to influence fiber diameter, often resulting in smaller fibers due to increased solution conductivity and a higher stretching force during electrospinning.64,65 Additionally, SEM images of biocomposite membranes incorporating GO and/or HAP revealed that these additives do not adversely affect the morphology of the composite. The structure remains intact and similar to that of the PCL-PEG and BCM1 composites, demonstrating that the incorporation of GO and/or HAP does not negatively impact the overall integrity of the composites. This finding is consistent with previous studies.66,67 Thus, RSV can be effectively loaded into the scaffolds while preserving their structural integrity.
Obtained results demonstrated that the mechanical properties of the modified membranes, including tensile strength, modulus, elongation at break, and toughness, were favorable, indicating that the scaffolds are robust enough for load-bearing bone defects (Figure 3 and Table 1). The incorporation of GO and HAP enhanced both the mechanical properties and biological activity, which are critical for scaffolds in BTE. These improvements are significant, as scaffolds must mimic the mechanical properties of natural bone to support physiological loads and promote cell attachment and proliferation. 68 Notably, the integration of GO into the PCL-PEG matrix not only improved the mechanical properties but also enhanced cell growth due to its high surface area and biocompatibility. 68 Obtained results showed that the BCM4 scaffold loaded with HAP exhibited the following optimal properties: the highest elongation at 9.8%, the greatest strength at 105.2 MPa, the best toughening effect at 3895 MPa, the best elongation performance at 8.4%, and the highest toughness value at 5.88 MJ/m³. These findings demonstrated the synergistic effect of combining GO, HAP, and RSV, which is consistent with previous studies.8,60 For example, in other studies, GO nanosheets containing Mg/Se- HAP co-substitution for wound healing applications were incorporated into PCL to be produced in nanofibrous scaffolds. The mechanical properties of the nanofibrous scaffold were examined and it was shown that the fracture toughness increased from 0.66 ± 0.12 MJ/m3 to 3.10 ± 0.21 MJ/m3 at the lowest and highest GO, respectively, while the fracture strength increased from 0.81 ± 0.22 MPa to 4.4 ± 0.45 MPa. 69 In another study, mechanical results on PCL, HAp, GO nanocomposites showed that mechanical properties decreased when GO content was increased, while PCL regained its mechanical properties when HAP was added to the matrix. 70 Obtained results showed that the incorporation of GO, even at a low concentration, played a crucial role in promoting cellular adhesion to the RSV drug (Figures 6 and 7), leading to an enhanced degree of proliferation. Therefore, the inclusion of GO in the biocomposite construct had a positive impact on cellular adhesion and proliferation of the RSV drug, signifying its promising potential for beneficial applications in bone tissue engineering. The improvements observed in this study are consistent with previous research, which demonstrates that GO enhances scaffold performance by creating a more supportive environment for cellular activities.71,72 Similarly, it may be assumed that HAP, a mineral component resembling natural bone, boosts the bioactivity of the scaffold, fostering mineralization and better integration with surrounding bone tissue. 57 Previous studies demonstrated increased cell adhesion and proliferation over time, with HAP likely contributing to these effects due to its osteoconductive properties.73–76 Obtained results showed that the incorporation of GO and HAP into the substrate enhanced the pharmaceutical efficacy and tissue engineering applicability of the PCL-PEG polymeric material. These additions did not render the BCM structures cytotoxic. Furthermore, they improved the chemical stability of PCL-PEG/RSV by imparting the bioactive properties of HAP and enhanced tissue engineering applicability by introducing the hydrophilic properties of GO.46,48,54 Therefore, the synergistic effects of PCL, PEG, GO, and HAP can result in a scaffold that effectively balances biological activity and mechanical properties.
The controlled and sustained release of RSV, as demonstrated by high R² for Korsmeyer–Peppas, Higuchi, and Baker models (Figures 4 and 5), is crucial for maintaining therapeutic levels over time and ensuring effective bone regeneration. The overall results of the release studies indicated the suitability of RSV-loaded composite conjugates as a means of providing sustained amounts of RSV. The initial therapeutic burst release of RSV indicates that by delivering a high concentration of the drug immediately, the tissue can help to immediately control or reduce the spread of disease. The sustained release phase of RSV was observed due to the remaining of active drug over time after the initial burst. The obtained results indicated that the overall treatment may lead to improved therapeutic outcomes as well as more effective. Overall, the initial burst release functions as a loading dose that effectively controls disease progression. Moreover, the sustained release phase further enhances therapeutic efficacy and outcomes.26,46,77 However, in vitro release kinetics might not fully capture the complexities of the in vivo environment. Factors such as polymer degradation, RSV interactions with biological fluids, and enzyme activity could impact the drug release profile in a physiological setting.78–80
Several limitations of the study should be considered in the interpretation of these results. These results were obtained under controlled conditions and may not fully replicate the in vivo environment. Further research, including in vivo studies, is necessary to assess how these scaffolds interact with live tissue and support bone regeneration in a more complex biological context. Another limitation of the current study is the use of DAPI dye, which primarily stains the nuclear structure. While methods like F-actin staining are commonly employed to observe the longitudinal shape of the cell, the nuclear morphology observed with DAPI can still provide valuable insights into cell shape. Many studies have demonstrated the relationship between nuclear morphology and overall cell shape.81–83 Additionally, the long-term stability and degradation of the biocomposite membranes were not evaluated, which are critical for understanding the membranes’ performance over time. Moreover, while the release kinetics models offer insights into drug release, they do not account for variability due to physiological conditions such as pH changes, enzyme activity, and interactions with biological fluids.
Conclusion
The novelty of this study lies in its multifunctional biocomposite membrane (BCM4), which incorporates a well-optimized combination of PCL-PEG polymers with GO, HAP, and RSV, leading to an advanced scaffold with superior mechanical properties, morphological integrity, and sustained drug delivery potential. These scaffolds represent a significant improvement over existing models by offering both enhanced mechanical strength and sustained drug release, making them potentially ideal candidates for bone tissue engineering applications. In summary, RSV-loaded PCL-PEG/GO/HAP biocomposite membranes hold significant promise for bone tissue engineering since they improve mechanical and biological properties while providing a controlled and sustained drug release profile. This composite system can potentially enhance osteogenic differentiation, mechanical properties, and bioactivity, thereby improving the efficacy of scaffolds used for bone regeneration. Continued research and development are still crucial for refining these scaffolds and enhancing their clinical outcomes in bone repair and regeneration. Future research should address the limitations by conducting in vivo preclinical and clinical trials to evaluate the performance of RSV-loaded biocomposite membranes under real physiological conditions. Further optimization of scaffold designs and advanced imaging techniques could enhance the clinical applicability of these materials.
Footnotes
Acknowledgements
The authors would like to thank Fatih Sultan Mehmet Vakıf University Technology Transfer Office and Biomedical Department Biomaterials BİORGİNE Laboratory for supporting this study.
Author contributions
Betül Meryem Arpacay: Conceptualization, Methodology, Investigation, Formal analysis, Writing – original draft, Visualization. Fatih Ciftci: Conceptualization, Methodology, Investigation, Formal analysis, Writing – original draft, Visualization and Supervisor. Ali Can Özarslan: Conceptualization, Methodology, Formal analysis, Writing – review and editing. Mustafa Unal: Conceptualization, Methodology, Writing – original draft, Writing – review and editing. Mine Kucak: Conceptualization, Methodology, Writing – review and editing. Aslihan Yelkenci: Conceptualization, Methodology, Writing – review and editing.
Data and code availability
The authors confirm that the data supporting the findings of this study are available within the article. Raw data that support the findings of this study are available from the corresponding author, upon reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.